Abstract
An investigation combining thermogravimetric analysis/differential scanning calorimeter technique and a fundamental study of coal characteristics was conducted on different coal samples sourced from Chinese coalfields. This analysis investigated the impact of several factors, including heating rate, particle size, and the propensity of coal for spontaneous combustion, on the pyrolysis process of coal. In addition, the characteristic temperature points corresponding to each stage of the experimental process were defined. Thermogravimetric analysis study of coal samples indicates that heating rate, particle size, and coal spontaneous combustion propensity significantly influence the pyrolysis process, as reflected by variations in the thermogravimetric/differential thermogravimetric curves under different conditions. The weight loss rate increases when the heating rate decreases/particle size decreases/spontaneous ignition propensity increases. Kinetic analysis shows that the activation energy is positively correlated with the heating rate. The calculated findings reveal that the activation energy diminishes when the heating rate decreases/particle size decreases/spontaneous ignition propensity increases, which is in accordance with the experimental results. Our results suggest that the activation energy can be available to assess the spontaneous combustion of coal. This study not only deepens the understanding of the thermal reactivity of coal under varying conditions but also offers important insights for early warning and risk evaluation in coal mining and storage operations.
Keywords
Introduction
A significant threat is posed by the spontaneous ignition of coal to the activities of mining, storage, and transportation within the coal production chain, thereby impeding the overall advancement of the coal sector (Adamus et al., 2011; Song and Kuenzer, 2014). Not only are considerable coal resources wasted as a result of this phenomenon (Parsa et al., 2017; Zhang et al., 2018), but mine fires are also generated, which disrupt the airflow in the mine's ventilation system, consequently increasing the risk of gas explosions and causing potential harm to personnel (Li et al., 2020; Shi et al., 2021; Xie et al., 2011). Thus, it is crucial to research the mechanism of spontaneous ignition of coal and propose effective anti-ignition methods.
Physically, spontaneous combustion of coal results from its physicochemical reaction with atmospheric oxygen, during which heat accumulates and raises the internal temperature beyond the ignition point, ultimately triggering combustion (Ma et al., 2020; Singh, 2013). In laboratory settings, researchers have employed various means to investigate coal's tendency for spontaneous ignition (Banerjee, 2021; Banerjee and Chakraborty, 1967; Kök, 2008; Mohalik et al., 2017). Onifade and colleagues conducted a comprehensive assessment of predictive methods for spontaneous ignition widely utilized within academic, research, and industrial settings, and classified them into mathematical modeling, experimental, and statistical methods (Onifade and Genc, 2020a). Wen and his co-researchers used the oxidation and self-heating characteristics of loose coal to establish multiple large-scale experimental platforms of varying sizes, aiming to reproduce the phenomenon of spontaneous combustion in coal (Wen et al., 2017). Deng and collaborators utilized an experimental setup for coal spontaneous ignition to investigate how diverse factors influence the minimal duration of ignition (Deng et al., 2015; Deng et al., 2016). The coal temperature is recognized as the most precise and straightforward metric for assessing the extent of coal spontaneous ignition (Basil et al., 2000; Sun et al., 2019). Through the study of coal oxidation kinetics, it was found that coal temperature affects the apparent activation energy, and oxygen concentration affects the oxidation activity of coal (Wang et al., 2022). In recent years, innovative techniques such as thermal analysis using instruments like DTA, TGA, and DSC have been utilized to analyze coal properties and study its spontaneous combustion behavior using sample masses ranging from a few milligrams to 1 g (Kök, 2012; Mahajan and Walker, 1971; Marinov, 1977a, 1977b, 1977c; Marinov et al., 2010; Mohalik et al., 2009; Mohalik et al., 2010; Xu et al., 2021; Zhao et al., 2019). By conducting TGA experiments under different conditions and varying factors, researchers can gain valuable insights into the mass loss and exothermic properties of coal as it undergoes oxidation, thereby elucidating the influence mechanism of each factor from an exothermic perspective.
From the literature survey, it is evident that experiments utilizing TGA/DSC have delved into fundamental coal characteristics, including mass changes, combustion behavior, calorific value, kinetic study, and oxidation behavior interlinked with parameters influencing spontaneous combustion. Some researchers have identified specific operational conditions and characteristics within studies of oxidation and spontaneous ignition to evaluate the likelihood of spontaneous combustion. Zhang et al. (2020) also found that under oxygen-deficient conditions, coal exhibits reduced heat release, delayed characteristic temperature points, and prolonged reaction time. Previous studies have established a correlation between the inherent properties of coal-bearing shale samples and their spontaneous combustion behavior, demonstrating that these intrinsic characteristics significantly influence their propensity for spontaneous combustion (Onifade et al., 2020b; Onifade and Genc, 2018). It has been observed that a lower mass gain corresponds to reduced reaction time, leading to a decrease in ignition temperature and an increased likelihood of spontaneous combustion (Avila et al., 2014). Additionally, the observed rise in mass accumulation and associated temperature zones in TGA experimental data may be beneficial for classifying coal according to its reactivity. Some academics have made predictions about coal's susceptibility to spontaneous combustion based on the slopes of the DTG curves at various heating rates (Janković et al., 2020). Furthermore, the spontaneous combustion behavior of coal has been evaluated by utilizing the initiation temperature obtained from DSC tests within the first exothermic peak zone, as conducted by some researchers. Attempts have also been conducted to categorize coal types based on their likelihood of spontaneous combustion, using a limited set of samples via TGA and DSC experiments (Mohalik et al., 2021). However, this categorization may not be advantageous for mine planners, operators, and mining engineers because of varying experimental conditions such as sample size, spontaneous combustion propensity, heating rate, etc. Thus, additional investigation into the impact of different factors remains necessary to enhance understanding of coal's spontaneous combustion mechanism.
To address the aforementioned limitations, this research investigates the application of TGA/DSC methods on coal specimens obtained from various Chinese coalfields, including those with differing ranks and histories of combustion incidents. The TG experimental findings were analyzed to determine characteristic temperatures that help evaluate coal's susceptibility to spontaneous combustion. In comparison to traditional experimental methods, TGA experiments are notably uncomplicated and robust, requiring less time and exhibiting excellent repeatability. The sample size, the influence of the propensity for spontaneous ignition, and the heating rate on its spontaneous ignition of coal were comprehensively analyzed. Furthermore, a kinetic analysis was conducted to evaluate the activation energy under different conditions. Overall, this study not only deepens the multi-scale understanding of the mechanism of coal spontaneous combustion, but also lays the necessary theoretical and practical foundation for the accurate prediction and prevention of spontaneous combustion risks in safe mine operations.
Experimental methodology
Coal sample
Firstly, coal blocks were obtained from the working faces of different mines (Rongchang, Datong, and Tongchuan). Then, the coal blocks were broken down and screened for different particle sizes of 0.25 mm, 0.5 mm, 1 mm, and 2 mm under nitrogen protection in the laboratory. The coal used in this work was analyzed in a research institute and the parameters were calculated according to China's testing standards (GB/T 212-2008, GB/T 474-2008, GB/T 211-2007, GB/T 30733-2014, GB/T 214-2007, and GB/T 213-2008). Table 1 provided a detailed presentation of the analysis results. In this table,
Analysis of coal.

