Abstract
A new method is proposed based on temperature-controlled self-reaction to generate and release inhibitors in the form of foam at a specific temperature, which can overcome the disadvantages of short effective time and low efficiency in the inhibition of the spontaneous combustion of coal when inhibitors are released in advance, and greatly increase the action range of inhibitor through foam diffusion. The proposed temperature-controlled foaming system was prepared with hollow spheres as solution carriers, NaHCO3 and acetic acid as basic reactants, reaction-generated CO2 as foaming gas, sodium dodecyl sulfate and sodium carboxymethyl cellulose as foaming additives, NaCl, MgCl2 and CaCl2 as additives to enhance inhibition effect, and temperature sensitive paraffin as insulating material to generate and release inhibitor foam. The effects of releasing foam on restraining the spontaneous combustion of coal were studied based on the experimental analysis of the optimum ratio of reactants and additives, hollow sphere parameters, released temperature of the foam and variation rule of CO generated by coal oxidation. The obtained results showed that the released temperature of foam was 59–61°C and the covered area of pulverized coal by inhibitor foam was 12.9–13.9 times higher than when it was directly wetted by inhibitor solution. The total effects of inhibition and inerting were achieved after pulverized coal was wetted with inhibitor foam and the efficiency of restraining the spontaneous combustion of coal reached 88.51–97.06% when temperature was increased to 160°C.
Introduction
In major coal producing countries, such as China, the United States, Australia and India, coal fire disasters are very common and are considered as a global disaster problem (Havva, 2007; James et al., 2009; Kong et al., 2017; Mohalik et al., 2017; Parsa et al., 2017; Wang et al., 2018; Yang and Qiu, 2019). More than 95% of Chinese collieries perform underground coal mining, in which fire accidents due to spontaneous coal combustion account for more than 90% of the total number of mine fires (Deng et al., 2008; Fan et al., 2020, 2019). Especially, certain amounts of coal are left in goafs after mining as broken accumulations. Due to air leakage and oxygen supplement, slow oxidation and spontaneous combustion of coal can take place in goafs generating toxic gases which diffuse to working faces and seriously threaten the health of workers (Chu et al., 2017; Taraba and Zdeněk, 2011; Yuan and Smith, 2008). If coal contains high amounts of pyrite, it is more prone to spontaneous combustion (Yang et al., 2019).
Restraining the spontaneous combustion of coal with inhibitors is one of the common fire prevention methods. According to the inhibition mechanism of spontaneous coal combustion, inhibitors can be divided into two groups of physical and chemical inhibitors (Li et al., 2012a). Physical inhibitors are mostly water-absorbing salts such as CaCl2, MgCl2 and NaCl (Ma et al., 2015; Wang et al., 2015; Xiao et al., 2017). On the other hand, chemical inhibitors, such as antioxidant and ionic liquids, can react with active molecules and free radicals generated by coal at low temperatures, thus interrupting free radical reaction chain of oxidized coal (Li et al., 2017, 2018; Wang et al., 2014). Due to the low cost, high availability and simple operation process of physical inhibitors, they are much more commonly applied in collieries than chemical inhibitors. Many research works have been conducted on the inhibition mechanism of physical inhibitors and their effects on restraining coal oxidation. Smith et al. (1988) evaluated the performance of ten inhibitors in the inhibition of the spontaneous combustion of bituminous coal and found that NaNO3, NaCl and CaCO3 were the most effective inhibitors, followed by NH4H2PO4, CaCl2, NH4Cl, NaAc and KCl, while HCOONa and H3PO4 enhanced spontaneous coal combustion. Zhan et al. (2011) found that Na3PO4 improved the thermal stability of coal and effectively inhibited the generation of free radicals. Watanabe and Zhang (2001) experimentally studied seven inhibitors and found that NaCl, CaCl2 and Mg(Ac)2 inhibited the low-temperature oxidation reactions of coal while Ca(Ac)2 was only slightly effective. Václav and Taraba (2012) carried out thermoanalytical studies on the influence of CaCl2 and urea on coal oxidation and concluded that both inhibitors decreased oxidation heat at 100–250°C. Gao et al. (2012) showed that NaHCO3, KHCO3 and NH4HCO3 had good resistance to spontaneous coal combustions.
In the early stages of inhibitory action on coal, inhibitors play an important role in restraining spontaneous coal combustion. However, they have a certain effective resistance duration. With the loss of water, their inhibition efficiency is greatly decreased (Li et al., 2012b). In maintaining the high efficiency of inhibiting spontaneous coal combustion, the application of temperature sensitive materials as coating to allow the release of inhibitors at a specific temperature was investigated (Cui et al., 2017; Li et al., 2016a; Ma et al., 2016, 2017; Qi et al., 2016). However, they did not consider the effect of diffusion and action range of inhibitors, and it was found that the covered area of coal was extremely limited after solid or liquid inhibitors were released. The uncoated coal was still normally oxidized.
To solve the above-mentioned problems, a new method has been proposed in this paper for the spontaneous release of inhibitor solutions in the form of foam at a specific temperature through temperature-controlled self-reaction. Using hollow spheres as inhibitor solution carriers, foaming and stabilizing agents were added into the solution and the inert gas needed for foaming was generated by self-reaction. The effects of reactant concentration, temperature and halogenated salt additives on the foaming and stabilizing properties of the foam were evaluated through the chemical reactions of foaming. The optimum values of volume and placement angle of hollow spheres were determined by the release efficiency of inhibitor foam. Through temperature programmed experiments, the efficiency of inhibitor foam on restraining spontaneous coal combustion was found to be higher than unfoamed inhibitors.
Materials and methods
Experimental materials
Experimental coal samples were collected from Tashan colliery, Datong, China. Table 1 lists the proximate analysis of coal. The collected coal samples were ground and sieved to particle size of 0.18–0.38 mm and were kept under an inert atmosphere.
Proximate analysis (air-dried basis) of the coal sample.

NaHCO3 and acetic acid (HAc) were selected as basic reactants. The chemical equation of the reaction of NaHCO3 and HAc is shown in equation (1). Both NaAc and H2O in the reaction products could be used for the inhibition of spontaneous coal combustion. In addition to being used as foaming gas, CO2 also effectively inerted the atmosphere. The reaction was endothermic, avoiding the traditional exothermic reaction of carbonate and acid which promoted coal oxidation.

Polytetrafluoroethylene (PTFE) hollow spheres were used as solution carrier and circular paraffin slice was used to isolate the two reactants inside hollow spheres (Figure 1). 52# paraffin wax with melting point 52°C was used in all experiments. The diameter of circular paraffin slice was equal to that of hollow spheres and its thickness was 1.5 mm. Paraffin slice was melted at a certain temperature causing the contact and reaction of NaHCO3 and HAc. Then, a large amount of CO2 was generated and hollow spheres were split under high pressure, releasing the inhibitor. The wall thickness of hollow spheres was 2 mm and they were split into hemispheres A and B from the middle. Hemisphere B was used to hold HAc solution and cement paraffin slice. The cementation surface of hemisphere B was engraved with argyle to increase the cementing stability of paraffin slice. Hemisphere A was used to hold NaHCO3 solution and merged into the whole hollow sphere with hemisphere B. The interface of hemispheres was sealed with polyvinyl alcohol (PVAL) liquid adhesive.
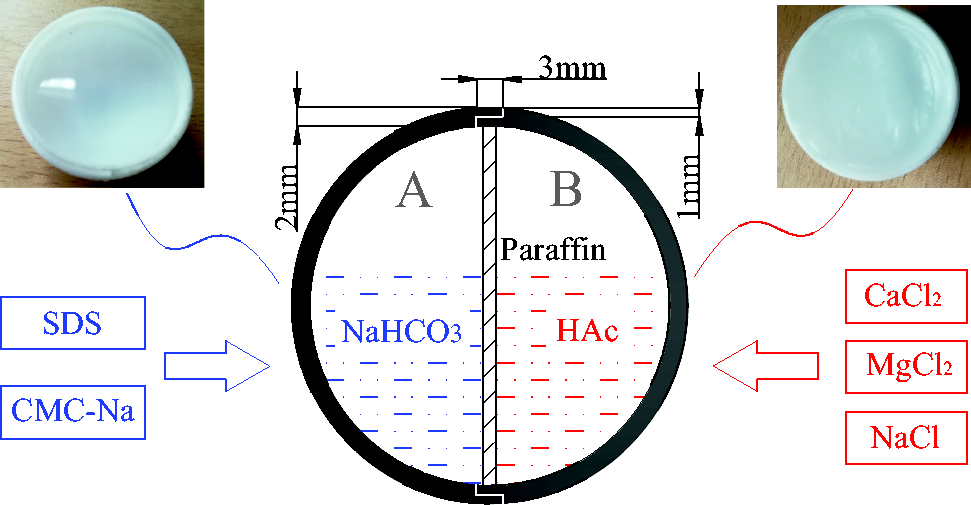
Temperature-controlled inhibitor forming system.
