Abstract
Objective
Several trials had compared the efficacy and safety between non-vitamin K antagonist oral anticoagulants and warfarin for acute venous thromboembolism, but the results were incomplete. This updated review comprehensively assessed the efficacy and safety of non-vitamin K antagonist oral anticoagulants for venous thromboembolism.
Design
Meta-analysis of randomised control trials. Six databases were searched from January 2000 to December 2018.
Setting
Adult patients had got non-vitamin K antagonist oral anticoagulants or warfarin for venous thromboembolism.
Participants
Randomised control trials that compared the efficacy and safety between non-vitamin K antagonist oral anticoagulants and warfarin.
Main outcome measures
The efficacy and safety of non-vitamin K antagonist oral anticoagulants .
Results
Seven studies involving 29,879 cases were included, among which 14,943 cases were assigned to non-vitamin K antagonist oral anticoagulants group and 14,936 cases to warfarin group. Meta-analysis showed that compared with warfarin, recurrent venous thromboembolism (odds ratio 0.94 [95% confidence interval 0.81 to 1.11]), death related to venous thromboembolism or fatal pulmonary embolism (odds ratio 1.00 [95% confidence interval 0.63 to 1.60]), symptomatic deep-vein thrombosis (odds ratio 0.88 [95% confidence interval 0.72 to 1.09]), symptomatic nonfatal pulmonary embolism (odds ratio 1.03 [(95% confidence interval 0.82 to 1.30]) and all deaths (odds ratio 0.92 [95% confidence interval 0.76 to 1.12]) are similar in non-vitamin K antagonist oral anticoagulants group, but major bleeding event (odds ratio 0.61 [95% confidence interval 0.50 to 0.75]) and clinically relevant non-major bleeding event (odds ratio [95% confidence interval 0.53 to 0.85]) are less in non-vitamin K antagonist oral anticoagulants group.
Conclusions
For the treatment of venous thromboembolism, non-vitamin K antagonist oral anticoagulants is as effective as warfarin, and has a better safety profile than warfarin.
Keywords
Introduction
Acute venous thromboembolism, including deep vein thrombosis and pulmonary embolism, is associated with substantial morbidity and mortality. 1 In the past decades, warfarin and other vitamin K antagonists had been the primary therapy for patients with venous thromboembolism. Although vitamin K antagonists are cheap, they have a narrow therapeutic window and require frequent monitoring, they also have many interactions with food and drugs, which can result in poor adherence. 2
Alternatively, there are non-vitamin K antagonist oral anticoagulants that have been approved by the US Food and Drug Administration (FDA) for use in venous thromboembolism, and their use has increased substantially. non–vitamin K antagonist oral anticoagulants do not require laboratory monitoring and have fewer food–drug interactions. 3 However, there are some concerns about non–vitamin K antagonist oral anticoagulants, including poor adherence in the absence of monitoring, more cost (three times more expensive than warfarin even when including laboratory monitoring), bleeding risk and current almost absence of a specific antidote.
There are several randomised controlled trials which compared the efficacy and safety between non–vitamin K antagonist oral anticoagulants and warfarin, and showed similar or non-inferiority effect and similar or superior safety of non–vitamin K antagonist oral anticoagulants in treatment of venous thromboembolism. Several meta-analyses have evaluated the efficacy and safety of non–vitamin K antagonist oral anticoagulants compared with vitamin K antagonists in venous thromboembolism, but only showed part results of the data about efficacy and safety.4–7 In the present study, we performed a meta-analysis to comprehensively assess the efficacy (recurrent venous thromboembolism, venous thromboembolism related death or fatal pulmonary embolism, symptomatic deep vein thrombosis, symptomatic nonfatal pulmonary embolism) and safety (all death, major bleeding event and clinically relevant bleeding event) of non–vitamin K antagonist oral anticoagulants in venous thromboembolism treatment.
Methods
PICOS identifiers from research questions (key terms) and database- and thesaurus-derived alternatives (additional terms) used to generate database searches.

