Abstract
Drug use has emerged as a significant global health concern, contributing to the burden of liver diseases, particularly hepatocellular carcinoma (HCC) and cirrhosis. This study aims to assess the global burden of drug use-related HCC and cirrhosis from 1990 to 2021, using data from the Global Burden of Disease (GBD) study, and to predict future trends up to 2045. Data on drug use-related cirrhosis and HCC were collected from the GBD 2021 database. Joinpoint regression analysis to evaluate temporal trends. Health inequalities were assessed via the slope index of inequality (SII) and the concentration index (CI). Future burden was projected via Bayesian Age-Period-Cohort (BAPC) models. From 1990 to 2021, global mortality and disability-adjusted life-years (DALYs) attributable to drug use-related HCC and cirrhosis increased gradually. The age-standardized death rate (ASDR) for drug use-related HCC increased by 1.96%, whereas the age-standardized DALY rate for HCC increased, with an average annual percentage change (AAPC) of 1.36%. In contrast, the DALY rate for drug use-related cirrhosis declined annually by −0.12%. High socio-demographic index (SDI) regions, particularly high-income North America, presented the highest burden of drug use-related HCC and cirrhosis. The burden of cirrhosis attributable to drug use increased in low-middle-SDI regions. The global burden of liver cancer due to drug use has become more concentrated in high-SDI regions. Future projections indicate that deaths from drug use-related HCC and cirrhosis will continue to increase, with HCC deaths expected to increase by 150.15% and cirrhosis deaths increasing by 43.03% by 2045. The burden of drug use-related cirrhosis and HCC has increased and is projected to continue increasing. This trend highlights the urgent need for targeted public health interventions, improved access to healthcare, and effective drug use prevention strategies, particularly in high-SDI regions.
Keywords
● This study provides the first comprehensive global assessment (1990–2021) and future projections (to 2045) of hepatocellular carcinoma (HCC) and cirrhosis attributable to drug use based on GBD 2021 data.
● Mortality and DALYs attributable to drug use-related HCC increased steadily, with the ASDR rising by 1.96% and DALY rates showing an AAPC of 1.36%, while drug use-related cirrhosis showed a slight annual decline (−0.12%).
● High-SDI regions—especially high-income North America—carried the highest burden, whereas low-middle-SDI regions experienced increasing cirrhosis burden; inequality analyses revealed growing concentration of HCC burden in high-SDI settings.
● Bayesian Age-Period-Cohort projections indicate a marked rise in future mortality, with HCC deaths expected to increase by 150.15% and cirrhosis deaths by 43.03% by 2045.
● The escalating global burden highlights the urgent need for targeted public health strategies, improved healthcare access, and strengthened drug use prevention programs, particularly in high-SDI regions.
Introduction
Drug use represents a pervasive global phenomenon that significantly impacts human health and social development. People often initiate drug use seeking euphoria without realizing that there is an enormous health crisis hidden behind it. 1 Since 1990, the global consumption of illicit drugs and its associated burden have increased significantly, 2 making illicit drug use one of the major causes of premature mortality and morbidity worldwide. This phenomenon not only has both acute and long-term effects on the health of populations but also poses a serious challenge to the stability and harmony of societies. 3 Drug use ranked 11th in global disability-adjusted life years (DALYs) attributable to behavioral risk in 2021, accounting for 1% of total DALYs. 4 Behind this figure is the suffering and loss of countless individuals and families due to drug use. Currently, drug use is widespread globally, and governments and organizations are actively taking measures to address it. Addressing this problem requires coordinated efforts across all sectors of society.
Drug use creates multiple health burdens, including severe liver damage. As the third leading cause of cancer death globally, liver cancer has become the third leading cause of carcinoma death after lung and colorectal cancer and the sixth most commonly diagnosed cancer. 5 Cirrhosis, on the other hand, is a major determinant of hepatocellular carcinoma (HCC), 6 whose main etiology is chronic liver inflammation caused by hepatitis B virus (HBV) and hepatitis C virus (HCV). 7 Globally, more than 250 million people suffer from chronic HBV, and more than 800 000 people die annually from complications of HBV. 8 In addition, the risk of HBV infection is significantly greater in the drug-using population than in the general population. 9 Moreover, approximately 71 million people worldwide suffer from chronic HCV infection. 10 Notably, since the early 1990s, HCV transmission via blood transfusion has been effectively controlled, while injecting drug use has become the main route of HCV transmission, especially in high-income countries. 11 A global meta-analysis revealed that 18% of people who inject drugs (PWID) are infected with human immunodeficiency virus (HIV), 52% are HCV seropositive, and 9% are positive for HBV surface antigen (HBsAg). 12 These data suggest that drug use is not only an important driver of viral hepatitis transmission but also an important factor in the development of cirrhosis and HCC. Thus, drug use can indirectly contribute to the global epidemic of these 2 diseases by increasing the transmission of viral hepatitis and HIV.
