Abstract
Kidney cancer (KC) is an important public-health challenge in China. Although benmelstobart plus anlotinib (BEN + AL) improves survival compared with sunitinib (SUN) for advanced renal cell carcinoma, its economic value in the Chinese healthcare system remains uncertain. Epidemiological trends in KC incidence and disability-adjusted life years (DALYs) in China from 1990 to 2023 were assessed using Global Burden of Disease 2023 data, including joinpoint regression, decomposition analysis, age- and sex-specific patterns. A partitioned survival model was developed to compare BEN + AL with SUN, informed by the phase 3 ETER100 trial. Outcomes included total costs, life-years (LYs), quality-adjusted life years (QALYs), and incremental cost-effectiveness ratios (ICERs) at a willingness-to-pay (WTP) threshold of $ 26 896 per QALY. Sensitivity and scenario analyses were conducted to assess model stability. From 1990 to 2023, incident KC cases in China increased from 19 500 to 61 323, with the age-standardized incidence rate rising from 1.99 to 3.24 per 100 000, particularly among males. DALYs nearly doubled from 326 005 to 582 244, while the age-standardized DALY rate remained stable. Decomposition analysis showed that incidence growth was driven mainly by epidemiological change and population aging, whereas increases in DALYs were largely attributable to aging, partially offset by epidemiological improvements. In the base-case cost-effectiveness analysis, BEN + AL yielded higher effectiveness (4.67 LYs and 3.48 QALYs) than SUN (3.81 LYs and 2.73 QALYs) but at markedly higher cost ($ 192 292.58 vs $ 11 400.59), resulting in an ICER of USD 240 961.36 per QALY. Sensitivity and scenario analyses consistently showed ICERs exceeding the WTP threshold, with progression-free survival value and benmelstobart cost emerged as the primary influencing factors. While the burden of KC in China continues to rise, particularly due to population aging, BEN + AL is unlikely to be cost-effective as first-line therapy for advanced renal cell carcinoma under current pricing.
Keywords
Introduction
Kidney cancer (KC) represents a growing global public-health challenge. In 2022, KC contributed 434 019 new diagnoses and 155 702 fatalities, corresponding to the 14th highest incidence and 16th highest mortality among all cancers. 1 The burden is especially heavy in China, which now accounts for the largest share of KC cases, deaths and disability-adjusted life years (DALYs) worldwide. 2 Renal cell carcinoma (RCC) constitutes more than 90% of all KC cases and represents the predominant histological subtype driving KC-related mortality. 3 Because RCC typically develops with minimal early symptoms, it is frequently diagnosed incidentally during abdominal imaging, resulting in 11% of patients being identified at stage IV at initial presentation. 4 Advanced (locally unresectable or metastatic) RCC is associated with markedly worse outcomes than localized disease.
Treatment of advanced RCC has evolved rapidly over the past 2 decades. Sunitinib (SUN), a multi-targeted vascular endothelial growth factor receptor (VEGFR) tyrosine kinase inhibitor (TKIs), has long served as a cornerstone first-line treatment for advanced RCC and remains an important comparator in contemporary clinical trials.5-7 The introduction of immune checkpoint inhibitors (ICIs), and subsequently of ICI + TKI combinations, reshaped first-line therapy: dual-ICI regimens (eg, nivolumab + ipilimumab) and ICI + TKI combinations (eg, pembrolizumab + axitinib, nivolumab + cabozantinib, lenvatinib + pembrolizumab) have delivered clinically meaningful improvements in progression-free survival (PFS) and overall survival (OS) compared with SUN in multiple randomized trials.4-9
Benmelstobart (BEN) is a novel humanized immunoglobulin G1 monoclonal antibody targeting PD-L1, designed to enhance antitumor immune responses by blocking the PD-1/PD-L1 signaling pathway.10,11 Anlotinib (AL) is an oral multi-target TKI with potent inhibitory activity against VEGFR, fibroblast growth factor receptor (FGFR), platelet-derived growth factor receptor (PDGFR), and c-Kit, thereby exerting both anti-angiogenic and antiproliferative effects.10,12 The phase 3 ETER100 trial randomized Chinese patients to first-line benmelstobart plus anlotinib (BEN + AL) versus SUN and demonstrated a marked improvement in PFS. 10 In the full analysis set (527 patients), median PFS was 19.0 months (95% CI 15.3-22.8) with BEN + AL versus 9.8 months (95% CI 8.4-12.4) with SUN (hazard ratio [HR] 0.53; 95% CI 0.42-0.67; P < .0001). 10 The 2025 Guidelines of the Chinese Society of Clinical Oncology (CSCO) have incorporated BEN + AL into the recommended first-line treatment strategies for advanced RCC. Nevertheless, the widespread adoption of innovative immune–targeted combinations raise critical concerns regarding affordability and sustainability, particularly in countries with constrained healthcare resources. Although the survival benefits of BEN + AL in patients with advanced RCC are compelling, the high acquisition costs of these agents cannot be ignored. To date, evidence regarding the economic value of BEN + AL remains limited, and its cost-effectiveness relative to established therapies such as SUN has not been fully elucidated within the context of the Chinese healthcare system.
In this context, we performed an integrated analysis that links up-to-date disease burden estimates with a formal economic evaluation. Specifically, we used 2023 Global Burden of Disease (GBD) estimates to update 34-year epidemiological trends of KC incidence and DALY, then performed the cost-effectiveness BEN + AL versus SUN as first-line therapy for advanced RCC in China. By integrating a partitioned survival model (PSM) with the latest GBD 2023 data on KC burden, this study seeks to provide comprehensive evidence on both the clinical and economic implications of adopting BEN + AL. The findings are intended to support evidence-based decision-making for clinicians, policymakers, and payers, and to inform sustainable cancer care strategies in China.
Materials and Methods
Overview
This study was conducted in China and comprised 2 complementary analytical components: (i) a population-level assessment of KC burden and long-term trends, and (ii) a trial-informed pharmacoeconomic evaluation of first-line therapy for advanced RCC from the perspective of the Chinese healthcare system. As illustrated in Figure 1, we first characterized national trends in KC incidence and DALYs from 1990 to 2023 using data from the GBD 2023 Study.13-15 These GBD estimates were used to provide epidemiologic context and decision relevance and were not used to parameterize the economic model. We then constructed a PSM to evaluate the cost-effectiveness of BEN + AL versus SUN as first-line treatment for advanced RCC, based on aggregated efficacy and safety data from the ETER100 phase III randomized controlled trial. 10 Collectively, this dual approach integrates antitumor pharmacoeconomic with the broader public-health burden, informing affordability and resource allocation decisions while preserving the methodological independence of economic and epidemiological assessments.
