Abstract
Vitamin K antagonists, such as warfarin, have been the mainstay in treatment and prophylaxis of venous thromboembolism. However, warfarin has many disadvantages including a narrow therapeutic window, numerous potential drug interactions, modulation of effect by alcohol and foods containing vitamin K and genetic variation in metabolism of warfarin, all of which contribute to the unpredictability of therapy. This has provided the impetus for developing new oral anticoagulants with a rapid onset of action, wide therapeutic window, predictable and reversible action, with few drug or dietary interactions, no requirement for routine coagulation monitoring or dose adjustment and acceptable cost. No single agent incorporates all these characteristics, but new factor Xa and direct thrombin inhibitors are being introduced into clinical practice that fulfil some of these aims. Here, we briefly discuss the current practice with its limitations and pitfalls, and then review important trials that have launched new oral anticoagulants into clinical practice.
Keywords
Introduction: the clinical problem, current therapy and its limitations
Thrombus originating in the veins of the lower limbs or pelvic circulation, termed deep vein thrombosis (DVT), may propagate to the lungs as pulmonary embolism (PE). PE is common, often asymptomatic, sometimes misdiagnosed and a leading cause of mortality.
1
In Europe, venous thromboembolism (VTE) accounts for 12% of deaths annually,
2
and is the third most common cause of vascular death after myocardial infarction and stroke. Virchow's triad of venous stasis, hypercoaguability and endothelial damage to the blood vessel wall represent predisposing factors for developing DVT,
3
and the causes can be categorized under these headings (Figure 1).
Risk factors for venous thromboembolism.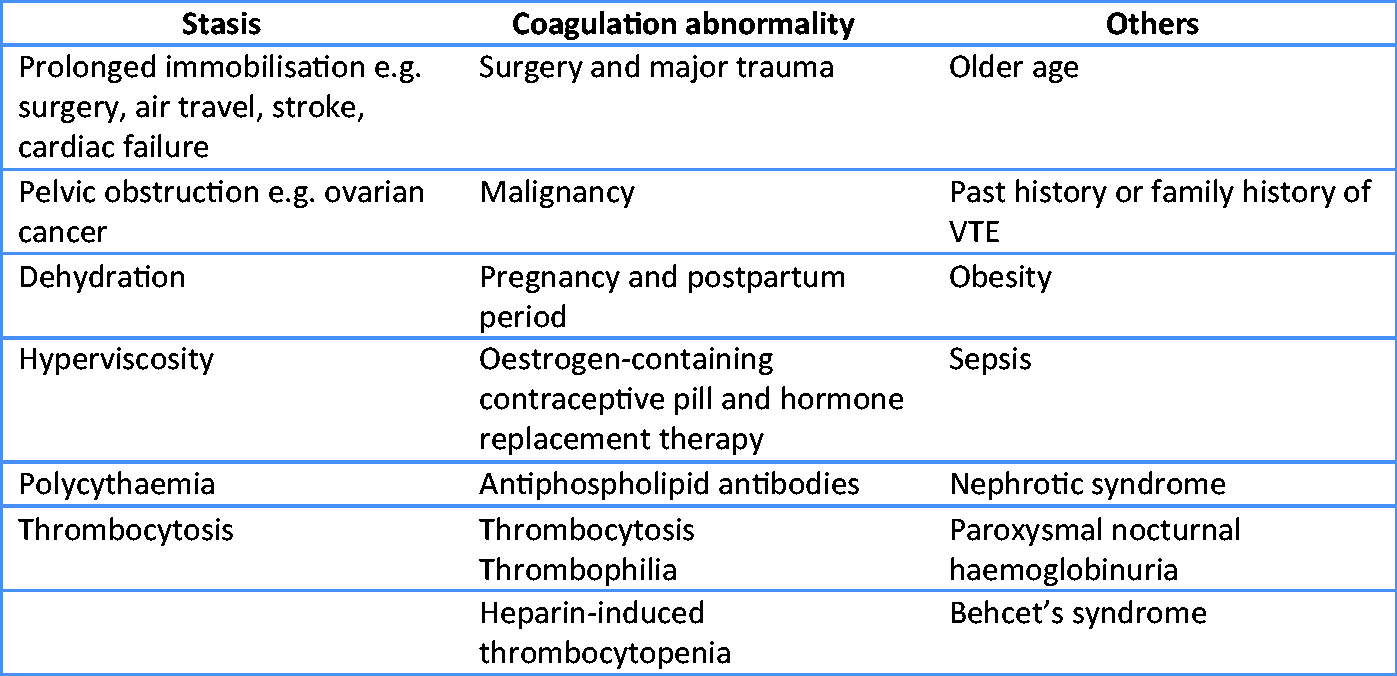
VTE is especially common during and after hospitalization for surgery and acute medical illness, with the elderly at greatest risk.4,5 Without thromboprophylaxis, sensitive radiolabelling studies reveal evidence of subclinical DVT occurring in up to 60% of patients undergoing orthopaedic surgery, 15–40% undergoing major general surgery, and is a complication in as many as 20% of patients on a general medical ward. 4 Under-utilization of VTE prophylaxis among hospitalized patients is a global problem. 5
