Abstract
Metabolic-associated fatty liver disease (MAFLD) is a prevalent condition associated with significant hepatic morbidity, yet evidence-based community interventions remain limited. This mixed-methods study first assessed self-management capacity and quality of life (QOL) among MAFLD patients (n = 218) in community settings, revealing suboptimal self-management (mean score 2.84 ± 0.73) and moderate QOL (3.23 ± 0.77), with education level and occupation identified as predictors. Subsequently, a randomized controlled trial (RCT) evaluated a novel community-based multidisciplinary intervention (nutritional counseling, behavioral coaching, clinical monitoring) versus standard education (n = 100). At 3-month follow-up, the intervention group demonstrated clinically significant improvements versus controls: greater weight reduction (−9.76 vs 0.11 kg; P = .028), body mass index (BMI) decrease (−3.01 vs 0.28 kg/m²; P = .006), and QOL enhancement (+8.97 vs. +2.83 points; P = .002). Significant metabolic and hepatic improvements included reduced liver steatosis (controlled attenuation parameter [CAP]: −29 dB/m; P = .001), lower liver enzymes (alanine aminotransferase [ALT]: −12 U/L, P = .039; aspartate aminotransferase [AST]: −5.5 U/L, P = .015), and decreased fasting glucose (−0.97 mmol/L; P = .026). This scalable multidisciplinary model effectively enhances self-management adherence, improves metabolic health and hepatic parameters, and boosts QOL in MAFLD, offering a resource-efficient strategy for reducing chronic disease burden in primary care.
Keywords
Introduction
Metabolic-associated fatty liver disease (MAFLD) places a substantial daily burden on millions globally, causing fatigue, reduced productivity, and anxiety about disease progression. In China alone, it affects 30.4% of adults, yet only 33% of at-risk individuals are aware of their condition.1,2 This gap leaves patients struggling to manage their health amid confusing nutritional information and sedentary lifestyles.3,4 Our research directly addresses these human challenges through a patient-centered intervention designed to simplify daily self-management. By integrating practical tools like WeChat-based tracking with community health worker support, we empower patients to take control of their health in culturally relevant ways.5 -7
MAFLD’s progression to serious liver complications constitutes a critical global health challenge, with annual economic costs exceeding $100 billion. 8 Its silent early stages and complex self-care requirements create particular vulnerabilities for underserved populations.9 -11 While self-management programs have transformed diabetes and hypertension care, MAFLD-specific approaches remain scarce—especially in resource-limited settings.12,13 Grounded in behavioral change theory, our intervention systematically tackles 3 barriers: (1) knowledge gaps about disease-lifestyle connections, (2) limited self-monitoring skills, and (3) socioeconomic obstacles to sustained habit change.14 -17 We achieve this through metabolic education, goal-setting tools, and community-based peer support networks.14 -17
The study rigorously assesses our multidisciplinary model using clinical indicators (liver stiffness measurements, blood markers) alongside patient-reported outcomes like quality of life.18,19 Our 90-day evaluation examines lasting impacts while identifying adherence patterns across different patient groups. 20 By bridging hospital-based diagnostics with community-delivered behavioral support, this approach establishes sustainable MAFLD management ecosystems accessible beyond specialized clinics.21,22 Ultimately, we provide evidence for scalable metabolic health management—a cost-effective solution for regions confronting the dual challenges of nutritional transition and healthcare constraints.23,24
Methodology
This mixed-methods study integrated a cross-sectional survey with a randomized controlled trial. A cohort of 220 eligible participants (age 18-65 years, confirmed MAFLD diagnosis, intact cognition) underwent baseline assessments. Sample size was calculated using variable-to-respondent ratios (1:10) and power analysis (α = .05, β = .2, δ = .5 standard deviation), accounting for an anticipated 20% attrition rate. For the RCT phase, 100 participants were randomized via stratified block design (1:1 allocation, computer-generated sequence) to either the intervention group (n = 50) or standard care control (n = 50). Allocation concealment used sequentially numbered opaque envelopes. Final analysis included 80 participants (40 per group) after attrition, maintaining sufficient statistical power (1−β = .8) to detect clinically meaningful effect sizes. Outcome assessors and statisticians were blinded, though participant blinding was limited by the behavioral nature of the intervention. Primary outcomes (self-management capacity, QOL) were assessed using validated instruments and analyzed via intention-to-treat principles. Methodological rigor was ensured through eligibility screening, covariate adjustment, and standardized protocols. Figure 1 presents the Consolidated Standards of Reporting Trials (CONSORT)-compliant participant flowchart.