Data source and searches
We searched six databases and the reference lists of retrieved reports from January 2000 to December 2018 for studies of efficacy and safety of non–vitamin K antagonist oral anticoagulants versus warfarin in treatment of patients with venous thromboembolism using the terms identified by PICOS (Table 1).
Study selection
Two investigators (Y.Z and L.F.D) independently screened all titles and abstracts to identify studies that examined the efficacy and safety of non–vitamin K antagonist oral anticoagulants in patients with venous thromboembolism. Only reports in English were included in this study. Studies were excluded if the research met any one of the following criteria: (1) the efficacy and safety of non–vitamin K antagonist oral anticoagulants versus warfarin were not reported, (2) publication only as an abstract and (3) duplicate publication or ongoing/unpublished study.
Data extraction
Two reviewers (Y.Z and L.F.D) extracted relevant data from the included studies using a standardised data extraction form. Randomised studies with follow-up duration at least three months were considered for inclusion. Primary outcome measures were recurrent venous thromboembolism, venous thromboembolism-related death or fatal pulmonary embolism, symptomatic deep vein thrombosis, symptomatic nonfatal pulmonary embolism, all death, major bleeding event and clinically relevant bleeding event. Studies reporting efficacy and safety of non–vitamin K antagonist oral anticoagulants on the basis of different drugs were analysed separately, and the total efficacy and safety for all studies (dabigatran, rivaroxaban, endoxaban and apixban) were also analysed.
Quality assessment
The quality of included studies was assessed by Cochrane Collaboration Tool, which consisted of seven sections as follows: (1) random sequence generation, (2) allocation concealment, (3) blinding of participants and personnel, (4) blinding of outcome assessment, (5) incomplete outcome data, (6) selective reporting and (7) other biases.
Data analysis
Statistical analyses were performed using RevMan version 5.3 (The Cochrane Collaboration, Oxford, England), and the results are expressed as odds ratio for dichotomous outcomes, with 95% confidence intervals. We calculated the I2 statistic to assess the heterogeneity across the trials, and a value greater than 50% was considered substantial heterogeneity then data were pooled using the random-effects model. The efficacy of non–vitamin K antagonist oral anticoagulants in this meta-analysis was assessed using RCTs which were designed as non-inferiority studies with associated non-inferiority margins used to interpret the comparison results, so we also try to use non-inferiority margins to interpret the meta-analysis results. The noninferiority margins were estimated from studies which evaluated the efficacy of warfarin as compared with no anticoagulation. If the upper boundary of 95% confidence interval for the pooled odds ratio was within reasonable non-inferiority margin, the result may be interpreted as ‘similar efficacy’. We conducted sensitivity analyses by comparing the outcomes using the fixed- versus random-effects model. Publication bias was explored by visual inspection of a funnel plot. p < 0.05 was used for statistical significance.
Results
We identified seven studies8–14 (enrolling 29,879 patients, among which 14,943 cases were assigned to the treatment group and 14,936 cases to the control group) that reported the effects and safety of non–vitamin K antagonist oral anticoagulants in patients with venous thromboembolism. Reports evaluated for inclusion in meta-analysis are shown in Figure 1.
Reports evaluated for inclusion in meta-analysis.
Study characteristics
Characteristics of studies included in the meta-analysis.