Drug use has been shown to vary by age and region, and the burden of cirrhosis and HCC due to drug use must therefore have its own distinctive age and regional patterns. From an age perspective, there are significant differences in the risk of liver disease due to drug use among different age groups; from a regional distribution point of view, the associated burden of cirrhosis and HCC due to drug use also shows significant geographic variations across countries and regions owing to differences in socio-economic status, cultural background, accessibility of health-care resources and public health policies. However, despite the growing magnitude of the problem, there is still a relative paucity of studies on the impact of drug use on the global burden of cirrhosis and HCC. While recent studies have examined drug-attributable liver cancer burden using GBD data, 13 comprehensive analyses simultaneously incorporating both cirrhosis and hepatocellular carcinoma with health inequality assessments and detailed temporal trend analysis remain limited. Therefore, this study, which is based on the Global Burden of Disease (GBD) database, aims to provide a scientific basis for the formulation of regional or even global health policies with a more comprehensive perspective by analyzing the burden of cirrhosis and HCC due to drug use and revealing its association with socio-economic status in 204 countries and 21 regions around the world from 1990 to 2021.
Materials and Methods
Study design This study employed a retrospective, cross-sectional design using secondary data from the Global Burden of Disease (GBD) 2021 database to assess temporal trends and predict future burden.
Data Collection
Data on the burden of cirrhosis and HCC due to drug use from 1990 to 2021 were obtained from the GBD Study (GBD 2021) results tool (http://ghdx.healthdata.org/gbd-results-tool), a comprehensive global health assessment program that covers more than 360 diseases and injuries and over 80 risk factors. The tool also provides a comprehensive assessment of these diseases and injuries by country, region, sex, and age. On the basis of the socio-demographic index (SDI, an indicator based on fertility, education, and income), the 204 countries and territories can be categorized into 5 regions, namely low, low-medium, medium, high-medium, and high regions. The countries and areas can be categorized into 21 regions on the basis of geographical coherence, such as East Asia and Central Europe. The data results are presented as numbers and 95% uncertainty intervals (UIs) and 95% confidence intervals (CIs). Inclusion criteria: All countries and territories with available data in the GBD 2021 database (n = 204); Study period: 1990 to 2021. Diseases: Hepatocellular carcinoma (HCC) and cirrhosis cases with drug use as an attributable risk factor. Age groups: All age groups as defined in the GBD study. Both sexes included in the analysis. Exclusion criteria: Countries or regions with incomplete or unavailable data in the GBD 2021 database; Liver diseases not attributable to drug use; Data points with uncertainty intervals that could not be reliably estimated.
Assessment of the Burden of Drug Use
Definitions of drug use include injecting drug use (IDU), which is the current behavior of ingesting drugs by injection among people aged 15 to 64. Drug Use Disorder (DUD): opioid, amphetamine, and cocaine use disorders defined by DSM or ICD criteria, with 100% of the burden attributable to drug use. Theoretical minimum risk exposure level (TMREL): zero exposure, that is, any level of drug use is considered harmful. Methods of assessing drug use have been detailed on the following website: https://www.healthdata.org/gbd/methods-appendices-2021/drug-use. To quantify the proportion of each risk factor contributing to the burden of a particular disease, comparative risk assessment methods were included in the GBD study. 14 The following key steps are involved: The following key steps are involved: 1. Define risk and outcomes → 2. Estimate exposure → 3. Set the TMREL → 4. Calculate the relative risk (RR) →5. Derive population attribution Population Attributable Fraction scores (PAFs) →6. Integrate disease burden data →7. Analyze uncertainty →8. Validate and apply results.
Data Analysis
We assessed trends in the burden of drug use-related cirrhosis and hepatocellular carcinoma using DALYs, age-standardized death rate (ASDR), and DALY rates. DALYs equal years of disability (YLDs) plus years of life lost (YLLs). 15 When morbidity, mortality and DALYs are compared, the ASDR is an important indicator, with the ASDR being a measure of the age-standardized incidence of death from specific causes in a given population. The aim is to eliminate the effect of differences in age structure on mortality rates between different populations, thus allowing for fairer comparisons of mortality rates in different regions or at different points in time. The DALY rate is a standardized form of DALYs that provides a comprehensive assessment of the burden of disease by adjusting for population size, which allows the burden of disease to be comparable across regions or over time. In addition, we applied joinpoint analysis, which is a method for identifying the change points of trends in time-series data to more accurately reflect the phasing of trends and provide more detailed temporal pattern analysis than simple linear trend approaches. Using the joinpoint regression model, significant change points (joinpoints) in the time series are identified, which indicate significant changes in the trend. Each joinpoint splits the time series into multiple linear segments, and the slope of each line segment indicates the rate of change in the trend for that segment. On the basis of the analytical approach described above, we quantified temporal trends in ASDR and DALY rate for drug use-associated cirrhosis and HCC from 1990 to 2021 via average annual percentage change (AAPC). AAPC can identify continuous changes in disease data throughout the study period, and when the AAPC value and its 95% CI > 0, the trend is defined as increasing. Conversely, when the AAPC value and its 95% CI < 0, the trend decreased. Otherwise, the burden was considered relatively stable over time. 16
Measurement of Health Inequalities
To address gaps in previous analyses of drug use-related liver disease burden, we conducted comprehensive health inequality assessments using standardized methodological approaches recommended by the World Health Organization (WHO), 17 with both crude DALY rates and age-standardized DALY rates extracted from the GBD 2021 database for all 204 countries and territories for inequality analysis. Countries were ranked according to their SDI values, which served as the primary socioeconomic ranking variable, where SDI ranges from 0 to 1 as a composite measure incorporating fertility rates under 25, mean education level, and lag-distributed income per capita. The relative rank (R) for each country was calculated as R = (Cumulative population up to countryi − 0.5 × population of countryi)/Total population, which formed the basis for calculating 2 standardized inequality indicators.