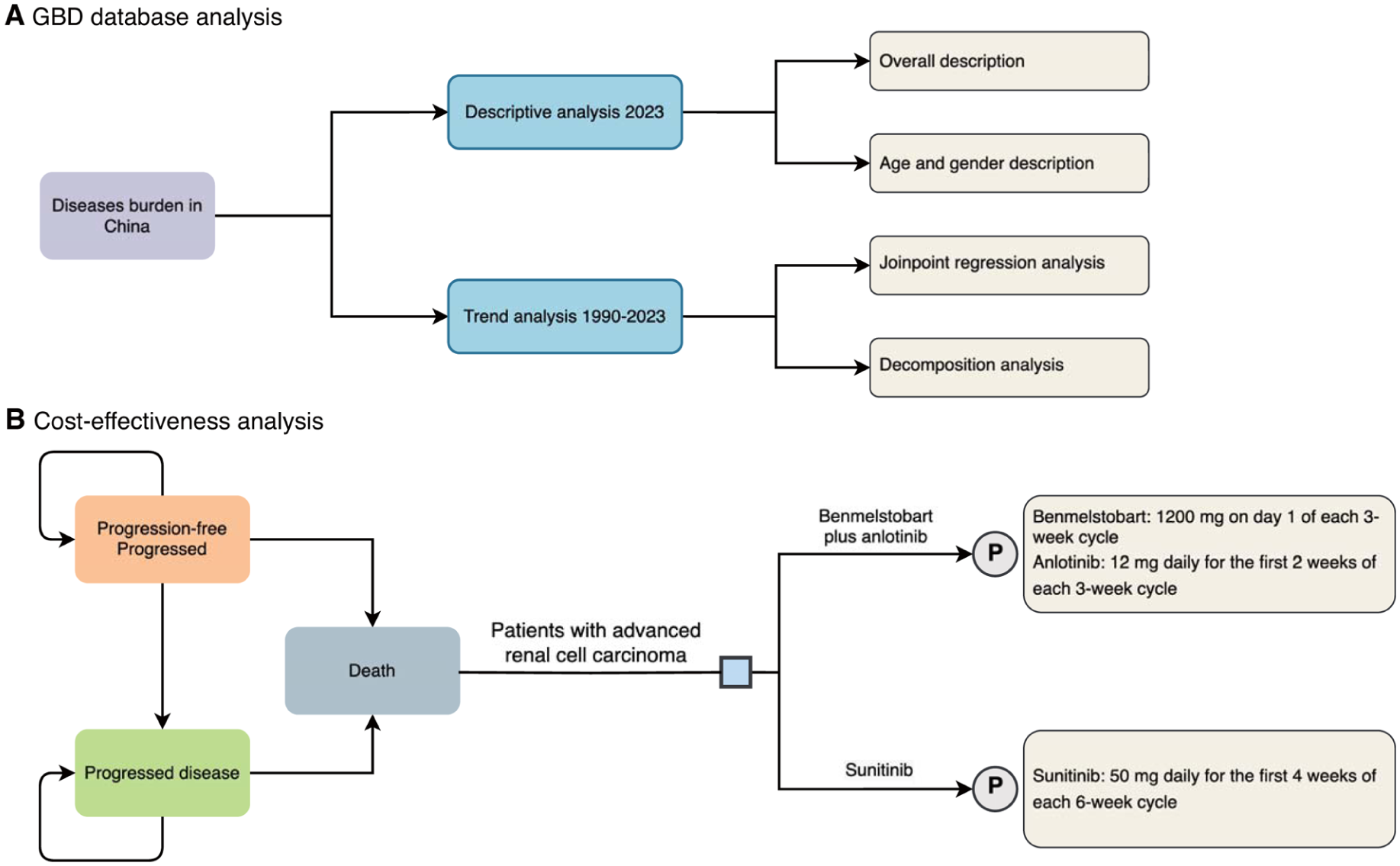
Overall study design: (A) schematic diagram of Global Burden of Disease analysis in China and (B) schematic diagram of 3 state partitioned survival model.
GBD Database Analysis
KC burden data were obtained from the GBD 2023 study, a large-scale international epidemiological initiative that systematically estimates incidence, mortality, and DALYs across diseases, geographic locations, age groups, and sexes using standardized analytical frameworks and comparative risk assessment methods.13-15 For the present population-based epidemiological analysis, we extracted data specific to China and examined incidence and DALYs of KC over the 34-year period from 1990 to 2023 (Figure 1A). Inclusion criteria comprised all cases classified under kidney and renal pelvis malignancies according to the International Classification of Diseases (ICD), including codes C64-C65.9, D30.0-D30.1 and D41.0-D41.1 in ICD-10, and 189.0-189.1, 189.5-189.6, and 223.0-223.1 in ICD-9. 14 Both sexes and all age groups were included to ensure comprehensive national representation. No additional exclusion criteria were applied, as the GBD framework provides standardized, de-identified, and quality-controlled aggregated estimates. All rates were age-standardized to the GBD world standard population and are presented with 95% uncertainty intervals (UIs).
Joinpoint Regression Analysis
Temporal trends in age-standardized incidence and DALY rates of KC in China from 1990 to 2023 were assessed using joinpoint regression analysis. This method identifies statistically significant inflection points (“joinpoints”) at which temporal trends change in direction or magnitude. The maximum number of joinpoints was set to 6, in accordance with the recommendations of the Joinpoint Regression Program for time series comprising more than 30 observation points. 16 The final number of joinpoints was determined automatically using permutation tests implemented in the software, thereby minimizing the risk of overfitting. Annual percentage changes (APCs) and average annual percentage changes (AAPCs) were estimated with corresponding 95% confidence intervals (CIs) under a log-linear model. Statistical significance was evaluated using Monte Carlo permutation tests. All analyses were performed using the Joinpoint Regression Program (version 5.3.0; National Cancer Institute, Bethesda, USA). 17
Decomposition Analysis
Changes in KC incidence and DALYs over time were decomposed using the Das Gupta decomposition method,18,19 which partitions the total change into contributions from population growth, population aging, and changes in age-specific rates. This approach enables clear attribution of observed burden changes to demographic versus epidemiological factors, providing a population-level context for interpreting the value of emerging immunotherapy strategies.