Testing techniques
In this study, a thermogravimetric analyzer, capable of measuring changes in both the sample's weight and temperature, was employed, as depicted in Figure 1. The system contains the following components: a thermobalance, several gas sources, a furnace, a holder, a control panel, and a supervisory control computer. The quality of the sample could be continuously recorded under different heating rates. Each time samples of about 10 mg were utilized for testing. Temperature was regulated from ambient levels to 900°C, with the heating rates adjusted correspondingly. An amount of oxygen (21%) was mixed with
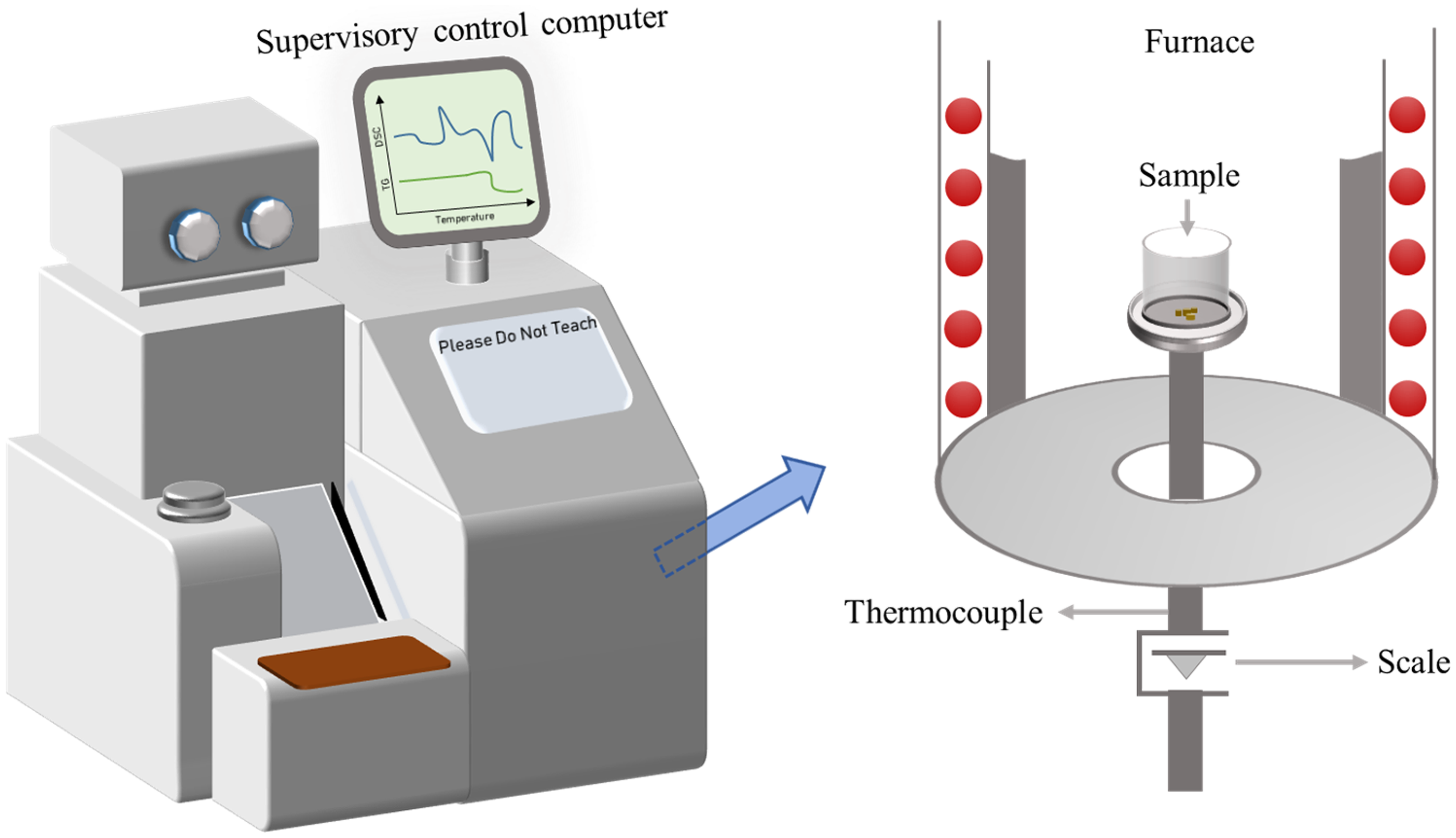
The thermogravimetric analyzer.
Test results and discussion
Thermogravimetric analysis
Characteristic temperature
The primary mechanism of coal spontaneous combustion involves a complex series of reactions between coal and oxygen. To attain a more accurate analysis, it is essential to delve into the mechanisms of the coal pyrolysis process initially. The coal structure, as depicted in Figure 2 (Fletcher et al., 1990; Yan et al., 2013). Throughout the pyrolysis process, the unstable bonds within the coal structure undergo fracture or transformation, liberating small free radicals that subsequently react with hydrogen sourced from both the coal and the gasification agent. This fusion results in the production of light gases. Some segments of the coal separate from the lattice and convert into tar with higher molecular weight. Stabilized charcoal bridges are formed by the remaining materials within the coal lattice, ultimately developing into char. Therefore, the quality of coal continuously changes during the pyrolysis process.
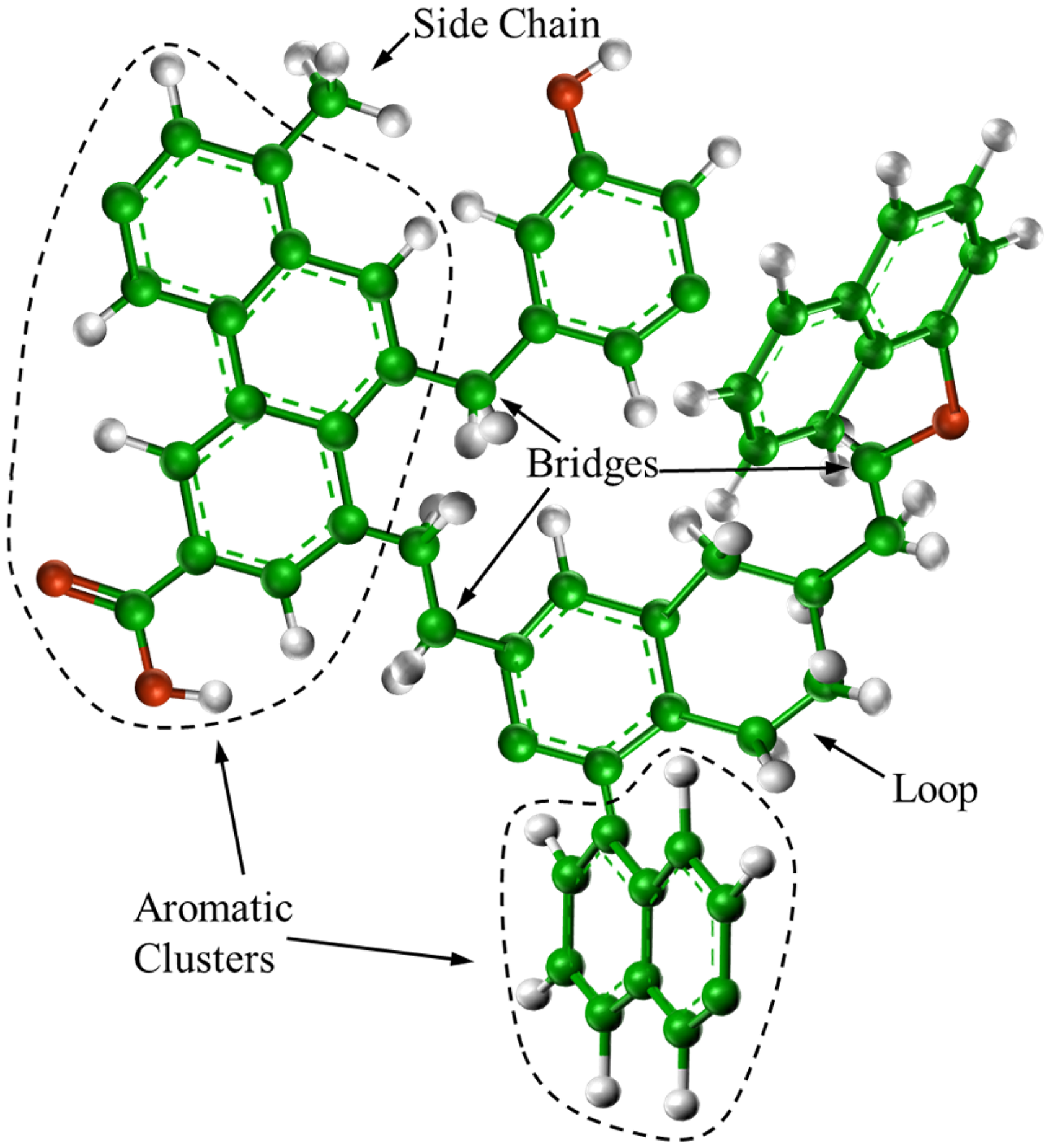
Chemical structures of coal.
Different stages in the combustion process correspond to distinct characteristic temperatures. Characteristic temperature serves as a crucial parameter in the oxidation and decomposition processes of coal, with changes in its value reflecting the stages of combustion and providing insights into the reactivity and combustion behavior of coal samples. Taking the Rongchang coal sample with a particle diameter of 2 mm as an example, Figure 3 illustrates the TG (Thermogravimetry), DTG, and DSC curves for this coal specimen, measured under a heating rate of 10°C/min. Variations in quality are represented by the weight loss curve (TG) as oxidation and decomposition occur. The DTG curve represents the rate of weight loss per unit of time and temperature, indicating the intensity of coal oxidation reactions at different temperatures. Peaks on this curve correspond to temperature ranges where significant reactions occur.
