Abstract
Propane-oxidizing bacteria in surface soils are often used to indicate the position of oil and gas reservoirs. As a potential replacement for the laborious traditional culture-dependent counting method, we applied real-time fluorescent quantitative polymerase chain reaction detection as a quick and accurate technology for quantification of propane-oxidizing bacteria. The propane monooxygenase gene was set as the target and the assay is based on SYBR Green I dye. The detection range was from 9.75 × 108 to 9.75 × 101 gene copies/µl, with the lowest detected concentration of 9.75 copies/µl. All coefficient of variation values of the threshold cycle in the reproducibility test were better than 1%. The technique showed good sensitivity, specificity, and reproducibility. We also quantified the propane-oxidizing bacteria in soils from three vertical 250 cm profiles collected from an oil field, a gas field, and a nonoil gas field using the established technique. The results indicated that the presence of propane monooxygenase A genes in soils can indicate an oil or gas reservoir. Therefore, this technique can satisfy the requirements for microbial exploration of oil and gas.
Keywords
Introduction
With the extensive exploration of oil and gas reservoir, the distribution of remaining oil and gas resources increasingly dispersed, and reservoirs are getting smaller and most of the reservoirs belong to atectonic reservoirs. Therefore, it is difficult and costly to explore the reservoir with the geophysical and geochemistry exploration only (Yuan, 2011). Gaseous hydrocarbons present in subsurface reservoirs can migrate to the surface soil through upward diffusion and effusion in response to the formation pressure (Saunders et al., 1999). Some microbes in soil can exclusively use these hydrocarbons as the carbon source for their metabolic activities and growth (Mogilewskii, 1938). As a result, there is a positive correlation between hydrocarbon concentration and hydrocarbon-oxidizing bacteria in the soil (Hitzman et al., 2002). These light hydrocarbon-oxidizing bacteria have been exploited for petroleum exploration for a long time (Rasheed et al., 2011). The microbial prospecting methods can give us the cheap and effective early exploration to reduce drilling risks. The methods can also grade the hydrocarbon contents in various geologic structures. Especially, it is of great significance in the prediction of subtle and deep reservoir (Liu et al., 2014).
Methane is the dominant component of light hydrocarbons in oil and therefore methane-oxidizing bacteria have been intensively investigated (Xu et al., 2013). However, in addition to thermogenic methane, biogenic methane originates from the decomposition of organic matter by methanogens in anaerobic environments and can occur under appropriate substrate and environmental conditions regardless of the presence of oil or gas reservoirs (Schoell, 1988). As a result of the multifactorial nature of methane formation, methanotrophs alone are not reliable indicators of petroleum reservoirs (Brisbane and Ladd, 1965). In contrast, propane, another important light hydrocarbon, is appropriate for petroleum reservoir prospecting because biologically produced propane is much less prevalent than propane originating from seepage from geological sources (Ashraf et al., 1994).
There are two different traditional methods of determining the abundance of gaseous hydrocarbon-oxidizing bacteria: the plate method and the maximum probability number method (including the culture flask test) (Yi et al., 2006). The current two microbiological techniques used to explore for oil and gas, the Microbial Oil Survey Technique and Microbial Prospecting for Oil and Gas, are examples of a plate method and a maximum probability number method, respectively (Chan, 2011). Both methods can be called culture-dependent microbial approaches, which are laborious and time consuming and cannot represent the uncultured microorganisms that account for more than 99% of the microorganisms naturally occurring in the environment (Hugenholtz et al., 1998).
The development of a technology that detects a population of bacteria without culturing is widely preferred. Molecular techniques offer speed and specificity that culturing cannot. Molecular techniques related to functional genes have been proven to be effective as a basis for understanding the microbial diversity in environmental communities (Anderson and Cairney, 2004). Methods to indicate the presence of propane-oxidizing microorganisms based on polymerase chain reaction (PCR) technology have been developed (Chan, 2011). However, there is currently no culture-independent quantification method for propane-oxidizing microorganisms. Fluorescent quantitative real-time PCR (RT-PCR), an important molecular technique, can quantify the specific functional genes from environmental samples within a matter of hours (Bustin et al., 2005). Molecular techniques, including RT-PCR, which can detect methanotrophic bacteria are currently available and have been intensively used in petroleum reservoir prospection (Miqueletto et al., 2011; Xu et al., 2013; Zhang et al., 2010). However, as an important indicator of oil and gas reservoirs, the quantification of propane-oxidizing bacteria has not been studied in detail.
Bacterial propane oxidation is catalyzed by a reduced nicotinamide adenine dinucleotide-dependent multicomponent enzyme system belonging to the family of dinuclear-iron oxygenases. There are three components in this dinuclear-iron-containing monooxygenases, including a hydroxylase, a reductase, and a coupling protein. Hydroxylation of propane occurs at a dinuclear iron of the large subunit (propane monooxygenase A (
In this study, we developed the RT-PCR technique for the quantification of propane-oxidizing bacteria based on the
Materials and methods
Sample collection and DNA preparation
Gas field soil samples were taken from the Xiaoquan-Xinchang gas exploration area in western Sichuan Province, China. This gas field is a composite field that was composed of multiple Jurassic secondary gas pools and has a shallow burial depth (Ren and Xia, 2006). Oil field soil samples were taken from the Yangxin oil exploration area in the Huabei oil field, China. The site is located above an anticlinal structural reservoir, which is an anticlinal structural trap that was formed by the gravitational effects and the differential compaction of Paleogene Shahejie formation sediments that were deposited above the early formed horst. The nonoil gas field samples were collected from a farmland in Zhengding County, Hebei Province, China. It is a contact zone of the Shanxi platform and the Bohai depression. The sampling sites are pinpointed in Figure 1.