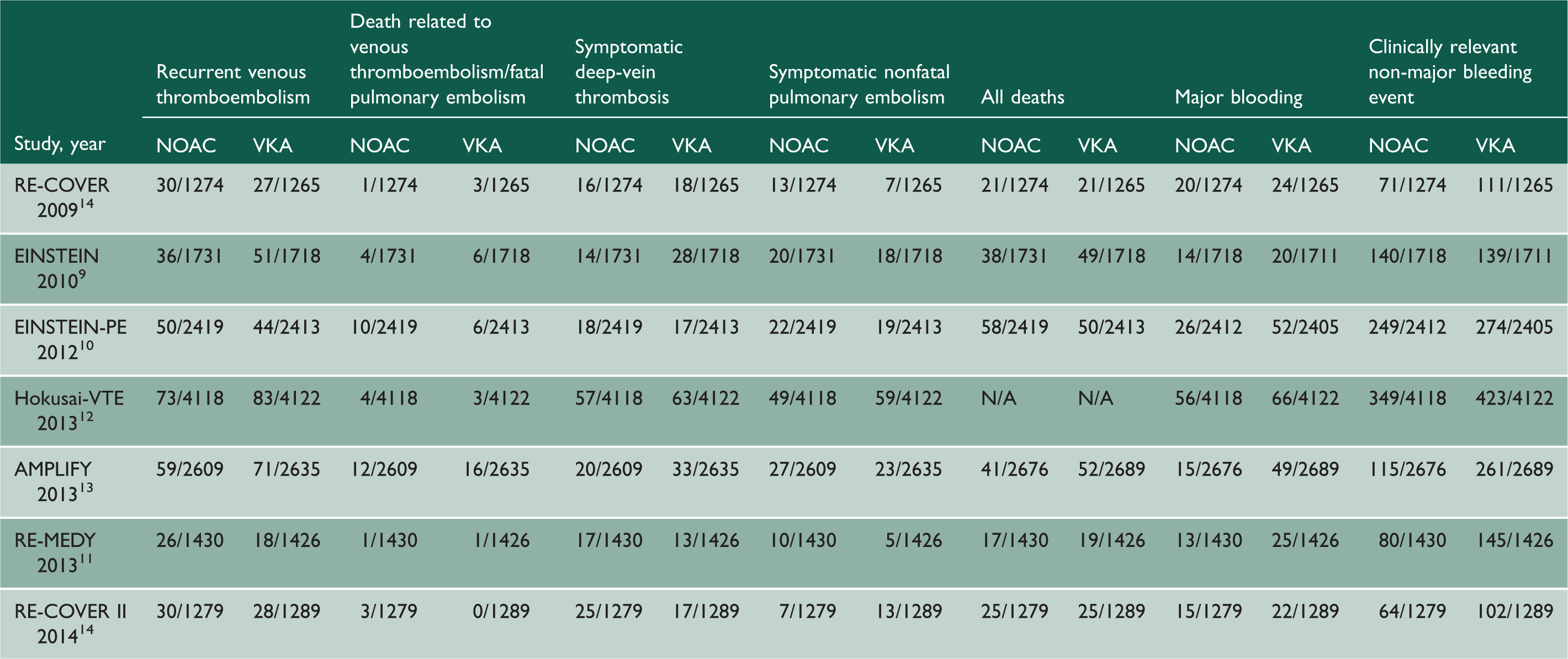
The primary outcomes of included studies.

Risk of bias assessment
The details on risk for bias are shown in Figure 2. Seven studies8–14 were judged to be at low risk for bias in the random sequence generation, allocation concealment, blinding of outcome assessment, incomplete outcome data and selective reporting. Five studies8,11–14 were judged to have low risk for bias in blinding of participants and personnel. Three studies8–10 had unclear risk of other bias.
Risk of bias summary of all included studies.
Non-inferiority margin assessment
In order to assess non-inferiority margin to interpret the meta-analysis results, we performed a meta-analysis to assess efficacy of warfarin as compared with no anticoagulation in four studies.15–18 The results are expressed as odds ratio with 95% confidence intervals. Pooled analysis showed that warfarin could decrease the rate of recurrent venous thromboembolism than no anticoagulation, and the difference was significant (odds ratio 0.55 [95%confidence interval, 0.39 to 0.76], p = 0.0004). The noninferiority margin was estimated to correspond to preservation 50% (for assessment of odds ratio) of the lower boundary of the 95% confidence interval for the efficacy of warfarin as compared with no anticoagulation, so the assessed noninferiority margin of odds ratio was 1.14.
