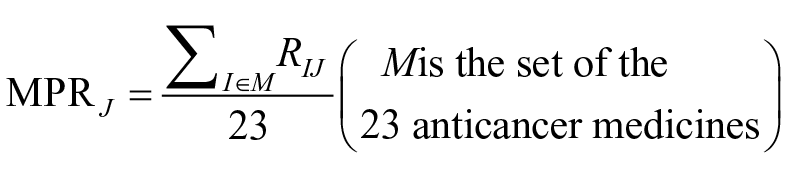Abstract
The study aimed to evaluate the change in accessibility of essential anticancer medicines, from 2015 to 2018 in a pilot province for health care reform in China. Data on access to 23 essential anticancer medicines was obtained from 6 provincial tertiary hospitals. A comprehensive analysis was applied to explore these trends. The total utilization of anticancer medicines had increased by an average of 2.57 times (P < .001) during the study period, of which targeted anticancer medicines had the fastest growth rate of 6.45 times (P < .001). The prices of all targeted medicines and original brands (OBs) were showing a downward trend, with the average change rate of −32% and −28% respectively (both P < .001). In contrast, the price of non-targeted medicines and lowest-price generics (LPG) increased by an average of 98% (P < .001) and 117% (P < .004) respectively. All targeted anticancer medicines were found to be unaffordable under this standard of this study, but the affordability of these medicines is on the rise. The study suggested positive changes in the utilization, price, and affordability of the most essential anticancer medicines. In the future, comprehensive strategies need to be conducted to further increase the affordability of targeted anticancer medicines.
The accessibility of anticancer medicines, especially novel targeted anti-cancer medicines, is low in some low- and middle-income countries. How to solve this worldwide inequality is a topic of great concern to the academic community.
The utilization of anticancer medicines in the sample areas of China shows an increasing trend, indicating that the availability of anticancer medicines is gradually improving. However, most anticancer medicines are still not affordable for patients. In addition, the affordability of novel targeted anticancer medicines is improving.
At present, China’s medicine policy is very effective in improving the accessibility of anticancer medicines. In the future, the authorities need to continue to include more novel targeted anticancer medicines in the list of essential medicines, focusing on the problem of rising prices of LPG anticancer medicines.
Introduction
Cancer is one of the non-communicable diseases with the highest morbidity and mortality in the world today. 1 According to the latest research data from International Agency for Research on Cancer (IARC), there were over 1.8 million new cases of cancer and 9 555 027 deaths worldwide in 2018, with a morbidity of 101.1 per 100 000 and a mortality of 197.9 per 100 000. 2 Unfortunately, with the consolidation of the aging population, these data will continue to expand in the future. 3 Therefore, strengthening cancer prevention and ensuring universal access to essential cancer treatments are important tasks of the global public health sector. Ensuring the accessibility of essential anticancer medicines (EAM) is a necessary condition for cancer treatment. The 19th revision of the WHO Essential Medicine List (WHOEML) included dozens of anticancer medicines that the population from every country should be equal access to. Nevertheless, some EAMs are almost unaffordable in low- and middle-income countries (LMICs), especially novel anticancer medicines. 4 Where monthly medicine expenditure often exceeds annual income. For instance, in 2017, Pakistanis need to spend 71 months to pay 1 month’s cost of Nilotinib that was added to the 20th version of WHOEML. 5
China has a population of more than 1.4 billion and an elderly population of nearly 300 million. In 2009, the Chinese government initiated nationwide health care reforms. 6 This is a comprehensive reform of the health system, which mainly includes 5 top priority areas: optimizing the primary health service system, establishing a national essential medicine system, promoting the reform of public hospitals, building an essential medical insurance system, and improving the equalization of public health services. The progress of China’s healthcare reform has been internationally recognized. Over the decade from 2009 to 2018, the reform improved access to health services, reduced healthcare costs, and increased the efficiency of health services.7,8 The fact that the life expectancy of the Chinese population has increased from 74.8 years in 2010 to 77 years in 2018 can also be indicative of the effectiveness of the reform. 9 Nevertheless, China has the largest number of cancer patients in the world, with 4 285 003 new cases and 2 865 174 deaths in 2018, exceeding a quarter of the new cases and deaths worldwide. 2 The cost of expensive cancer treatment in China has become a major problem in the field of social security. An empirical study from China found that the annual direct medical expenses of cancer patients after reimbursement by essential medical insurance amounted to 86 100 renminbi (RMB). 10 Of these costs, the expenditure on anticancer medicines is the main one. The low availability of anticancer medicines has become an important obstacle to the treatment of cancer patients in China. To alleviate this problem, the Chinese government added 26 anticancer medicines to the national essential medicines list (NEML) for the first time in 2013. Then twelve anticancer medicines were added to the latest version of NEML, including 6 targeted anticancer medicines (it can target specific lesions in the body and accumulates or releases active ingredients there) especially. The supporting policies played a synergy to decrease the price, including a zero-mark-up policy, province-wide centralized bidding procurement policy, and government price negotiation.