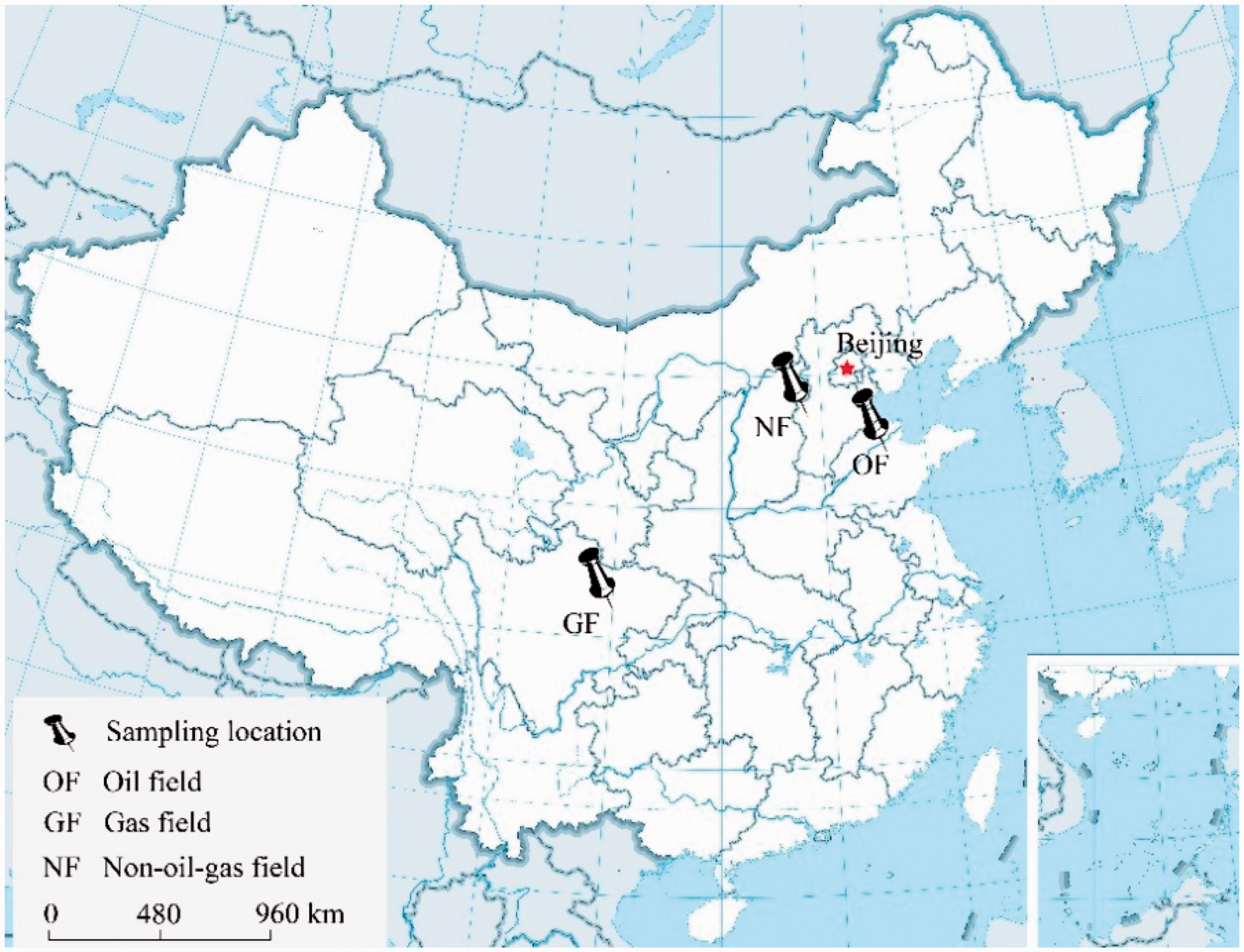
The locations of the sampling sites.
In the oil and gas fields, a vertical profile in the vadose zone was collected, 50 m from the oil or gas well to avoid disturbance during well completion. A vadose zone vertical profile was also chosen in the nonoil gas field in an area identified as least disturbed by human activities. Eight soil samples retrieved from depths of 0.05, 0.2, 0.5, 1.0, 1.5, 2.0, and 2.5 m were collected from each profile.
When sampling, the soil cores were initially taken out using Luoyang shovel which is a special hand auger and is used for drilling soil profiles and sampling the soil. Subsequently, a part of the soil samples was collected from the core center using a sterile knife and placed in aluminum boxes previously sterilized at high temperatures. Collected samples were immediately stored in an incubator with an ice pack and then transferred to the laboratory and stored at −70°C prior to extraction of the genomic DNA within two days. Another subsample of the soils was stored in plastic automatic sealing bags for analysis of physical and chemical characteristics. In addition, the soils at 150 cm depth were chosen for light gaseous hydrocarbon analyses. These soil samples were placed into a 1000 ml plastic bottle, which was prefilled with saturated brine and then sealed with a screw cap.
