Abstract
Introduction:
Tracer medicines are medicines that must be available in sufficient quantities at all times to satisfy the priority health care needs of the population. Inventory mismanagement of these medicines poses significant challenges to public health systems, especially in countries like Ethiopia, where access to healthcare services is already limited.
Objective:
This study aims to assess inventory management performance for tracer medicines at public health facilities of the southwest Shewa zone, Oromia region, Ethiopia.
Method:
Concurrent triangulation mixed-method study was conducted from 1 to 30 June 2022. The quantitative data were collected using interviewer-administered semistructured questionnaires and observational checklists. The qualitative data were gathered through in-depth interviews with key informants. The quantitative data were analyzed using the SPSS version 26 and Excel spreadsheet version 16. We calculated the stock-out rate, inventory accuracy rate, percentage of facilities that fulfill appropriate storage conditions, percentage of facilities that submitted reports on time, and received the exact quantity of drugs they ordered. Qualitative data were analyzed manually using the thematic content analysis technique.
Results:
The inventory accuracy rate for tracer medicines was 76% for hospitals and 72.5% for health centers. The overall mean stock-out rate was 24.99%. Bin card updating practice was 93.3%, and only 25% health facilities met the acceptable storage conditions criteria. Among submitted reports, 88.8% were timely reported, 72.2% were accurate, and 75% were complete. Inventory management challenges include a shortage of supplies from the supplier and in the market, price inflation, inadequate training, lack of supportive supervision, insufficient IT and storage infrastructures, lack of communication, and budget constraints.
Conclusion:
From this finding, we concluded that facilities’ report quality, inventory record accuracy, storage conditions, and logistics management information system needs improvements. Therefore, Ministry of Health, assessed facilities and Ethiopian pharmaceuticals Supply Services should take measures to improve them.
Introduction
Tracer medicines are medicines that satisfy the priority healthcare needs of the majority of the population. Every disease management strategy requires the availability and accessibility of medicines for prevention, diagnosis, treatment, palliative care, and rehabilitation. Access to tracer medicines is a global priority. The availability, accessibility, and affordability of these medicines need to be addressed to achieve the sustainable development goal. Therefore, essential medicine policies are crucial for promoting health and achieving sustainable development.1,2 To improve access to tracer medicines, the World Health Organization (WHO) has ratified framework work such as rational selection, sustainable financing, reliable health, and supply of quality products. 3
Inventory represents the highest current and liquid asset in health facilities, and its value continues to rise because of the growth in the variety and cost of medicines. 4 Around 40%–60% of the public health budget is allotted for medicines and supplies in many countries. 5 Despite this substantial investment, about one-third of the world population lacks access to essential medicines.2,6 Among the factors contributing to the lack of access to tracer medicines are poor inventory management and lack of funds. 7 Poor inventory management leads to an overstock of certain items, an understock of others, and inefficient operations. This could lead to a decrease in responsiveness to customer demand and an increase in operating costs.8–10 Public health facilities mainly rely on the availability and timely arrival of essential medicines from their key suppliers to satisfy their clients’ demands. However, assessments conducted at different levels of the supply chain identified frequent interruptions of essential medicines’ supply attributed to the dissatisfaction of health providers, patients, and the community at large.3,11
In the Ethiopian current supply system, program pharmaceuticals are reported and ordered every 2 months by hospitals and health centers. Then, Ethiopian Pharmaceuticals Supply Services (EPSS) deliver their orders directly or indirectly through Woreda Health Offices. For revolving drug fund (RDF) pharmaceuticals, health centers, and hospitals complete Report and Resupply Forms (RRFs) as per the facilities review period (every 2 months, every quarter, or every 6 months) and collect products from the affiliated EPSS branch. During these deliveries and collections, upper-level facilities provide feedback to lower-level facilities. In the feedback reports, facilities will be able to see how they are performing compared to other facilities in their area and will be able to facilitate stock transfer. 12
Improving inventory management at public health facilities will improve availability of medicines, reduce inventory holding cost and medicines expiry, and ultimately increase patients’ satisfaction. Regular assessment of health facilities’ performance is essential to track the root causes of problems and can improve inventory management. 13 To the best of our knowledge through literature review, there are very limited empirical studies published on inventory management of tracer medicines in Ethiopia, and there is no empirical data in current specific study area. Therefore, the objective of this study is to examine inventory management performance for tracer medicines in public health facilities of the southwest Shewa zone. It assessed the availability of tracer medicines, stockout rate, storage condition, and inventory management challenges. The findings of this study can serve as foundational data for upcoming researchers and offer valuable insights for healthcare policymakers and local decision-makers. This information enhances the accessibility of essential medicines, minimizes costs linked to inadequate inventory control, and addresses the complexities of inventory management.