Different additives were added into the solutions of NaHCO3 and HAc to achieve the desirable foaming reaction and improve inhibition efficiency. 0.6% sodium dodecyl sulfate (SDS) as foaming agent and 0.5% sodium carboxymethyl cellulose (CMC-Na) as foam stabilizing agent were added into NaHCO3 solution (Liu et al., 2009). NaCl, MgCl2 and CaCl2 as inhibitors were added into HAc solution.
Experimental system
Measuring cylinders of 5, 50, and 250 ml were used to measure the volumes of solutions and foams generated by their reaction. The half-life of foam was the main indicator for the evaluation of foam stability, which was measured by stopwatch. An electronic scale was used to measure the masses of the additives added into the solution. Thermostatic water bath was used to heat the solution to specific temperatures to evaluate the effect of temperature on foam stability. In a round glass petri dish with diameter 150 mm and height 33 mm, pulverized coal with thickness 10 mm was uniformly dispersed to measure the diffusion and wetting area of foam after it was released. The following two experimental tests were also conducted:
Test system for the inhibition efficiency of foam
The experimental system used for the evaluation of the inhibition effect of foam on spontaneous coal combustion is shown in Figure 2. 100 g pulverized coal was put into cylindrical reaction vessel with diameter × height of 100 × 100 mm and the vessel was placed in a temperature programmed chamber. Hollow spheres filled with reaction solution were placed on a wire mesh with hole diameter of 10 mm above the pulverized coal. Air flow of 100 ml/min was supplied from an external air system to the bottom of reaction vessel through preheating tube. The gas discharged from reaction vessel flowed into gas detection system equipped with O2, CO2 and CO sensors and detection signals were transmitted to PC. Temperature probes were set inside temperature programmed chamber and pulverized coal. The temperature signal collector was connected to PC and values were recorded in real time. Once the inhibitor foam was released at the specific temperature, CO2 was discharged from reaction vessel due to the continuous supplement of air and the inhibitory effect of foam on the oxidation of pulverized coal was measured.
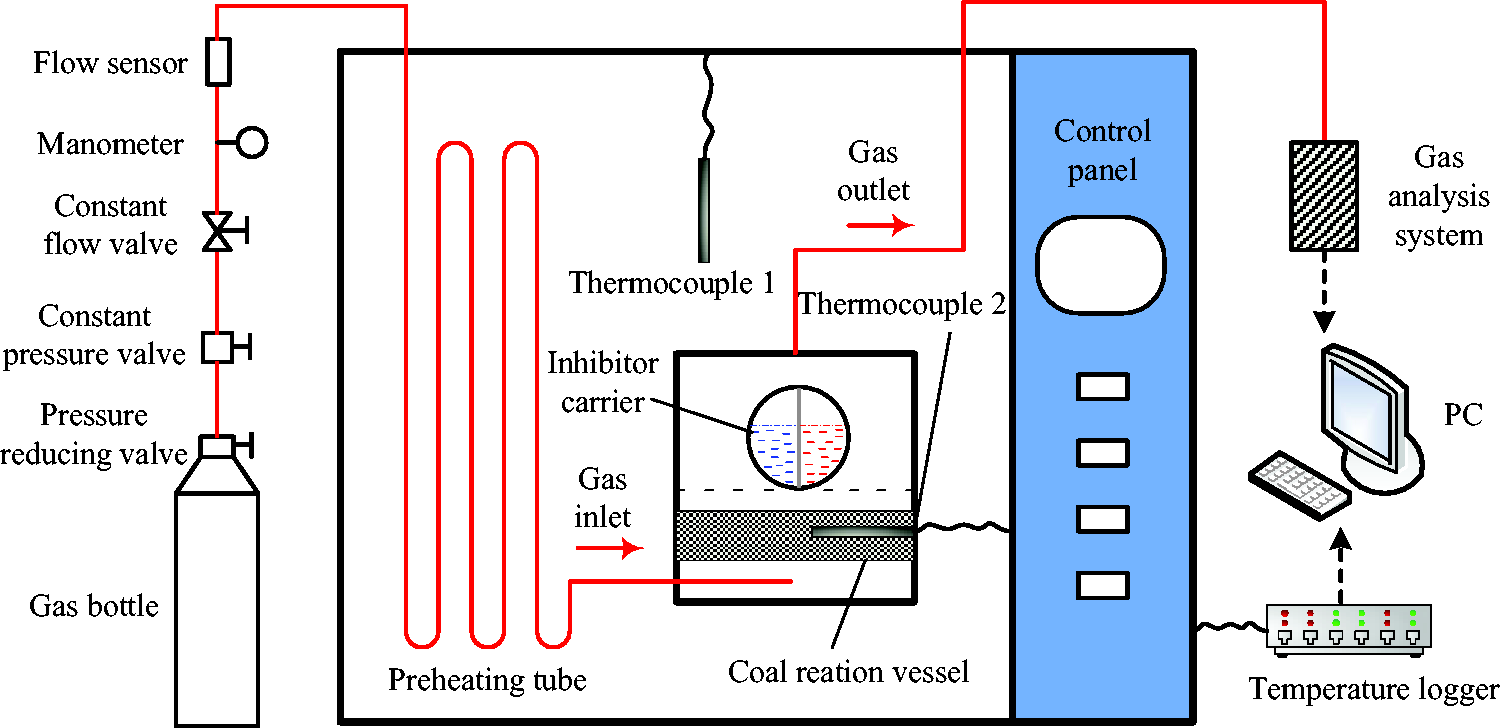
Test system for inhibition effect of foam.
Test system for inhibition and inerting efficiency of foam
The experimental system shown in Figure 2 was modified to a closed circulation system and used for the evaluation of the comprehensive inhibition and inerting effects of foam on coal oxidation (Figure 3). External air supply system was eliminated and air supply tube was connected to a miniature air pump. The gas discharged from the reaction vessel was pressurized by a miniature air pump and injected into preheating tube and then, it was fed into the reaction vessel again. After the release of inhibitor foam at the specific temperature, CO2 was not expelled, but inerted the air inside the reaction vessel.