Cost-Effectiveness Analysis (CEA)
This pharmacoeconomic evaluation was conducted in accordance with the Consolidated Health Economic Evaluation Reporting Standards (CHEERS 2022; Supplemental Table 1). 20 Analyses were performed from the perspective of the Chinese healthcare system, with outcomes expressed as total costs, life-years (LYs), quality-adjusted life years (QALYs), incremental net health benefit (INHB), incremental net monetary benefit (INMB), and incremental cost-effectiveness ratios (ICERs). A willingness-to-pay (WTP) threshold was set at 2 times China’s 2024 gross domestic product (GDP) per capita (USD 26 896 per QALY gained). 21 This threshold was selected in accordance with the 2025 China Guidelines for Pharmacoeconomic Evaluations, which recommend using a national WTP threshold of less than 2 times per capita GDP per QALY for incremental analysis. Recent studies indicate that health opportunity cost–based thresholds in China generally remain under twice per-capita GDP.22-24 Consequently, the revised guideline incorporates these findings, replacing the historical 3 times GDP benchmark with a more policy-relevant standard.
Model Population and Inclusion Criteria
The target population of the pharmacoeconomic model comprised patients with advanced RCC, consistent with the eligibility criteria of the ETER100 phase III randomized controlled trial. 10 Patients were assumed to have baseline characteristics representative of the trial population. Key inclusion criteria for the model were therefore defined as: (i) Histologically confirmed advanced RCC with a clear-cell component 10 ; (ii) No systemic therapy for locally advanced/metastatic disease (except for failure/resistance on prior cytokine therapy) 10 ; (iii) Receipt of either BEN + AL or SUN as initial treatment. Key exclusion criteria included: (i) Central nervous system metastases, active hepatitis B or hepatitis C, autoimmune disease, the use of glucocorticoids or immunosuppressants, evidence of active bleeding or bleeding susceptibility, and poorly controlled hypertension 10 ; (ii) Previous therapy with antiangiogenic drugs (such as sunitinib, sorafenib, pazopanib, axitinib, lenvatinib, cabozantinib, bevacizumab, etc.) and related immunotherapies (targeting PD-1, PD-L1, etc.) 10 ; (iii) Adverse events (AEs) of grade <3 or with incidence <5%, which were excluded from costing to avoid overestimation of economic burden.
Model Construction
A PSM was developed to compare BEN + AL with SUN in patients with advanced RCC (Figure 1B). The model included 3 mutually exclusive health states: PFS, progressed disease (PD), and death. All patients were assumed to enter the model in the PFS state. A lifetime time horizon was adopted to capture all relevant long-term costs and health outcomes, assuming that more than 99% of patients would transition to death during the simulation period. 25 This assumption has been widely adopted in prior economic evaluations with lifetime horizons.26 -28 A 21-day cycle length was applied to align with treatment administration schedules, and half-cycle correction was implemented to account for transitions occurring, on average, midway through each cycle. In the ETER100 randomized controlled trial, SUN was administered on a 4-weeks-on/2-weeks-off (4/2) schedule. To maintain consistency with the 21-day model cycle, SUN administration in the base-case analysis was assumed to occur for 14 of 21 days (2-weeks-on/1-week-off, 2/1 schedule). This approach has been adopted in prior pharmacoeconomic modeling studies to ensure structural compatibility between dosing patterns and cycle length. 29 Drug acquisition costs were calculated accordingly. All model calculations were performed using Microsoft Excel 2025.
Clinical Data and Survival Modeling
PFS and OS probabilities were extracted from published Kaplan–Meier (KM) curves of the ETER100 phase 3 trial using WebPlotDigitizer (version 4.8), 30 following the reconstruction approach proposed by Guyot et al. 31 Individual patient data (IPD) were subsequently reconstructed (Supplemental Tables 2 and 3, Supplemental Figures 1 and 2), and survival extrapolation was conducted in R Studio. To characterize long-term survival, multiple parametric distributions, including Weibull, exponential, log-logistic, log-normal, gompertz, and gamma models, were fitted to the reconstructed IPD.32-34 Model selection was guided by quantitative goodness-of-fit metrics (AIC and BIC), together with visual inspection of the overlap between observed KM curves and model-based fits, as well as the shape of long-term extrapolations to avoid implausible survival patterns. 35 Detailed AIC and BIC results are presented in Supplemental Table 4, and the extrapolated OS and PFS curves are shown in Figure 2. For the BEN + AL group, log-logistic distributions demonstrated the most favorable fit for OS, whereas log-normal distributions were selected for PFS (Figure 2, Supplemental Table 5). In the SUN group, log-normal distributions provided the most robust fit for both endpoints (Figure 2, Supplemental Table 5).

Results of the survival curve fit the benmelstobart plus anlotinib group and sunitinib group for 10-year time: (A) OS for the benmelstobart plus anlotinib group, (B) PFS for the benmelstobart plus anlotinib group, (C) OS for the sunitinib group, and (D) PFS for the sunitinib group.
Costs and Utilities
The analysis incorporated direct medical costs, including costs of drugs, follow-up, 36 administration,37,38 management of severe AEs (grade ≥ 3 with ≥5% incidence),27,36,39-41 best supportive care, 42 and end-of-life care (Table 1).36,43 Drug prices were obtained from the YAOZHI database (https://data.yaozh.com/) in 2025, reflecting the most recent nationally available bidding and negotiated prices at the time of analysis. 44 As the full-year 2025 average exchange rate was not yet finalized when the model was developed, all costs were converted into US dollars using the officially reported 2024 annual average exchange rate (USD 1 = CNY 7.12). 45 This approach ensured the use of a stable and complete exchange-rate reference while preserving the most up-to-date drug pricing information.
Parameters of Partitioned Survival Model: Baseline Values, Ranges, and Distributions for Sensitivity Analysis.