The SII serves as an absolute inequality indicator that quantifies differences in disease burden across socio-economic levels using weighted linear regression where SII = β1, derived from the equation Y = β0 + β1(R) + ε, with Y representing the health outcome (DALY rate), R representing the relative rank based on SDI, and each country weighted by population size, where a positive SII indicates greater disease burden in higher socio-economic groups while negative SII indicates higher burden in lower socio-economic groups.
Complementing this absolute measure, the CI functions as a relative inequality indicator ranging from −1 to +1 that assesses unequal distribution of health outcomes across socioeconomic levels, calculated using CI = (2/μ) × Cov(Y, R), where μ represents the population-weighted mean of the health outcome and Cov(Y, R) represents the covariance between health outcome and relative rank, with the CI expressing inequality through the area under the concentration curve where CI > 0 indicates outcomes concentrated in higher socioeconomic groups and CI < 0 indicates concentration in lower socioeconomic groups. 18
Evaluation of Drug Use-related Burden
We used a Bayesian age-period-cohort (BAPC) model to predict future patterns of the burden of cirrhosis and HCC due to drug use. The BAPC model is based on the age-period-cohort (APC) model, which assumes that morbidity or mortality is related to age structure and population size. It is able to simultaneously account for the effects of age, period, and birth cohort on outcome metrics, and provides predictions and uncertainty estimates through a Bayesian framework.19 -21 Briefly, the BAPC model is expressed as log(λij) = μ + αi + βj + γk + ε. In the model, μ represents the intercept; ε represents random error; α, β, and γ represent age, period, and cohort, respectively; and i, j, and k represent the coefficients of the age, period, and cohort effects, respectively. To account for overdispersion, we used the BAPC model, which was implemented via the INLA and BAPC packages in R software. All the statistical analyses and visualizations of the results were performed via R software (R version 4.4.1), and P < .05 was considered statistically significant.
Results
Burden and Temporal Trends of Cirrhosis and HCC Attributable to Drug Use
In 2021, the number of deaths attributable to drug use globally was estimated to be 463 360.16 (95% UI 408 450.59-516 078.54),of which 65 499.15 (95% UI 50 430.35-83 777.75) were due to drug-use-related HCC and 177 279.02 (95% UI 137 545.01-218 698.55) were due to drug-use-related cirrhosis. The global DALYs attributable to drug use in 2021 was 27 666 344.32 (95% UI 24 217 536.77-30 956 951.40), of which the DALYs attributable to cirrhosis due to drug use was 6 270 773.11 (95% UI 4 917 528.19-7 676 389.40), and the DALYs attributable to HCC due to drug use had a DALYs of 1 611 233.55 (95% UI 1 254 877.04-2 052 507.05). From 1990 to 2021, the global ASDR of drug use increased by 0.51% per year (95% CI 0.09-0.92), the ASDR of HCC due to drug use increased by 1.96% per year (95% CI 1.76-2.16), and the ASDR OF cirrhosis due to drug use increased by 0.09% per year ( 95% CI −0.05 to 0.24). Meanwhile, the global age-standardized DALY rate due to drug use continued to increase, with an AAPC of 0.20%, the age-standardized DALY rate for HCC due to drug use continued to increase, with an AAPC of 1.36% (95% CI 1.2 to 1.52), and the age-standardized DALY rate for cirrhosis due to drug use declined each year with an AAPC of −0.12 (95% CI –0.26 to 0.03) (Tables 1 and 2), for more details, please see the attached Supplemental Tables S1 to S4.
The Number, Age-Standardized Rate, and Temporal Trend of HCC Burden Attributable to Drug Use From 1990 to 2021.

The Number, Age-Standardized Rate, and Temporal Trend of Cirrhosis Burden Attributable to Drug Use From 1990 to 2021.