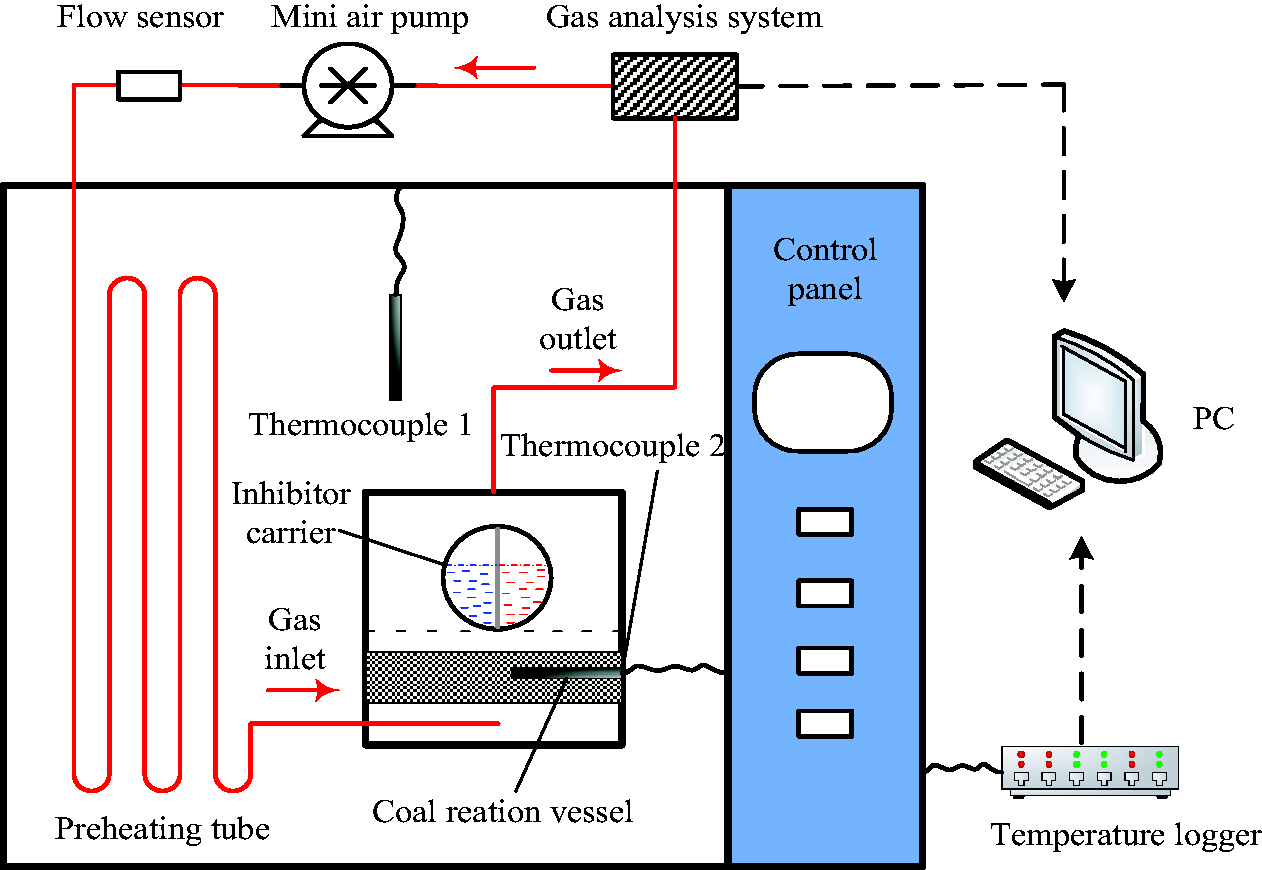
Test system for inhibition and inerting effects of foam.
Experimental methods
Experiment on the foaming and foam stability of the reaction system
Both NaHCO3 and HAc solutions of 10 ml were used. NaHCO3 solutions of 1 to 12% were prepared at the interval of 1% and the concentrations of HAc solution fully reacted with NaHCO3 were 0.72–9.8%. After the addition of SDS and CMC-Na into NaHCO3 solution, the mixture was mixed with HAc in a 250 ml measuring cylinder. The test was repeated three times and the change rule of volume and half-life of foam were evaluated to determine the optimum reactant concentrations. NaCl, MgCl2 and CaCl2 powders of 0.1–1.5 g were added into HAc solution. The NaHCO3 reaction test was repeated three times and the effects of halogenated salts on the volume and half-life of foam were investigated to determine the optimum masses of additives. Measuring cylinders containing NaHCO3 (with SDS and CMC-Na) and HAc (with or without NaCl, MgCl2 and CaCl2) solutions were placed in thermostatic water bath in the temperature range of 20 to 70°C. The temperature interval was 5°C and the solutions were heated for 5 minutes. The reactions were carried out with three replications in constant temperature chamber at temperatures set according to that of the solution. The influence of temperature on the volume and half-life of foam were analyzed to determine the allowable upper temperature limit.
Experiment of the effects of hollow sphere parameters on foam release
Hollow spheres of 24.4, 28.7, 36.1, 44.6 and 50.9 ml with diameters 40, 42, 45, 48 and 50 mm, respectively, were used in experiments. Hollow spheres filled with 20 ml reaction solution were placed at the center of a pulverized coal sample in a glass petri dish and heated in the temperature programmed chamber at the rate of 1°C/min. The temperature at which hollow spheres released the inhibitor foam was recorded. The wetted area of pulverized coal was measured after three repeated tests to determine foam diffusion. In the system shown in Figure 3, the concentration of CO2 released from hollow spheres was measured to calculate the ratio of solution reactions using equation (2).
where R is reaction ratio (%), C1 is the measured concentration of CO2 (%), and C0 is the concentration of CO2 when the reactants were completely reacted (%).
From experiments (a) and (b), hollow spheres with optimum diameter were selected as reaction solution carrier.
c. Another major factor affecting foam release is the angle at which hollow spheres are placed. When paraffin slice was placed along horizontal direction (hemisphere A at the top and hemisphere B at the bottom), the angle was considered to be 0°. The spheres were rotated by 15° until the angle reached 180° (hemisphere A at the bottom and hemisphere B at the top). Through temperature programmed experiments, the volumes of residual solution in hemispheres A and B were measured using a 5 ml high-precision measuring cylinder after the release of foam. Based on residual solution volume, the release ratios of foam at different placement angles of hollow spheres were calculated using equation (3) to determine the optimum angle.

where S is the release ratio of inhibitor foam (%), W1 is the volume of residual solution (ml), and W0 is the volume of reaction solution which was 20 ml in this work.
Experiment on restraining spontaneous coal combustion by inhibitor foam
Temperature-programmed experiments were carried out by the system shown in Figure 2 to study the change of temperature increase rate of pulverized coal. CO, which was generated by raw coal and coal covered with inhibitor foam, was used as an index gas and its concentration was monitored. The inhibition efficiency of foam on coal oxidation was calculated based on equation (4).
b. In the experimental system shown in Figure 3, changes of the concentrations of O2 and CO2 in reaction vessel were recorded before and after the release of foam. The concentration changes of the CO generated by raw coal and coal covered with inhibitor foam were compared to evaluate the comprehensive inhibition and inerting effects of foam on coal oxidation.
where Z is the inhibition efficiency of foam (%), X1 is the concentration of CO generated by the coal which was covered by inhibitor foam (ppm), and X0 is the concentration of CO generated by raw coal (ppm).

Results and discussion
Foaming reaction and foam stability
Foaming properties of reactants at different concentrations
When 10 ml NaHCO3 and 10 ml HAc solutions were directly mixed, they violently reacted and a large volume of CO2 was generated. NaAc solution of 20 ml was formed and the volume of solution was not significantly changed during the whole reaction process (Figure 4(a)). The addition of SDS into NaHCO3 solution significantly enhanced the foaming capacity of the reaction but it was immediately decreased after reaching the highest point (Figure 4(b) and (c)). As an anionic surfactant with excellent performance, SDS effectively reduced the surface tension of the liquid and created a double electron layer on liquid film surface to surround CO2 gas (Li et al., 2016b). Bubbles were finally formed and joined together creating foam. However, the formation of foam was unstable. It had a decay process due to liquid film drainage and gas diffusion. Therefore, it was necessary to add a stabilizing agent to slow foam decay. CMC-Na is a typical thickener which can effectively improve the intensity of liquid film and slow down drainage time. Moreover, CMC-Na is resistant to high temperatures and preserves its high viscosity after heating to 150°C (Wang and Du, 2008).

(a) Non-foaming; (b) complete foaming; (c) defoaming.