In China, the cost of health services is shared between the government, patients, and government-led essential medical insurance. The financing and payment of essential medical insurance is mainly coordinated by provincial governments, and the sources of financing mainly include local governments and residents. Nevertheless, some studies reported that several EAMs were completely unaffordable for low-income patients.11-13 Such as the treatment cost of trastuzumab, icotinib, and gefitinib far exceed the ability of Wuhan residents to pay.12,13 The high cost of anticancer medicines is one of the important factors leading to or returning to poverty. Therefore, Anhui province which was approved as one of the provinces included in the first round of pilots of the health care reform policy was selected as a broadly representative sample to survey the utilization, price, and affordability of EAMs.
Materials and Methods
Samples Selected and Study Design
The study was conducted in Anhui province, with a registered population of about 70 million during 2015 to 2018. The proportion of the population in rural areas was 72.41% (50.3 million), 70.47% (49.5 million), 68.93% (48.7 million), and 67.34% (47.7 million) respectively from 2014 to 2018. It has a medium economic level in mainland China. Anhui was one of the earliest provinces chosen by China’s State Council to pilot a new healthcare reform policy, broadly representative of the reform status of other provinces. 14 Anhui Province has a huge burden of cancer, with residents having a risk of premature death from cancer of more than 7%, ranking first among non-communicable diseases. 15
Five provincial tertiary comprehensive hospitals and 1 tertiary tumor hospital in Anhui Province were selected by convenient sampling. These sample hospitals are affiliated with the Anhui Provincial Health Commission, with over 10 000 beds and 15 000 health workers, and provide high-level specialized medical and health services for residents in the province and undertake higher education and scientific research tasks. Each medicine procurement and consumption data from sample hospitals from 2015 to 2018 was collected. According to the EML, WHO EML, and previous research experience, 23 anticancer medicines were identified for statistical analysis. Trend analysis and significance tests of the price, utilization, and affordability of these medicines between 2015 and 2018 were carried out. Moreover, the changing trend of different categories of medicines was also analyzed separately.
Selection of Medicines
A comprehensive strategy was conducted to select the representative medicines. First, all anticancer medicines were identified based on China EML or WHO EML. Second, at least 1 anticancer medicine was included in each classification. For example, daunorubicin was selected in the list as the antibiotic anticancer medicine. Third, these factors of local importance and expert advice were considered. Finally, a list of 23 anticancer medicines was included in this survey, including 2 alkylating agents, 6 antimetabolites, 2 antibiotics, 2 phytomedicines, 3 platinums, and 8 targeted medicines. The characteristics of each medicine are shown in Table S1.
Data Collection and Processing
Data on utilization and price for each medicine was collected from the health information system (HIS) of each sampled hospital, covering the period from January 1, 2015, to December 31, 2018. Two investigators were hired to collect data in January 2019. Initially, a total of 38 306 electronic records (trade name, generic name, batch number, patient price, specification, target pack size, quantity used, dosage form, manufacturer) from sample hospitals were identified. After data cleaning and removing outliers, 37 924 records were obtained. Finally, 1112 records of these 23 anticancer medicines in this dataset were screened out. Two analysts independently convert the data of medicine utilization and price to the minimum package size (bag, tablet, bottle. . .), and identify these medicines as originator brands (OB) or lowest price generics (LPG) according to the trade name and manufacture. In addition, the per capita disposable incomes of residents of Anhui province from 2015 to 2018 were gathered from the statistical yearbook published on the official website of the Statistics Bureau of Anhui Province. The survey was approved by Anhui medical university, the School of Pharmacy, and the information management department of each hospital.