Outcomes
Efficacy outcomes
Recurrent venous thromboembolism
Seven studies8–14 that included 14,860 patients from non–vitamin K antagonist oral anticoagulants group and 14,868 patients from warfarin group reported recurrent venous thromboembolism. Pooled analysis showed that there was no significant difference in recurrent venous thromboembolism between non–vitamin K antagonist oral anticoagulants and warfarin groups in the fixed-effects model (odds ratio 0.94 [95%confidence interval, 0.81 to 1.11], p = 0.47). The upper boundary of 95% confidence interval was within the non-inferiority margin, which indicated that the non–vitamin K antagonist oral anticoagulants were noninferior with regard to the prevention of recurrent venous thromboembolism to warfarin. There was no substantial heterogeneity among the studies (I2 = 0%, p = 0.43). When analysed on the basis of different types of non–vitamin K antagonist oral anticoagulants, there were no significant differences in recurrent venous thromboembolism in dabigatran and rivaroxaban treatment trials (Figure 3).
Forest plot of comparison of recurrent venous thromboembolism between NOACs and warfarin.
Venous thromboembolism-related death/ fatal pulmonary embolism
Seven studies8–14 that included 14,860 patients from non–vitamin K antagonist oral anticoagulants group and 14,868 patients from warfarin group reported venous thromboembolism-related death or fatal pulmonary embolism. Pooled analysis showed that there was no significant difference in venous thromboembolism-related death or fatal pulmonary embolism between non–vitamin K antagonist oral anticoagulants and warfarin groups in the fixed-effects model (odds ratio 1.00 [95%confidence interval 0.63 to 1.60], p = 0.99). The upper boundary of 95% confidence interval was beyond non-inferiority margin, which indicated that the non–vitamin K antagonist oral anticoagulants were not similar with regard to the prevention of venous thromboembolism-related death or fatal pulmonary embolism to warfarin. There was no substantial heterogeneity among the studies (I2 = 0%, p = 0.59). When analysed on the basis of different types of non–vitamin K antagonist oral anticoagulants, there were no significant difference in venous thromboembolism-related death or fatal pulmonary embolism in dabigatran and rivaroxaban treatment trials (Figure 4).
Forest plot of comparison of death related to venous thromboembolism between NOACs and warfarin.
Symptomatic deep-vein thrombosis
Seven studies8–14 that included 14,860 patients from non–vitamin K antagonist oral anticoagulants group and 14,868 patients from warfarin group reported symptomatic deep vein thrombosis. Pooled analysis showed that there was no significant difference in symptomatic deep vein thrombosis between non–vitamin K antagonist oral anticoagulants and warfarin groups in the fixed-effects model (odds ratio 0.88 [95%confidence interval 0.72 to 1.09], p = 0.24). The upper boundary of 95% confidence interval was within non-inferiority margin, which indicated that the non–vitamin K antagonist oral anticoagulants were noninferior with regard to the prevention of symptomatic deep vein thrombosis to warfarin. There was no substantial heterogeneity among the studies (I2 = 33.4%, p = 0.17). When analysed on the basis of different types of non–vitamin K antagonist oral anticoagulants, there were no significant differences in symptomatic deep vein thrombosis in dabigatran and rivaroxaban treatment trials (Figure 5).
Forest plot of comparison of symptomatic deep-vein thrombosis between NOACs and warfarin.
Symptomatic nonfatal pulmonary embolism
Seven studies8–14 that included 14,860 patients from non–vitamin K antagonist oral anticoagulants group and 14,868 patients from warfarin group reported symptomatic nonfatal pulmonary embolism. Pooled analysis showed that there was no significant difference in symptomatic nonfatal pulmonary embolism between non–vitamin K antagonist oral anticoagulants and warfarin groups in the fixed-effects model (odds ratio 1.03 [95%confidence interval 0.82 to 1.30], p = 0.81). The upper boundary of 95% confidence interval was beyond non-inferiority margin, which indicated that the non–vitamin K antagonist oral anticoagulants were not similar with regard to the prevention of symptomatic nonfatal pulmonary embolism to warfarin. There was no substantial heterogeneity among the studies (I2 = 8.7%, p = 0.36). When analysed on the basis of different types of non–vitamin K antagonist oral anticoagulants, there were no significant differences in symptomatic nonfatal pulmonary embolism in dabigatran and rivaroxaban treatment trials (Figure 6).