Figure 5 shows the foaming effects of reaction with HAc solution after the addition of 0.6% SDS and 0.5% CMC-Na into NaHCO3 solution. At NaHCO3 concentrations of below 4%, the amount of CO2 generated by the reaction was not high enough to completely foam 20 ml of solution. At the concentration of 4%, the solution reached fully foaming state with the CO2 volume of 111.2 ml, foam volume of 78 ml and the foaming multiple of 3.9. Moreover, the generated foam had a good compactness and its half-life reached 39 seconds. As the concentration was gradually increased from 4% to 12%, the amount of the CO2 generated by the reaction was increased from 111.2 to 363 ml, leading to the continuous increase of foam volume with maximum value of 206 ml. However, foam half-life was first increased and then decreased. At the concentration of 8%, the maximum foam half-life of 95 seconds and foam volume of 166 ml were obtained with bubbles of uniform sizes and the highest stability. By increasing concentration from 9% and 10%, foam volume was also increased but its half-life was slightly decreased. By further increase of concentration from 11% and 12%, foam half-life was significantly and rapidly decreased to 65 seconds. During foaming process, some large bubbles were created and foam uniformity was decreased. The main reason for this was the very high volume of CO2, which increased gas diffusion ability through foam liquid film and decreased foam stability. Considering foam volume and half-life as well as the amount of CO2 generated by the reaction, 10% NaHCO3 was chosen as the optimum concentration. Also, the optimum volumes of foam and CO2, half-life of foam, and concentration of NaAc were 190 ml, 294.2 ml, 92 seconds and 5.3%, respectively.
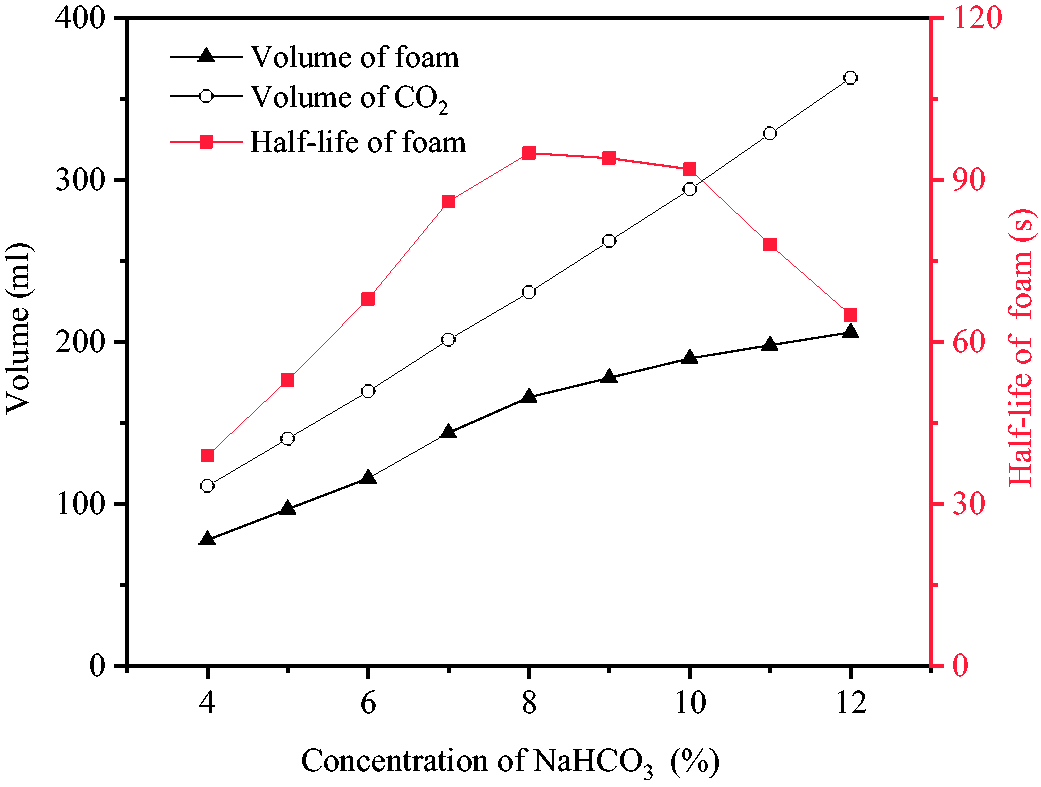
Effects of different NaHCO3 concentrations on foaming and foam stability.
Effects of adding halogenated salts on foaming and foam stability
The volume of foam generated by the reaction was less affected by NaCl, MgCl2 and CaCl2 and the main effective factor on the volume of foam was the amount of CO2, but the stability of foam was significantly fluctuated by increasing the mass of additives, as shown in Figure 6. Low amounts of halogenated salts, especially NaCl and CaCl2, promoted foam stability. By increasing the amount of these additives from 0.1 to 0.4 g, foam half-life was increased from 91 and 89 seconds to 124 and 122 seconds, respectively. The reason for this observation was that, the addition of inorganic salts compressed the double electric layer of bubbles and increased electrostatic repulsion between bubbles. The merging of bubbles was restrained and their size was reduced (Shen, 2018). As the amount of the additives was further increased, salting-out effect on the foam was witnessed continuously decreasing foam half-life. NaCl was the most effective additive, followed by CaCl2 and MgCl2. In order to ensure stable diffusion time of foam, half-life of 50 seconds was taken as the lower limit index (considering subsequent effects of temperature) and the optimal masses of NaCl, MgCl2 and CaCl2 added into NaHCO3 solution were found to be 0.9, 1.1 and 1.4 g, respectively. After mixed reaction of the solutions, the concentrations of the above halogenated salts were 4.1, 5.0 and 6.2%, respectively.
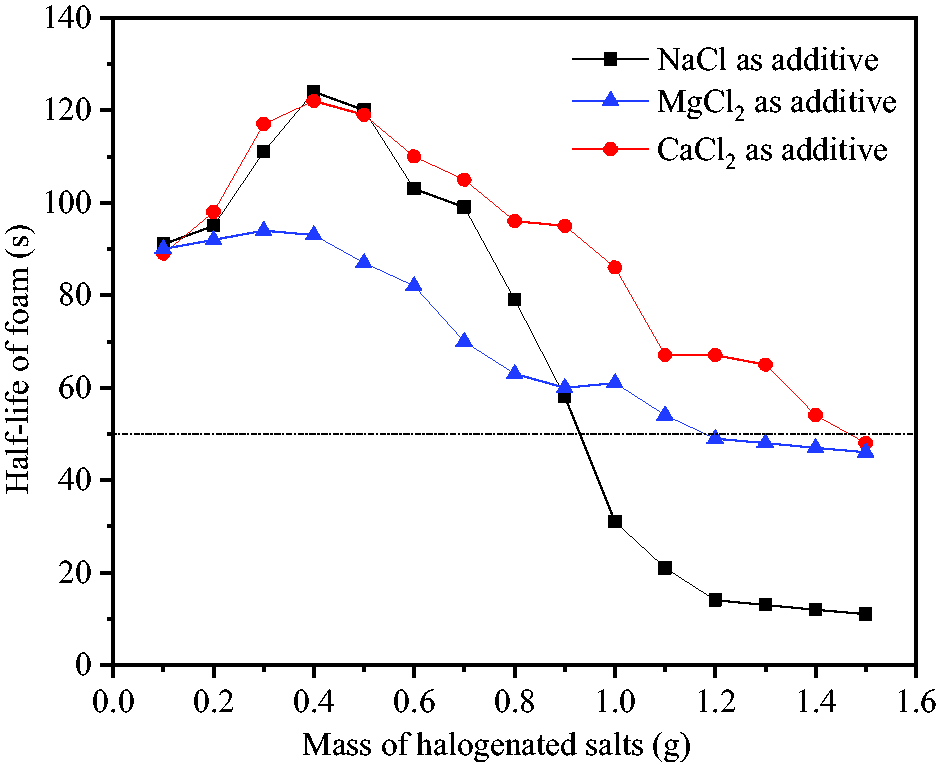
Effects of adding halogenated salts on foam stability.
Effects of temperature on foaming and foam stability
The volume of foam was not significantly changed at different temperatures and the main factor affecting foam volume was still the amount of CO2. However, foam half-life continued to sharply decrease, as shown in Figure 7.