Genomic DNA from approximately 0.5 g of fresh soil for each of the samples was extracted using the PowerSoil® DNA Isolation Kit (Mo Bio Laboratories, USA) according to the manufacturer’s instructions. The obtained DNA was quantified with a Biodropsis BD-2000 spectrophotometer. The DNA extracts were stored in Tris–EDTA buffer (pH 7.0) at −20°C for further analysis.
The soil temperature was measured using a mercurial thermometer and the pH was measured using a portable pH meter (Leici PHB-4, China). The soil water content was determined by drying soil overnight at 105°C. The soil lithology was determined according to the
Primer design and standard plasmid preparation
The forward and reverse primers of 955F25 (TGG CAC CGG TGG ATC TAC GAC T) and 1517R22 (GCG CGA TCA GCG TCT TGC CGT C) were designed using conserved nucleotide sequences in genes encoding the large hydroxylase subunit of propane monooxygenases,
The genomic DNA from the oil field at 50 cm soil depth was chosen as the template for the PCR procedure. The PCR was performed in an Eppendorf, 5333 PCR Thermocycle Instrument (Eppendorf AG) and followed the optimized protocol for 50 µl reactions (Chan, 2011). The PCR reaction products were examined by 1.0% agarose gel electrophoresis (DYY-6C, Beijing Liuyi, China) and visualized with the Gel Documentation and Analysis System (WD-9413B, Beijing Liuyi, China). If the PCR products were verified as the target fragment,
Optimization of the RT-PCR reaction system
First, according to the procedures and reaction system of the SYBR® Premix Ex Taq™ kit, the
The total volume of the reaction mixture system was 20 µl, including 2× SYBR Premix Ex Taq™ (10 µl), 10 µM PCR Forward Primer (0.2 µl), 10 µM PCR Reverse Primer (0.2 µl), 50× ROX Reference Dye (0.4 µl), DNA (2 µl), and dH2O (7.6 µl). The mixtures were processed at 94°C for 30 s, followed by 40 cycles of 5 s at 94°C, 30 s at 66°C, and 60 s at 72°C and then a read step at 88°C for 15 s. Subsequently, in the melt stage, the temperature was held for 15 s at 95°C and for 60 s at 60°C, and then the temperature was gradually increased to 95°C (for every 0.3°C increase, the fluorescence intensity was read for 15 s).
Sensitivity evaluation
Tenfold serial dilutions of standard plasmid DNA, acting as templates in duplicate, were used as a RT-PCR assay to prepare a standard curve for the enumeration of propane-oxidizing bacteria. The relationship between
Repeatability evaluation
The standard plasmid DNA was diluted to three concentrations from high to low concentrations (9.75 × 105, 9.75 × 103, and 9.75 × 101 copies/µl). Each of the three concentrations was measured six times. The standard deviation and coefficient of variation of the
Specificity evaluation
The specificities of the technique were tested with template DNA from strains not containing propane monooxygenase from our laboratory culture collection, which include aniline-degrading bacteria, quinoline-degrading bacteria, ethyl sulfonic acid-degrading bacteria, and oil-degrading bacteria. Plasmid DNA at the concentration of 9.75 copies/µl and sterile water were set as the positive and negative controls, respectively. Subsequently, the
Further, the diversity of propane-oxidizing bacteria in the RT-PCR product was examined and the phylogenetic tree was generated using clone library analysis (Jones et al., 2009).
Quantification of the prmA gene in soil samples
The purified DNA fragments from soil samples collected from the oil field, gas field, and nonoil gas field were used as template to perform the RT-PCR amplification to quantify the
To determine the relative abundance of
Results
Physical and chemical characteristics
The results of physical and chemical characteristics of the soil samples are shown in Table 1. All samples have similar salt content ranging from 0.02 to 0.07%. The gas field samples had the highest temperature by a small margin. The oil field samples had higher pH values (8.92–9.38) than the gas field samples (7.61–8.08). The surface samples had lower pH values than samples at depth in both fields. The moisture content of gas field samples was nearly double that of the oil field samples, which might be because of the climate (Chen et al., 2005). The surface soil samples (0–50 cm) in the gas field are silty light clay while in the oil field they are silty light loam. At the depth of about 200 cm, the gas field samples are medium sand while the oil field samples are sandy silt. In general, the Yangxin oil field soil is characterized by weak alkaline pH, low moisture, and low temperature.
The physical and chemical characteristics of the soil samples.