In 2005, the UK Department of Health reported that 25,000 people were dying from preventable hospital-acquired VTE every year, and that nationally, 70% of individuals considered to be at risk of developing DVT did not receive thromboprophylaxis. 6 In 2007, 16,670 deaths were recorded in England and Wales where PE and DVT were mentioned on the death certificate. 7 The overall death rate from VTE in hospital and the community was likely to have been significantly higher given that the condition is often clinically silent and the diagnosis missed due to the limits on postmortem examinations being undertaken nationally. Preventing VTE is a major goal for the NHS to save lives and avoid chronic disability due to post-thrombotic syndrome (chronic venous insufficiency, venous leg ulcers and pulmonary hypertension).
Historically, the prevention and initial treatment of VTE is based on the administration of rapidly acting parenteral anticoagulants, such as unfractionated heparin, with transition to oral coumarin therapy, usually warfarin, after adequate concurrent use of the two anticoagulants. 4 The availability of low-molecular weight heparin (LMWH) and fondaparinux has been a major step forward. Unlike heparin, these agents can be given subcutaneously and without coagulation monitoring.8,9 Furthermore, the incidence of heparin-induced thrombocytopenia is much lower with LMWH than with unfractionated heparin, and absent with fondaparinux.
The vitamin K antagonist, warfarin, has been a cornerstone of VTE treatment 10 and stroke prevention in patients with atrial fibrillation. 11 It is relatively safe, well tolerated and, in general, has an excellent safety record. Its anticoagulant effects can be reversed with vitamin K, and in major bleeding, by the administration of fresh frozen plasma (FFP) and/or human prothrombin complex. However, the drawback of warfarin is its slow onset of action of three to five days necessitating an overlap with a parenteral anticoagulant, and a narrow therapeutic window, which increases the risk of bleeding with over-anticoagulation, and risk of thrombosis with under-anticoagulation. Other issues include numerous drug interactions, modulation of effect by alcohol and foods containing vitamin K, and genetic variation in metabolism of warfarin, all of which contribute to the unpredictability of therapy. 10 Frequent and expensive monitoring of prothrombin time translated into the International Normalized Ratio (INR) is needed when prescribing warfarin.
Methods
The authors agreed upon the scope and extent of the article. A search was conducted using Medline and PubMed to identify current and relevant papers to gather evidence for this review.