Outcome Measures
Utilization
In China, whether an anticancer medicine is used clinically is determined by complex external factors. The 23 medicines in this study were selected from the NEML and they were recommended for clinical priority. Doctors can determine the appropriate medicines for actual treatment based on their own experience and clinical application guidelines for anticancer medicines, such as OB or LPG, traditional, or novel medicines. In addition, medication regimens are affected by the cost of treatment and the adequacy of supply. Here, utilization refers to the cumulative times a medicine has been used clinically in 1 year. Horizontal comparison can reflect which anticancer medicines are more clinically favored. Longitudinal comparison can reflect the changing trend of frequency. The utilization combined with the price of the medicine can truly reflect patients’ economic burden. The utilization was expressed as annual defined daily doses (DDDs) and calculated according to the following equation:
DDD is defined in this study as the average maintenance dosage of an anticancer medicine used for its main indication in adults on a daily basis. The DDD value could be determined by searching the WHO website, authoritative medicine specifications, and clinical guidelines.
Price
According to real-world data, most medicines have slightly different prices in different hospitals at the same time node. The weighted mean unit price (MUP) was applied to represent the average price level of the medicine. The MUP was used to replace the mean unit price or the median unit price, aiming to get closer to the real patient price. The MUP of medicine I was calculated as follows:
where
To facilitate price comparisons between different medicines, all medicine prices were calculated as defined daily dose cost (DDDc), which refers to the average maintenance dosage cost on a daily basis. The formula used to calculate DDDc was:
To analysis the changes in medicine prices in different years, the change rate of medicine was adopted to describe the speed of price change. 16 The price change rate of medicine I in year J (2015, 2016, 2017, and 2018) was calculated according to the following equation:
Where PI2015 means the MUP of medicine I in the first year (2015) for 6 hospitals; PIJ means the MUP of medicine I in the J year. The annual average price change rate covering all anti-cancer medicines is defined as MPR. And the MPR in year J was calculated as follows:
Similarly, the average utilization change rate (MFR) and average economic burden change rate (MBR) were calculated.
Moreover, taking into account the impact of inflation on medicine prices, 2016, 2017, and 2018 medicine prices to 2012 prices by deflating them by 2.0%, 3.6%, and 5.8% respectively.
Affordability
Affordability in this study refers to the financial ability to purchase a certain anticancer medicine. According to the WHO/HAI methodology, if the cost of course treatment of a medicine is less than the daily wage of the lowest-paid unskilled government worker (LPGW), which is considered as affordable. 17 However, due to the complexity of this wage calculation in China, it is difficult to determine a representative wage. Moreover, considering the difference in income structure between rural residents and urban residents. Therefore, another widely used methodology was chosen to calculate affordability. The medicines were deemed affordable if the total monthly cost of the medicines was less than 20% of the households’ monthly capacity to pay. 18 The monthly cost of an anticancer medicine is equal to the DDDc of the medicine multiplied by 30 days. In this study, the affordability of urban households and rural households in different years was compared. The affordability is calculated as the monthly cost of an anticancer medicine divided by 20% of the households’ monthly income. The larger the MBR, the lower the affordability of the medicines.
Data Analysis
The original data is extracted, screened, and analyzed using EXCEL 2016 software, SPSS 25.0 (Stata Corporation, College Station, Texas, USA). The Kolmogorov-Smirnov test is used to examine whether the data conforms to the normal distribution. Comparison of differences between different groups, analysis of variance (ANOVA), and Friedman nonparametric test are used when the data are normal and non-normal respectively. The price and affordability are calculated in Chinese legal tender (RMB), which equals approximately 1 US dollar to 6.5 RMB between 2015 and 2018. A P-value <.05 was considered to be statistically significant.
Results
Utilization
Table 1 shows the cumulative utilization of 23 medicines and each category in different years. The overall utilization of anticancer medicines had nearly doubled during this period, from 1 150 498 times in 2015 to 2 093 404 times in 2018. The frequency of anticancer medicine in 4 subcategories has increased to varying degrees. Among them, it is worth noting that the frequency of targeted anticancer medicines had increased by about 5 times from 2015 to 2018. The proportion of targeted anticancer medicines in the total utilization of anticancer medicine use increased from 9.34% to 24.5% from 2015 to 2018 (Figure 1). In terms of individual medicines, the highest change rate of utilization compared to 2015 is icotinib (OB) of 7.57 in 2016, icotinib (OB) of 15.95 in 2017, and icotinib (OB) of 24.30 in 2018, and the lowest rate is gemcitabine (OB) of 0.06 in 2016, paclitaxel (OB) of 0.04 in 2016, and gemcitabine (OB) of 0.04 in 2018. On average, the MFR of all anticancer medicines also showed an upward trend from a 75% increase in 2016 to a 257% increase in 2018 (P < .001). The MFR of non-targeted anticancer medicines was found to have a slight increase, but it was not statistically significant (P = .191). Nevertheless, the MFR of OBs in 2018 was increased by 355%, but no significant change in OBs (P = .116). On the contrary, the MFR of LPGs and targeted anticancer medicines increased significantly among 4-year groups (both P < .001). The results were presented in Table 2.