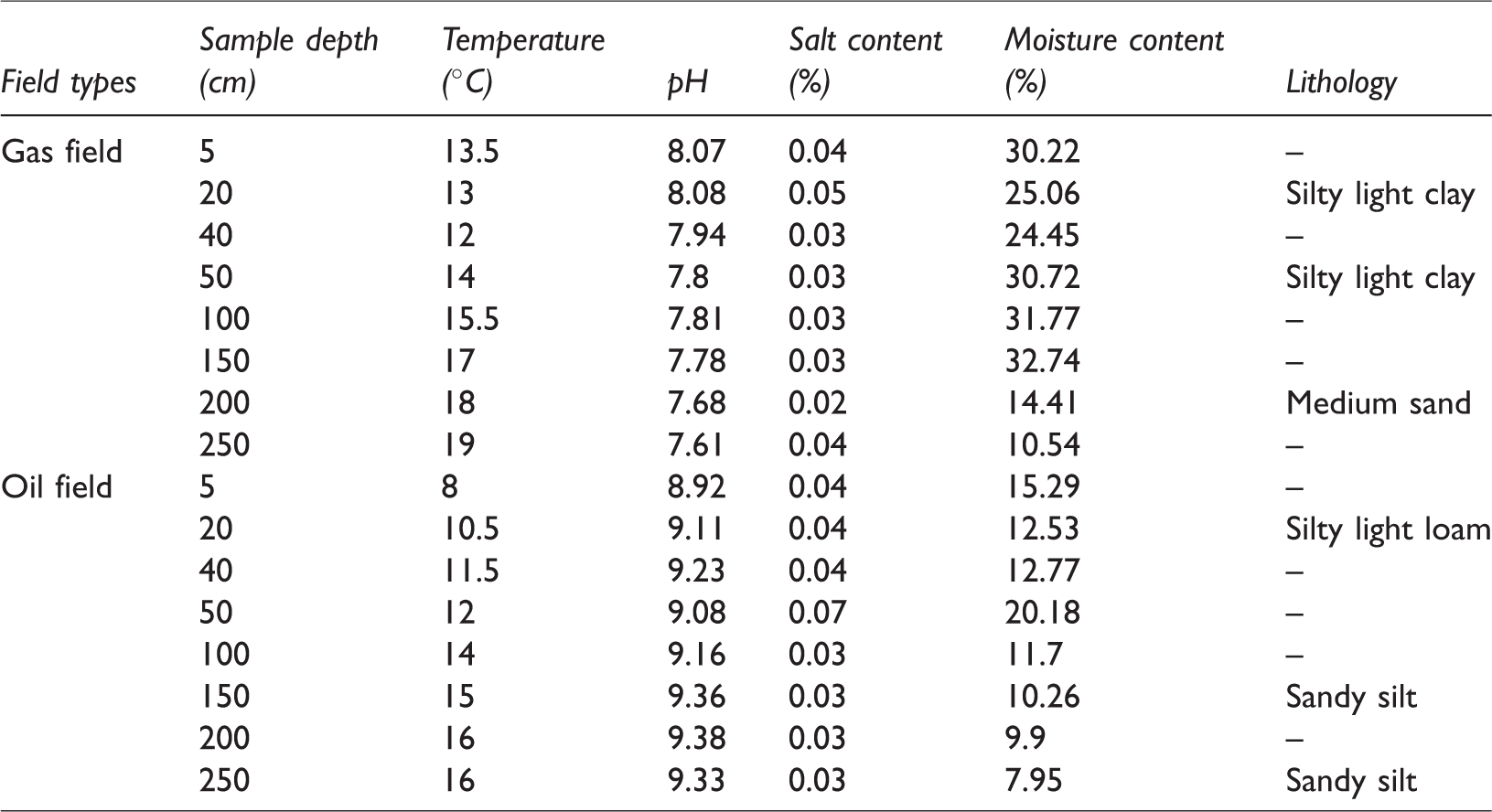
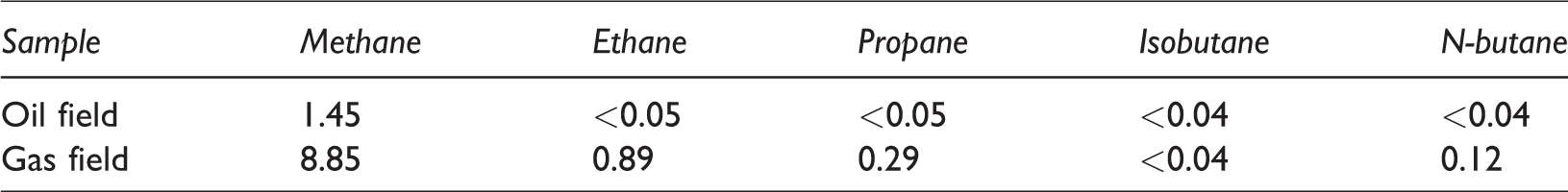
Table 2 shows the concentrations of light gaseous hydrocarbons adsorbed in soil particles at 150 cm depth. We found that the soil in the gas fields had more light gaseous hydrocarbons than those in the oil field. This is because the gas reservoir might effuse more propane than the oil reservoir and the surface clay soil might retard the gas seeping out of the soil in the gas field.
The concentrations of C1–C4 hydrocarbons in soil samples’ particles µl/l.