Gender Differences in the Burden of Cirrhosis and HCC Due to Drug Use
In 2021, the global number of deaths due to HCC and cirrhosis caused by drug use in females was 21 651.39 (95% UI 15 292.51-29 522.80) versus 39 451.43 (95% UI 27 615.87-52 991.88), and the global number of deaths due to HCC and cirrhosis in males attributable to drug use was 43 847.76 (95% UI 34 291.05-55 641.33) and 137 827.59 (95% UI 109 559.67-168 390.49), respectively. From 1990 to 2021, the global ASDR for HCC due to drug use in women increased from 0.22 (95% UI 0.14-0.34) to 0.47 (95% UI 0.33-0.64), with an AAPC of 2.4% (95% CI 2.22-2.58), whereas the ASDR for cirrhosis due to drug use increased from 0.75 (95% UI 0.50-1.09) to 0.88 (95% UI 0.62-1.19), with an AAPC of 0.49% (95% CI 0.28-0.71). The global ASDR for HCC due to drug use in men increased from 0.63 (95% UI 0.44-0.85) to 1.10 (95% UI 0.85-1.40), with an AAPC of 1.87% (95% CI 1.66-2.08).The global age-standardized DALY rate of HCC due to drug use for females was 6.37 (95% UI 4.15-9.66) versus 11.43 (95% UI 8.14-15.22) in 1990 and 2021, respectively, and its age-standardized DALY rate continued to increase, with an AAPC of 1.91% (95% CI 1.69-2.12). The global age-standardized DALY rate for HCC due to drug use also continued to increase in males, with an AAPC of 1.22% (95% CI 1.01-1.42) and DALY rate of 18.13 (95% UI 13.27-24.27) and 26.15 (95% UI 20.52-33.20) in 1990 and 2021, respectively. In 2021, the global age-standardized DALY rate for cirrhosis due to drug use in women was 30.95 (95% UI 21.68-41.65) with an AAPC of 0.36% (95% CI 0.09-0.64). In contrast, the global age-standardized DALY rate for cirrhosis due to drug use among men declined from 1990 to 2021, with an AAPC of −0.18% (95% CI −0.3 to −0.07) (Tables 1 and 2), and more details were provided in the Supplemental Table S1 to S4.
Age Composition of the Burden of Cirrhosis and HCC Attributable to Drug Use
Globally, the number of liver cancer deaths due to drug use as well as DALYs showed an increasing trend over time. In 1990, the number of liver cancer deaths due to drug use peaked at 3446.40 (95% UI 2254.54-4483.86) in the 60 to 64 years age group. The mortality rate increased incrementally with age group and peaked at 2.4/100 000 in the 65 to 69 age group. Moreover, the global DALYs associated with HCC due to drug use also increased with age, peaking at 100 315.06 (95% UI 65 633.35-140 586.84) in the age group of 60 to 64 years, whereas the DALY rate peaked at 62.46/100 000 in this age group (Figure 1A and B). In 2021, the number of global HCC deaths, DALYs and DALY rate attributable to drug use all reached highest in the 65 to 69 year age group, which were 11 133.48 (95%UI 8170.97-14 793.68), 27 338 550.25 (95%UI 200 361.79-3 634 440.25), and 99.11/100 000, respectively. The mortality rate peaked at 7.35/100 000 in the 80 to 84 year age group (Figure 1C and D). For cirrhosis, in 1990, global deaths of cirrhosis due to drug use were highest in the 50 to 54 year age group at 12 720.87 (95% UI 8346.19-19 040.90), and the mortality rate peaked in the 55 to 59 year age group at 6.68/100 000. The global DALYs associated with cirrhosis due to drug use peaked at 496 471.73 (95% UI 349 309.20-691 144.55) in the 40 to 44 year age group, and the DALY rate peaked at 230.14/100 000 in the 50 to 54 year age group (Figure 2A and B). In 2021, global cirrhosis deaths due to drug use peaked at 23 528.89 (95% UI 16 487.22-30 895.62) in the 55 to 59 year age group, and the mortality rate peaked at 6.68/100 000 in the 65 to 69 year age group. The global DALYs associated with cirrhosis attributable to drug use peaked at 844 394.60 (95% UI 599 836.81-1 173 778.48) in the of 50 to 54 year age group and the DALY rate peaked at 200.96/100 000 in the 55 to 59 year age group (Figure 2C and D), more details were available in Supplemental Tables S5 to S8.