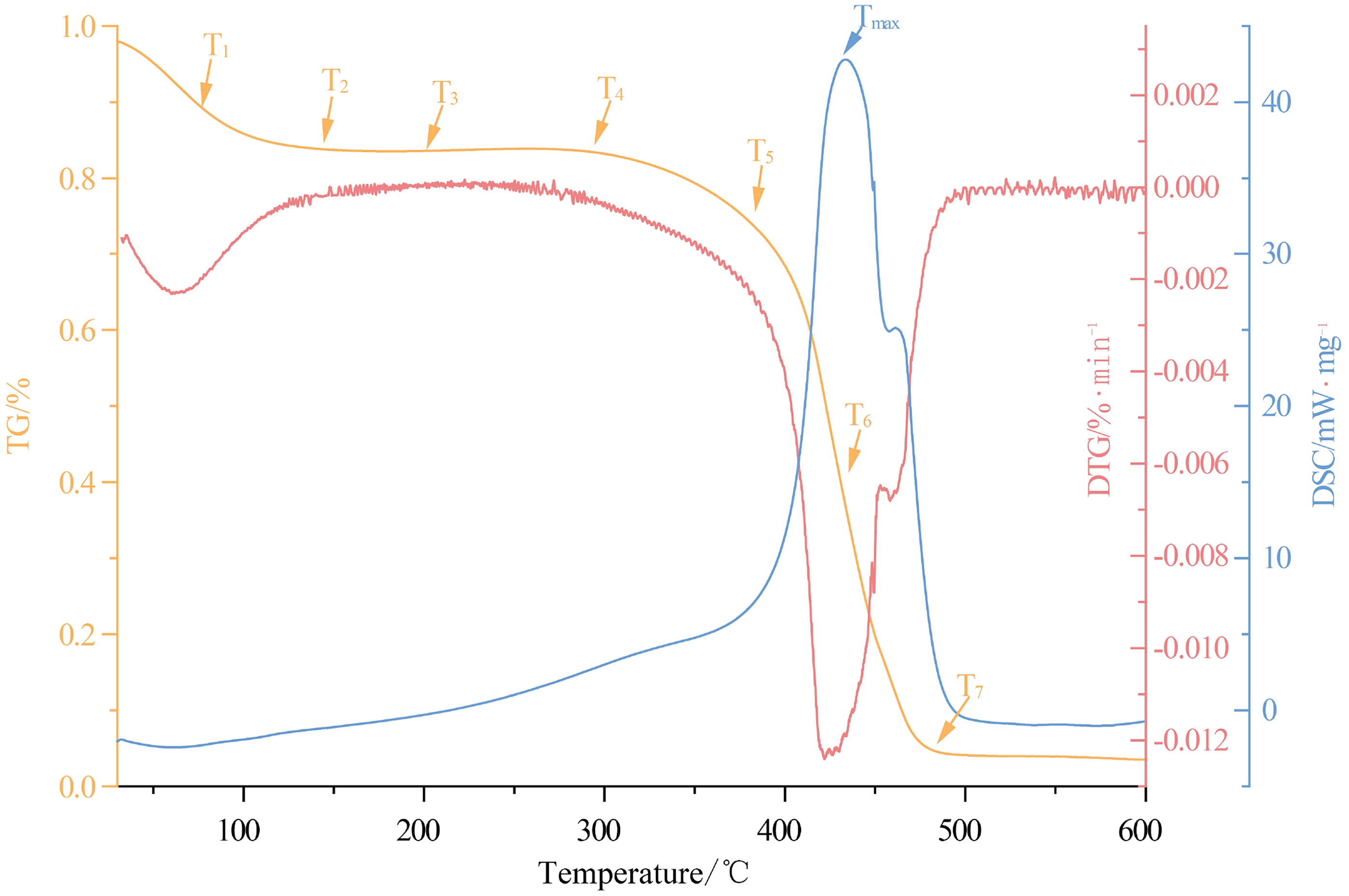
Characteristic temperatures in TG curves.
In Figure 3, it is observed that the TG and DTG curves exhibit significant differences which depend on the combustion stages. Therefore, characteristic temperatures can be defined based on these observations. (a) Critical temperature
Heating rate effect
As shown in Figure 4, with an increase in heating rate, the distinction between curves becomes progressively more discernible. The TG and DTG curves consistently exhibit a lagging behavior, which is associated with an elevation in coal's characteristic temperature (Figure 5). This phenomenon can be attributed to the fact that different active structures or functional groups within coal require a certain amount of time for their reaction processes. As the heating rate increases, this reaction time gradually diminishes, leading to a progressive rise in activation energy required by these active structures or functional groups. Consequently, fewer active structures participate in reactions and some conditionally involved structures fail to fully decompose. Furthermore, it is worth highlighting that a temperature disparity exists between the coal's interior and its surrounding environment. This temperature difference results in a lag effect where the interior takes longer to reach its surrounding environment temperature during heating. An increase in the heating rate leads to a more rapid rise in coal temperature per unit time and a corresponding increase in temperature disparity, causing both the TG and DTG curves to shift toward the higher temperature region. Similar trends have been reported by Gao et al. (2017). As the heating rate increases, this lag effect becomes stronger, which results in an increasing trend exhibited by each characteristic temperature of the coal. Each characteristic temperature of the coal is caused to exhibit a rising trend with higher heating rates by the slower response of the coal's interior.
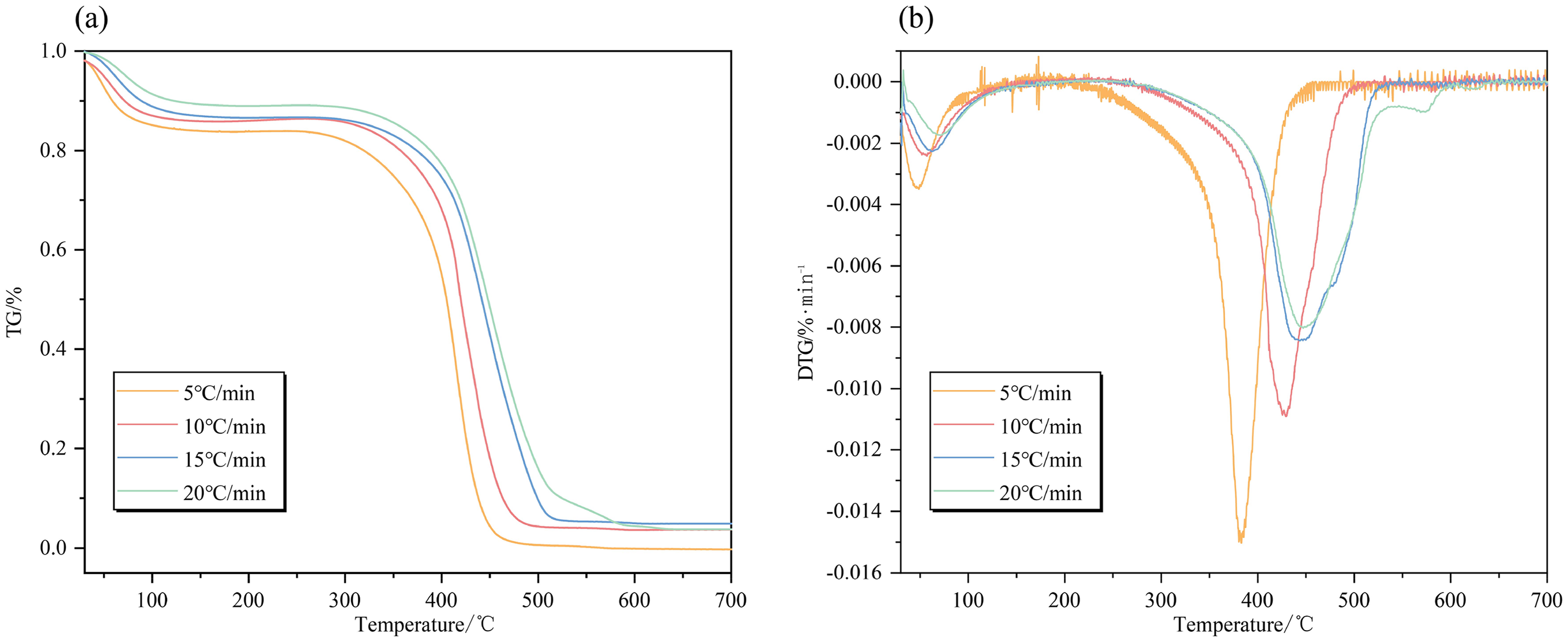
(a) TG curves and (b) DTG curves of coal samples at various heating rates.