New orally active anticoagulants: direct factor Xa and direct thrombin inhibitors
The unpredictable pharmacokinetics and pharmacodynamics of warfarin has been the stimulus for the development of new oral anticoagulants with a rapid onset of action, wide therapeutic window, predictable and reversible action, with few drug or dietary interactions, no requirement for routine coagulation monitoring or dose adjustment and acceptable cost. 9 No single agent incorporates all these characteristics, but the new factor Xa and direct thrombin inhibitors fulfil some of these aims. The rationale for developing factor Xa and thrombin inhibitors is that they are common to both the intrinsic and extrinsic pathways of coagulation. Thrombin not only converts fibrinogen to fibrin but also activates platelets. Thrombin inhibition both attenuates fibrin formation and inhibits platelet activation. 12 Some consider that factor Xa inhibition has greater potential because of a wider therapeutic window. 13
Many candidate orally-active anticoagulants are being investigated in clinical trials (Figure 2), but this discussion will be limited to those with the greatest available clinical data and are most likely to be established early in clinical practice. These are the direct thrombin inhibitor, dabigatran etexilate and the factor Xa inhibitors, rivaroxaban and idrabiotaparinux, none of which require routine coagulation monitoring for efficacy.
New anticoagulants inhibiting thrombin and factor Xa.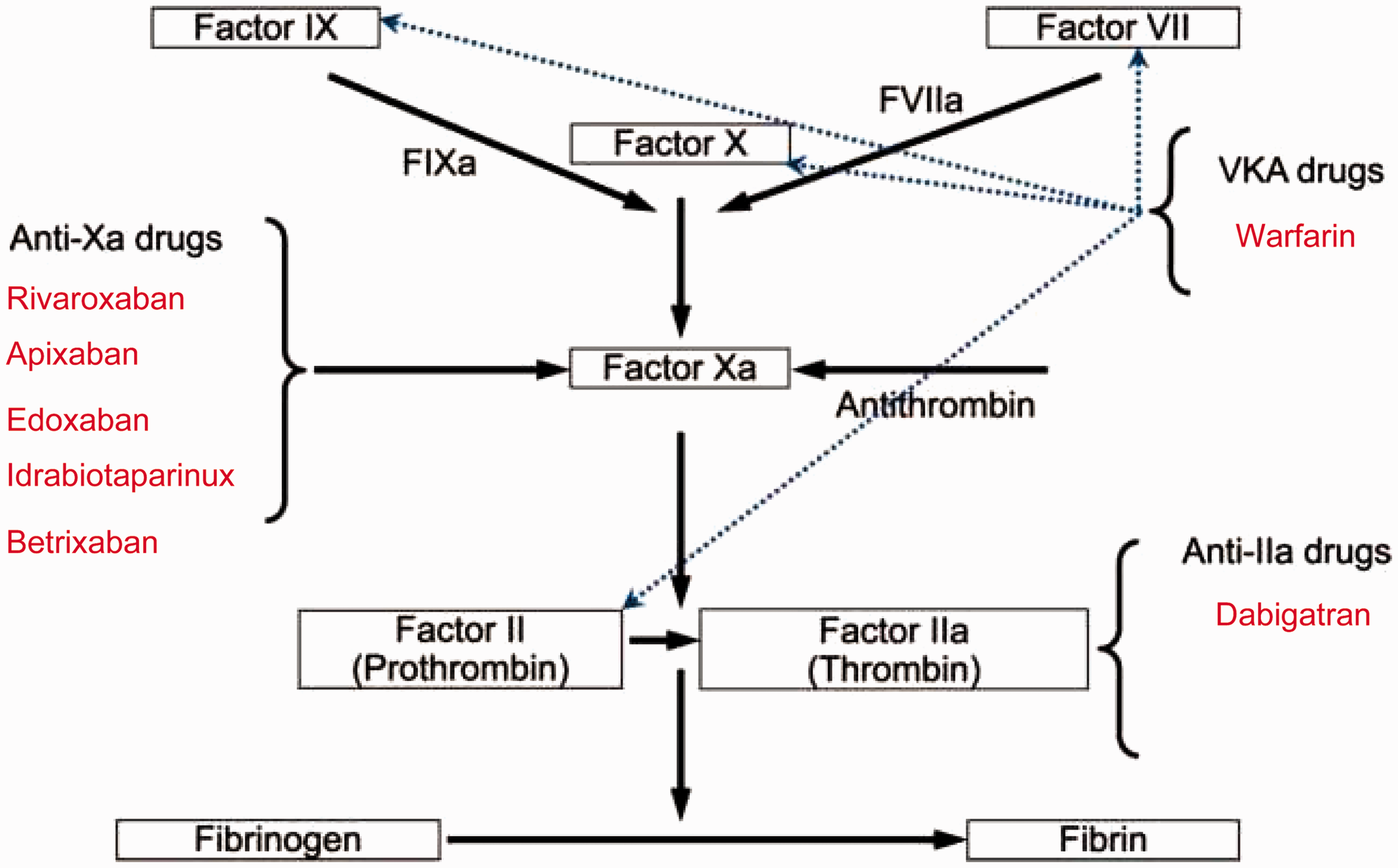
Dabigatran etexilate, a reversible direct thrombin inhibitor, is converted to the active drug, dabigatran, with a half-life of 14–17 h. Peak plasma concentrations are reached within 0.5 and 2 h of oral administration, and 80% is eliminated by the kidneys.8,13 A meta-analysis of two large international trials exploring the efficacy of dabigatran versus enoxaparin in preventing VTE after major orthopaedic surgery showed that dabigatran given in doses of 150 mg and 220 mg once daily was not inferior to enoxaparin 40 mg daily. 14 A randomized, double-blind, non-inferiority trial in patients with acute VTE (the RECOVER trial) compared dabigatran and warfarin. 15 Patients were randomized to either dabigatran 150 mg twice daily or warfarin to target INR (two to three). The six-month incidence of recurrent VTE was similar, 2.4% and 2.1%, respectively, and dabigatran was non-inferior to warfarin with respect to thrombotic deaths. Although there was no statistically significant difference in bleeding complications, the data trend favoured dabigatran. In this study, patients in both groups were initially treated with a parenteral anticoagulant for five days. The prospect of utilizing dabigatran without a parenteral anticoagulant would be a significant advance and would simplify the treatment of acute VTE. The speed of action of the thrombin inhibitor is no different to that of the LMWH.