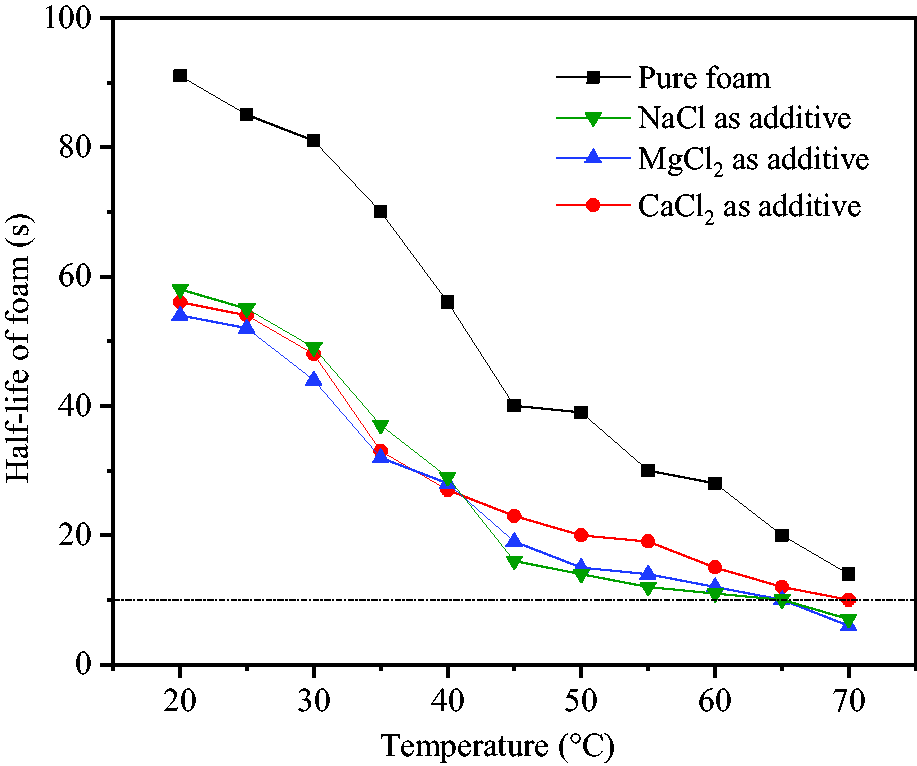
Effects of temperature on foam stability.
At temperatures of above 30°C, foam half-life began to rapidly decrease and reached 14 seconds at 70°C. By adding NaCl, MgCl2 and CaCl2, foam stabilization times became even shorter with foam half-life decreasing to 7, 6 and 10 seconds, respectively, at 70°C. This indicated that, temperature was the main factor affecting foam stability, except the salting-out effect of halogenated salts. By increasing temperature, the thermal motion of molecules was intensified and the interaction between the hydrophilic groups of surfactants were weakened decreasing liquid film intensity which made it difficult for the foam to remain stable for a long time. Half-life of 10 seconds was taken as the lower limit index to ensure the stable diffusion time of foam. The upper temperature limit was set at 70°C in the absence of halogenated salts and the corresponding values were changed to 65, 65 and 70°C after the addition of NaCl, MgCl2 and CaCl2 into the reaction mixture, respectively. Therefore, 52# paraffin wax with melting point 52°C was reasonably selected as the temperature-sensitive insulating material inside hollow spheres.
Release and diffusion of foam from hollow spheres
Effect of the volume of hollow spheres on foam release and diffusion
The critical temperature of foams released from hollow spheres was in the range of 58–59°C. Figure 8 shows the wetted area of pulverized coal. The wetted regions were roughly circular with the areas of 50.2, 75.4, 98.5, 98.5 and 102.1 cm2 for hollow spheres with diameters 40, 42, 45, 48 and 50 mm, respectively. Compared with the area of 7.1 cm2 when 20 ml solution wetted pulverized coal directly, increase multiples were 7.1, 10.6, 13.9, 13.9 and 14.4, respectively. Therefore, foam diffusion effect was very remarkable.
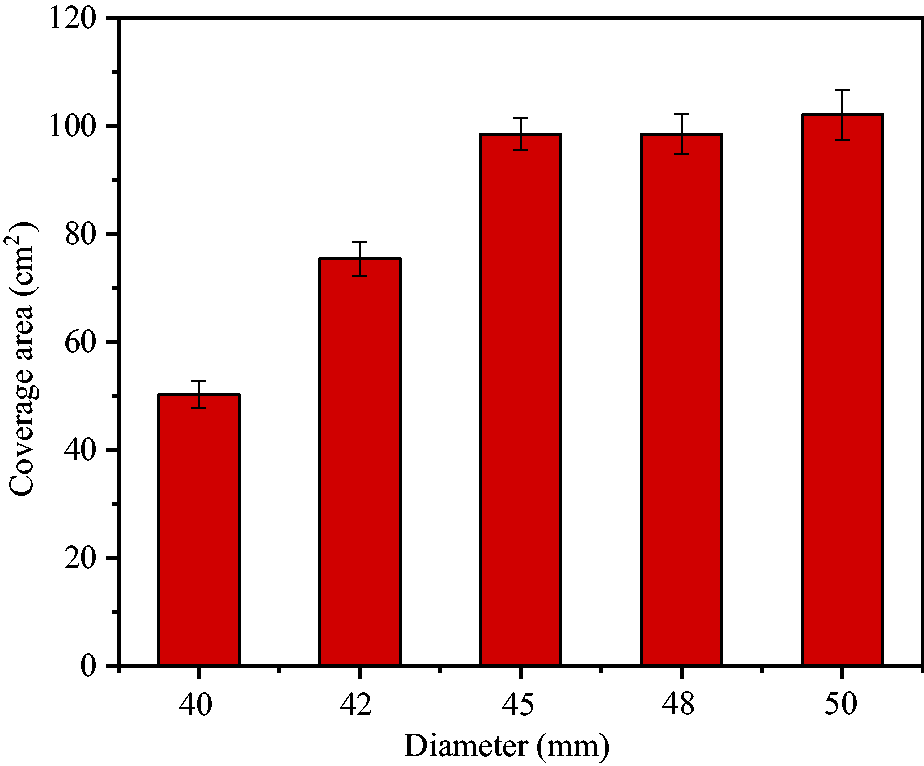
Coverage area of foam released by hollow spheres.
The concentrations of CO2 released by hollow spheres with different diameters were evaluated in the system shown in Figure 3. CO2 concentration was recorded every 5 seconds from 58°C and the obtained results are shown in Figure 9. The time nodes of releasing CO2 from hollow spheres were slightly delayed with the increase of sphere diameter and concentration change trend was transformed from sudden surge to slow increase, which was stabilized after 25–30 seconds. This indicated that a part of CO2 was wrapped in the foam in the early stage of foam release and was completely released in 25–30 seconds time range. Then, foam was disappeared and coal body was wetted. If the mixture of NaHCO3 and HAc reached the state of complete reaction, the theoretical volume of CO2 would be 294.2 ml and the concentration of CO2 in the reaction vessel would be C0 = 29.40%. The actual measured concentrations C1 of CO2 released from hollow spheres with diameters 40, 42, 45, 48 and 50 mm were 15.01, 20.71, 27.30, 27.69 and 28.17%, respectively. From equation (2), the reaction ratios R of solutions in hollow spheres were 51.1, 70.4, 92.9, 94.2 and 95.8%, respectively.
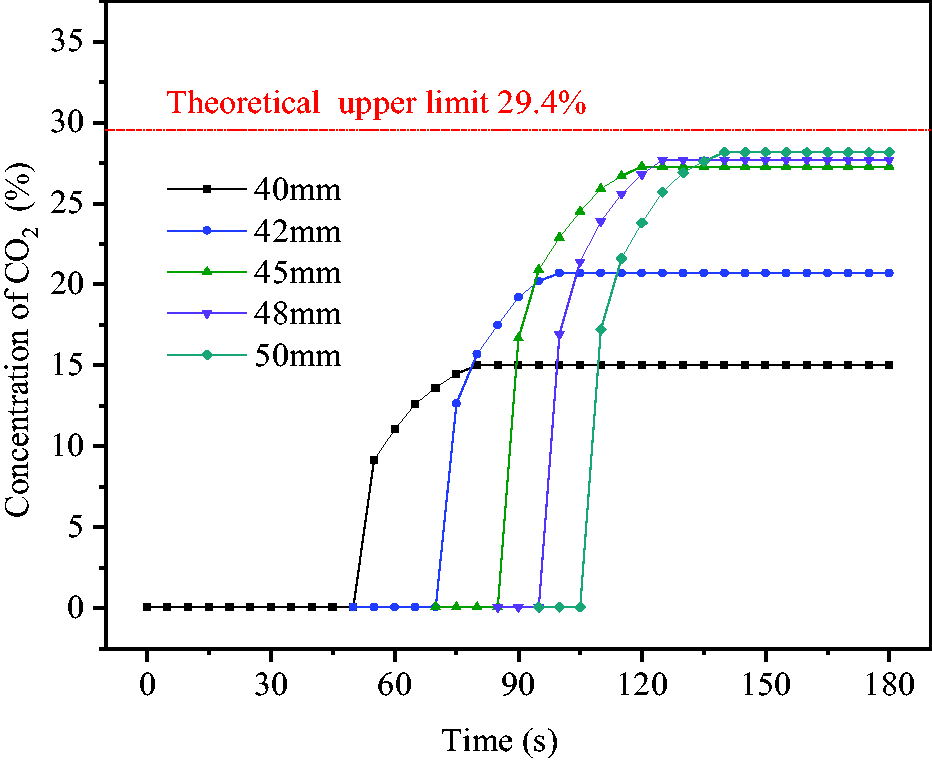
Concentration change of CO2 released by hollow spheres.