Forest plot of comparison of symptomatic nonfatal pulmonary embolism between NOACs and warfarin.
Safety outcomes
All deaths
Six studies8–11,13,14 that included 10,809 patients from non–vitamin K antagonist oral anticoagulants group and 10,800 patients from warfarin group reported all death. Pooled analysis showed that there was no significant difference in all death between non–vitamin K antagonist oral anticoagulants and warfarin groups in the fixed-effects model (odds ratio 0.92 [95%confidence interval 0.76 to 1.12], p = 0.42). Since the upper boundary of 95% confidence interval was within non-inferiority margin, the indicated non–vitamin K antagonist oral anticoagulants were noninferior with regard to the prevention of all death to warfarin. There was no substantial heterogeneity among the studies (I2 = 0%, p = 0.73). When analysed on the basis of different types of non–vitamin K antagonist oral anticoagulants, there were no significant differences in all death in dabigatran and rivaroxaban treatment trials (Figure 7).
Forest plot of comparison of all deaths between NOACs and warfarin.
Major bleeding event
Seven studies8–14 that included 14,907 patients from non–vitamin K antagonist oral anticoagulants group and 14,907 patients from warfarin group reported major bleeding event. Pooled analysis showed that there was significant decrease in major bleeding event in non–vitamin K antagonist oral anticoagulants group in the fixed-effects model (odds ratio 0.61 [95%confidence interval 0.50 to 0.75], p < 0.00001). There was no substantial heterogeneity among the studies (I2 = 45.4%, p = 0.09). When analysed on the basis of different types of non–vitamin K antagonist oral anticoagulants, there were significant decreases in major bleeding event in dabigatran and rivaroxaban treatment trials (Figure 8).