Rivaroxaban is an orally active direct factor Xa inhibitor which is rapidly and almost completely absorbed, with plasma concentration peaking at 2–3 h after oral intake and the terminal half-life is 7–11 h. 8 One-third of the unchanged drug is eliminated by the kidneys, one-third is metabolized in liver via CYP3A4-dependent pathways prior to excretion in faeces. The remainder is metabolized in liver and then renally excreted. Potent inhibitors of CYP3A4 such as ketoconazole and the anti-HIV drug, ritanovir, should not therefore be co-prescribed with rivaroxaban. 8 The superiority of rivaroxaban over enoxaparin in a series of international trials examining VTE prevention after major orthopaedic surgery has led to its widespread use. 16
In the treatment of acute DVT, the EINSTEIN investigators compared oral rivaroxaban with standard sequential anticoagulation (enoxaparin followed by warfarin) over three, six or 12 months. 17 Rivaroxaban was non-inferior to the standard anticoagulation regimen for the prevention of recurrent DVT (2.1% vs. 3% respectively, P < 0.001). Major bleeding occurred in 0.8 and 1% of patients given rivaroxaban and the conventional regimen, respectively. All-cause mortality did not differ between the two groups, 2.2% (rivaroxaban) versus 2.9%. In the extended 12-month study, rivaroxaban produced an 82% reduction in the risk of recurrent VTE compared to placebo. Finally, in a large randomized trial, patients with acute PE (with or without DVT), were treated with rivaroxaban (15 mg twice daily for three weeks, followed by 20 mg once daily) without receiving LMWH initially. These were compared with those treated with conventional therapy of enoxaparin overlapping with warfarin for the first few days of treatment. 18 In this study spanning between three and 12 months, rivaroxaban was found to be as efficacious and safe as conventional therapy. The incidence of symptomatic recurrent VTE was not significantly different in the two groups, occurring in 2.1% of patients treated with rivaroxaban compared with 1.8% (conventional therapy). Major bleeding was observed in 1.1% of patients in the rivaroxaban group and 2.2% in the standard-therapy group, which was statistically significant (P = 0.003), favouring rivaroxaban over conventional therapy.