Research flowchart.
The intervention group received a 12-week program implemented through 3 pillars: (1) structured psychoeducation targeting disease misconceptions, (2) technology-enhanced self-regulation using Specific, Measurable, Achievable, Relevant, Time-bound (SMART) goals and digital trackers, and (3) moderated peer engagement via WeChat support groups. The 12-week duration was selected to align with established metabolic response timelines in lifestyle interventions and enable assessment of sustained behavioral change.25,26 Implementation fidelity was maintained through weekly adherence feedback (month 1) and longitudinal biomarker monitoring (month 3), with full protocol specifications detailed in Table 1. Control participants received standard MAFLD pamphlets with monthly educational materials, reflecting routine practice. This pragmatic design facilitated direct comparison of multidisciplinary care versus conventional approaches.
Multidimensional Intervention Framework Based on Integrated Theory of Health Behavior Change.

Outcome measurements spanned clinical biomarkers (anthropometrics via SECA 284 scales, hepatic/metabolic panels using Cobas 8000 analyzers, FibroScan B0X9 assessments with body mass index [BMI]-adjusted probes), psychometric evaluations (31-item MAFLD Self-Management Questionnaire [α = .927], WHO-aligned 26-item QoL scale [α = .922]), and digital behavioral tracking through a WeChat-based AI dietary logger.27,28 Potential self-report bias was mitigated through triangulation with clinical biomarkers and digital tracking data. 29 Rigorous quality control protocols governed data collection: fasting blood samples (>8 h) collected by certified phlebotomists, elastography by hepatologists with ≥5 years’ FibroScan experience, and triphasic assessments (baseline/30-day/90-day) ensuring temporal consistency. 30 A dual-data system integrated Questionnaire Star platform responses with hospital electronic health records, cross-validated by independent researchers.
This methodology balances scientific rigor with real-world applicability through 3 key innovations: (1) blended digital-clinical data capture enabling multidimensional outcome analysis, (2) behavioral intervention components tailored to China’s mobile health ecosystem, and (3) equity-sensitive implementation addressing literacy and access barriers. While limited by single-center recruitment and short-term follow-up, the design provides a template for evaluating complex interventions in chronic metabolic disease management.
Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics version 29.0. Scale reliability was evaluated through Cronbach’s alpha coefficients, confirming robust internal consistency across all measurement instruments. Descriptive statistics summarized demographic profiles, self-management capabilities, and quality-of-life indicators, with continuous variables expressed as mean ± standard deviation and categorical variables as frequencies (percentages). Missing data were addressed using listwise deletion, with most variables exhibiting ≤5% missingness. This level was considered unlikely to introduce significant bias. Notably, objective indicators (e.g., ALT, CAP) contained no missing data. Comparative analyses utilized independent samples t-tests or 1-way analysis of variance (ANOVA) for normally distributed data, with non-parametric alternatives (Mann-Whitney U, Kruskal-Wallis) applied when parametric assumptions were violated. Bivariate relationships between self-management capacity and quality of life were examined using Pearson correlation coefficients. Multivariate stepwise regression analysis identified determinants of self-management capacity. Moderating effects of disease duration on self-management and quality-of-life relationships were tested using the SPSS PROCESS macro (version 4.2). Intervention efficacy was assessed via chi-square tests for categorical outcomes and independent *t*-tests or Mann-Whitney U tests for continuous measures, contingent on distributional properties. All analyses maintained a 2-tailed significance threshold of α = .05, with appropriate corrections for multiple comparisons. Effect sizes were calculated to complement statistical significance testing, providing comprehensive assessment of clinical meaningfulness.
Results
Participant Characteristics
This cohort exhibited a substantial metabolic burden, with 84.9% classified as overweight or obese. Sociodemographic profiling revealed predominantly married participants (79.8%) with a bachelor’s degree or higher education (66.5%; Supplemental Table C1).
For the subsequent randomized controlled trial phase, baseline assessments confirmed no significant differences (P > .05) between the intervention and control groups across key domains. These included anthropometric measures (BMI, waist circumference), biochemical parameters (ALT, AST, lipid profiles), and functional assessments encompassing comprehensive self-management capacity scores and quality-of-life metrics. This established essential comparability prior to intervention initiation(Supplemental Table C2).
Self-Management Capacity and Quality of Life Association Mechanisms
Baseline assessments revealed suboptimal self-management capacity across the cohort, with mean scores falling below the moderate threshold (<3 points), while quality of life (QoL) metrics indicated moderate-to-above-average levels (slightly > 3 points; Supplemental Table C3).