Forest plot of comparison of major bleeding event between NOACs and warfarin.
Clinically relevant non-major bleeding event
Seven studies8–14 that included 14,907 patients from non–vitamin K antagonist oral anticoagulants group and 14,907 patients from warfarin group reported clinically relevant non-major bleeding event. Pooled analysis showed that there was significant decrease in clinically relevant non-major bleeding event in non–vitamin K antagonist oral anticoagulants group in the random-effects model (odds ratio 0.67 [95%confidence interval 0.53 to 0.85], p = 0.001). There was substantial heterogeneity among the studies (I2 = 86.4%, p < 0.00001). When analysed on the basis of different types of non–vitamin K antagonist oral anticoagulants, there were significant decreases in clinically relevant non-major bleeding event in dabigatran treatment trials, but no significant difference in rivaroxaban treatment trials (Figure 9).
Forest plot of comparison of clinically relevant non-major bleeding event between NOACs and warfarin.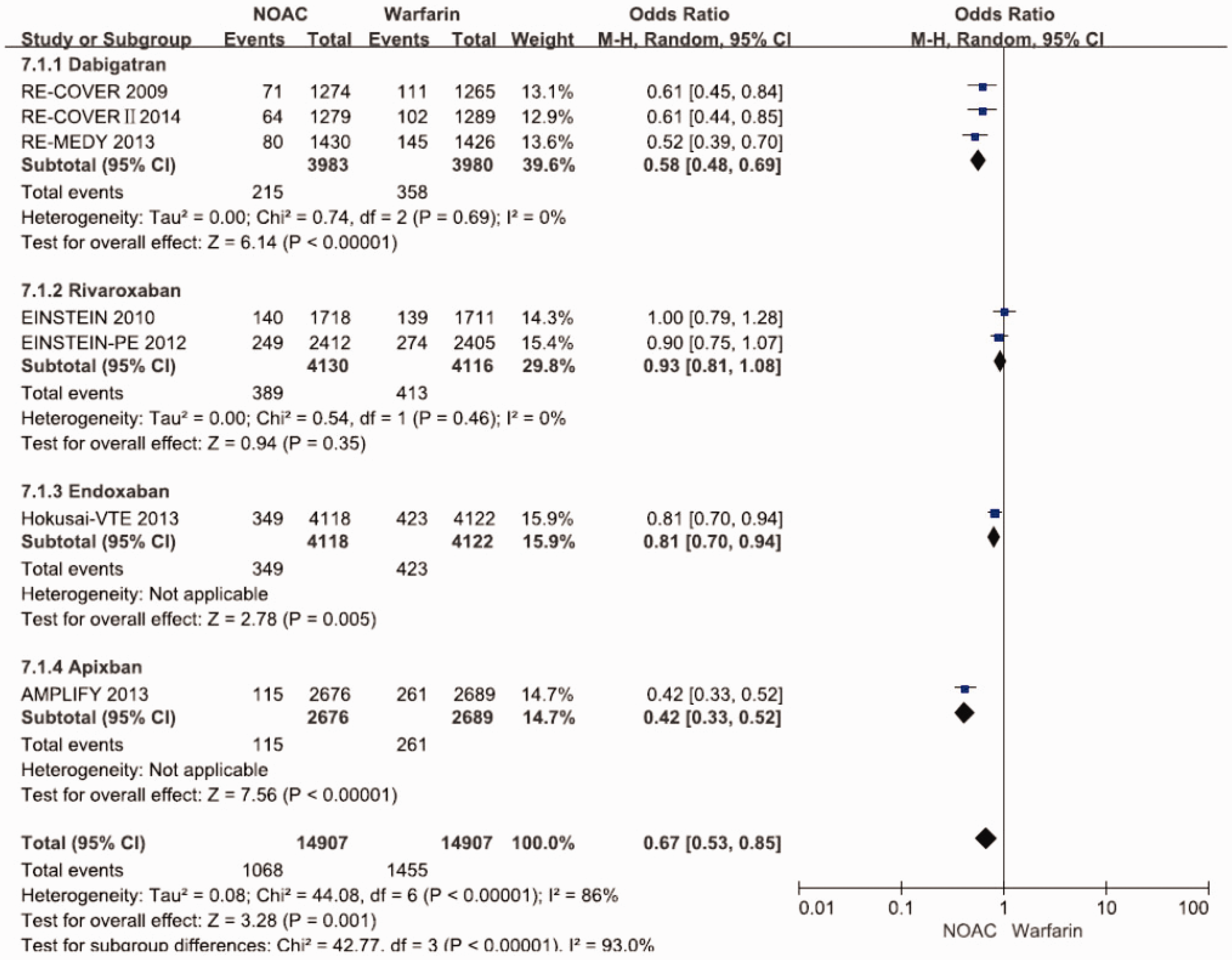
Heterogeneity assessment
The results of efficacy and safety of NOACs by using random- or fixed-effects model.

Publication bias assessment
On the basis of funnel plot analysis, the effect points of the seven studies are roughly the inverted funnel type with the centre of the combined effect and the roughly symmetrical distribution, but the number of studies is too small to completely exclude the publication bias of the literature (Figure 10).