Method
Study area
This study was conducted in public health facilities of southwest Shewa zone of Oromia region, located 116 km southwest of Addis Ababa (the country’s capital). Southwest Shewa comprises 11 rural districts, with a total projected population of 1,110,011 residing in 268 villages (data from Southwest Shewa zone administration office). There were two general hospitals, three primary hospitals, and 56 health centers during the study period. The Ethiopian pharmaceutical supply agency Addis Ababa hub-1 is the main supplier of medicines for these public health facilities. This study was conducted from 1 to 30 June 2022.
Study design
A concurrent triangulation mixed-study design was employed to assess the inventory management performance for tracer medicines and associated challenges.
Population
The source populations were all public health facilities in the Southwest Shewa zone, health professionals working in those health facilities, essential medicines, and logistics documents. The study units are selected public health facilities, selected health professionals, selected key informants (KIs), selected tracer medicines, and selected logistics reporting and recording tools.
Sample size and sampling procedure
Sample size for facilities: There were eight health centers and four public hospitals, which manage antiretoviral therapy (ART) drugs during the study period in Southwest Shewa zone. Since ART drugs are part of selected tracer medicines, those facilities that didn’t manage ART drugs were not included in this study. Thus, 12 public health facilities were included in this study.
Sample size for medicines and documents: Regarding the sample size for tracer medicines, we included a list of 25 tracer medicines taken from Ethiopian Federal Ministry of Health (EFMOH) monitoring and evaluation document. 13 For reviewing documents, each medicine has one bin card in each facility; therefore, 300 (12*25) bin cards were reviewed. According to integrated pharmaceutical logistics system (IPLS) of Ethiopia, each facility should submit completed RRFs every 2 months; therefore, 36 RRFs were reported from the assessed (12 facilities) in the last 6 months preceding the study period. Therefore, for evaluating reporting timeliness, completeness, and accuracy, three consecutive reports per facility (a total of 36 reports) were reviewed.
Sample size for personnel: For the quantitative study, we interviewed all store managers of selected public health facilities. For the qualitative study, we selected KIs from the primary health care unit (PHCU) director, pharmacy head, medical store managers, Zonal health department (ZHD) logistics officer, Oromia Regional Health Bureau (ORHB) supply case team, and Ethiopian Pharmaceutical Supply Service (EPSS) capacity-building directorate. Different stakeholders were included to avoid early data saturation. Selected KIs were four store managers, four pharmacy heads, two PHCU directors, two medical directors, one ZHD logistics Officer, one pharmacist from ORHB supply case team, and one pharmacist from EPSS hub-1 capacity building directorate. They were selected because they are in charge of the overall pharmaceutical logistics activities and are expected to provide detailed information from all perspectives. Generally, we interviewed 14 KIs. The interviews were conducted by investigators to maintain consistency throughout the interviews.
Inclusion criteria: All health centers that manage ART drugs are included in this study.
Exclusion criteria: Private health facilities were excluded, since they do not manage program medicines most of the time.
Data collection method, procedure, tool, and quality assurance
We used semistructured questionnaires and observational checklists to collect data from primary and secondary data sources. The primary data sources were store managers, pharmaceutical logistics officers, and documents (bin cards, RRFs). A physical count of tracer drugs, observation of records on bin cards, and RRFs were used to determine inventory accuracy rate, stockout rate, and reporting accuracy and timelines. Pharmaceutical warehouses were inspected using a storage condition observation checklist adopted from the monitoring and evaluation tool. 14
An interview guide with flexible and probing questions was designed to explore inventory management challenges. The interview guide was adapted from the logistics system assessment tool (LSAT) 15 and translated into the region’s working language, Afaan Oromo, to facilitate conversation and eliminate the language barrier. The interview was face to face and took 30 min on average. The interviews were audio-recorded and transcribed.