Significant positive correlations emerged between self-management capacity and quality of life (r = .441, P < .01), with particularly strong associations in psychological (r = 0.453-0.553) and environmental domains (r = 0.523-0.838; Supplemental Table C4).
Multivariate regression analysis identified educational attainment (β = .59, P < .01) and occupational status (β = .11, P < .05) as independent predictors of self-management capacity, collectively explaining 38% of the observed variance (R² = 0.38, F = 11.33, P < .001; Table 2).
Multifactorial Regression Analysis of Patient Self-Management Ability.

Bold statistical values: Their corresponding p-values are statistically significant (i.e., p < 0.05 or p < 0.01).
Indicates p < 0.05, meaning the result is statistically significant at the 0.05 level.
Indicates p < 0.01, meaning the result is statistically significant at the 0.01 level.
Crucially, disease duration significantly moderated the relationship between self-management capacity and QoL outcomes. The beneficial impact of self-management on QoL was most pronounced among patients with 3 to 5 years of disease duration (coefficient = 0.466, P < .001), whereas this positive association attenuated substantially beyond 10 years’ duration, showing non-significant negative moderation (coefficient = –.092, P = .542; Supplemental Table C5).
Longitudinal Intervention Outcomes
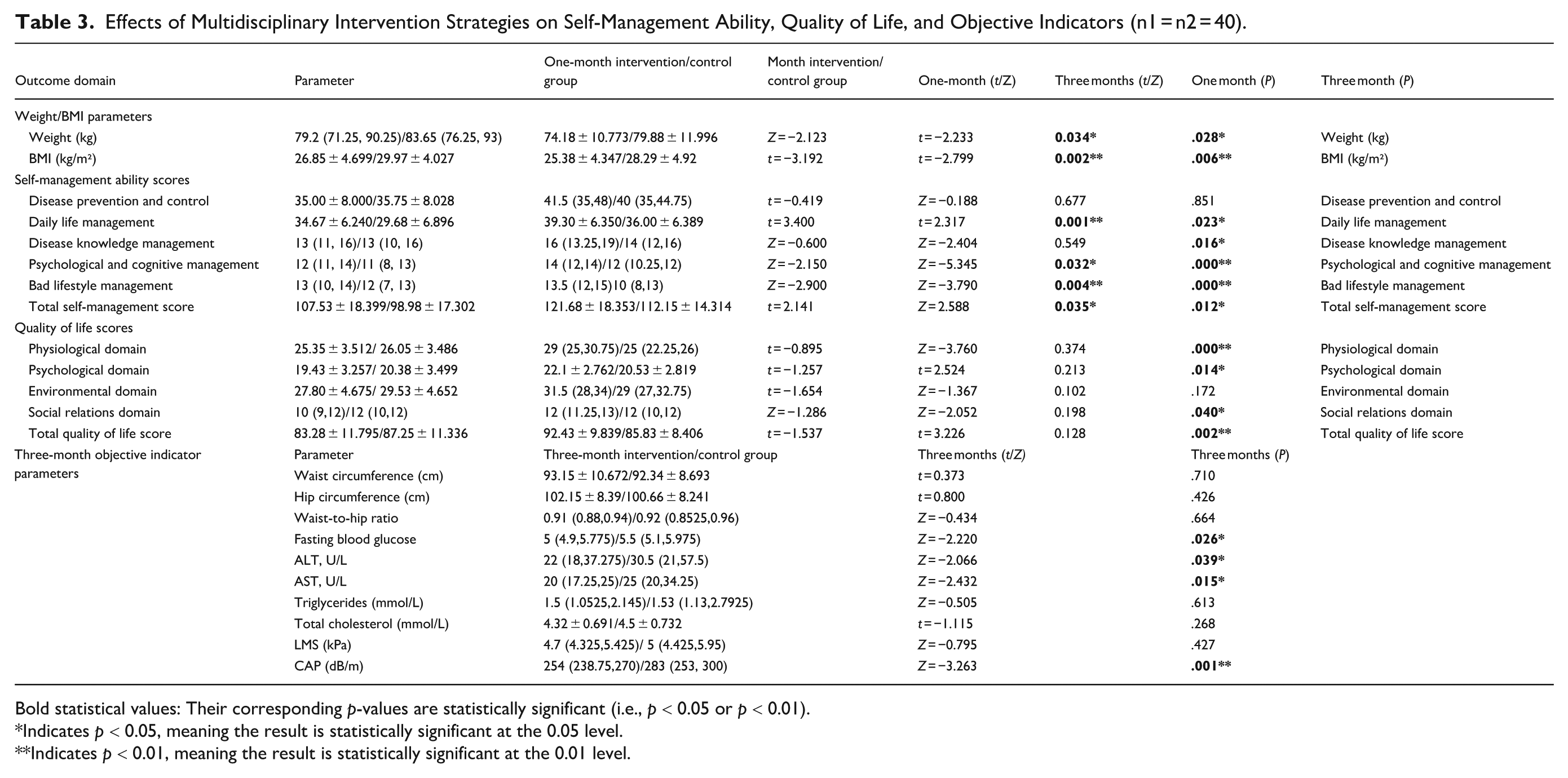
At 1-month follow-up, the intervention group demonstrated significant improvements versus controls in: ① Weight reduction (−4.74 kg vs. 3.88 kg; P = .034); ② BMI decrease (−1.54 vs 1.96 kg/m²; P = .002); ③ Self-management domains: Daily life management (P < .05), Psychological cognitive management (P < .05), Bad lifestyle management (P < .05), Overall Self-Management Ability (P < .05) (Table 3).