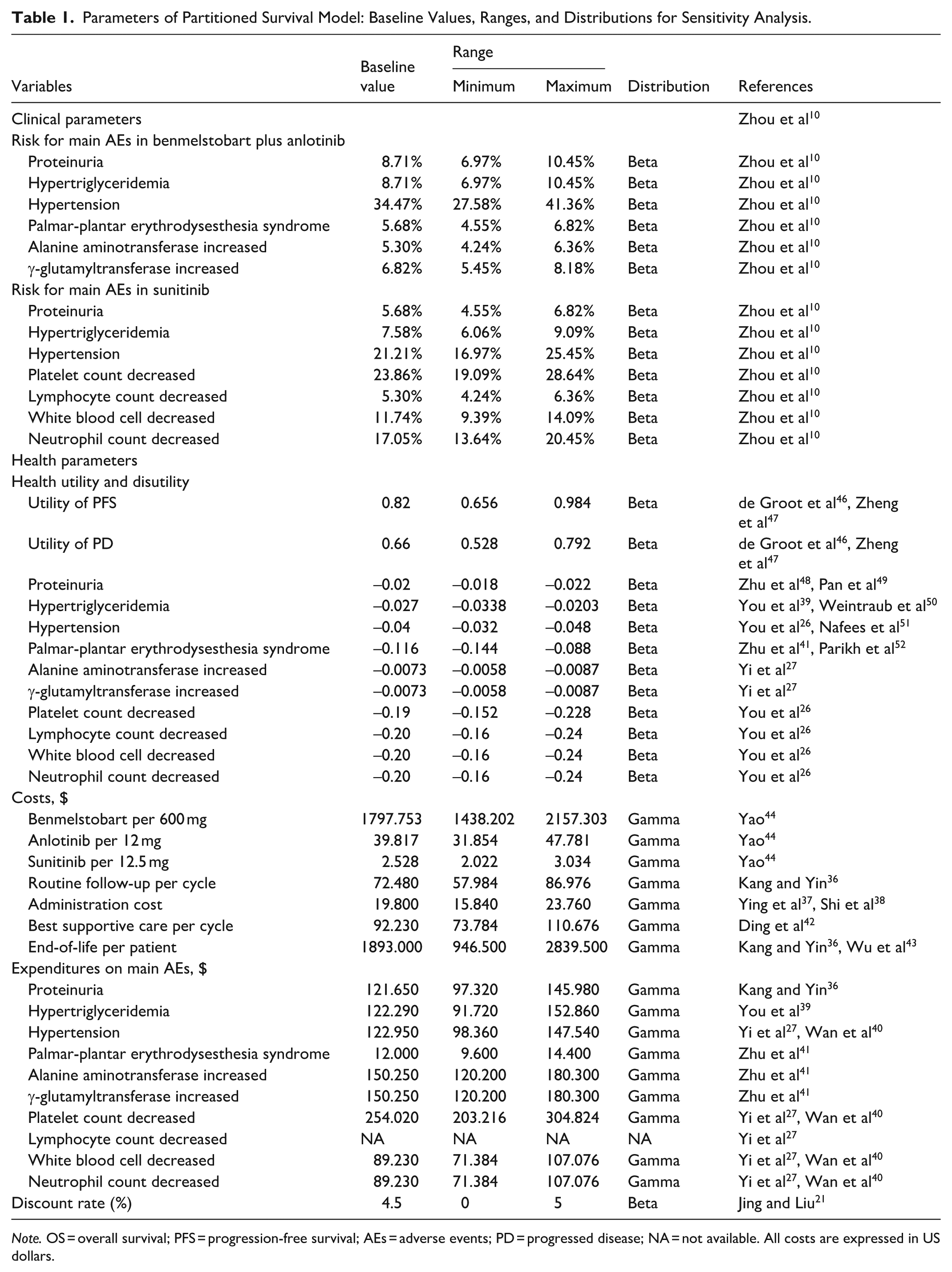
Note. OS = overall survival; PFS = progression-free survival; AEs = adverse events; PD = progressed disease; NA = not available. All costs are expressed in US dollars.
Health state utilities were assigned according to previously published literature on advanced RCC. Utility weights of 0.82 for the PFS state and 0.66 for the PD state were applied, reflecting health-related quality of life differences across disease stages (Table 1).46,47 Utility decrements associated with AEs were applied during the corresponding cycles. Both costs and health outcomes were discounted at an annual rate of 4.5%, in accordance with the 2025 China Guidelines for Pharmacoeconomic Evaluations. 21 All parameters applied in the PSM are summarized in Table 1.
Sensitivity and Scenario Analysis
Deterministic sensitivity analysis (DSA) was conducted to assess the robustness of the base-case results. Plausible ranges for each parameter were derived from published literature or official drug bidding price lists. When empirical ranges were unavailable, parameters were varied by ±20% around their base-case values (Table 1). Probabilistic sensitivity analysis (PSA) was performed using Monte Carlo simulation with 1000 iterations, simultaneously sampling all model parameters from appropriate probability distributions (Table 1).
Scenario analyses were additionally conducted to evaluate the impact of alternative pricing assumptions, dosing schedules, discount rates, and survival extrapolation strategies. Specifically, reductions of 25% (scenario analysis 1), 50% (scenario analysis 2), and 75% (scenario analysis 3) in the price of BEN were examined, alongside alternative discount rates of 3% (scenario analysis 4) and 5% (scenario analysis 5). To address structural uncertainty related to treatment dosing, scenario analysis 6 was conducted using the original 4/2 dosing schedule for SUN as administered in the ETER100 randomized trial. The corresponding SUN drug costs were recalculated based on the 4/2 regimen, and total costs, QALYs, and ICERs were re-estimated to assess the impact of this modeling assumption. Furthermore, to evaluate uncertainty associated with parametric survival model selection, scenario analysis 7 was performed using the second-best fitting parametric distributions (those with the second-lowest AIC and BIC values) for both OS and PFS (Supplemental Table 4), replacing the base-case survival functions. The alternative survival estimates were incorporated into the PSM to recalculate incremental costs, QALYs, and ICERs. These scenario analyses were designed to examine the stability of cost-effectiveness conclusions under both policy-relevant assumptions and methodological uncertainty.
Statistical Software
All data processing, analysis, and visualization were executed using R (version 4.4.1; The R Foundation for Statistical Computing, Vienna, Austria). The packages used for this study included “ggplot2” (version 3.5.1), “tidyverse” (version 2.0.0), “flexsurv” (version 2.3.2) and “survHE” (version 2.0.5). A P-value of less than .05 was deemed statistically significant.
Results
Disease Burden in China
Descriptive and Joinpoint Regression Analyses
In 1990, China recorded 19 500 incident cases of KC, increasing to 61 323 cases in 2023 (Table 2). The age-standardized incidence rates (ASIR) rose from 1.99 per 100 000 to 3.24 per 100 000, consistent with a significant long-term increase (AAPC = 1.55%, 95% CI: 1.27-1.83; Figure 3A). Sex-stratified analyses demonstrated a consistently higher incidence burden among males (Table 2, Figure 3A). Joinpoint regression revealed that the ASIR remained stable during 1990 to 1997, followed by a rapid increase from 1997 to 2005, a continued but slower rise during 2005 to 2010, a transient stable phase during 2010 to 2015, and a renewed increase from 2015 to 2023 (Figure 3A, Supplemental Table 6). Similar temporal patterns were observed in both sexes, although incidence levels and growth rates were consistently higher in males (Figure 3A, Supplemental Table 6).
Incidence and DALYs of Kidney Cancer by Sex in 1990 and 2023, and Their Average Annual Percentage Changes From 1990 to 2023.

Note. DALYs = disability-adjusted life-years; UI = uncertainty interval; ASR = age-standardized rate; AAPC = average annual percentage change; CI = confidence interval.