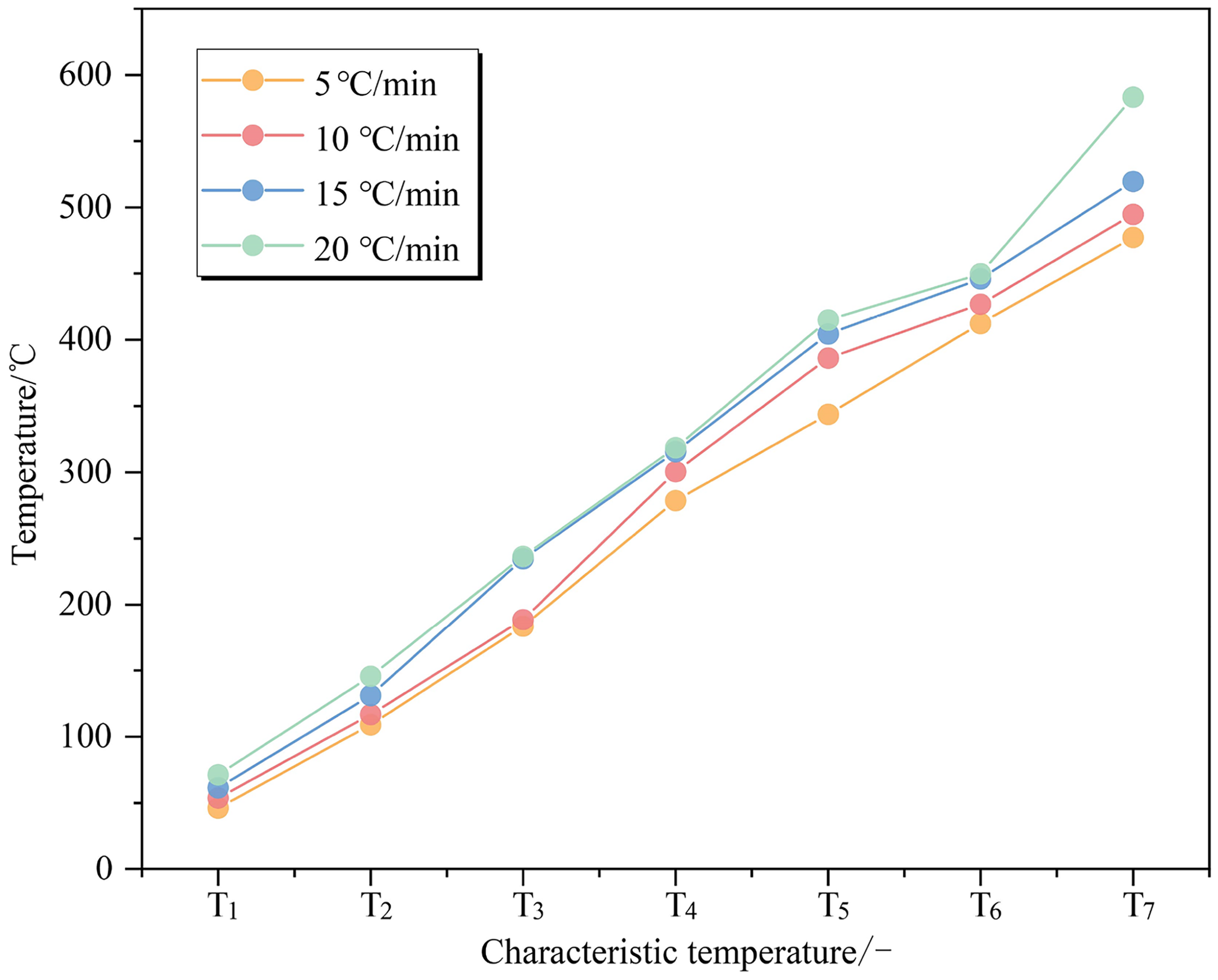
Characteristic temperature curves of coal samples at different heating rates.
The DSC curves exhibit a consistent pattern at different heating rates, as depicted in Figure 6. In Figure 6, the DSC curve displays two distinct peaks at heating rates of 5 °C/min, 10 °C/min, and 15 °C/min. The first peak demonstrates a relatively gentle height and shape, while the second peak exhibits a sharp increase in both height and shape. This phenomenon exactly mirrors the two stages of coal's spontaneous combustion, namely oxidation pyrolysis and intense combustion. Furthermore, when the heating rate is adjusted to 20 K/min, a third small yet sharp peak emerges at the end of intense combustion on the DSC curve. Some active structures require exceedingly high activation energy for their initiation reactions; thus, they struggle to react within the low-temperature environment. Excessively rapid heating rates result in an increased temperature disparity between the interior of coal and its surrounding environment. Consequently, active structures with exceptionally high activation energy can only be activated at temperatures higher than their typical reaction temperatures. Additionally, some active structures do not have sufficient time for reaction due to excessive heating speed, leading to temporary accumulation within coal samples throughout the process of combustion. These accumulated structures would be activated at the end of combustion, showing the third peak.
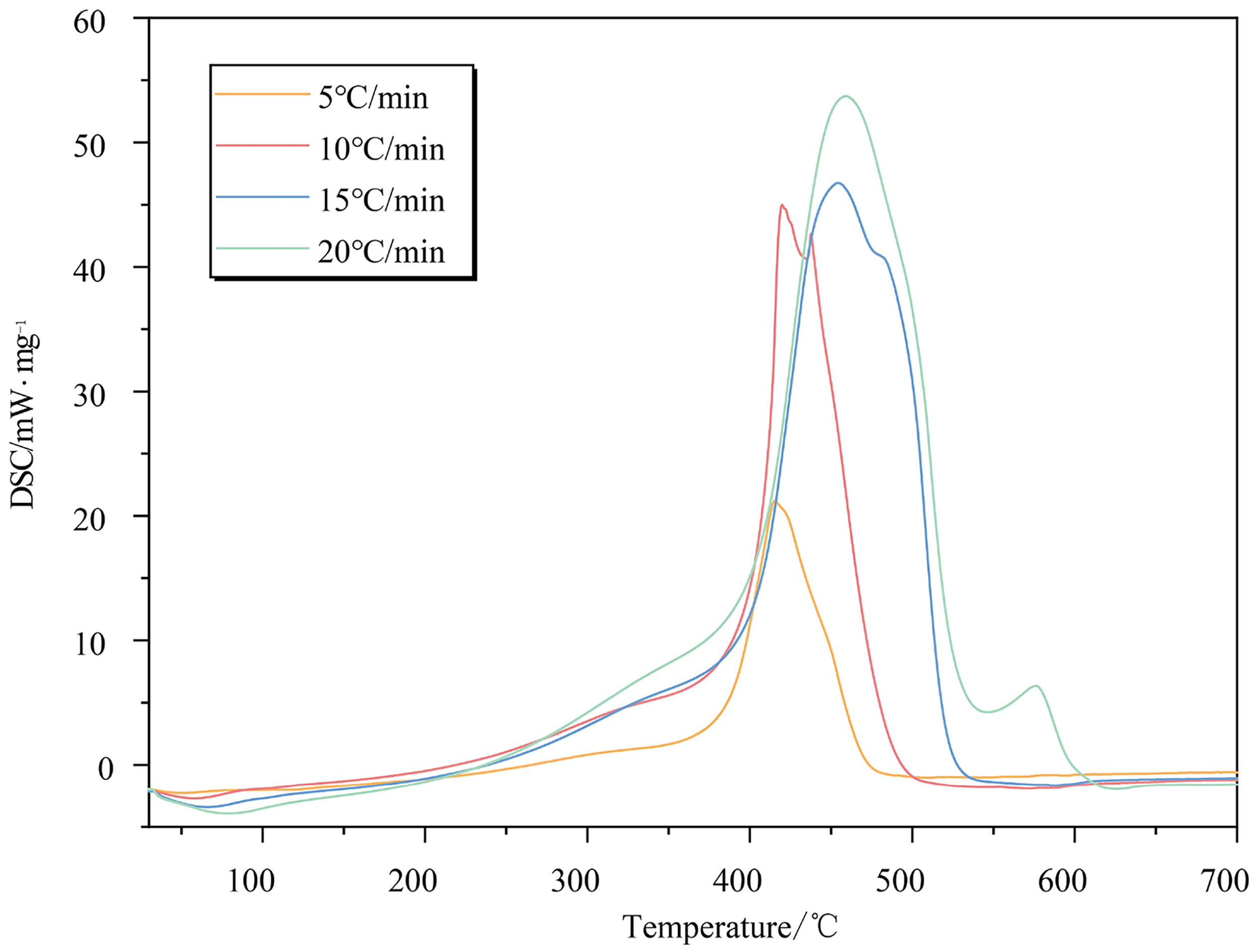
DSC curves of coal samples at different heating rates.