Effects of Multidisciplinary Intervention Strategies on Self-Management Ability, Quality of Life, and Objective Indicators (n1 = n2 = 40).
Bold statistical values: Their corresponding p-values are statistically significant (i.e., p < 0.05 or p < 0.01).
Indicates p < 0.05, meaning the result is statistically significant at the 0.05 level.
Indicates p < 0.01, meaning the result is statistically significant at the 0.01 level.
No significant changes occurred in disease prevention, knowledge management, or quality-of-life metrics during this phase.
By 3-month assessment, the intervention group maintained anthropometric benefits and showed expanded therapeutic gains: ①
Disease prevention (self-management), environmental QoL, waist-hip ratio, triglycerides/total cholesterol (TG/TC), and liver stiffness measurement (LSM) showed no significant changes.
Discussion
This mixed-methods study demonstrates that a primary care-anchored multidisciplinary intervention (MDI) significantly enhances self-management capabilities, quality of life (QoL), and clinical outcomes in metabolic-associated fatty liver disease (MAFLD) patients. Cross-sectional findings identified high-risk subgroups requiring targeted approaches: sedentary working-age males (67.4%) and those with disparities between educational attainment and self-management abilities.31,32 To address these modifiable factors, the intervention utilized digital peer support through WeChat communities, effectively bridging the gap between education and self-efficacy. Specifically, among patients with a bachelor’s degree, 66.5% demonstrated suboptimal disease knowledge (scoring 2.42 ± 0.98).3,33,34
Notable clinical improvements included 5% to 10% weight loss in 68% of participants—a threshold proven to reverse steatosis—along with significant reductions in hepatic fat (as measured by CAP), liver enzymes, and fasting glucose levels. 35 These outcomes counter obesity-driven pathophysiology prevalent in 84.9% of participants and address the risks associated with sedentary occupations, which affected 95.9% of the group. 36 Effect sizes demonstrated robust clinical relevance: weight reduction (Cohen’s d = 0.69), CAP reduction (d = 0.86), and QoL gains (d = 0.72) all exceeding minimal clinically important differences. The enhancements in QoL specifically targeted the low baseline health satisfaction reported (2.60 ± 0.91), with the most pronounced improvements seen in psychological domains. These changes aligned with previously established relationships between psychological factors and QoL (r = 0.407-0.553).37,38
Implementation Considerations
Sustainability and Adaptability
Though the 3-month outcomes are promising, the long-term sustainability remains unclear, as returns tend to diminish in cases where disease duration exceeds 10 years. 25 Future research should assess the value of booster sessions and digital adherence monitoring to help maintain these effects. 39 The control group here received only minimal standard care (educational pamphlets plus monthly sessions), which may have led to an underestimation of the intervention’s efficacy. Using active comparators—such as standard behavioral counseling—would strengthen the rigor of future trials. 40
Equity and Generalizability
Educational attainment was found to predict self-management capacity (β = .59, P < .01), and non-manual workers showed better performance than manual laborers (β = .117, P < .05).41,42 Due to sample size constraints, subgroup analyses by education or occupation could not be conducted, but such investigations are needed in larger study populations. 43 The sample, consisting of urban and educated individuals, limits the generalizability of findings to rural settings. To adapt the intervention for rural use, simplified interfaces (such as voice-based AI), deployment of community health workers, and integration with mobile clinics would be necessary, particularly for populations with low literacy levels.44 -46
Methodological Rigor
Weekly engagement with the digital platform averaged 85%, as tracked through login data, with automated reminders and technical support helping to maintain consistent use. 47 Despite adjusting for covariates, some residual confounding factors—such as motivation and health literacy—may still influence results. 48 Liver stiffness measurements (LSM) showed no significant changes, which aligns with the short duration of the intervention, as this timeframe is unlikely to affect fibrosis. Future studies should therefore track fibrosis biomarkers over a longer period. 49