Joinpoint regression analysis of kidney cancer incidence (A), and DALYs (B) in China by sex from 1990 to 2023. (C) Decomposition of changes in the number of incidences, and DALYs of kidney cancer attributable to population aging, population growth, and age-specific rates by sex in China between 1990 and 2023.
In contrast, DALYs showed a different temporal pattern. Although the total number of DALYs increased from 326 005 in 1990 to 582 244 in 2023, the age-standardized DALY (ASDR) declined slightly from 32.69 per 100 000 to 29.48 per 100 000 (Table 2). The overall AAPC (−0.25%, 95% CI: −0.56 to 0.06) crossed zero, indicating a stable long-term trend. Males consistently exhibited higher DALYs than females in both years, with sex-specific ASDR patterns differing notably (Table 2, Figure 3B). In males, the ASDR remained largely unchanged, decreasing slightly from 43.29 per 100 000 to 41.06 per 100 000, with no significant temporal trend (AAPC = −0.07%, 95% CI: −0.36 to 0.22; Table 2, Figure 3B). By contrast, females experienced a significant decline in ASDR, from 22.29 per 100 000 in 1990 to 18.16 per 100 000 in 2023 (AAPC = −0.63%, 95% CI: −0.90 to −0.36; Table 2, Figure 3B). Joinpoint results revealed alternating periods of decline and increase (Figure 3B. Supplemental Table 6). For both sexes combined, ASDR decreased significantly during 1990 to 1997 and 2010 to 2015, increased during 1997 to 2004, and remained stable from 2015 to 2023 (Figure 3B. Supplemental Table 6). Male ASDR declined during 1990 to 1997 and 2004 to 2017, followed by a stable phase after 2017 (Figure 3B, Supplemental Table 6). Females experienced larger early declines, a sharper increase during 1997 to 2003, and a significant reduction during 2009 to 2015, with stability thereafter (Figure 3B, Supplemental Table 6).
Decomposition Analysis Between 1990 and 2023
Decomposition analysis showed that the number of incident KC cases increased by 41 823 between 1990 and 2023 in China, representing a 214.48% rise (Figure 3C, Supplemental Table 7). Epidemiological change was the dominant driver (91.65%), followed by population aging (86.09%) and population growth (36.75%). Sex-specific analyses indicated similar patterns. In males, incident cases increased by 239.67%, with aging (101.84%) and epidemiological change (100.38%) contributing almost equally (Figure 3C, Supplemental Table 7), whereas in females, the increase (173.24%) was mainly driven by epidemiological change (77.09%), with smaller contributions from aging (61.42%) and population growth (34.73%).
In contrast to incidence, the decomposition of DALYs demonstrated a distinct structure (Figure 3C, Supplemental Table 7). The overall increase in DALYs (78.60%) was predominantly driven by population aging, which accounted for approximately 62.86% of the total change, followed by population growth (26.09%). Notably, epidemiological change exerted a mitigating effect, contributing a relative 10.35% reduction in DALYs (Figure 3C, Supplemental Table 7). This pattern was consistent across sexes. In males (91.02%), population aging accounted for 69.91% of the increase in DALYs, with population growth contributing approximately 26.09%, while epidemiological change reduced DALYs by about 5.15%. Among females (54.58%), the contribution of population aging was comparatively smaller (49.45%), whereas the offsetting effect of epidemiological change was more substantial, accounting for an approximate 19.72% reduction in DALYs.
Sex and Age Specific Burden in 2023
In 2023, KC incidence in China exhibited 2 distinct peaks across the 20 age groups (Figure 4, Supplemental Table 8). A smaller peak occurred in early childhood (0-4 years), with 2636 cases and an age-specific incidence rate of 4.24 per 100 000 (Supplemental Table 8). The second, more pronounced peak was observed in older adults, with the largest number of incidence (7905) at 65 to 69 years and the highest age-specific incidence rate at 85 to 89 years (14.85 per 100 000). DALYs followed a similar bimodal pattern, with early childhood contributing 16 153 DALYs, while the largest DALYs was observed in adults aged 55 to 59 years (76 488) and highest an age-specific DALYs rate at 75 to 79 years (131.69 per 100 000; Supplemental Table 8). Across nearly all age groups, males consistently exhibited higher incidence and DALYs than females, with the sex differences particularly pronounced in older adults (Figure 4, Supplemental Table 8). For example, males aged 75 to 79 years had an age-specific incidence rate of 20.91 per 100 000, compared with 9.19 per 100 000 in females, and at 80 to 84 years, rates were 21.46 versus 8.88 per 100 000, respectively (Figure 4A and B, Supplemental Table 8). Regarding DALYs, the greatest male–female differences were observed in the 70 to 79 age range, where males aged 75 to 79 had an age-specific DALY rate of 186.23 per 100 000 compared with 83.63 in females, and at 70 to 74 years, 144.13 versus 79.55. In early adulthood (50-69 years), males also had substantially higher DALY rates than females, while in early childhood, females showed slightly higher rates (Figure 4C and D, Supplemental Table 8).