Utilization of Each Anticancer Medicine From 2015 to 2018.

Note. OB = originator brands; LPG = lowest price generics.
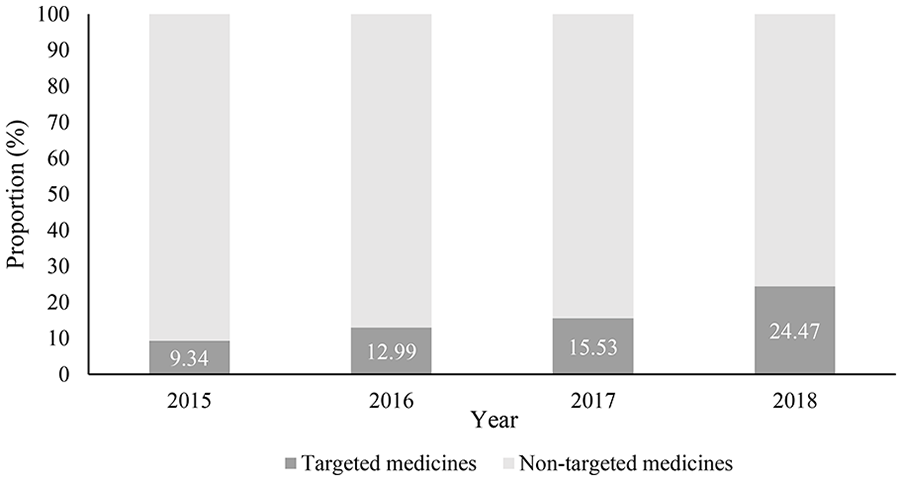
The changes in the proportion of the utilization of targeted and non-targeted anticancer medicines from 2015 to 2018 (%).
The Average Change Rate of Each Category Compared to the Variable in 2015.

Note. OB = originator brands; LPG = lowest price generics.
Price
Table 3 details the price information of each medicine from 2015 to 2018. Oxaliplatin (OB) ranked the highest DDDc in 2015, 2016, 2017, and 2018 with 10 495.63 RMB, 10 207.84 RMB, 10 047.28 RMB, and 9840.28 RMB respectively. Methotrexate (LPG) ranked the lowest DDDc in 2015, 2016, 2017, and 2018 with 0.77 RMB, 1.80 RMB, 1.75 RMB, and 1.70 RMB respectively. During the 4-year groups, the average DDDc of the OBs was 5 to 7 times higher than the LPGs during the 4 years; the average DDDc of targeted anticancer medicines was 1.5 to 2 times higher than the non-targeted anticancer medicines.
Defined Daily Dose Cost of Each Anticancer Medicine From 2015 to 2018 (RMB).

Note. RMB = renminbi, China’s legal tender; OB = originator brands; LPG = lowest price generics.
As shown in Table 2, the MPR of all medicines surveyed showed a slight upward trend compared to 2015 (6% in 2016, 35% in 2017, and 53% in 2018, P < .001). Fluorouracil (LPG) was ranked with the highest growth rate (15.88 times), and the price ranged from 8.48 RMB in 2015 to 134.69 RMB in 2018. Dasatinib (OB) was ranked with the highest drop rate (−70.56%), and the price ranged from 1066.97 RMB in 2015 to 314.17 RMB in 2018. For 4 subcategories, the MPR of OBs had significantly increased from a 10% drop in 2016 to a 28% drop in 2018 (P < .001). Similarly, the MPR of novel target medicines had also decreased from −11% in 2016 to a −32% drop in 2018 significantly (P < .001). In contrast, the MPR of LPGs and non-targeted anticancer medicines had increased by 98% (P < .001) and 117% (P = .004) respectively in 2018. Finally, it is worth mentioning that the DDDc of all OBs and targeted medicines has decreased to varying degrees during this period.