Size effect
Significant variations are observed in the TG curves of coal samples with varying particle sizes, as illustrated in Figure 7. It shows that an increase in particle size leads to a lagging effect on both the TG and DTG curves, which results in a continuous decrease in weight loss rate for coal samples. However, it is noteworthy that for the initial peak on the DTG curves, the curve of 2 mm particle size possesses the highest weight loss rate. This can be attributed to their higher water content and subsequent evaporation-induced weight loss. As the reaction proceeded further, the TG and DTG curves of coal samples with various particle sizes tended to converge at the combustion stage without any discernible differences. This phenomenon can be explained by the continuous rise in temperature and ongoing evaporation of water and volatiles within coal samples, leading to the formation of internal fractures. Upon reaching the combustion stage, all coal samples exhibit relatively well-developed internal pore structures irrespective of their particle sizes, enabling them to participate more extensively in reactions. Moreover, it is also worth noting that all coal samples with different particle sizes display similar burnout temperatures and quality in the end. Although characteristic temperatures generally show an increasing trend with larger particle sizes, this increase is marginal without significant implications (Figure 8).
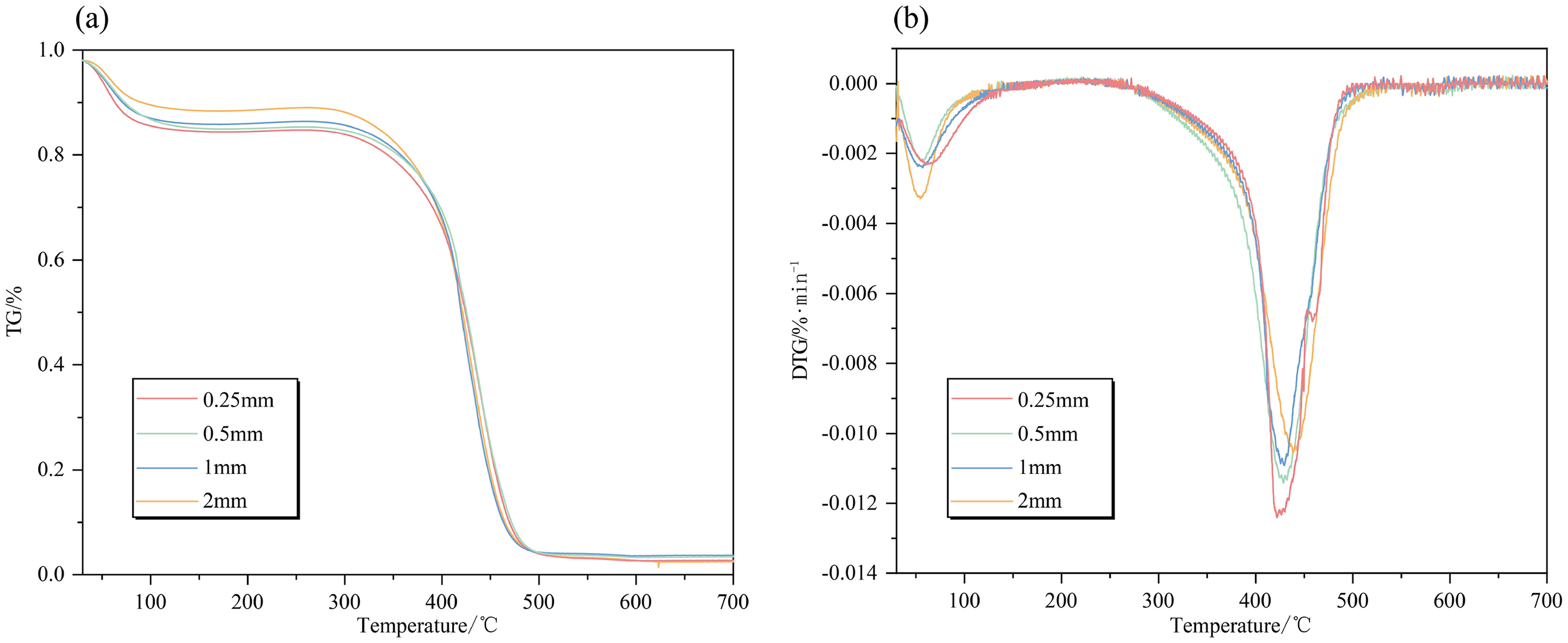
(a) TG and (b) DTG pyrolysis curves of coal samples for different particle sizes.
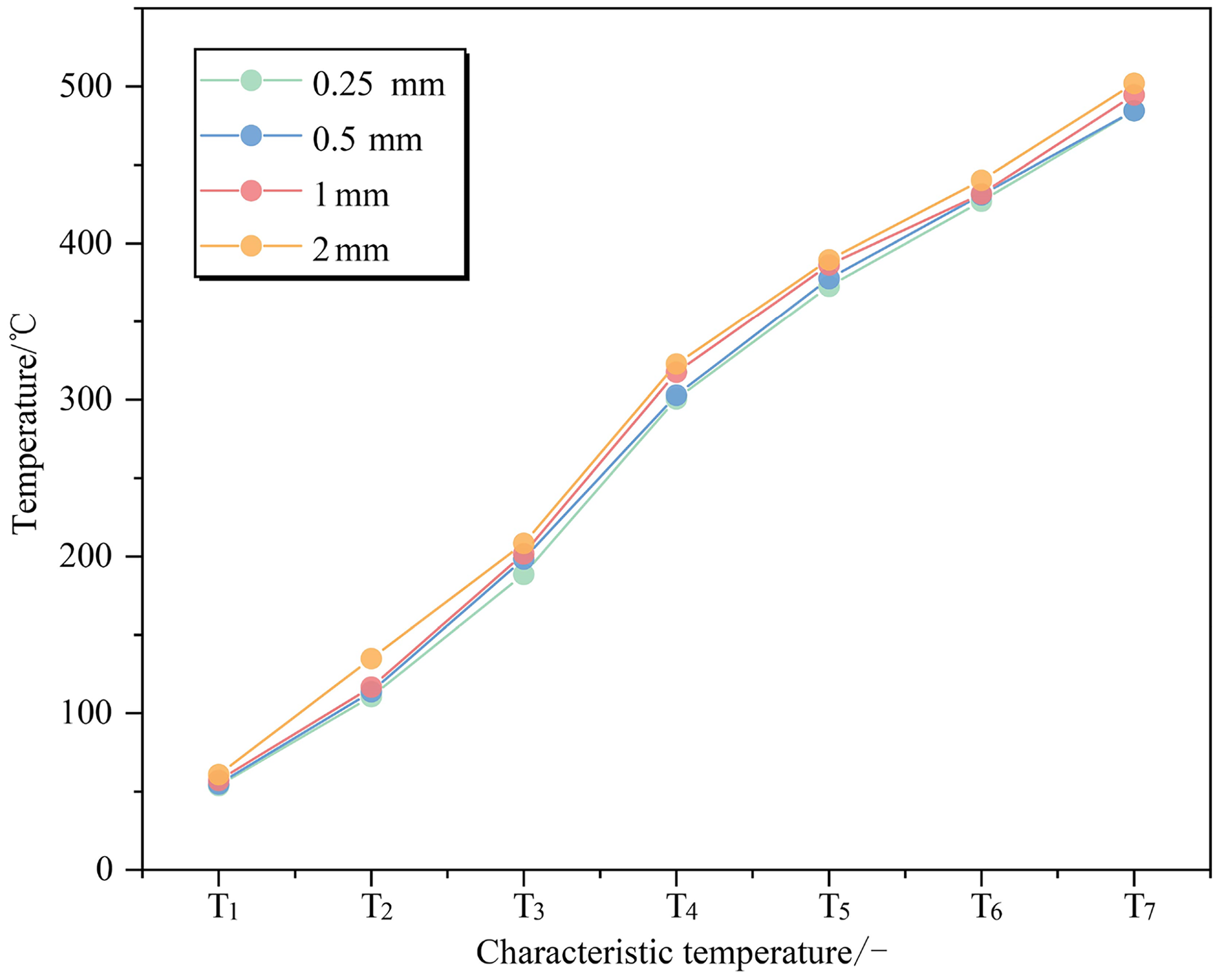
Characteristic temperature curves of coal samples for different particle sizes.