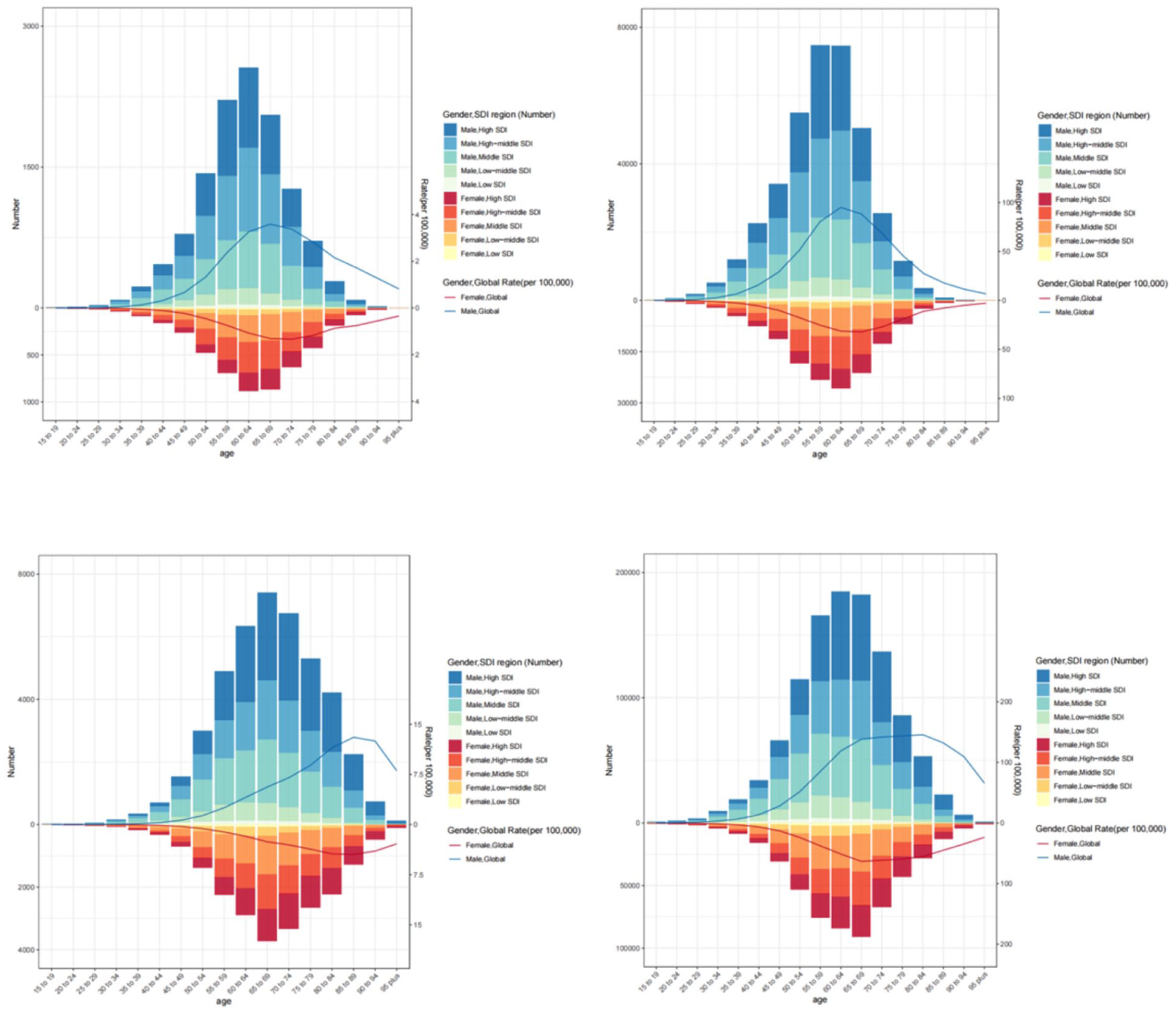
The burden of liver cancer attributable to drug use for both genders in global by age group and SDI index in 1990 and 2021. (A) Global number of death cases and death rate of HCC in 1990; (B) Global number of DALYs and DALY rate of HCC in 1990; (C) Global number of death cases and death rate of HCC in 2021; (D) Global number of DALYs and DALY rate of HCC in 2021, DALYs disability-adjusted life-years.

The burden of cirrhosis attributable to drug use for both genders in global by age group and SDI index in 1990 and 2021. (A) Global number of death cases and death rate of cirrhosis in 1990; (B) Global number of DALYs and DALY rate of cirrhosis in 1990; (C) Global number of death cases and death rate of cirrhosis in 2021; (D) Global number of DALYs and DALY rate of cirrhosis in 2021, DALYs disability-adjusted life-years.