Affordability
The results of affordability are shown in Table 4. In general, the affordability of each anticancer medicine in rural households is much lower than that in urban households, but compared with urban households, the affordability trend of rural households is more positive. In terms of specific affordability, methotrexate (LPG) and oxaliplatin (OB) ranked the lowest (0.04-0.09 times) and highest (181.71-577.74 times) economic burden of anticancer medicines during the 4 years, respectively. The highest increase in economic burden during the survey period was for fluorouracil (LPG), which increased 12.76 times and 14.16 times for rural and urban households respectively, while dasatinib (OB) ranked the largest decrease, about 4.23 times and 3.81 times for rural and urban households respectively. All OBs and targeted anticancer medicines are determined to be unaffordable in the 2 groups of families. Nevertheless, about 1/2 and 1/3 of the anticancer medicines are affordable every year for urban and rural households respectively.
Affordability for Rural and Urban Households During the Period Surveyed (Times).
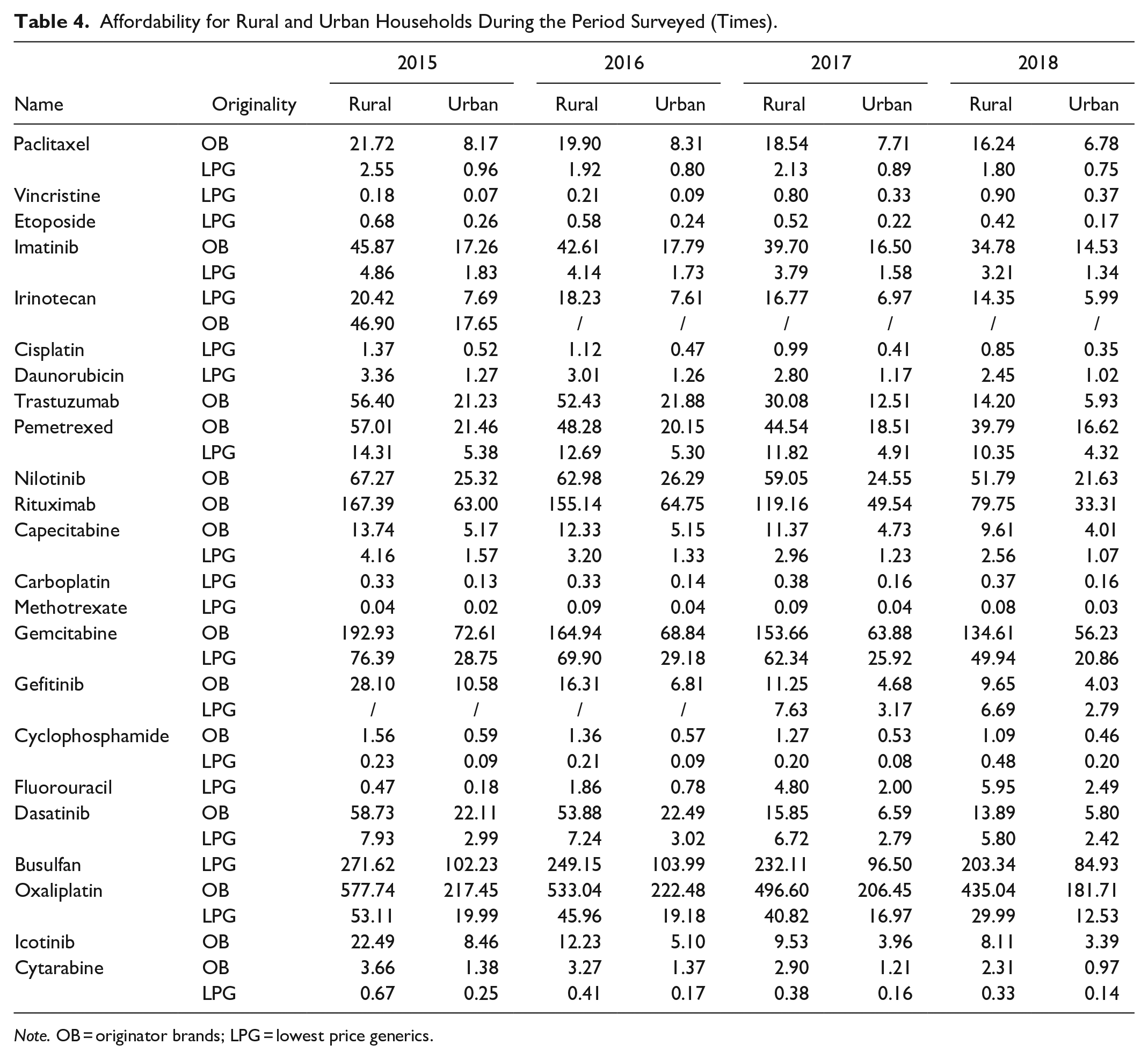
Note. OB = originator brands; LPG = lowest price generics.