The shape and development trend of DSC curves of coal specimens with varying particle sizes exhibit remarkable consistency, as depicted in Figure 9. Generally, the curves demonstrate a “double ladder” pattern, and the laws governing DSC curves for coal samples with varying particle sizes remain consistently observed at each experimental stage. However, particle size significantly impacts the maximum heat release rate of coal samples; specifically, larger particles result in higher heat release rates and lower peak temperatures. This phenomenon can be attributed to the less developed internal pore structure and smaller specific surface area for larger particles, which consequently reduces the number of active structures exposed to the surface and diminishes oxygen adsorption per unit mass of coal. As a result, the coal–oxygen combination becomes less severe and oxidation heat release decreases.
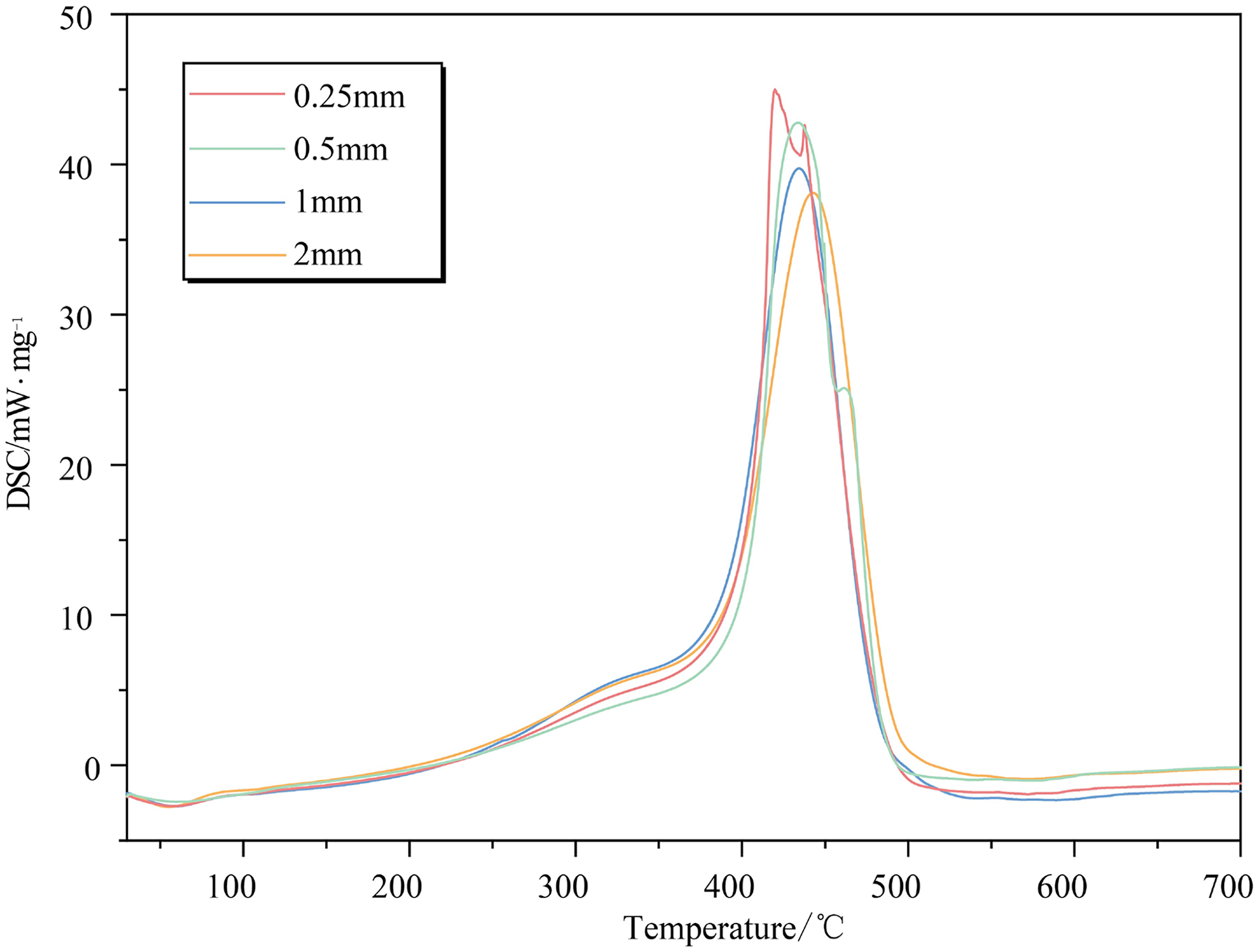
DSC curves of coal samples for different particle sizes.
The propensity effect
The tendency of coal for spontaneous combustion reflects its natural ability for self-heating and oxidation. To explore the influence of propensity on the spontaneous combustion of coal, three coal samples, gathered from Rongchang, Datong, and Tongchuan, were selected for analysis. The propensity for each coal sample was measured, with the largest to smallest being Rongchang, Tongchuan, and Datong. For convenience in analysis, their values (1, 2, 3) are given based on the order of propensity.
It is seen in Figure 10 that the TG and DTG curves exhibit significant differences, showing a pronounced forward phenomenon with the increase in spontaneous ignition propensity. The profiles of mass loss shift to elevated temperatures, and the final mass loss is larger than that associated with a smaller propensity. Each characteristic temperature gradually decreases as spontaneous ignition propensity increases (Figure 11). These observations indicate a shortening of the total time for coal oxidation and the ignition process. The higher spontaneous ignition propensity of coal corresponds to more active groups with lower activation energy requirements. Therefore, more generated gases escape leading to significant variations in mass loss rate and residual quality of coals.
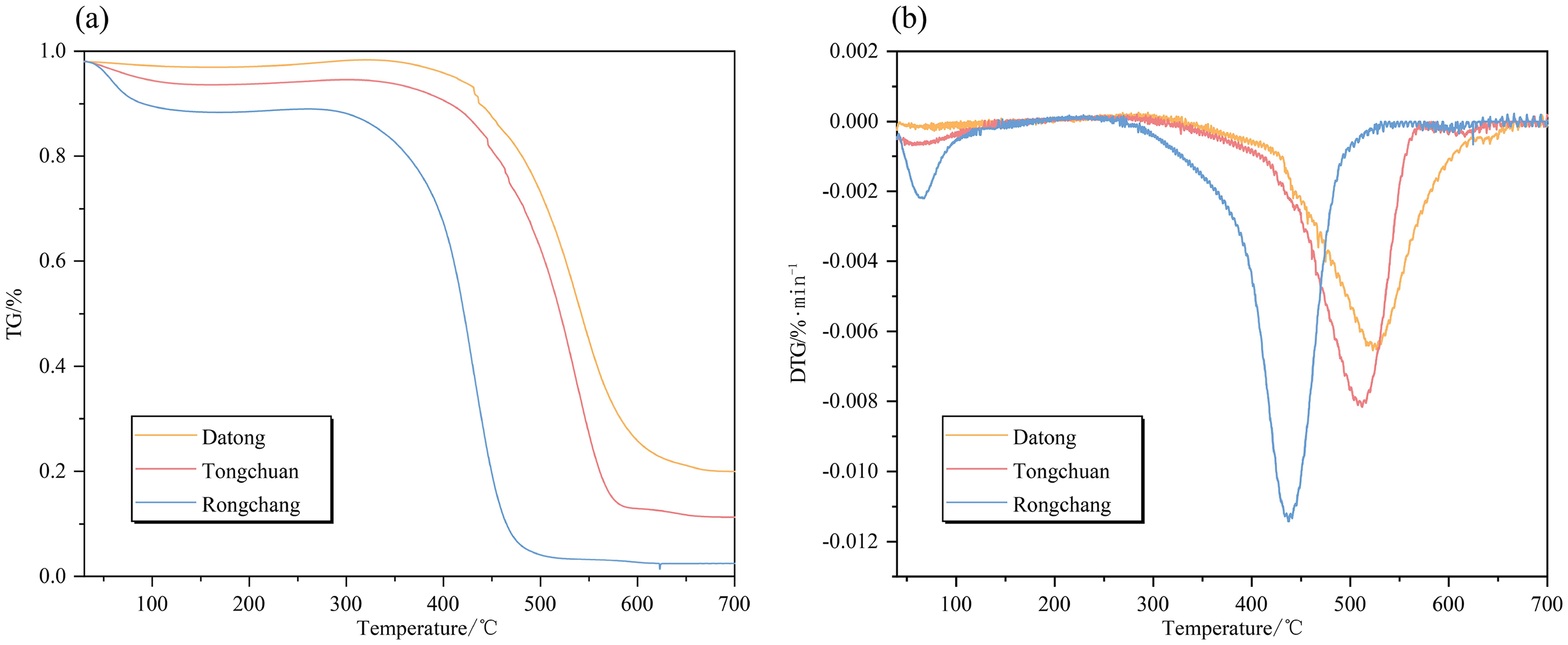
(a) TG and (b) DTG pyrolysis curves of coal samples for different spontaneous combustion propensities of coal.