Regional Composition of the Burden of Cirrhosis and HCC Attributable to Drug Use
In the terms of regions by SDI, the ASDR and age-standardized DALY rate for diseases attributable to drug use were highest in high-SDI regions and lowest in low-SDI regions. In particular, in 1990, the number of deaths from liver cancer due to drug use, DALYs, ASDR, and age-standardized DALY rates were highest in the high-middle SDI regions and lowest in the low-SDI regions. In 2021, the number of liver cancer deaths, DALYs, ASDR, and age-standardized DALY rate due to drug use
were highest in the high-SDI regions and remained lowest in the low-SDI regions. For cirrhosis due to drug use, in 1990, the number of deaths and DALYs for cirrhosis due to drug use were highest in the middle-SDI region, the ASDR and age-standardized DALY rate associated with cirrhosis due to drug use were highest in the low-middle-SDI region, and both were lowest in the low-SDI region. In 2021, the number of cirrhosis deaths and DALYs due to drug use was highest in the middle-SDI region, ASDR and age-standardized DALY rate were highest in the low-middle-SDI region, and DALY rate were lowest in the high-middle-SDI region (Table 1). In 2021, high-income North America had the highest burden of disease due to drug use, followed by Eastern Europe and Central Asia. The region with the largest increase in ASDR was high-income North America, whereas East Asia had the largest decrease (Supplemental Table S9). The 3 regions with the highest burden of hepatocellular carcinoma due to drug use were Central Asia, high-income North America, and high-income Asia-Pacific. The region with the largest increase in ASDR was high-income South Latin America, while Oceania had the smallest increase (Supplemental Table S10). The top 3 regions with the highest burden of cirrhosis due to drug use were Central Asia, high-income North America, and Central Latin America. The region with the largest increase in ASDR was Eastern Europe, whereas East Asia had the largest decrease (Supplemental Table S11). The 3 countries with the largest increases in the ASDR for diseases attributable to drug use were Lesotho, Swaziland, and Canada, whereas the 3 countries with the largest increases in age-standardized DALY rate were Lesotho, the United States, and Swaziland (Supplemental Figure S1 A and B). The 3 countries with the greatest increases in the ASDR for hepatocellular carcinoma due to drug use and the 3 countries with the largest increases in the age-standardized DALY rate were Canada, Greenland, and Uruguay (Figure 3A and B). The 3 countries with the largest increases in ASDR and age-standardized DALY rate for cirrhosis attributable to drug use were Canada, Greenland, and Lithuania,(Figure 3C and D), see Supplemental Figures S2 and S3 for more details.

The global disease burden of cirrhosis and liver cancer attributable to drug use for both genders in 204 countries and territories from 1990 to 2021. (A) AAPC of age-standardized DALY rate for liver cancer; (B) AAPC of ASDR for liver cancer; (C) AAPC of age-standardized DALY rate for cirrhosis. (D) AAPC of ASDR for cirrhosis. AAPC average annual percentage change; DALYs disability-adjusted life-years; ASDR age-standardized death rate.
Global Assessment of the Inequality of the Burden of Cirrhosis and HCC due to Drug Use
From 1990 to 2021, inequality in the burden of disease deaths and DALYs due to drug use increased (Supplemental Figure S4A and B), and the associated burden was concentrated in regions with higher SDI index (Supplemental Figure S4 C and D). SII indicated that the burden of HCC and cirrhosis due to drug use was positively associated with the SDI index. Moreover, the inequality of burden of disease for HCC due to drug use increased (Figure 4A), whereas the inequality of burden of cirrhosis due to drug use decreased (Figure 4B). Regions with higher SDI account for a greater proportion of the burden of liver cancer associated with

SII analysis and Concentration index analysis. (A) Health inequality regression curves for crude DALY rate of liver cancer attributable to drug use. (B) Health inequality regression curves for crude DALY rate of cirrhosis attributable to drug use. (C) Concentration curves for DALYs of HCC attributable to drug use. (D) Concentration curves for deaths of HCC attributable to drug use.
drug use, that is, the global burden of liver cancer disease attributable to drug use is progressively concentrated in regions with higher SDI (Figure 4C and D), and more details shown in Supplemental Figure S4A-D.
Prediction of the Burden of Cirrhosis and HCC due to Drug Use
We further projected the disease burden of HCC and cirrhosis due to drug use over the next 15 years via the GBD database. We found that by 2045, the number of global deaths from HCC and cirrhosis due to drug use would increase to 163 601.03 and 253 620.13, respectively, which is 150.15% and 43.03% higher than that in 2021. From 2021 to 2045, the global ASR related to liver cancer due to drug use would gradually increase and reach 1.08/100 000 in 2045 (Supplemental Tables S12 and 13). In China and the United States, the number of drug-induced deaths from hepatocellular carcinoma and cirrhosis would increase, and the ASR associated with drug-induced HCC and cirrhosis would also increased from 2021 to 2045 (Figure 5A and B). Fortunately, the global ASR of cirrhosis due to drug use would remain largely unchanged (Supplemental Table S12 and 13).

The observed (solid line) and predicted (dashed line) deaths of HCC (A) and cirrhosis (B) attributable to drug use from 1990 to 2045.
Discussion
The global burden of drug use-related liver disease represents a complex epidemiological challenge that intersects with evolving therapeutic landscapes and persistent health inequalities. Our analysis of 204 countries from 1990 to 2021 reveals distinct trajectories for hepatocellular carcinoma and cirrhosis that have profound implications for clinical practice and public health policy.
Recent studies have examined drug use-attributable liver disease using GBD data, with Chen et al analyzing drug-induced liver cancer mortality trends from 1990 to 2021. 13 Our study significantly expands this scope by providing comprehensive assessment of both cirrhosis and hepatocellular carcinoma, revealing distinct epidemiological patterns with important clinical implications. The dramatic divergence we observed—HCC burden increasing 150.15% by 2045 versus stabilizing cirrhosis DALY rates (−0.12% AAPC)—occurs when the therapeutic landscape for hepatocellular carcinoma has undergone significant transformation through immune checkpoint inhibitors, targeted therapies, and novel combination treatments.22 -26 This epidemiological-therapeutic intersection creates important considerations for resource planning. While advances in systemic treatments have improved survival outcomes, the projected doubling of drug-related cases suggests that treatment innovations alone cannot address this public health challenge. The pathophysiology underlying drug-related liver disease involves multiple mechanisms including direct hepatotoxicity, viral hepatitis transmission, and immune dysfunction that creates particularly aggressive disease phenotypes.27 -31 Understanding these mechanistic differences becomes crucial as personalized approaches gain prominence in oncological care.