Stage-Tailored Management
Disease duration significantly moderated outcomes, with the greatest improvement in quality of life observed among patients 3 to 5 years after diagnosis (β = .466, P < .001). 50 This finding necessitates differentiated treatment protocols: mobile health coaching for early-stage disease (<5 years), comorbidity-focused care for mid-stage disease (5-10 years), and integrated palliative support for advanced disease (>10 years).51 -53
Policy and Practice Integration
Four key priorities stand out: (1) Risk-stratified screening targeting sedentary males aged 31-50 54 ; (2) Digital stepped-care platforms that automate interventions tailored to disease stage 55 ; (3) “MAFLD-smart workplace” partnerships with a focus on manual laborers 46 ; (4) Value-based payment models that incentivize improvements in quality of life, aligned with China’s primary care reforms. 56
Strengths and Limitations
The Integrated Theory of Health Behavior Change (ITHBC)-based multidisciplinary intervention effectively integrates behavioral theory with metabolic targeting. Limitations include single-center recruitment restricting rural generalizability, self-reporting biases despite digital supplementation, and the 3-month timeframe inadequate for assessing fibrosis progression. Advanced fibrosis exclusion necessitates validation in severe disease. Future multicenter studies should implement ≥12-month follow-ups with active comparators in primary care settings.
Conclusions
This integrated analysis demonstrates that primary care-anchored multidisciplinary interventions significantly enhance self-management capabilities, quality of life (QoL), and clinical outcomes in MAFLD by addressing population-specific determinants identified in our cross-sectional study. The intervention successfully bridged the education-self-efficacy gap among high-risk subgroups through WeChat-based support, while stage-adapted protocols maximized impact during the 3 to 5-year diagnostic window. By achieving 5% to 10% weight loss in 68% of participants—directly countering sedentary occupational risks—and improving psychological well-being (strongly correlated with QoL), this model validates scalable strategies for frontline care. Educational attainment and occupational status remain key predictors of self-management success, necessitating tiered approaches: simplified tools for low-literacy groups, workplace partnerships for manual laborers, and digital solutions ensuring universal accessibility. These findings advocate for policy reforms integrating value-based payments into China’s primary care modernization to reward demonstrated QoL improvements. Embedding such multidimensional strategies into primary care ecosystems offers a sustainable pathway to reducing MAFLD’s chronic disease burden through patient-centered innovation.
Supplemental Material
sj-docx-1-inq-10.1177_00469580251380071 – Supplemental material for Enhancing MAFLD Care: Efficacy of a Multidisciplinary Community Intervention on Self-Management and Hepatic Outcomes
Supplemental material, sj-docx-1-inq-10.1177_00469580251380071 for Enhancing MAFLD Care: Efficacy of a Multidisciplinary Community Intervention on Self-Management and Hepatic Outcomes by Huan-Jing Cai, Hai-Lun Liang, Lei-Yu Shi, Yi-Hui Rong, Jing Li, Yan Zhang and Rui-Fang Ma in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Ethical Considerations
The study was conducted in accordance with the guidelines of the Declaration of Helsinki and approved by the Institutional Ethics Committee of Peking University International Hospital (approval number 2024-KY-0070-01, dated September 29, 2024).
Consent to Participate
Consent to Participate was obtained from all Subjects involved in the study.
Author Contributions
Conceptualization, Huan-Jing Cai and Hai-Lun Liang; Methodology, Lei-Yu Shi; Software, Jing Li and Rui-Fang Ma; Formal Analysis, Huan-Jing Cai; Investigation, Yan Zhang, Jing Li and Rui-Fang Ma; Resources, Yi-Hui Rong; Writing – Original Draft Preparation, Huan-Jing Cai; Writing – Review & Editing, Huan-Jing Cai and Hai-Lun Liang; Supervision, Jing Li and Yan Zhang; Project Administration, Huan-Jing Cai; Funding Acquisition, Yi-Hui Rong. All authors have read and agreed to the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Peking University International Hospital, grant number YN2024HL03.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available from the first author, Huan-Jing Cai, upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