In acute DVT, three to six month duration of anticoagulation is recommended, 10 but Prandoni et al. 19 reported that the risk of recurrent VTE after stopping anticoagulation in patients with proximal DVT or PE was 11%, 20%, 30% and 40% at one, three, five and 10 years, respectively. Rivaroxaban may offer a safe method of providing thromboprophylaxis into the long term and may reduce the risk of developing complications such as post-thrombotic syndrome and pulmonary hypertension. Long-term trials are needed to confirm this.
Idrabiotaparinux is a derivative of fondaparinux, and binds antithrombin with high affinity. It has a half-life of 130 h. 20 This permits once-weekly subcutaneous administration. The advantage of idrabiotaparinux over idraparinux is that it contains a biotin moiety which permits rapid reversal of anticoagulant action with intravenous avidin as the biotin–avidin complex terminates the drug action. 20 In a multi-centre trial, patients with an objectively confirmed PE received 5–10 days treatment with enoxaparin before being randomly allocated to treatment with idrabiotaparinux given once weekly or to the group treated conventionally with warfarin for three to six months. 21 Outcome was assessed at 99 days after randomization. Recurrent VTE was determined in 2% allocated to receive idrabiotaparinux compared to 3% treated conventionally, confirming non-inferiority of the new treatment (p for non-inferiority = 0.0001). Clinically relevant bleeding was significantly less in those given idrabiotaparinux compared to the warfarin treated group (5% vs. 7%, p = 0.0098). Similar differences in outcome were observed at six months. Idrabiotaparinux may provide an attractive alternative to warfarin for the long-term treatment of PE.
Options for reversing the effect of the new directly acting oral anticoagulants
The effects of direct thrombin and factor Xa inhibitors on clotting factors have been reviewed by the British Committee for Standards in Haematology,
22
and are tabulated in Figure 3.
Pattern of prolongation of coagulation assays by directly acting oral anticoagulants.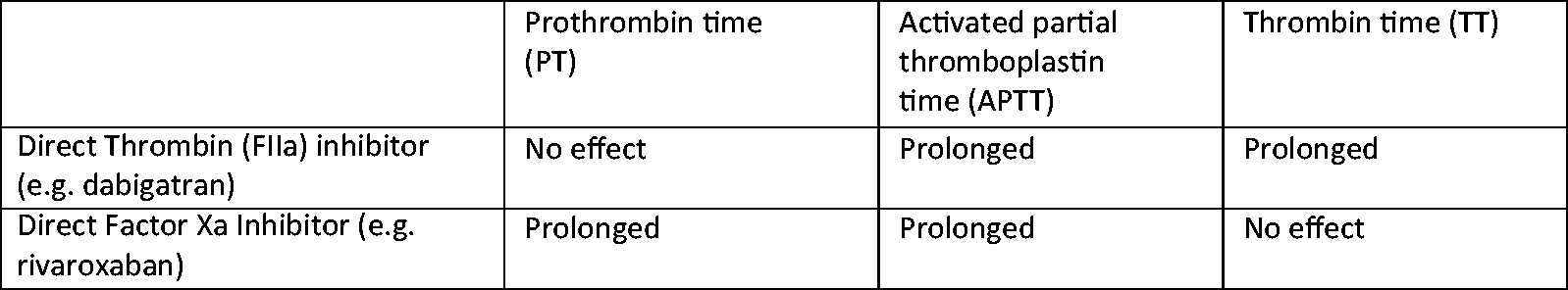
In contrast to warfarin, there are currently no specific antidotes for reversing the anticoagulant effect of these new agents.9,23 Whereas fresh frozen plasma (FFP) has been central for urgent reversal of potentially life-threatening bleeding due to the anticoagulant effects of warfarin, it has no reversal effect on either class of directly acting anticoagulant.23–25 In due course, it is anticipated that the pharmaceutical industry will develop specific antidote technologies. This may take the form of either small inert blocking molecules or perhaps use of antibody-based or strept-avidin based means of drug effect reversal, as historically deployed to neutralize potent drugs such as digoxin. 24 These developments may take several years to enter routine clinical practice.