The data collection tool for quantitative data was carefully adopted from logistics indicator assessment tool (LIAT) 16 and amended to the local context. Professionals with pharmacy background were recruited for data collection and principal investigator gave half-day training for data collectors. During data collection, there was a close supervision by the principal investigators to ensure consistency and completeness of the data. The saturation of data was ensured by rehearsing the recorded data throughout the data collection time.
Data analysis
The quantitative data were analyzed using the Excel spreadsheet version 16 and Statistical package for Social Science (SPSS) version 26. First cleaned data were entered into epidata version 3.6 and exported to excel spreadsheet and SPSS. Stockout days, stockout duration, and report accuracy were calculated by excel spread sheet. Sociodemographic characteristics, facility characteristics, and frequencies and percentage were calculated by SPSS. The results were then presented using tables, graphs, and texts.
The qualitative data were analyzed manually using thematic content analysis technique. Transcribed results were then translated into English by investigators. Then, the results were coded and categorized. We constructed themes for similar categories and summarized results under selected themes (emerging themes and subthemes are illustrated in flow chart 1). The transcribed data were brought back to participants and their comments were incorporated in the final report. Then, the results were presented in the form of narration under each theme. Finally, the qualitative results were used to triangulate the quantitative findings in the discussion.
Operational definition
Logistics management information system: The collecting, processing, and reporting of pharmaceuticals supply chain data to the concerned body, which can be either a manual system or an automated system.
Storage conditions: Ensuring the safety and integrity of the products and its package until drugs are dispensed to the clients.
Measurement of variables
Inventory management performances of health facilities for tracer medicines which are measured by different indicators was an outcome variable. Details of indicators is depicted on Table 1.
List of indicators and their data sources.

Source: Ministry of Health-Ethiopia. 13
Variables in this study were measured using standard indicators adopted from EFMOH monitoring and evaluation manual. 14
Description of core logistics indicators
i. Inventory accuracy rate:

ii. Reporting rate:
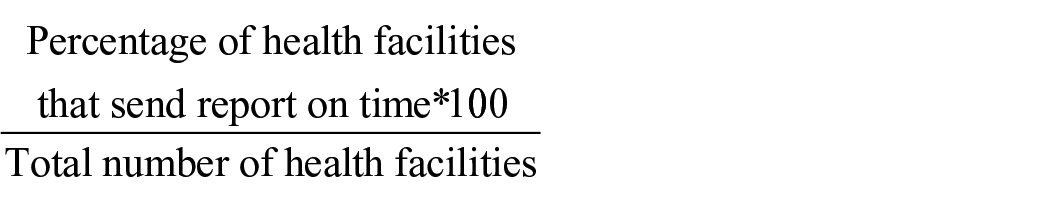
iii. Order fill rate:
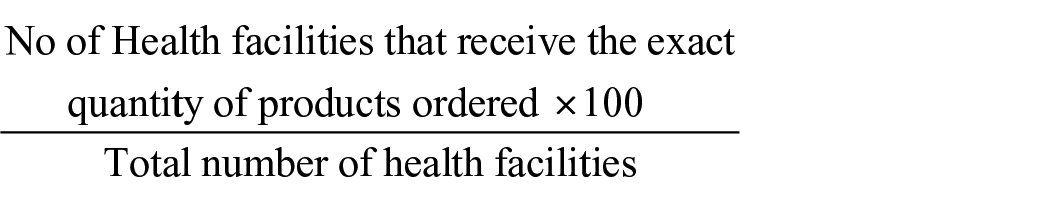
iv. Percentage of health facilities that meet adequate storage conditions

v. Stock out rate:

vi. Order Lead time: day order is received by facility—day order is placed by facility to the supplier
Results
Characteristics of health facilities and respondents
Forty-eight professionals were working in pharmacy units in assessed health facilities. Among them, 20 (43%) were degree holders, and 30 (68.2%) of the staff had service years between 1 and 5 years. Four (33.3%) of the study facilities had received supportive supervision 3 months before data collection, 2 (16.6%) of health centers had never received supervision within the last 6 months, and (41.66%) had never received feedback from higher levels. Of the total 48 staff in study facilities, 37 (80.4%) never received formal IPLS training, 11 (23.9%) received preservice training, and none of them received on-the-job training (Table 2).