Standard plasmid preparation
The PCR reaction was performed using the template DNA from the oil field soil and the product was determined to be the target fragment, the
Sensitivity evaluation
When the concentrations of standard plasmid ranged from 9.75 × 108 to 9.75 × 101 copies/µl, the 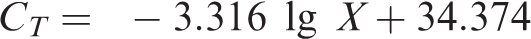
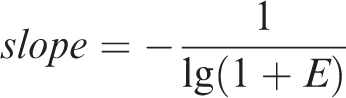
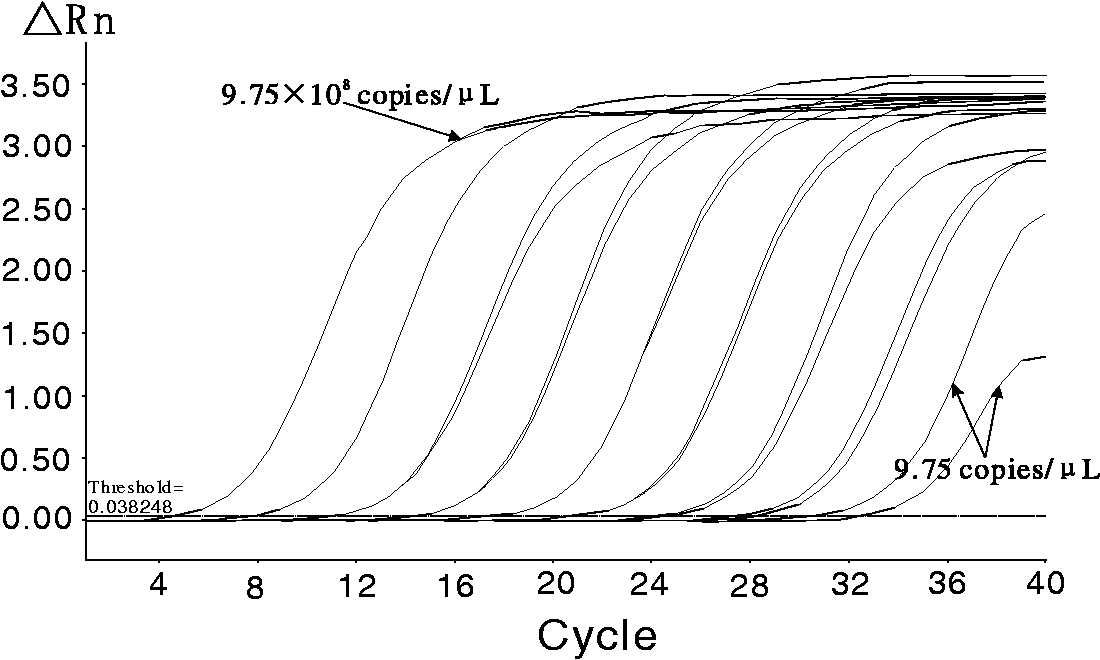
Amplification curves of standard plasmid with
As a result, the measurement range of this method was shown to be from 9.75 × 108 to 9.75 × 101 copies/µl.
We also found that the amplification curve could still be detected even if the concentration of the template was only 9.75 copies/µl. Therefore, the limit of detection of the method was 9.75 copies/µl.
Repeatability evaluation
The
The
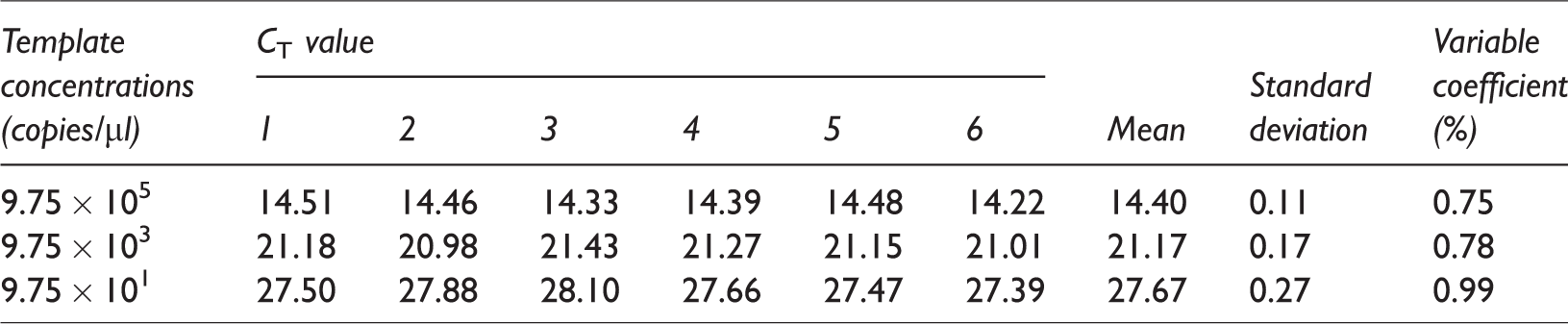
The template concentrations measured in repeatability evaluation.