Until drug-specific or class-specific antidotes are developed, the options for urgent reversal rely upon: (i) physical compression of the bleeding site or the achievement of tamponade, (ii) awaiting the spontaneous decay in activity of the shorter half-life direct coagulation inhibitors, (iii) haemodialysis, or (iv) the use of activated or non-activated human coagulation factor concentrates. 24 However, there is concern over the use of activated coagulation factor concentrates because they may carry a significant risk of paradoxical thrombosis, particularly in individuals with underlying thrombotic predisposition.26,27
None of the available human coagulation factor concentrates are licensed in Europe specifically for the emergency reversal of the new oral directly acting coagulation inhibitors. Their deployment in this setting is at present an ‘off-label’ use. The available coagulation factor concentrates are activated prothrombin-complex concentrate (APCC) as FEIBA (Baxter Healthcare), recombinant factor VIIa (rFVIIa, Novoseven, Novo Nordisk), and prothrombin complex concentrates (PCCs). Two PCCs that are licensed for coumarin anticoagulant reversal in Europe are Beriplex (CSL Behring) and Octaplex (Octapharma).
FEIBA (Factor Eight Inhibitor Bypassing Activity) contains coagulation factors II, IX and X, mainly in the non-activated state, together with activated plasma-derived factor VII(a). It is licensed for use in the management of bleeding in Haemophilia A patients with acquired auto-antibodies (inhibitors) to native factor VIII. In animals treated with higher dose dabigatran, the bleeding time was significantly reduced by administration of FEIBA at doses of 50 or 100 U/kg, with pre-FEIBA bleeding times of 1455 s reduced to 186 s post-FEIBA. FEIBA had no effect on shortening the prolonged APTT.24,25
Recombinant factor VIIa was used in the same animal model and a similar shortening of dabigatran-induced bleeding time was observed with rFVIIa at doses of 0.5 or 1 mg/kg. 25 Recombinant FVIIa also reduced the prolonged APTT from 58.8 s to about 30 s, in contrast to the lack of effect of FEIBA.
Prothrombin complex concentrates (PCC) are available as Beriplex and Octaplex. Beriplex PCC contains three largely non-activated plasma-derived coagulation factors, such as factors II, IX and X together with Protein C and Protein S. Octaplex PCC additionally contains largely non-activated factor VII forming a ‘4-factor’ PCC. In a study of normal human volunteers treated with therapeutic dose dabigatran, there was neither in vivo nor in vitro reversal of the anticoagulant drug effect using a 4-factor PCC given at a dose of 50 iU/kg. 25 The same authors repeated the study with volunteers receiving therapeutic dose rivaroxaban and reported both a significant in vivo and in vitro attenuation of the anticoagulant drug effect using the same dosing of 4-factor PCC.
Final remarks and conclusions
If the current clinical trials and long-term data of the new orally active thrombin and factor Xa inhibitors establish their safety and efficacy in thromboprophylaxis, in treatment of VTE and prevention of stroke in atrial fibrillation, then the future role of vitamin K antagonists, such as warfarin, seem likely to steadily diminish. The new oral anticoagulants offer fixed dosing without routine monitoring of coagulation, fewer opportunities for significant drug interactions and rapid onset of action. Like warfarin, these agents cannot be used in pregnancy. Dose adjustment is necessary in mild renal impairment, but they are contraindicated if the creatinine clearance falls below 15 mL/min.
The initial drug cost may be greater than for warfarin, but this may be offset by fewer patients needing hospital admission and the costs of warfarin monitoring. The annual cost of the novel anticoagulants is approximately £800 compared to between £115 and £415 per patient in those maintained on warfarin (including INR monitoring).28,29 Treatment can be initiated in the community prior to confirmation of VTE and lengthy inpatient stay pending adequacy of anticoagulation with warfarin, avoided. The issues of the emergency reversal of these new agents need addressing.