Assessment of publication bias by using funnel plot.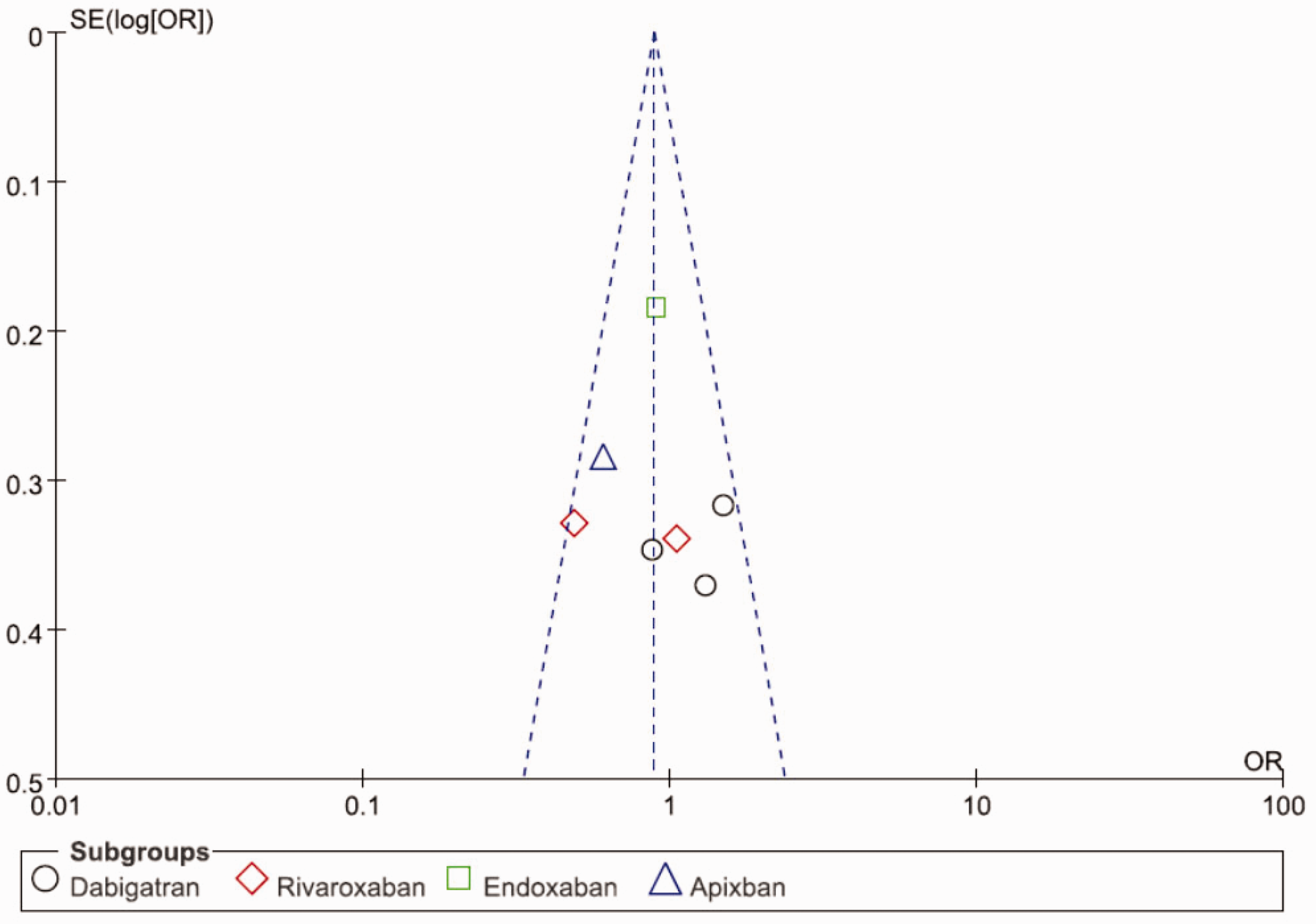
Discussion
Summary of evidence
Our study showed that compared with warfarin, recurrent venous thromboembolism, death related to venous thromboembolism or fatal pulmonary embolism, symptomatic deep vein thrombosis, symptomatic nonfatal pulmonary embolism and all deaths are similar in non–vitamin K antagonist oral anticoagulants group, but major bleeding event and clinically relevant non-major bleeding event decreased in non–vitamin K antagonist oral anticoagulants group. When studies were separately analysed on the basis of types of non–vitamin K antagonist oral anticoagulants, effects and safety showed the same trends.