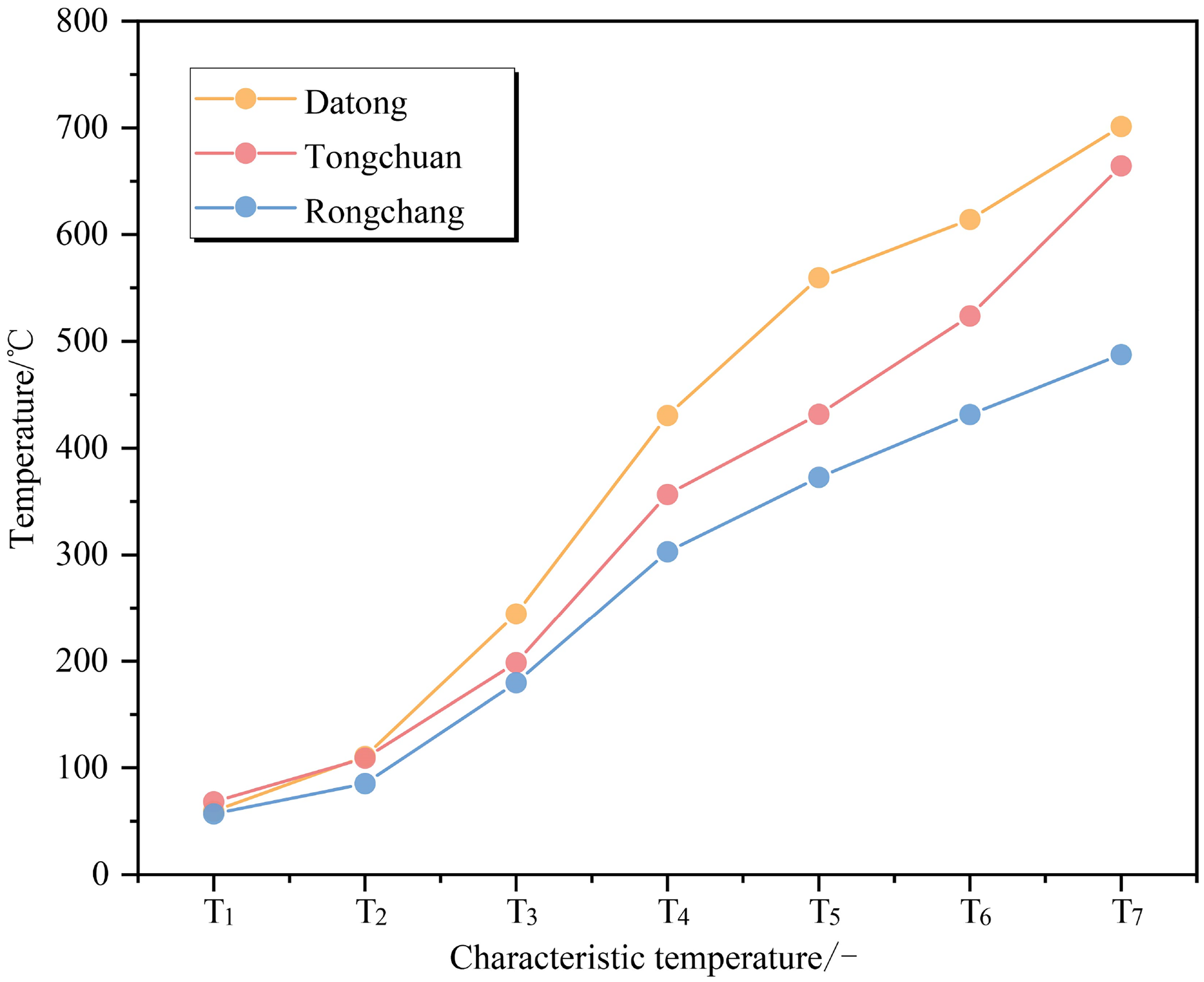
Characteristic temperature curves for different spontaneous combustion propensities of coal.
As illustrated in Figure 12, it can be observed that the shape and development trend of DSC curves for different levels of spontaneous combustion propensities are essentially similar. During the initial stage, both Datong and Tongchuan coal samples exhibit no significant changes and have a similar temperature to the first peak. However, Rongchang coal samples with a weaker propensity exhibit significant distinctions prior to the first peak, showing an increasing-then-decreasing trend. This is ascribed to continuous heat absorption of active groups with larger activation energy requirements. During the middle and final stages of the experiment, all three coal samples display consistent patterns. Overall, as spontaneous combustion propensity decreases, the peak temperature increases, leading to a decrease in heat release rate.
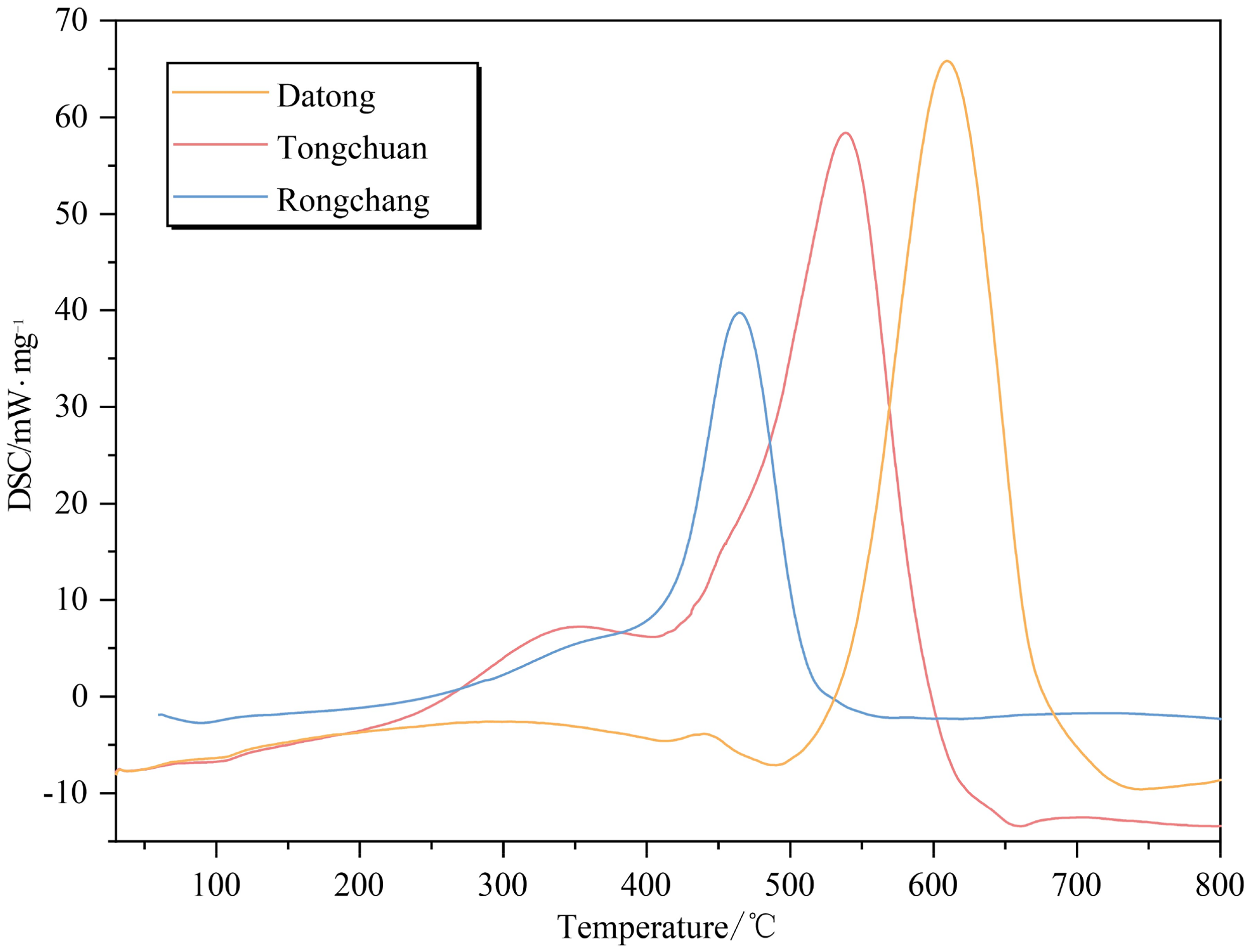
DSC curves of coal samples for different spontaneous combustion propensities of coal.