The obtained results showed that reactants were released before adequately reacting inside hollow spheres with diameters 40 and 42 mm, which led to lower reaction ratios and wetting areas of pulverized coal. Therefore, hollow spheres with diameter 45 mm were selected as the optimum solution carriers due to their proper volume, large coverage areas and high reaction ratios. At this time, solution occupied 55.4% of the space volume and the remaining 44.6% was used as reaction foaming space.
Effect of the placement angle of hollow spheres on foam release
Figure 10 shows the residual volumes of solution in hemispheres A and B after foam release from hollow spheres placed with different angles. At 0° angle, residual solution was mainly in the lower hemisphere B with volume W1 of 1.6 ml. According to equation (3), the release ratio S of foam was calculated to be 92%. With the increase of angle, the volume of residual solution was decreased. At 90° angle, W1 was decreased to a minimum value of 0.2 ml and the release ratio of foam was increased to the maximum value of 99%. By further increase of angle, the volume of residual solution maintained its increasing trend. At 180° angle, the residual solution was mainly located in the lower hemisphere A with volume W1 increasing to 1.5 ml and the release ratio S of foam decreasing to 92.5%. This demonstrated that, the release ratio of foam was maximum when the paraffin slice inside the hollow spheres was in vertical state. All experiments carried out in this paper were performed at 90° placement angle of hollow spheres.
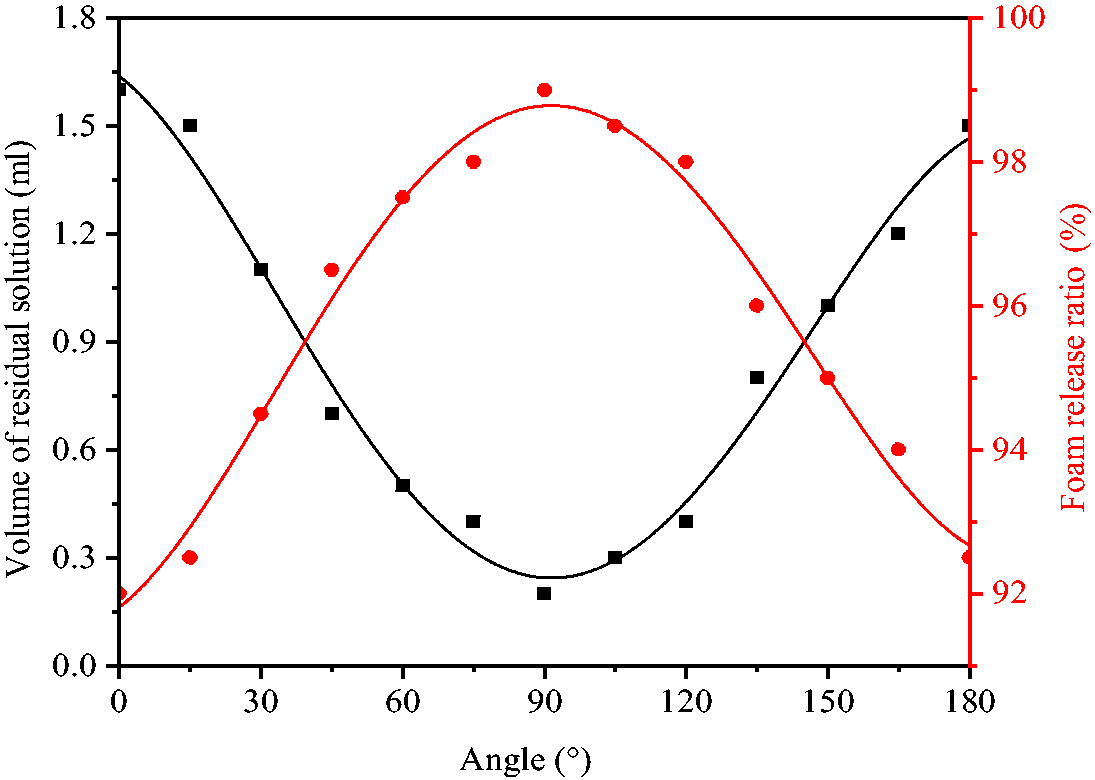
Foam release at different placement angles of hollow spheres.
Effect of halogenated salts on foam release
After the addition of halogenated salts into the solution, the release temperature of foam was slightly increased as shown in Table 2. The main reason was that, the addition of halogenated salts reduced the thermal conductivity of the solution, which delayed the melting of paraffin and further increased foam release temperature maximum by 2°C. At the same time, foam stability was reduced due to the salting-out action of halogenated salts, leading to a slight decrease in the area of wetted coal by foam. However, it was still 12.9 times larger than the area of coal directly wetted by 20 ml solution.
Release parameters of foam under the action of halogenated salts.

Restraining effect of spontaneous coal combustion
Inhibition effect of foam on spontaneous coal combustion
Figure 11 shows the temperature changes of pulverized coal during temperature programmed process. The temperature of raw coal was always lower than ambient temperature in the chamber until it reached crossing point temperature (CPT), which was 152°C. The average temperature increase rate of coal was only 0.67°C/min before 70°C, which was lower than the programmed temperature increase rate of 1°C/min. However, temperature increase rate of coal was basically consistent with the programmed temperature increase rate from 70 to 110°C. Above 110°C, coal temperature was rapidly increased with the rate of 1.52°C/min until 152°C. This indicated that, when coal temperature reached 110°C, it began to enter the stage of intense oxidation where the processes of coal oxidation and self-heating were accelerated. After the pulverized coal was wetted by the foam released from hollow spheres at the specific temperature, the thermal conductivity of coal was increased and its temperature was slightly higher than that of raw coal before 100°C. However, the rate of temperature increase was slowed down after 100°C due to the inhibition effects on coal oxidation and continuous water evaporation. Coal CPT never appeared within the programmed temperature up to 160°C. The highest temperature of the coal wetted by pure foam and foam containing NaCl, MgCl2, CaCl2 were 140.1, 130.2, 125.4 and 122.2°C, respectively. The obtained results showed that temperature and temperature increase rate of coal wetted by inhibitor foam were effectively reduced.