Characteristics of health facilities and respondents of the selected health facility of Southwest Shewa zone, Oromia, Ethiopia, 2022.

Availability and stock out duration of key essential medicines
The mean availability of tracer medicines on the day of the visit was 86%, and the mean stock out of tracer drugs in assessed facilities within 6 months preceding the study period was 1.96 (16.3%) with a mean stock out duration of 62.2 days. Out of 25 tracer medicines, 14 (56%) of them were never stocked out in the preceding 6 months of the study period in any of the assessed facilities. Zinc dispersible tablets were stocked out in 75% of the health facilities, with an average stockout duration of 113.3 days, and 40% dextrose were stocked out in 50% of the assessed health facilities with a stock-out duration of 110 days within the preceding 6 months (Tables 3 and 4).
Availability on the day of visit and stockout of tracer medicines in selected public health facilities of Southwest Shewa zone, Oromia, Ethiopia, 2022.

Number of health facilities that encountered stockout, frequency, and duration of stockout of tracer medicines in the last 6 months in selected health facilities of Southwest Shewa zone, Oromia, Ethiopia, 2022.

Logistics management information system
Logistics recording and reporting (bin cards, RRFs, internal facility reports, and requisition forms) were available in all the assessed health facilities. However, only 4 (33.3%) of assessed health facilities (75%) of the hospitals, and only 12.5% of the health centers had electronic logistics information management systems. Only 50% of the facilities had an IPLS standard operating procedure manual at their store (Table 2).
Inventory accuracy rate
The discrepancy between the quantity recorded on the bin card, and the physical count was calculated to assess inventory accuracy. Accordingly, the inventory accuracy rate was 74.5% in assessed public health facilities (Figure 1).

Inventory accuracy rate for tracer medicines in selected public health facilities of Southwest Shewa zone, Oromia region, Ethiopia, 2022.
Logistics reporting timeliness, completeness, and accuracy
RRFs were reviewed to assess the report’s timeliness, accuracy, and completeness. From assessed RRFs, 27 (75%) were complete, 26 (72.2%) were accurately filled, and 32 (88.8)% of them were reported on time (Figure 2).
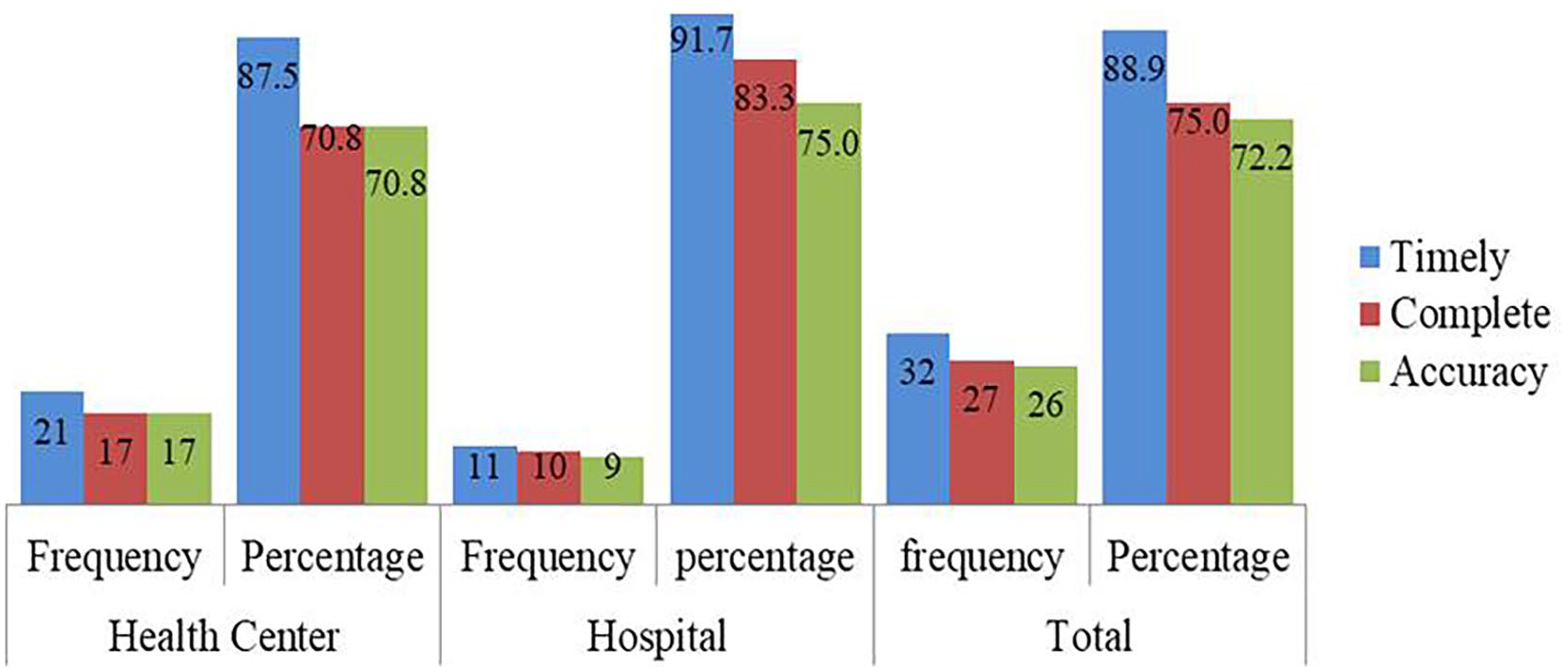
Timeliness, completeness, and accuracy of tracer medicines logistics report in selected public health facility of Southwest Shewa zone, Oromia, Ethiopia, 2022.
Storage conditions
This study showed that only 3 (25%) of assessed public health facilities met at least 80% of acceptable storage conditions. Three (75%) of the assessed hospitals and none of the assessed health centers fulfilled >80% of the criteria (Figure 3).