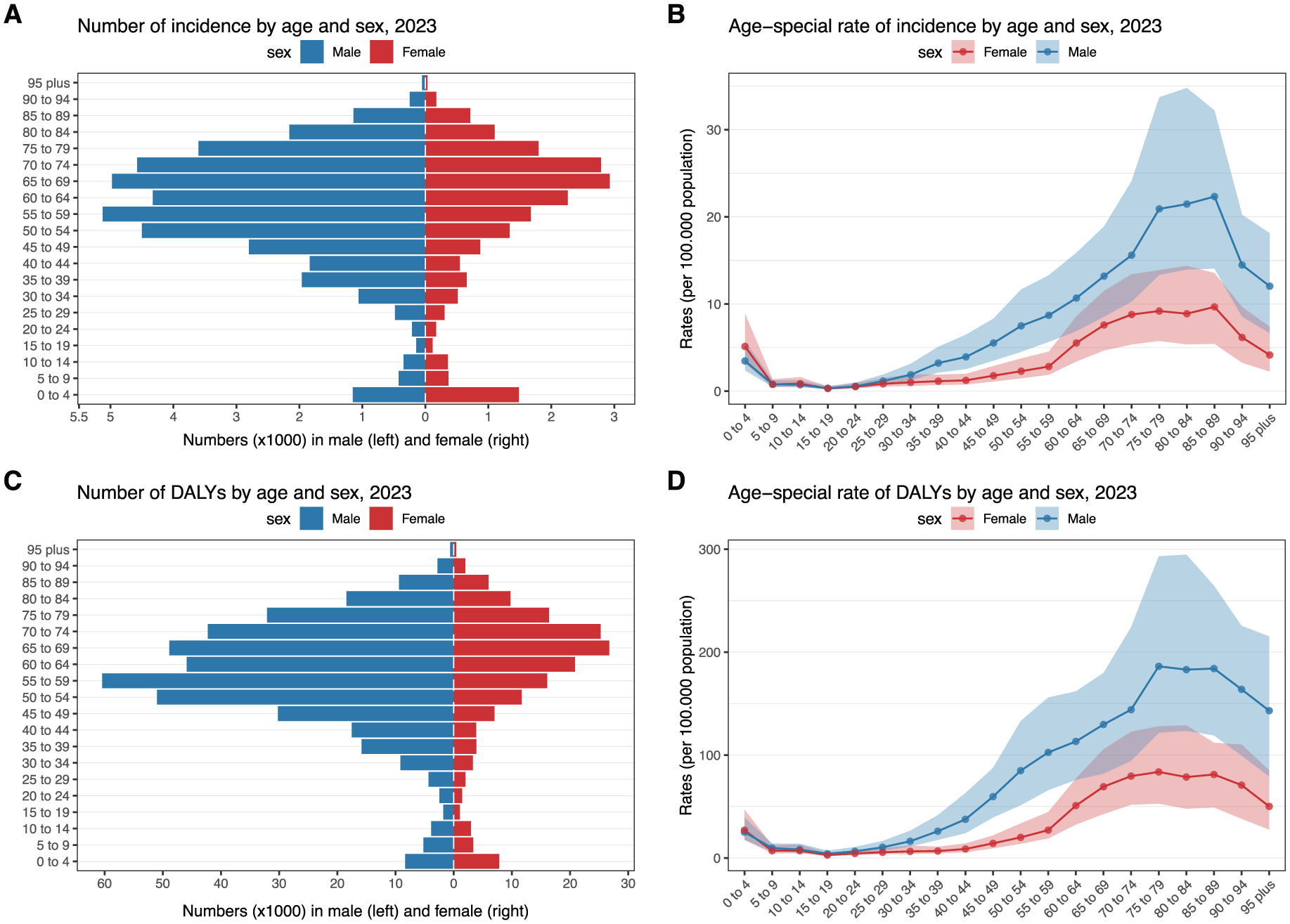
The incidence, and DALYs of kidney cancer by age and sex in China, 2023: (A) number of incidence, (B) age-special rate of incidence, (C) number of DALYs, and (D) age-special rate of DALYs.
Cost-Effectiveness Analysis
Basic Analysis Results
The base-case analysis results are summarized in Table 3. Over the modeled time horizon, treatment with BEN + AL was associated with higher total costs and greater health benefits than SUN. Specifically, the BEN + AL strategy accrued a total cost of $192 292.58, yielding 4.67 LYs and 3.48 QALYs, whereas SUN resulted in a substantially lower total cost of $11 400.59, with 3.81 LYs and 2.73 QALYs. Compared with SUN, BEN + AL generated incremental gains of 0.86 LYs and 0.75 QALYs, at an additional cost of $180 891.99. This translated into an ICER of $240 961.36 per QALY gained, indicating a substantial increase in expenditure per unit of health benefit. Consistent with this finding, both the INHB and INMB were negative, with values of –5.97 QALYs and –$170 796.52, respectively, suggesting that BEN + AL did not provide an economic advantage over SUN under the assumed willingness-to-pay threshold.
The Results in the Base-Case and Scenario Analyses.

Note. LY = life-year; QALY = quality-adjusted life year; ICER = incremental cost-effectiveness ratio; INMB = incremental net monetary benefits; INHB = incremental net health benefits; BEN + AL = benmelstobart plus anlotinib; SUN = sunitinib.
Sensitivity Analyses Results
The results of the one-way sensitivity analysis are presented in the tornado diagram (Figure 5A). The utility of PFS was the most influential parameter, with the ICER ranging from $201 276.44 to $300 138.55 when varied across its plausible lower and upper bounds. The cost of BEN (per 1200 mg) was the second most influential determinant, yielding ICERs between $198 392.95 and $283 529.77. In addition, the cost of anlotinib (per 12 mg) and the discount rate exerted moderate effects on the ICER, with corresponding ranges of $234 361.59-$247 561.13 and $207 125.75-$244 698.71, respectively. By contrast, variations in other parameters, including the utility of PD, the cost of SUN, adverse event–related utilities and disutilities, and costs related to administration and routine follow-up, had relatively limited impact on the ICER. Importantly, for all parameters examined and throughout their predefined ranges, the ICER consistently exceeded the WTP threshold of $26 896 per QALY.

(A) One-way sensitivity analysis tornado diagrams showing the parameters with the greatest impact on the ICER of the BEN + AL group compared with the SUN group. (B) A probabilistic scatter plot of the ICER between the BEN + AL group and the SUN group. (C) The cost-effectiveness acceptability curves for the BEN + AL group compared with the SUN group.
The results of PSA are illustrated by the incremental cost-effectiveness scatter plot and the cost-effectiveness acceptability curves (Figure 5B and C). In the scatter plot, most of the 1000 Monte Carlo simulation results were located in the northeast quadrant of the cost-effectiveness plane, indicating higher costs and greater effectiveness for BEN + AL relative to SUN (Figure 5B). Consistent with this finding, the cost-effectiveness acceptability curve showed that, at WTP thresholds up to $135 000 per QALY, the probability of BEN + AL being cost-effective remained 0%, while SUN alone retained a 100% probability of being the preferred strategy (Figure 5C). As the WTP threshold increased, the probability of BEN + AL being cost-effective rose gradually, reaching approximately 50.7% at a threshold of $240 000 per QALY. At substantially higher thresholds, this probability continued to increase, exceeding 80% at around $280 000 per QALY and approaching 99.8% at $500 000 per QALY.
Scenario Analyses Results
The results of the scenario analyses are summarized in Table 3. Across all scenarios, BEN + AL consistently generated higher LYs and QALYs than SUN, with incremental gains ranging from 0.60 to 0.96 LYs and from 0.56 to 0.82 QALYs. However, these health benefits were accompanied by substantial incremental costs, resulting in ICERs that remained above the prespecified WTP threshold in all scenarios.