Specificity evaluation
The
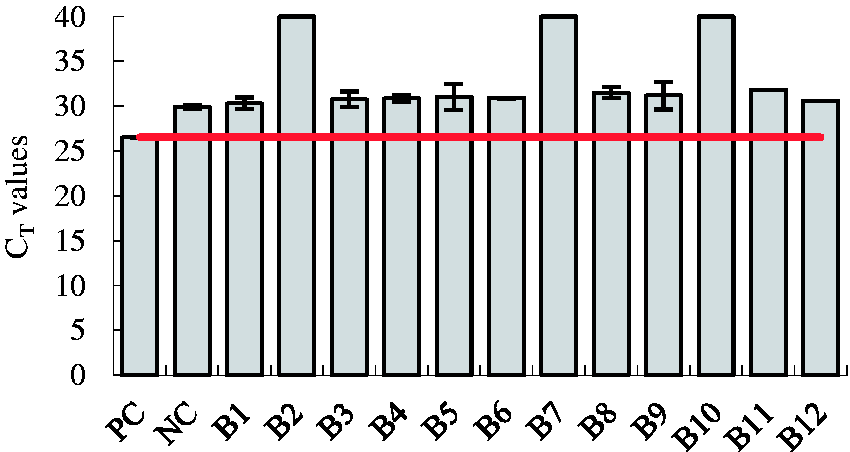
The
In addition, we examined the diversity of propane-oxidizing bacteria in the RT-PCR product using clone library analysis of the soil sample collected at 50 cm soil depth in the Yangxin oil field (Figure 4). There were 145 clones in the library, all of which contained the propane monooxygenase gene. Most of these clones have the same sequence as BYX-1, whose sequence is similar to an uncultured soil bacterium, clone SL01 (JX045657.1), propane monooxygenase gene, according to the NCBI database. The percentage identity value was 90%. The SL01 clone was obtained during an investigation of the activity and diversity of the microbial community in the presence of light hydrocarbons. The BYX-13, BYX-53, BYX-66 clones have similar sequences to
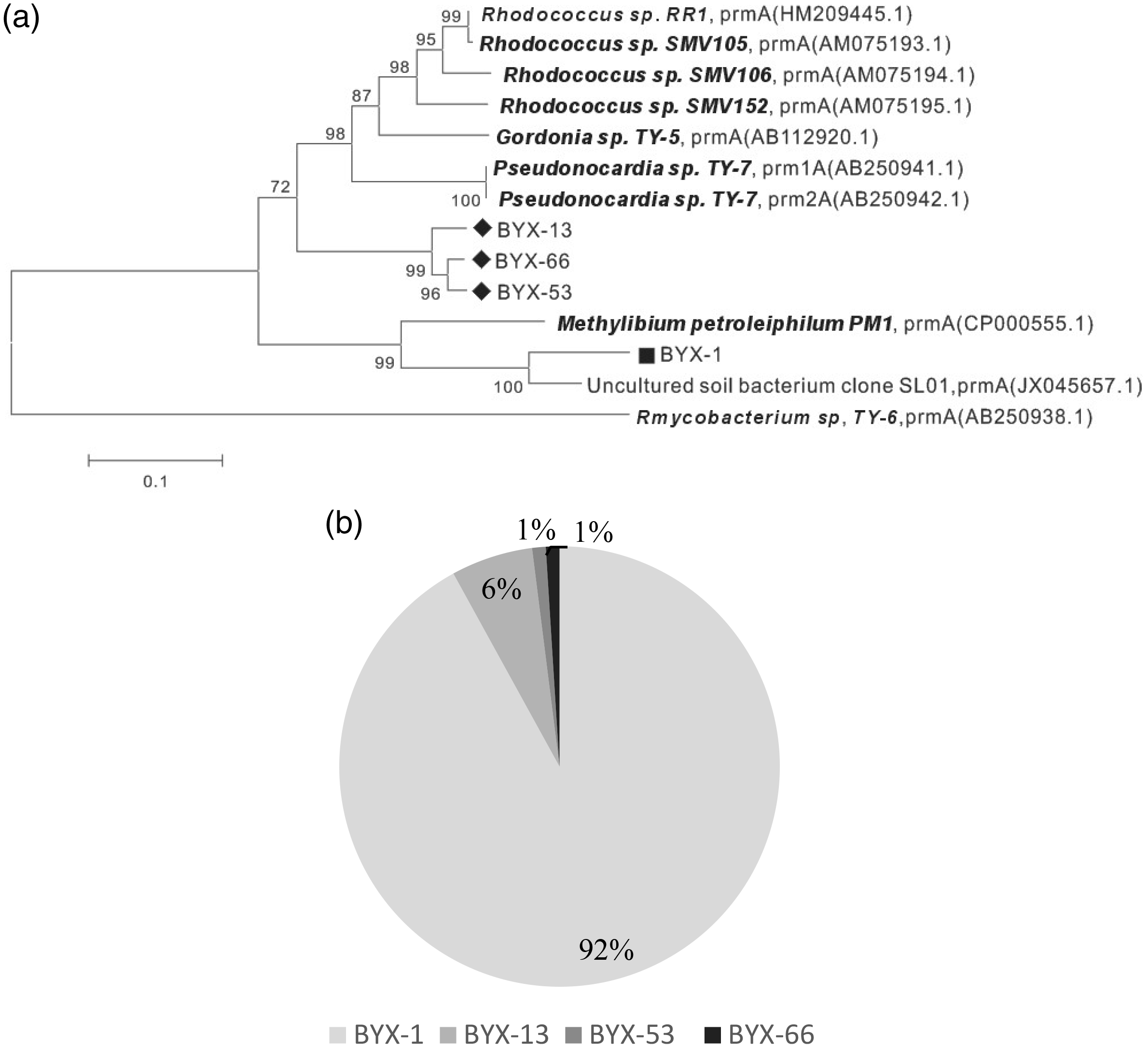
The diversity of bacteria in the RT-PCR products amplified from Yangxin oil field soil. Part a is the phylogenetic tree constructed through the clone library. Part b indicates the percentage of each clone that has the same sequences in the clone library. BYX-1, BYX-13, BYX-53, and BYX-66 are clones that have different sequences in the clone library.