Pharmaceutical stores fulfillment of the required drug storage conditions criteria in public health facilities of Southwest Shewa, Oromia region, Ethiopia, 2022.
All of the assessed health facilities fulfilled some conditions like protecting products from direct sunlight at all times, protecting cartoons from water and humidity at all times, storing products at appropriate temperatures all the time, and keeping the storage area secured with lock and key. The most unfulfilled criteria were the availability of fire extinguishers (2 (16.6%)) and the availability of sufficient space and organization for current products (25%) (Table 5).
Health facilities’ adherence to good storage condition standards for tracer medicines in selected public Health facilities of Southwest Shewa zone, Oromia, Ethiopia, 2022.

Lead time
Nine (75%) of the facilities reported that their lead time was 2–4 weeks, and none of the public health facilities reported a lead time of less than 2 weeks (Figure 4).

Estimated average order lead time for tracer medicines in public health facilities of Southwest Shewa, Oromia Region, Ethiopia, 2022.
Challenges of inventory management for tracer medicines
Purposively selected KIs were interviewed to explore the challenges of tracer medicines inventory management. All KIs were males with the age range of 30 and 40 years old. In addition, their work experience ranged from 4 to 10 years.
All challenges were categorized under four broad themes: financial, human resources and capacity building related, supplier-related, and infrastructure and IT-related (flow chart 1).
Financial and administrative-related
When asked about inventory management challenges, KIs mentioned budget deficit as the major challenge. Mainly during the end of the fiscal year (budget closing period), there is a high budget deficit because social health insurance does not pay us on time. One KI mentioned that:
There has been a high medicine price inflation in the last 6 months. Moreover, the majority of the costs are covered by health insurance. During the budget closing period, the insurance does not pay on time. So we cannot purchase medicines on time. (PHCU Director, male, 6 years experience)
Another KI from the EPSS also revealed the administrative and financial problems by expressing the following:
Some programs lack clear leadership and a clear system of accountability. There is a budget constraint for regular supervision, gap identification, and supportive supervision. (EPSS, male, 7 years experience)
He also stated structural and administrative challenges as follows:
We do not have direct communication with some of the health centers. They report to a Woreda health office, and the Woreda health office sends us their report. We do not give them supportive supervision. So their quantification data is usually poor.