Our demographic analysis reveals how biological, behavioral, and social factors intersect to shape disease burden. The gender-specific trends—males showing higher absolute burden but females demonstrating faster increase rates (2.4% AAPC for HCC ASDR)—reflect complex interactions between exposure patterns, biological susceptibility, and evolving substance use behaviors. These patterns connect with the strong associations between drug use, particularly injecting drug use, and HIV infection, which exacerbates liver disease risk through CD4+ T cell depletion, increased viral hepatitis susceptibility, and accelerated progression through immunosuppression. 32 The temporal shifts in peak disease age—HCC deaths moving from 60-64 to 65-69 years, with mortality peaks extending to 80 to 84 years—reflect interactions between population aging, treatment advances, and evolving substance use patterns. The delayed peak ages for cirrhosis-related deaths (from 50-54 to 55-59 years) alongside declining DALY rates suggest that early intervention advances have been more effective for cirrhosis management than preventing HCC progression, underscoring the importance of prevention strategies targeting earlier causal pathway stages.
Our implementation of health inequality measures using the Slope Index of Inequality (SII) and Concentration Index (CI) reveals critical dimensions that fundamentally challenge conventional understanding of global disease distribution patterns. The progressive concentration of drug use-related liver disease burden in high-SDI regions represents a paradigmatic shift from traditional global health frameworks where infectious diseases and poverty-related conditions typically concentrate in low-resource settings. This concentration pattern in high-SDI regions, particularly high-income North America, creates a paradoxical epidemiological situation where regions with the most advanced healthcare infrastructure, sophisticated diagnostic capabilities, and abundant medical resources simultaneously face the greatest absolute increases in disease burden. Our analysis demonstrates that high-SDI regions account for disproportionately large shares of both HCC and cirrhosis burden, with concentration curves consistently falling below the line of equality, indicating systematic concentration among more advantaged populations. The mechanisms underlying this concentration reflect complex interactions between multiple risk factor pathways. High-SDI regions demonstrate elevated drug accessibility through sophisticated distribution networks, higher purchasing power enabling sustained substance use, and lifestyle factors including concurrent alcohol consumption that synergistically increase liver injury risk. Additionally, the prevalence of viral hepatitis among people who inject drugs in these regions—with HCV incidence reaching 30% to 80%—creates self-reinforcing epidemiological cycles where high drug use prevalence intersects with efficient viral transmission networks, dramatically accelerating liver fibrosis progression and HCC development. 33 Furthermore, the relative abundance of medical resources in high-SDI areas enables more accurate disease detection, diagnosis, and attribution, potentially amplifying apparent burden differences through surveillance bias. However, our inequality analysis suggests that these detection differences alone cannot explain the magnitude of concentration observed, indicating genuine epidemiological disparities driven by differential exposure patterns and risk factor clustering. The regional heterogeneity within SDI categories adds additional complexity to inequality patterns. Middle-SDI regions, exemplified by Central Asia, demonstrate the highest absolute numbers of cirrhosis deaths and DALYs, reflecting rapid socioeconomic transitions characterized by increasing drug accessibility, changing lifestyle patterns, and insufficient development of prevention infrastructure. These regions face dual challenges of expanding substance use prevalence without corresponding harm reduction capacity, creating conditions for explosive disease burden growth. Conversely, low-SDI regions show the lowest burden levels, which may reflect genuinely lower drug accessibility and use prevalence, but also substantial underreporting due to weaker healthcare systems, competing health priorities, and limited diagnostic capabilities. The potential for systematic underestimation in these regions means that global inequality patterns may be even more pronounced than our analysis suggests. These inequality patterns have profound implications for resource allocation, intervention targeting, and global health priority-setting. The concentration of burden in high-resource settings challenges traditional development aid models and suggests needs for innovative financing mechanisms that address drug-related liver disease burden regardless of national economic status.
Our projections of 150.15% and 43.03% increases in HCC and cirrhosis deaths by 2045 occur against WHO’s ambitious targets of reducing hepatitis B and C deaths by 95% and new infections by 90% by 2030. 34 The temporal disconnect between prevention interventions and cancer outcomes creates immediate challenges for health systems implementing harm reduction while preparing for increased treatment demand. Interventions including needle exchange programs, opioid agonist therapy, and HIV antiretroviral therapy have demonstrated effectiveness in reducing infectious disease burden among people who inject drugs, yet coverage remains inadequate globally. 31 Integration of prevention approaches with emerging therapeutic options requires coordinated responses addressing both immediate treatment needs and long-term prevention goals.