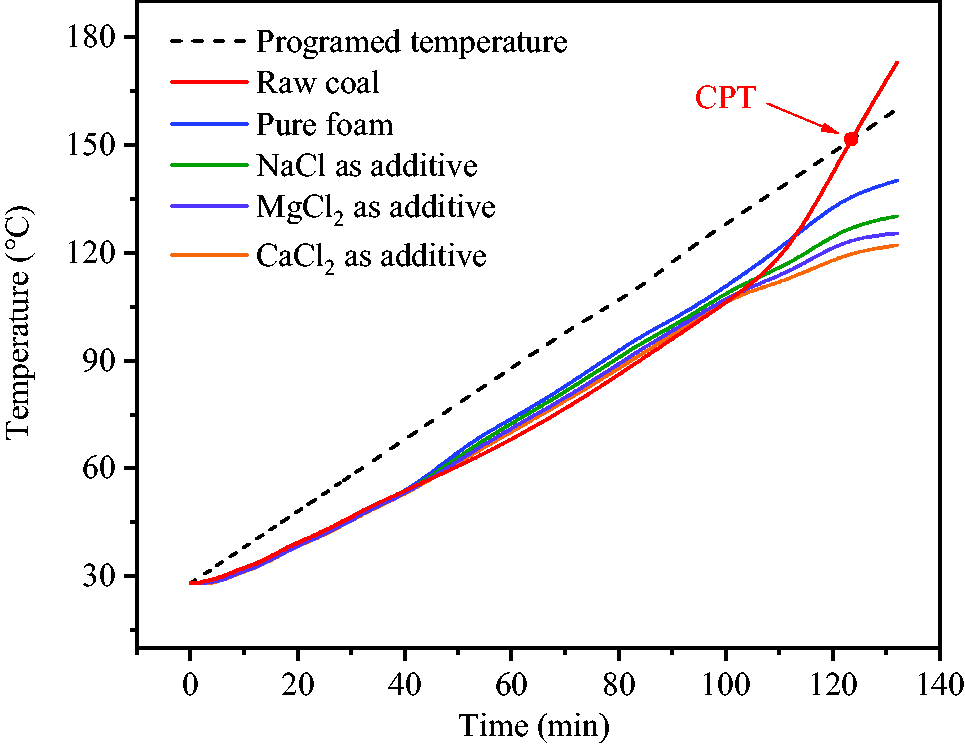
Temperature variations of coal during temperature programmed process.
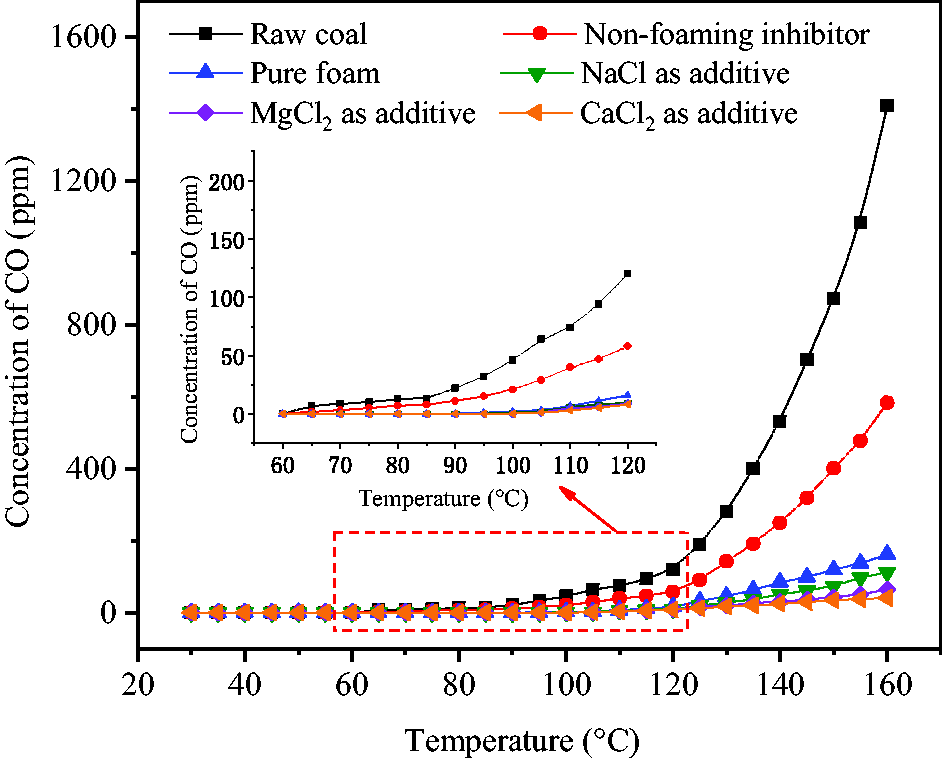
Figure 12 shows the concentration change of CO generated by coal oxidation. At the programed temperature of 90°C, the concentration of CO generated by raw coal began to steadily increase from 24 ppm and rapidly increased from 281 ppm to 1480 ppm by increasing temperature from 130°C to 160°C. Although NaAc solution could be released to wet coal body at 59°C without needing SDS and CMC-Na in hollow spheres, the coverage area of coal was very limited under this condition, such that the concentration of CO generated by coal oxidation was only slightly lower than that due to raw coal oxidation. After the addition of SDS and CMC-Na, NaAc was released in the form of foam which could completely cover and wet the coal body. The concentration of CO was very low at temperatures below 100°C. Above 120°C, however, there was a significant release of CO at the concentration of 43 ppm and the highest value was 490 ppm which was achieved when the programed temperature reached 160°C. The obtained results showed that inhibition effect was greatly increased by releasing inhibitor solution in the form of foam. After the addition of halogenated salts, the inhibitory efficiency of foam was further increased. Due to different inhibition abilities of NaCl, MgCl2, and CaCl2 and their different added mass, 6.2% CaCl2 in the foam had the strongest effect in inhibiting coal oxidation, followed by 5.0% MgCl2 and 4.1% NaCl.
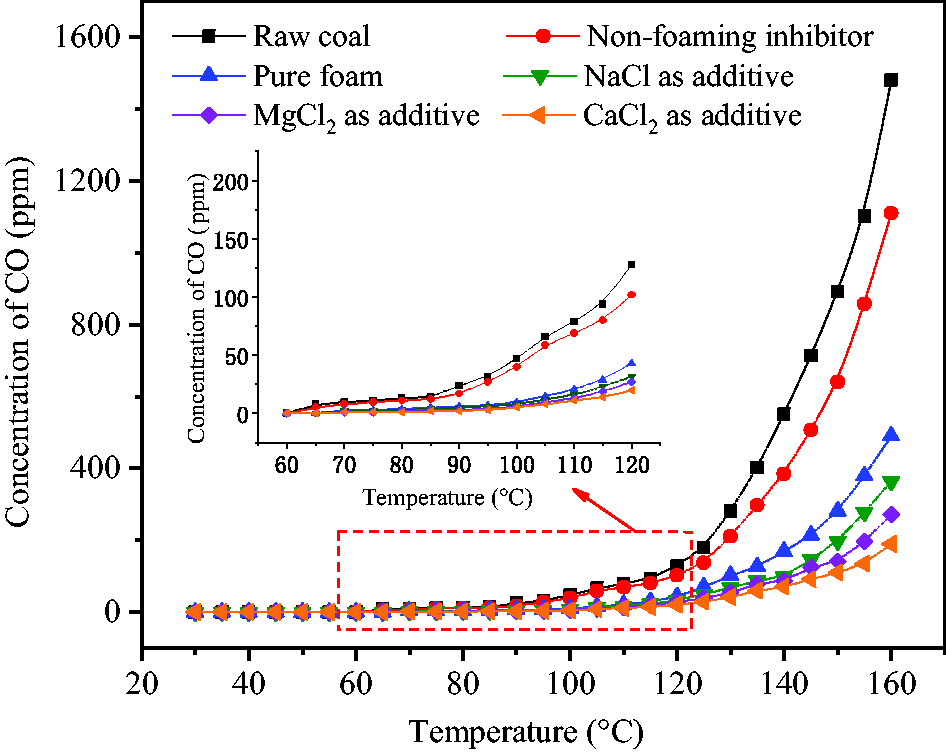
Concentration variations of CO generated by coal oxidation.
The highest inhibition efficiencies Z were calculated by equation (4) to be 91.6% for CaCl2, 87.5% for MgCl2, 82.9% for NaCl and 78.7% for NaAc alone when the programed temperature reached 90–100°C. At this time, the temperature of pulverized coal reached 70–85°C which was at the key point of the early stage of oxidation. Temperature-controlled foam release can accurately inhibit coal oxidation, which is difficult to achieve by the traditional method of wetting coal by inhibitor solution in advance. With continuous temperature increase and water evaporation, inhibition efficiency was slightly decreased and coal began to resume oxidation. Based on the concentration of CO generated by wetted coal at 160°C and comparing with the temperature at which the same concentration of CO was generated by raw coal, it was found that coal oxidation was postponed by 35°C (CaCl2 as additive), 31°C (MgCl2 as additive), 27°C (NaCl as additive) and 22°C (NaAc alone).