Supplier-related
The majority of the KIs agreed that stockout of medicines in their suppliers’ stock and on the market were among the challenges affecting the availability of the commodities at their facilities. KI stated:
For RDF drugs, we can purchase when we have a budget, but the non-availability of drugs on the market is the main challenge. For program drugs, once they (woreda) notify us of a product’s arrival, we go to the woreda health office to collect it. However, the product is out of stock, when we arrive there. (Manager, male, 7 years experience)
Most of the KIs agreed that the availability of the products at the private suppliers (wholesalers) was relatively good, but the procurement process was too long. One of the KIs articulated that “The drug procurement process from private wholesalers is too long, so it is difficult to get the drug instantly” (Pharmacist, male, 4 years experience).
Human resource and capacity building-related
Regarding human resources, the shortage of trained pharmacy professionals, especially in health centers is another challenge mentioned by the participants. For example, one of the KIs revealed
Our facility pharmacy workforce is currently around 42% of the standard. Thus, there is a lack of adequate workforce. (PHCU director, male, 8 years experience)
Again, the Zonal logistics officer also reported that
We have a deficiency of human resources, especially in health centers. The majority of our health centers do not have enough pharmacy professionals. (ZHB logistics officer, male, 10 years of experience).
In addition to professional deficiency, available staff also didn’t take on-the-job training. One KI said:
Usually, the supervisors from higher levels have been taken checklists for identified challenges; however, there is no training or regular feedback. (Pharmacist, male, 5 years experience)
Zonal logistics officer asked about the capacity building challenges, and reported that
Concerning the training, this year only a few store managers got a chance of IPLS training. (ZHB, male, 10 years experience)
Another KI from the EPSS also added:
There is a huge gap regarding the capacity building for our health professionals, due to programmatic area shifts of EPSS to only supply chain-related issues. RHB sometimes invites us for training; however, there is also a conflict of interest. (EPSS, male, 7 years experience)
Infrastructure and IT-related
KIs also reported nonstandardized pharmaceutical warehouses with inadequate space and lack of computers as other inventory management challenges. For example, one of the respondents said; “Our pharmaceutical warehouse space is not adequate. Having a big warehouse helps us to ensure safety, better categorization, and arrangement of drugs. We also store medical equipment, supplies, and drugs in the same store” (Pharmacist, male, 6 years experience).
Again, KIs stated that lack of automated (computer-based) information management system have an impact on improving the availability of the tracer medicines. One KI stated that
Since the inventory management affect the availability of key essential medicine we are focusing on it and give special attention. The software is advantageous to trace the dead stock, over stock and highly consumed items. The software support the manual and good for timely decision making despite our store the existing pharmaceutical logistics system is not automated and lacks more robust information management system at our facility. (Pharmacist, male, 10 years experience)
Zonal logistics officer also reported that.
The main challenge is quality of report from health facilities. Majority of the reports received from health facilities are incomplete and with poor quality. It is good if there is automated reporting mechanism, which could improve the quality and timeliness of the report. (ZHB, male, 10 years experience)
ORHB supply case team were asked about their commitment and challenges, and reported as
We have trained around hundred pharmacy professionals in 2013 E.C(2020 G.C) in Oromia region. The main challenges hindering availability in our facilities is problem with foreign currency for imported drugs, report quality from health facilities and lack of infrastructure specially in remote area. (RHB, male, 9 years experience)
KIs were also asked about the possible solutions and provided these solutions, regular supervision, electronic logistics management information system, continue “quick win initiative,” having pharmacy specific policy, procedure, and plan to procure from private supplier and skill-based capacity building to improve the logistics management performances of key essential medicine.