Kinetic analysis
The kinetic triplet, which includes the mechanism function, pre-exponential factor, and activation energy, effectively encapsulates the essence of the kinetic process. Activation energy is employed in the subsequent analysis to systematically analyze the quantitative characteristics of the coal pyrolysis process. The coal oxidation reaction rate can be expressed as follows using the principles of chemical reaction kinetics (Łabojko et al., 2012; Sbirrazzuoli, 2013):

Using the non-isothermal thermogravimetric approach, the heating rate is formulated as:
The integral representation of 
According to equations. (1), (3) and (4), 
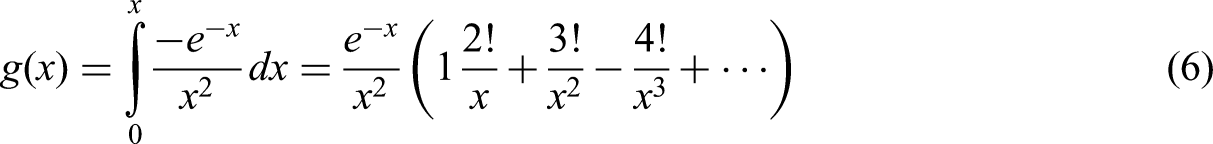
In fact, 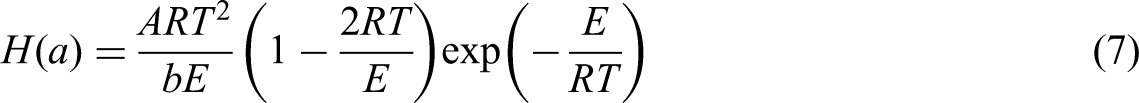
Given that 
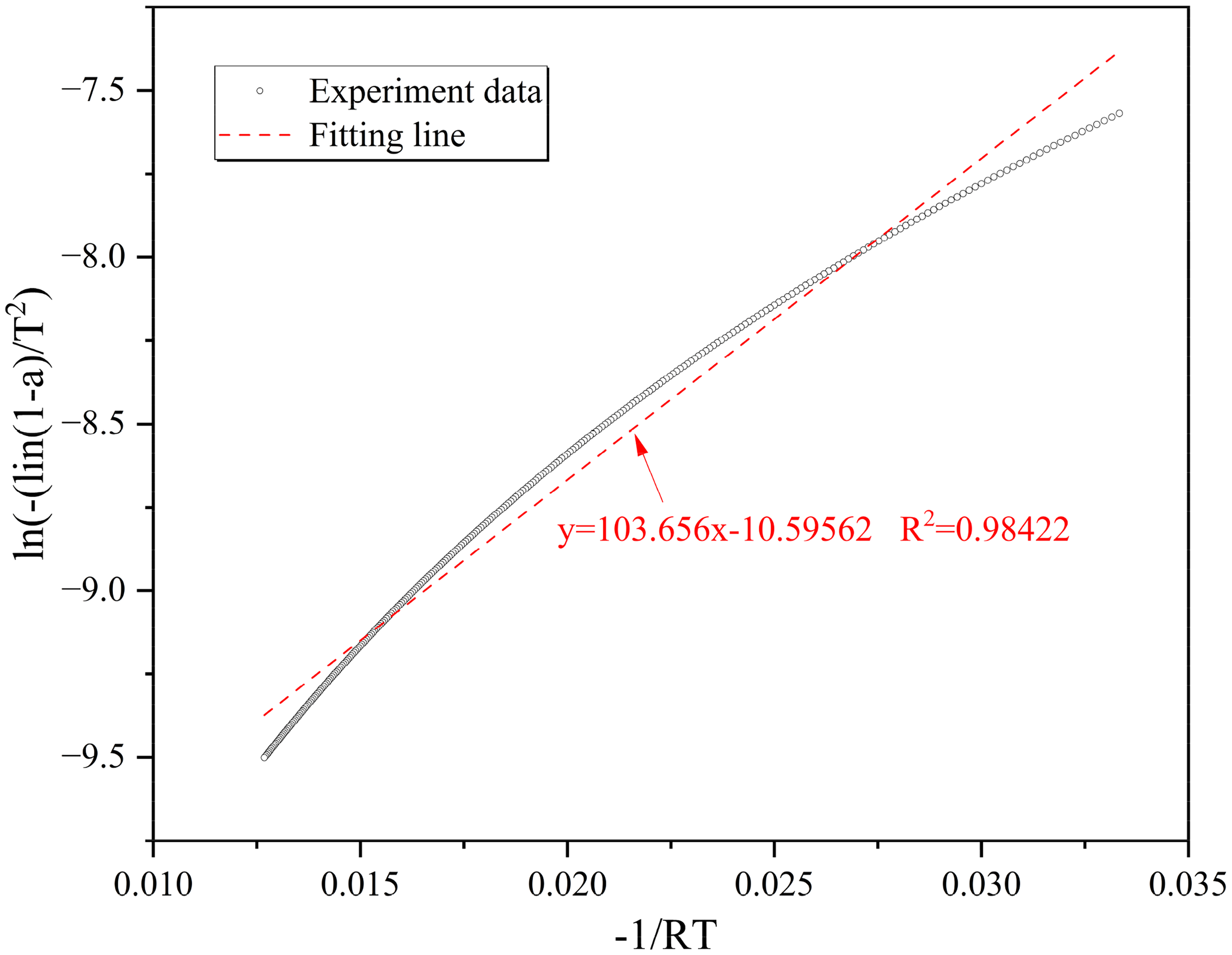
An example of fitting process for the activation energy (5 °C/min in Rongchang).
Analysis of coal.

As shown in Table 2, increasing the heating rate from 5 °C/min to 20 °C/min leads to an increase in activation energy from 103.656 to 125.846 J/mol, representing a relative increase of approximately 21.41%. This can be attributed to the fact that at higher heating rates, the energy required to initiate the combustion reaction also increases. This phenomenon can be explained by the Arrhenius equation, which states that the rate of a chemical reaction is exponentially dependent on the activation energy. Therefore, a rise in the heating rate results in an elevation of the activation energy. Moreover, the experimental data show that as the particle size increases, the activation energy also increases. This can be comprehended by considering the ratio of surface area to volume in the particles. Particles of smaller size possess a larger surface area relative to their volume, which promotes faster reaction rates and lower activation energies. In contrast, larger particles have a smaller surface area relative to their volume, leading to slower reaction rates and higher activation energies. Besides, it can also be seen that as the spontaneous combustion propensity increases, the activation energy decreases. This behavior can be elucidated by the observation that materials with a greater propensity for spontaneous combustion have a lower activation energy requirement for combustion to occur. This suggests that the presence of certain chemical compounds or conditions can enhance the reactivity of the material, thereby lowering the activation energy needed for combustion. In conclusion, our experimental data support the notion that the relationship between heating rate, particle size, spontaneous combustion propensity, and activation energy is indeed complex. These findings provide valuable insights into the mechanisms underlying combustion processes and can contribute to the development of safer and more efficient fire prevention measures.
Conclusions
This study conducts a comprehensive analysis of the factors affecting coal spontaneous combustion (heating rate, particle size, spontaneous combustion propensity) using a thermogravimetric analyzer. The characteristic temperature points at each phase of the experimental procedure are defined, leading to the following conclusions:
As the heating rate increases, continuous hysteresis is observed in the TG and DTG curves, resulting in a rise in the characteristic temperature of coal. As the particle size increases, both the rate at which the coal sample loses weight diminishes and the characteristic temperatures decrease, indicating that the TG and DTG curves lag. As the spontaneous combustion propensity increases, a notable distinction exists between the TG and DTG curves, showing a significant forward shift phenomenon. The characteristic temperatures progressively decrease, while the weight loss rate of the coal specimen increases. The activation energy, which is determined through thermogravimetric analysis, increases when heating rates and particle sizes are larger. In contrast, it diminishes with a rise in the propensity for spontaneous combustion, aligning with earlier experimental findings.
Footnotes
Nomenclature
Ethical considerations
The authors confirm that the work described in this paper has not been published elsewhere, nor is it under consideration by another publisher.
Consent to publication
Depending on the ownership of the journal and its policies, the authors grant the Publisher an exclusive license to publish this article.
Author contributions
Junchao Chen: conceptualization, methodology, supervision. Xiaoyuan Shen: data curation, methodology, writing-original draft. Ruiqing Bi: conceptualization, software. Liang Zhang: methodology, validation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research is financially supported by the National Natural Science Foundation of China (52204212; 52204219) and S&T Program of Hebei (22375401D).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data that support the findings of this study are available from the corresponding author upon reasonable request.