The total MBR of 23 medicines surveyed increased significantly to 0.23 in rural households and 0.37 in urban households during the period (both P < .001). For 4 subcategories, the MBR trends of LPGs and non-targeted anticancer medicines increased significantly (Rural: P = .003, P < .001; Urban: P = .005, P < .001), while that of OBs and new target anticancer medicines steady decreased (both P < .001).
Discussion
To the best of our knowledge, this was the first comprehensive analysis to evaluate the temporal variation of the utilization, price, and affordability of essential anticancer medicines in China. The analysis of utilization can facilitate the understanding of the structure and clinical preference of anticancer medicines use in a region. The evaluation of the changes in medicine prices can reflect the current situation of local market competition and the effectiveness of government intervention. The assessment of affordability can be used to describe whether the price of the medicine matches the local income level. Therefore, comprehensive indicators were applied to evaluate the accessibility of anticancer medicines. There are several main findings in the study. First, the utilization of targeted anticancer medicines had a higher growth rate compared to non-targeted anticancer medicines. Second, the prices of targeted anticancer medicines and OBs show a downward trend, while LPGs and non-targeted anti-cancer drugs have the opposite performance. Third, all targeted anticancer medicines and OBs were unaffordable for the sample population, but the affordability of these medicines was showing an upward trend.
In China, the low accessibility of anticancer medicines is a long-term problem, especially targeted anticancer medicines. Therefore, the Chinese government initiated a health care reform and released a series of health policies to improve the accessibility of essential medicines for low-income individuals, including essential anticancer medicines. The findings from this study are inextricably associated with these policies. We will discuss the changes in these indicators from a policy perspective, as follows.
Utilization
In recent years, the increase in the demand volumes for anticancer medicines is a common phenomenon across many countries. 19 The fast-growing utilization of anticancer medicines is a positive performance for cancer patients in China, which means that the availability of these medicines is increasing. There are several main factors associated with changes in utilization. Firstly, the increase in new cancer cases directly promotes the use of anticancer medicines. On the one hand, the prevalence is increasing of some cancers due to smoking, being overweight and physical inactivity. 1 On the other hand, the ever-expanding scale of cancer screening has allowed more potential cases to be diagnosed with cancer. Secondly, the accessibility of cancer treatment services is increasing. In the 10 years since the initiation of the health care reform, the number of hospitals in China has increased by 69.31%. 9 Intensive health service coverage guarantees more cancer patients receive treatment. Meanwhile, Anhui as a comprehensive medical reform pilot province takes the lead in the implementation of the “volume-based procurement policy” in 2015, 20 which operating mechanism is to compress the profit margin of each unit by raising an order containing the quantity of the medicine demanded in the region. Therefore, this demand-oriented medicine procurement model not only improves the availability of medicines but also reduces the price of medicines. Thirdly, lower prices drive higher demand. For example, the price of trastuzumab dropped by 69% in 4 years, which directly led its utilization to rising 15 times. Similar associations were performed in most other targeted anticancer medicines surveyed. Moreover, previous studies have reported the same findings for the targeted anticancer medicines in other provinces of China (Zhejiang and Jiangsu).21,22
The Price of Anticancer Medicines
The results from the study suggested a decreasing trend of price among all OBs and targeted medicines surveyed during the period, but 5 non-targeted anticancer medicines (LPG) were on an upward trend, ranging from 1.4 to 15.9 times. Several policy factors contribute to the decline in medicine prices. Firstly, the cancellation of medicine markups policy has been implemented by all public hospitals in Anhui province in 2015, which directly eliminates the 15% to 30% profit margin between the medicine purchase price and the patient’s price (authorized by the government). 23 Secondly, the first national-level medicine price negotiation with pharmaceutical companies was conducted by the National Health and Family Planning Commission in 2016, which uses the entire mainland China market as a “bargaining chip” in exchange for the price. The results of the negotiation showed that the price of Gefitinib (OB) and Icotinib (OB) dropped directly by 54% and 55% respectively. 24 Thirdly, the involvement of local brands of generics has increased the level of competition in the market. The price of LPGs is much lower than that of OBs because of the avoidance of huge research and development costs. Taking imatinib as an example, the price of the OB is about 10 times that of the LPG. Other related policies include the “two-invoices system” formally implemented across China in 2018, 25 and the volume-based procurement policy mentioned above. This price reduction mechanism is that the former streamlines public sector procurement and supply systems, while the latter adopts a similar “group buying” approach.