Quantification of the prmA gene content in various soil samples
The
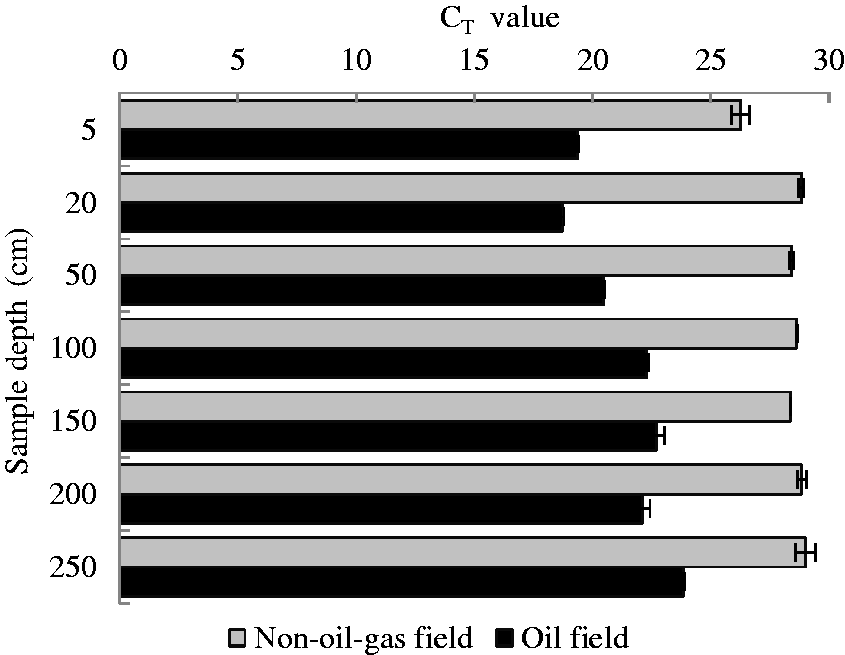
The
According to the above comparison and analysis, we found that the
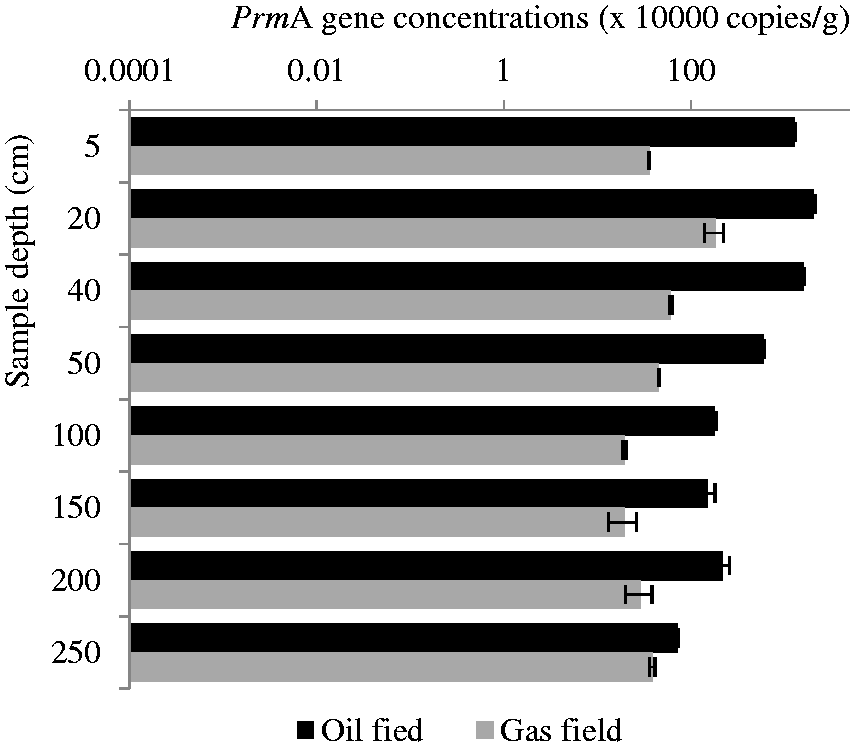
The ratio of the
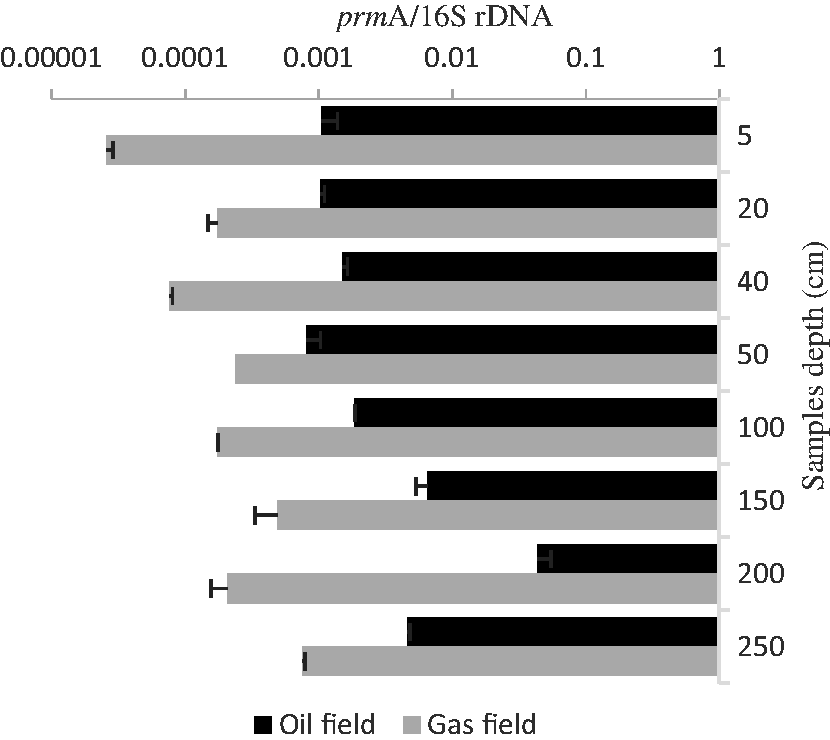
The ratio of
Discussion
The site selected in the Yangxin oil field is located above an anticlinal structural reservoir, which is an anticlinal structural trap that was formed by the gravitational effects and the differential compaction of Paleogene Shahejie formation sediments that were deposited above the early formed horst. The trap has the advantage of allowing the easy transportation, aggregation, and preservation of the oil and gas around it. The underground oil reservoir is 1390–2570 m beneath the surface. Furthermore, mature oil is predominantly distributed in the oil reservoir in this area (Yang et al., 2003). Mature oil has the characteristics of low density and viscosity, which means that the short-chain hydrocarbon content is high and light hydrocarbon gas can easily escape into the surface soils. Therefore, we detected relatively high concentrations of the
The gas field from which the samples were collected is called the Xiaoquan-Xinchang gas field, which is composed of multiple secondary gas reservoirs in the Jurassic system. The Xiaoquan-Xinchang structure is an anticline, which formed a northeast-east-striking Palaeohigh belt. This belt is the migration zone for natural gas in the subsidence formation. Moreover, there are good conditions for the formation of structural-lithologic composite traps, producing the optimal conditions for the formation of a productive gas field in the area (Ren and Xia, 2006). From the field sample, we detected high concentrations of propane according to the headspace gas chromatography of soil samples (Table 2). However, the gas field had lower concentrations of the
The ratio of functional gene to 16S rRNA genes might solve the problem. 16S rRNA genes are present in all bacteria, while the
The geological background of the nonoil gas field at the site indicated that there were no oil or gas reserves and covering strata. As a result, all samples in the 2.5 m profile collected in this area, except for the 5 cm deep soil, had no
As indicated above, it is important to determine the optimal sampling depth for prospecting oil and gas reservoirs. If the sampling depth is too shallow, numerous factors such as human activities could influence the result. However, if the depth is too deep, sampling would become a laborious process. Furthermore, as shown in Figure 6,