Based on our analysis, we believe the patterns observed reflect fundamental shifts in how drug use-related liver disease manifests globally, with direct implications for clinical practice and health system organization. The concentration in high-SDI regions challenges traditional global health priority frameworks and demands new intervention approaches accounting for drug-related liver disease population characteristics. The faster female increase rates, particularly for HCC DALYs, suggests an emerging concern intersecting with changing substance use patterns that may require gender-specific approaches. 35 We advocate for specialized treatment and prevention protocols accounting for complex comorbidity patterns, social circumstances, and behavioral factors common among individuals with drug use histories. Such approaches must integrate addiction medicine principles with advancing oncological care while addressing substantial healthcare access barriers characterizing this population.
Our analysis faces several important limitations. First, liver disease attribution to drug use relies on GBD modeling assumptions that may not capture evolving polysubstance use patterns or novel psychoactive substances with unknown hepatotoxic profiles. Second, projections assume current trend continuation without accounting for potential disruptions from policy changes, therapeutic breakthroughs, or large-scale interventions. Third, burden concentration in high-SDI regions may partially reflect diagnostic capacity rather than true epidemiological differences, with advanced healthcare systems detecting drug-related liver disease more frequently while emerging economies may systematically underreport. Fourth, our national-level inequality measures cannot capture within-country disparities including urban-rural differences and socioeconomic gradients that may create significant treatment access inequalities. Finally, temporal lag between drug use initiation and liver cancer development means projections may not reflect recent substance use pattern changes, particularly among younger populations. These limitations underscore needs for enhanced surveillance systems and regular projection updates.
Conclusion
This study revealed the remarkable impact of drug use on the global burden of both cirrhosis and HCC, especially in high-SDI regions and among male populations. Unlike previous studies focusing solely on liver cancer, our comprehensive analysis demonstrates distinct epidemiological patterns between cirrhosis and HCC, with important implications for targeted interventions. The implementation of inequality measures reveals that the burden is progressively concentrated in high-SDI regions, while joinpoint analysis identifies critical change points in disease trends. In the future, the global burden of HCC and cirrhosis due to drug abuse is expected to continue evolving differently, with HCC showing dramatic increases while cirrhosis patterns stabilize, especially in high-income countries. This trend highlights the seriousness of drug use as a threat to global health, therefore, countries around the world need to exert every effort to alleviate this public health burden and proactively address this global challenge by adopting more active prevention and intervention approaches to reduce drug use, improve access to healthcare services for people who use drugs, improve the prevention and treatment of viral hepatitis, as well as raise public awareness of the harms of drug use. Through the present study, we call upon governments and international organizations to intensify their collaboration in developing targeted interventions to reduce the burden of liver disease caused by drug use, with the expectation of achieving global health equity and sustainable development.
Supplemental Material
sj-docx-1-inq-10.1177_00469580251399106 – Supplemental material for Global Contribution of Drug Use to Cirrhosis and Hepatocellular Carcinoma Worldwide From 1990 to 2021 and Projections to 2045
Supplemental material, sj-docx-1-inq-10.1177_00469580251399106 for Global Contribution of Drug Use to Cirrhosis and Hepatocellular Carcinoma Worldwide From 1990 to 2021 and Projections to 2045 by Jingru Han, Yang Zhao, Simeng Gao, Yan Zhang, Yida Wang and Jianfu Zhao in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-2-inq-10.1177_00469580251399106 – Supplemental material for Global Contribution of Drug Use to Cirrhosis and Hepatocellular Carcinoma Worldwide From 1990 to 2021 and Projections to 2045
Supplemental material, sj-docx-2-inq-10.1177_00469580251399106 for Global Contribution of Drug Use to Cirrhosis and Hepatocellular Carcinoma Worldwide From 1990 to 2021 and Projections to 2045 by Jingru Han, Yang Zhao, Simeng Gao, Yan Zhang, Yida Wang and Jianfu Zhao in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-xlsx-3-inq-10.1177_00469580251399106 – Supplemental material for Global Contribution of Drug Use to Cirrhosis and Hepatocellular Carcinoma Worldwide From 1990 to 2021 and Projections to 2045
Supplemental material, sj-xlsx-3-inq-10.1177_00469580251399106 for Global Contribution of Drug Use to Cirrhosis and Hepatocellular Carcinoma Worldwide From 1990 to 2021 and Projections to 2045 by Jingru Han, Yang Zhao, Simeng Gao, Yan Zhang, Yida Wang and Jianfu Zhao in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Acknowledgements
We appreciate all the academics who have contributed to the GBD study.
Author Contributions
HJR and ZJF designed the study and edited the manuscript. ZY and GSM analyzed and processed the data. ZY and WYD reviewed and edited the manuscript. All authors contributed to the article and approved the submitted version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China [grant numbers 8237070526].
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The GBD 2021 study is an open database, and all data is anonymous.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