Inhibition and inerting effects of foam on spontaneous coal combustion
Figures 13 and 14 show the concentration changes of CO2 and O2 in the reaction vessel after foam release. CO2 was released at 59°C from hollow spheres without SDS and CMC-Na. Due to uneven gas distribution in reaction vessel, the initial release concentration of CO2 was slightly higher with the value of 29.1%. After 20 seconds, the final stable concentration was 27.25% and the corresponding concentration of O2 was reduced from 20.6 to 14.8%. When inhibitor was released in the form of foam, the concentration of CO2 was suddenly increased to 16.51–16.84%, and then to 27.1–27.3% since a part of the gas was wrapped in foam and slowly released. The corresponding concentration of O2 was decreased and stabilized at 14.8–14.85%. This indicated that the release of foam effectively reduced the concentration of O2 in the reaction vessel and played an inerting role in air.
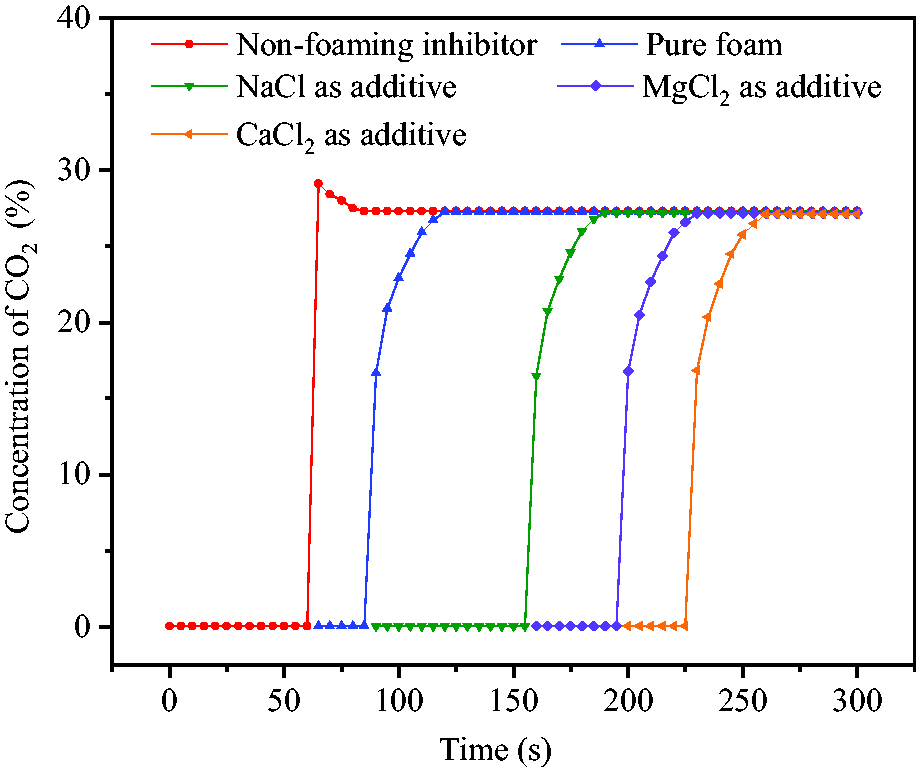
Concentration variations of CO2 in reaction vessel.
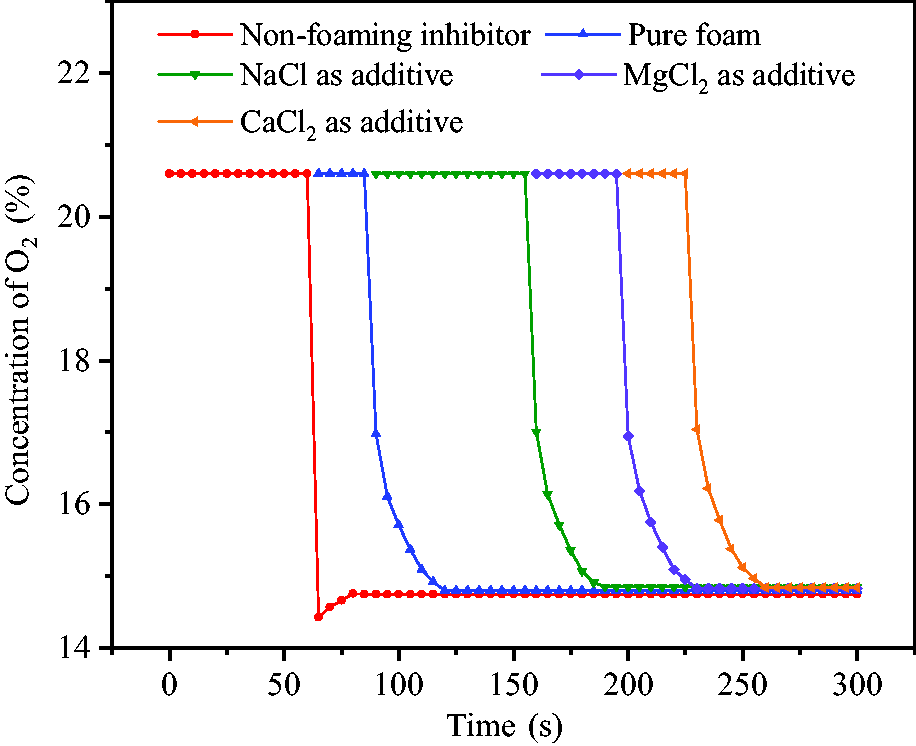
Concentration variations of O2 in reaction vessel.
Figure 15 shows the concentration change of CO generated by coal oxidation due to the inhibition and inerting effects of foam. Raw coal was partially wetted by inhibitor foam in the absence of SDS and CMC-Na in the solution of hollow spheres, but the release of CO2 effectively inerted the air which led to the concentration of CO generated by coal oxidation to be significantly lower than that of the mere action of inhibition. With the addition of SDS and CMC-Na, inhibitor solution was released in the form of foam. Due to the joint inhibition and inerting actions of foam, only low amounts of CO were generated at temperatures below 130°C and it showed a slow increasing trend with the increase of temperature. According to equation (4), at 160°C, the inhibitory effect of foam on coal oxidation was 97.06% (CaCl2 as additive), 95.46% (MgCl2 as additive), 92.06% (NaCl as additive) and 88.51% (NaAc alone). Therefore, under the combined action of inhibition and inerting, the restraining effect of foam on coal oxidation was more significant.
Concentration variations of CO generated by coal oxidation under the action of inhibition and inerting.
Conclusion
In this paper, a new method was proposed for releasing inhibitor solution in the form of foam at a specific temperature. Using temperature-controlled self-reaction of two solutions, a large amount of CO2 was released, which could be used as the gas source of inhibitor foam. It could effectively expand the action scope of inhibitors and reduce the concentration of O2, playing an important role in inhibition and inerting efficiency.
With SDS as foaming and CMC-Na as foam stabilizing agents, the optimum concentrations of NaHCO3 and HAc solutions were 10 and 7.9%, and the optimum concentrations of NaCl, MgCl2 and CaCl2 were 4.1, 5.0 and 6.2%, respectively, with the upper limit of temperature set at 65–70°C. Hollow spheres with diameter 45 mm were selected as the carrier of 20 ml solution and sphere placement angle of 90° was found to be the optimum angle when paraffin slice was in vertical state. Foam release temperature was 59–61°C and the covered area of pulverized coal was 12.9–13.9 times higher than that by directly wetting coal with solution. The generation and release of foam by temperature-controlled self-reaction provided high release efficiency of inhibitor at a specific temperature and large wetting area of coal body.
After the release of inhibitor foam, oxidation heating rate of coal was effectively suppressed with maximum inhibition efficiency of 78.7–91.6% and the process of coal oxidation was delayed by 22–35°C. Under the joint action of inhibition and inerting of foam, the efficiency of restraining coal oxidation reached 88.51–97.06% at 160°C, which was significant.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (51604144, 51774171), and the Natural Science Foundation of Liaoning Province in China (20170540430).