Comparison with other studies
Our findings were generally consistent with the previous meta-analysis,5,6 which showed similar or superior efficacy and safety of non–vitamin K antagonist oral anticoagulants compared with warfarin. But indirect comparisons indicate differences in the risk of clinically relevant bleeding events, which weredependent on the pharmacologic properties of non–vitamin K antagonist oral anticoagulants and diseases of patients. For example, dabigatran has a single renal route of elimination and a distinct variability in patients receiving the same dose, which may increase the bleeding risk, especially when using a higher dose. 19 So individualised therapy should be considered in patients with renal impairment. 4 Rivaroxaban has a short half-life, which cause less effective due to its rapid elimination, so the current once-daily regimen may result in insufficient concentrations at the end of a 24-h day. 20 Due to these pharmacologic properties, dabigatran and rivaroxaban may require more individualised dosing.
non–vitamin K antagonist oral anticoagulants’ pharmacokinetics is affected by obesity, and meta-analysis showed that compared to vitamin K antagonists/low molecular weight heparin, venous thromboembolism recurrence in patients with obesity and morbid obesity treated with non–vitamin K antagonist oral anticoagulants was similar and non–vitamin K antagonist oral anticoagulants could reduce the risk of major bleeding.21 When used in cancer-associated venous thromboembolism, oral factor Xa inhibitors reduced the risk of recurrent venous thromboembolism compared with low molecular weight heparin, but the likelihood of major bleeding was inconsistent.22,23 These characteristics of non–vitamin K antagonist oral anticoagulants indicate the importance of individualised therapy and it is also necessary to look for INR-like indicators to assess the target dose of non–vitamin K antagonist oral anticoagulants. 24
Strengths and limitations
This review updated the results of efficacy and safety of non–vitamin K antagonist oral anticoagulants for venous thromboembolism, and comprehensively assessed the efficacy (including recurrent venous thromboembolism, venous thromboembolism-related death or fatal pulmonary embolism, symptomatic deep vein thrombosis, symptomatic nonfatal pulmonary embolism) and safety (including all death, major bleeding event and clinically relevant bleeding event) between non–vitamin K antagonist oral anticoagulants and warfarin.
Our review had limitations that deserve further consideration. First, the results from the study may lack broad generalisability to patients treated in clinical settings due to the presence of highly selective patients in the included randomized controlled trials. Furthermore, several serious flaws in randomized controlled trials comparing non–vitamin K antagonist oral anticoagulants with vitamin K antagonists raised concerns about superiority claims for the non–vitamin K antagonist oral anticoagulants. For example, the outcomes of different studies were adjusted for different confounding factors, which made it difficult to compare the results across the included studies. Second, there are only seven studies included in our meta-analysis and the results for apixaban and edoxaban relied on a single randomized controlled trial, which may omit exact effects and safety of non–vitamin K antagonist oral anticoagulants. Third, we did not review non-English publications. Furthermore, the efficacy and safety of non–vitamin K antagonist oral anticoagulants may be influenced by actual adherence patterns, patient baseline risks and other real-world differences that may not be predicted by randomized controlled trial results. 25
Conclusions and implications
Our meta-analysis of randomized controlled trials showed that the efficacy of non–vitamin K antagonist oral anticoagulants for venous thromboembolism is comparable to that of warfarin, but the risk of clinical bleeding is lower than that in warfarin. However, randomized controlled trials excluded influences of patient baseline risks, actual adherence patterns and other real-world differences, so more evidence from observational studies is needed for non–vitamin K antagonist oral anticoagulants on their efficacy and safety in the real-world settings.
Supplemental Material
sj-pdf-1-shr-10.1177_20542704211010686 - Supplemental material for Efficacy and safety of non-vitamin K antagonist oral anticoagulants for venous thromboembolism: a meta-analysis
Supplemental material, sj-pdf-1-shr-10.1177_20542704211010686 for Efficacy and safety of non-vitamin K antagonist oral anticoagulants for venous thromboembolism: a meta-analysis by Yan Zhuang, Lin-feng Dai and Ming-qi Chen in JRSM Open
Footnotes
Declarations
Competing interests
The authors declare that there is no conflict of interest.
Funding
No funding.
Ethics approval
As a review of existing data, ethical approval was not required.
Guarantor
Yan Zhuang.
Contributorship
Yan Zhuang contributed to study design. Data were collected and analysed by Yan Zhuang and Lin-feng Dai. Yan Zhuang and Ming-qi Cheng drafted/revised the article. The final version has been approved by all authors.
Provenance
Not commissioned.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