To avoid the disturbance of human and plant activities as much as possible, we should not select depths above the 50 cm level, as these soils would be included within the plough and cultivated layer and might further be contaminated by organic pesticide and fertilizers and root exudations (Hutsch, 1998; Seghers et al., 2005; Stiehl-Braun et al., 2011). Therefore, to minimize the economic and resource costs of sampling work and prevent
When this newly established technique is used with samples at 50 cm depth to quantify propane-oxidizing bacteria in oil or gas fields, the
To investigate a specific field, the presence of an underlying gas or oil reservoir based on the presence of the
The RT-PCR technique could get the more accurate results than the geochemical data. For example, the geochemical data showed that there was no propane in the soil particles sampled in Xiliu field (Table 2), but there is an oil reservoir, while the
Conclusion
During this study, we established a robust method based on the real-time fluorescent quantitative PCR detection technology to detect the abundance of propane-oxidizing bacteria in oil and gas field soil. We considered 50 cm to be the optimal sampling depth for microbial prospecting of oil and gas reservoirs. This method can easily distinguish nonoil gas fields from those potentially containing oil or gas reservoirs.
Since these microorganisms are sensitive to hydrocarbons, the established PCR technique might detect the abnormal presence of these microorganisms in the field when gaseous hydrocarbon concentrations are below the detection limit. Compared with traditional culture-dependent microbial approaches, this technique has the advantage of saving labor, materials, and time and has more accurate results. Although we cannot identify whether the field is an oil field or a gas field through this procedure alone, the combination of the RT-PCR technique with geochemical and geophysical data could help us to do this. Therefore, the
Supplemental Material
Supplemental Material: Figures
Supplementary Figures, supplementary_material for Development of a
Footnotes
Acknowledgments
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by China Geological Survey project (No. 1212011120268) and the National Natural Science Foundation of China (No. 41402233).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