Discussion
In this study, we conducted 2 complementary analyses to inform KC control and treatment decision-making in China: a population-level assessment of KC burden and long-term trends using GBD 2023 estimates, and a trial-informed pharmacoeconomic evaluation comparing BEN + AL with SUN as first-line therapy for advanced RCC. Our burden findings show a substantial rise in KC incidence from 1990 to 2023, with annual new cases more than tripling and total DALYs nearly doubling. The ASIR increased from approximately 2.0 to 3.2 per 100 000 during this period, whereas the ASDR remained relatively stable. Decomposition analyses indicate that population aging was the dominant driver of the increasing burden, and older adults and males bore a disproportionately higher burden. These national burden patterns provide a population-level backdrop for interpreting the economic results, as the growing and aging patient population may amplify affordability pressures even when per-patient clinical benefits are substantial. Against this backdrop, our CEA showed that BEN + AL provides greater survival benefits than SUN, but at a dramatically higher cost, yielding an ICER of approximately $240 000 per QALY. This value far exceeds the assumed WTP threshold of $26 896 per QALY in China. Thus, despite its clinical benefit, the BEN + AL regimen is unlikely to be cost-effective as a first-line treatment for advanced RCC under current pricing. Sensitivity analyses and scenario analyses confirmed the robustness of the model outputs.
GBD 2023 Data
Our analysis of GBD 2023 data reveals a persistent rise in incidence in China, evident in both absolute case numbers and ASR. This increase likely reflects a combination of factors associated with socioeconomic development, including improved diagnostic imaging (leading to more incidental tumor detections) and greater exposure to established risk factors such as smoking, obesity, and hypertension. 53 The higher incidence observed in males, which aligns with prior studies, is supported by a global analysis revealing a stable male-to-female incidence ratio of approximately 2:1 for KC across all ages and regions. 54 This suggests that biological or genetic factors, rather than solely behavioral factors such as smoking, are primarily responsible for the sex disparity. 54 These epidemiological patterns reinforce the need for targeted prevention strategies, particularly among high-risk male populations, focusing on risk-factor modification and early detection. In contrast to incidence, the ASDR for KC showed no significant long-term increase, even as total DALYs nearly doubled between 1990 and 2023. This paradox could be attributed enhanced early detection and advancements in surgical techniques, systemic therapies, and overall cancer care. In China, the 5-year relative survival rate for KC improved from roughly 62% in 2003 to 2005 to nearly 70% by 2012 to 2015. 55 Such gains in survival mean that while more individuals are being diagnosed with KC, a smaller proportion are dying from it, thereby stabilizing the population-level DALY rates. Our decomposition analysis further supports this interpretation: the growth in total DALYs was driven largely by population growth and aging, partially offset by epidemiological improvements (reductions in per-case fatality or severity). These trends underscore the dual challenge of managing a growing patient population and reducing the severity or duration of disease through better care. In advanced RCC, while more patients are being diagnosed due to these demographic and epidemiological trends, ensuring that new therapies effectively reduce mortality and morbidity is vital to contain DALYs.
Age- and sex-specific patterns in 2023 further underscore the concentration of KC burden in older males. We observed a bimodal distribution of KC burden, with a minor peak in early childhood and a major peak in older adults. The incidence and DALYs are disproportionately concentrated in middle-aged and older adults, with both rates steadily increasing after age 50 and peaking in the seventh and eighth of life. Males had substantially higher incidence and DALY rates than females across almost all adult age groups, and this gap was most pronounced in the older ages. For example, among those aged 75 to 79, the incidence rate in men was more than double that in women. These findings underscore the need for targeted preventive measures, such as smoking cessation programs for high-risk men, early screening strategies, and investment in geriatric oncology to mitigate the growing burden.
Cost-Effectiveness Analysis
Within this context of a rising disease burden, our CEA provides timely insight into the value of a new first-line strategy for advanced RCC in China. We recognize that contemporary first-line RCC treatment increasingly includes multiple ICI–based combinations. However, SUN remains the standard comparator in pivotal trials, enabling consistent benchmarking of clinical benefits across regimens. This approach is exemplified by 3 phase 4 studies: CheckMate 214 (Nivolumab plus ipilimumab vs SUN), CLEAR (lenvatinib plus pembrolizumab vs SUN), CheckMate 9ER (nivolumab plus cabozantinib vs SUN), and KEYNOTE-426 (pembrolizumab plus axitinib vs SUN), each employing SUN as the common reference for final prespecified overall survival analyses.5-8 Against this evidence-aligned comparator framework, our base-case results show that BEN + AL increased 0.86 LYs and 0.75 QALYs versus SUN, echoing the OS benefit reported in clinical trials. However, these gains came at an incremental cost of nearly $181 000, leading to an ICER of approximately $240 961 per QALY gained. This ICER substantially exceeds the nationally recommended WTP threshold in China. According to the 2025 China Guidelines for Pharmacoeconomic Evaluations, 21 incremental analyses should adopt a threshold of less than 2 times per capita GDP per QALY, reflecting updated empirical estimates of health opportunity cost.22-24 Under this benchmark ($26 896 per QALY), BEN + AL yielded negative INHB and INMB. This indicates that, under current pricing, the combination regimen does not represent an efficient use of healthcare resources in the Chinese system. Sensitivity analyses confirmed the robustness of this conclusion. The DSA identified the utility value in the progression-free state and the cost of BEN as the most influential parameters on the ICER. Even under optimistic assumptions (for instance, using the highest plausible quality-of-life utility for PFS or substantially lowering the drug price), the ICER remained well above the $26 900 per QALY threshold. Likewise, the PSA showed that at WTP levels up to 5 times the current threshold, the probability of BEN + AL being cost-effective was essentially zero. Furthermore, scenario analyses consistently yielded ICERs above the threshold. These results underscore that without substantial price reductions or innovative financing strategies, broad adoption of BEN + AL would pose serious affordability challenges.
Our findings are in line with a growing body of evidence on immunotherapy-based combination treatments for cancers. A systematic review by Mahumud et al identified 69 economic evaluations of adjuvant immunotherapy published between 2015 and 2025. 56 Of these, only 40 studies (58%) reported favorable cost-effectiveness results. In China, several novel combination therapies have also demonstrated unfavorable cost-effectiveness when compared to the traditional standard.57-59 For example, amivantamab-lazertinib versus osimertinib in EGFR-mutated non-small cell lung cancer achieved 1.11 additional QALYs at $1 342 374 incremental cost (ICER $1 211 236 per QALY), 33-fold above the $36 887 WTP threshold. 58 Similarly, cadonilimab plus chemotherapy as first-line treatment for persistent, recurrent, or metastatic cervical cancer provided 0.61 additional QALYs versus chemotherapy alone at an incremental cost of $42 486.54. 57 However, this resulted in an ICER of $70 220.88 per QALY, surpassing the WTP threshold of $38 042 per QALY. 57 Furthermore, recent studies indicate that adding BEN + AL to etoposide/cisplatin as first-line therapy for extensive-stage small-cell lung cancer lacks cost-effectiveness relative to chemotherapy alone.26,60,61 A price reduction of 80% would be necessary to satisfy Chinese WTP thresholds. 40 Our analysis of BEN + AL aligns with this general trend, reinforcing the notion that price remains a decisive factor in the value proposition of innovative cancer therapies. Looking ahead, as China’s economy grows and healthcare spending increases, higher affordability thresholds and policy interventions (such as negotiated drug price cuts or patient assistance programs) could improve the cost-effectiveness of BEN + AL.