There are several possible reasons to explain the increase in medicine prices. The first is the cost factor. For example, due to the increase in the cost of raw materials, the price of Vincristine increased by 6.31 times during this period, which was reported in our previous study. 26 The second is the shortage of medicines. Fluorouracil injection almost disappeared in the entire Anhui market in 2016, which directly drove the immediate price increase of 3 times. The third is the policy factor. Vicious competition among pharmaceutical companies has caused the bidding price of medicines to be lower than the cost price. In the subsequent supply, the government has to agree to increase prices to ensure that medicines can be supplied sufficiently. Appropriate price increases can ensure the supply of medicines that are interrupted due to profit reasons. But the problem of excessive price increases must be curbed.
The Affordability of Anticancer Medicines
In terms of the findings from the study, all OBs and over half of the LPGs are unaffordable to local urban and rural households during the period, which cannot be ignored. Similarly, a recent systematic review reported that low-income patients from LMICs had low affordability of anticancer medicines. 27 Moreover, the affordability of most OBs and targeted medicines was far lower than that of LPGs and non-targeted medicines. Empirical evidence showed that targeted medicines could provide cancer patients with better outcomes for their treatment against cancer. A previous systematic review concerning cost-effectiveness analysis suggested that targeted oral anticancer medicines were more cost-effective than non-targeted anticancer medicines. 28 Fortunately, the affordability of all OBs and targeted anticancer medicines were showing an upward trend. The decrease in medicine prices is the main reason for the increase in affordability, while the increase in China’s per capita income in recent years has also contributed to the increase in the affordability of anticancer medicines. Nevertheless, the economic burden of several LPGs and non-targeted anticancer medicines is constantly rising during this period. This phenomenon reveals that the price of these medicines is rising faster than that of rural and urban residents’ income, which is an unhealthy manifestation for the health system.
There are several limitations in this study that have to be emphasized here. Firstly, this study was conducted in a province (Anhui) in eastern China and does not represent the current status and trends of the entire country. Attempts to generalize these findings should be cautious. Secondly, patients’ affordability was calculated based on the disposable income per capita of residents. In China, the income of residents does not meet the normal distribution (the median value is lower than the mean), which means that the affordability of more than half of the sample families is overestimated. Thirdly, lack of privacy settings as a control. In recent years, the growing gap in scale and technology between public settings and private settings cause many patients to prefer public hospitals, which may be a potential contributor to the increase in utilization.
Conclusion
The study suggested a positive change in the utilization, price, and affordability of essential anticancer medicines in Anhui province. In the anticancer medicine market in China, the proportion of targeted medicines being used is increasing, which can provide a reference for the formulation of the next edition of the essential medicine list and future price negotiations. Moreover, this study also proved that a series of drug price policies in the medical reform has played a positive role in medicine price reduction, especially OBs and targeted medicines. Although most anticancer medicines were not affordable for low-income families, their affordability for these medicines was increasing.
Supplemental Material
sj-docx-1-inq-10.1177_00469580231151783 – Supplemental material for Substantial Increase in Accessibility to Essential Anticancer Medicines in Anhui, China: A Longitudinal Study
Supplemental material, sj-docx-1-inq-10.1177_00469580231151783 for Substantial Increase in Accessibility to Essential Anticancer Medicines in Anhui, China: A Longitudinal Study by Rixiang Xu, Shuting Li, Tingyu Mu, Xuefeng Xie, Caiming Xu and Xiongwen Lv in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Consent to Participate
Not applicable. This study does not involve humans and experimental animals.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.