Strength
A notable strength of this work is the use of 2 complementary analyses—a population-level characterization of KC burden using GBD 2023 estimates and a trial-based cost-effectiveness model—to inform decision-making in China. As China’s population ages and more patients develop RCC, even a highly efficacious therapy can place unsustainable strain on healthcare budgets if priced exorbitantly. Our study therefore underscores the need to evaluate new treatments not only on their clinical outcomes but also on their affordability and opportunity costs in the real-world population. These considerations point to several strategies for balancing innovation with sustainability. One approach is to address drug pricing: substantial price reductions through national negotiation programs or value-based pricing agreements would directly improve the cost-effectiveness of BEN + AL. Indeed, price negotiation policies have consistently reduced anticancer medication costs across multiple studies.62-64 Zhou et al reported in 2024 that median treatment costs decreased substantially following price negotiation, from US$34 460.72 to US$13 688.79. 62 Similarly, Zhang et al demonstrated in 2021 that the 2017 medication price negotiation and reimbursement policy reduced average cost per defined daily dose (DDD) of 15 targeted anticancer agents by US$71.21, from US$169.24/DDD. 63 Cai et al further showed that 17 innovative anticancer medicines experienced an immediate DDD cost reduction of US$109.09 (95% CI 68.14-150.05; P < .001) upon implementation of China’s national drug price negotiation policy, with subsequent cost stability. 64 Similar negotiations for BEN + AL could bring their cost closer in line with the health benefits achieved, potentially making the combination an economically reasonable option. Another strategy is to strengthen prevention and early detection efforts. Effective tobacco control, weight management programs, and hypertension screening and treatment would not only reduce KC incidence but also confer broad health benefits. Overall, healthcare resources are finite and directing them toward interventions that provide the most health benefit for the cost is essential for maximizing population health.
Limitation
Several limitations of our analysis should be acknowledged. First, the cost-effectiveness model relied on clinical efficacy and safety data from a single phase III randomized controlled trial (ETER100), and survival beyond the observed follow-up period required parametric extrapolation. Although multiple parametric distributions were tested and selected based on goodness-of-fit criteria and clinical plausibility, long-term survival projections remain inherently uncertain. Different extrapolation assumptions could yield alternative estimates of lifetime benefits and ICERs. In addition, the PSM assumes that transitions between health states are determined solely by PFS and OS curves, without explicitly modeling state-to-state transition probabilities. This framework implicitly assumes independence between health states and does not account for potential heterogeneity in post-progression pathways, which may oversimplify real-world disease trajectories. Second, we derived health state utility values and some cost inputs from published literature rather than direct assessment in Chinese RCC patients. If these inputs do not accurately reflect the quality of life or medical costs in the Chinese setting, our results may be biased. Future studies would benefit from collecting country-specific utility measurements and real-world cost data for advanced RCC. Third, our analysis took the perspective of the healthcare payer and thus did not include indirect costs, which could be substantial for society, especially if younger patients are affected. Fourth, the GBD 2023 data we used for epidemiological trends are subject to uncertainties, particularly for earlier years when cancer reporting in China was less comprehensive. While these data enable assessment of long-term patterns, caution is warranted when interpreting the precise magnitude of changes over time. Fifth, our pharmacoeconomic model was limited to a comparison of BEN + AL versus SUN. Robust comparisons between BEN + AL and other ICI-based regimens would require either direct head-to-head randomized trials (currently unavailable) or a formal network meta-analysis/indirect comparison with careful handling of cross-trial heterogeneity, aligned endpoints, and harmonized China-specific cost assumptions across regimens. This represents an important priority for future research to better inform regimen selection and sequencing. Finally, our recommendations regarding price reduction and policy interventions are exploratory. While we suggest that significant price cuts could make the regimen cost-effective, determining the exact price threshold for cost-effectiveness would require further analysis.
Conclusion
In conclusion, China faces a growing KC burden primarily driven by an aging population. Meanwhile, the BEN + AL combination, despite extending survival for patients with advanced RCC, is unlikely to be cost-effective as a first-line therapy under current pricing. By combining epidemiological insights with pharmacoeconomic evaluation, our study provides a comprehensive evidence base to inform cancer control strategies. These findings emphasize the importance of aligning new therapeutic innovations with considerations of value and affordability. Striking a balance between innovation and sustainability will be crucial to ensure that advances in KC treatment translate into meaningful and equitable health gains for patients and society at large.
Supplemental Material
sj-pdf-1-inq-10.1177_00469580261440346 – Supplemental material for Benmelstobart Plus Anlotinib Is Unlikely to Be Cost-Effective for Advanced Renal Cell Carcinoma: An Integrated Disease Burden and Cost-Effectiveness Analysis
Supplemental material, sj-pdf-1-inq-10.1177_00469580261440346 for Benmelstobart Plus Anlotinib Is Unlikely to Be Cost-Effective for Advanced Renal Cell Carcinoma: An Integrated Disease Burden and Cost-Effectiveness Analysis by Haiyan Xu, Liejiong Wang, Ying Lou, Zhaoqi Qiu and Feng Xuan in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Acknowledgements
We extend our thanks to the Global Burden of Disease Study 2023 collaborators for their high-quality data.
Ethical Considerations
This study was conducted in accordance with the Declaration of Helsinki (2013 revision). As this research comprises (i) a secondary analysis of publicly available data from the Global Burden of Disease (GBD) database, and (ii) a cost-effectiveness analysis based on aggregated efficacy and safety data from published phase III clinical trial results, without direct human subject involvement or access to identifiable personal information in either component, institutional ethical review was not required.
Author Contributions
Haiyan Xu: contributed to conceptualization, formal analysis, methodology, software, visualization, validation, and writing original draft, review and editing. Liejiong Wang: contributed to conceptualization, investigation, formal analysis, methodology, and writing original draft. Ying Lou: contributed to conceptualization, investigation, methodology, validation, and writing original draft. Zhaoqi Qiu: contributed to conceptualization, methodology, and writing original draft. Feng Xuan: contributed to data curation, project administration, supervision, software, validation, and writing original draft, review and editing. All the authors have read and approved the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