The key informant also added his recommendation as
To improve the capacity of our pharmacy professionals’ information management, it is better to include IPLS and HCMIS dagu systems in the undergraduate pharmacy curriculum. (RHB, male, 9 years experience)
Discussion
Inventory management is an integral part of logistics management that safeguards healthcare against disruption of service provision. Therefore, evaluating inventory management performance helps to identify the weaknesses and challenges of managing inventory and the smooth running of logistics activities. There are several logistics management performance indicators to assess the efficiency of inventory management. This study emphasized the inventory management performance for tracer medicines using indicators such as availability, stock record accuracy, order fill rate, report accuracy, report completeness, and timeliness and storage condition of medicines. 13
Availability and stockout of key essential medicines
This study revealed that the mean availability of tracer medicines on the day of the visit was 86% with a range of 25%–100%. The present finding was higher than previous studies conducted in Ethiopia, nationwide, 11 Adama town, 17 and West Wollega zone 9 and the study done in Kenya. 18 The difference might be due to a number of medicines and the number health facilities included. The number of selected medicines and the number of health facilities in the previous studies were greater than the current sample size.
This study also revealed that the mean stockout rate was 24.99% with average stockout duration of 62.2 days. The result was comparable to study conducted in Bungoma county, Kenya, 18 and Adama city, Ethiopia. 17 Fourteen (56%) of the assessed medicines were stocked out at least in one facility on the day of the visit. This study done in the Gamo zone showed better results when compared to this finding. 19 The probable reason for the discrepancy might be the previous study included health posts, which manage a small number of tracer drugs and serve a small number of clients. Therefore, the risk of stockout might be less in health posts. KIs addressed the reasons for stockout. They complained about stockout of the products at the supplier (EPSA), unavailability of the products on the market, price inflation, and inadequacy of budget. Gurmu TG and Ibrahim AJ reported similar reasons for stockout in studies conducted in East Shewa, Ethiopia, 20 and Indonesia. 21 Stockout of tracer medicines could lead to unfavorable clinical outcomes, drug resistance, and increased mortality. 22
Order fill rate and lead time
Regarding the percentage of public facilities that receive the exact quantity of products ordered, 6 (50)% of the assessed public health facilities reported that they never received the exact quantities of medicines they requested. This result was higher compared to a result found in a study conducted in eastern Ethiopia, where only 10% of health facilities reported that they never received the exact quantity of medicines they ordered. 23 The difference could be because of the difference in the program. The previous study was conducted on ART drugs only, which are program drugs, but this study included nonprogram drugs.
The majority of assessed facilities reported that their order lead time was approximately less than 1 month. This finding was better than the study conducted in Uganda. 24 The difference could be due to the difference in the study setting. The previous study was a countrywide survey, while the current is a cross-sectional study on a specific zone. As revealed by qualitative results, shortage of medicines at EPSS (main supplier) is among the reasons for not supplying orders in full. 11
Logistics management information system
Poor LMIS data quality leads to discrepancies in logistics information and ultimately results in wrong supply decisions by the supplier. Keeping accurate records of the stock on hand is very important for effective quantification, procurement, picking, and distribution. 25 Poor logistics data and the absence of real-time logistics information result in the stock of essential medicines in sub-Saharan African countries. 26
Without accurate, complete, timely logistics information, the logistics system would not run smoothly. 25 This study revealed that 74.5% of the assessed bin cards were accurate. Similar result was reported by study done in Ethiopia, Diredawa city. 27 However, the current result was less than the study conducted in Ethiopia, East Wollega 28 and Addis Ababa city. 29 In this study, the bin card inaccuracy varies among all health facilities, highest in health centers. According to the qualitative result, the potential reason for this could be the absence of proactive supervision from a higher level, lack of training on IPLS, lack of staff commitments, and the absence of computer software systems at health centers.
Regarding the availability of logistics formats, the study showed that all of the study health facilities had logistic recording tools (bin cards) and reporting forms (RRF and Internal Facility Report and Resupply). A similar finding was reported by the study conducted in Ethiopia, west Wollega. 9 However, the current result is better than the studies conducted in Ethiopia, National survey. 11 The reason might be a degree of commitment of the staff and support from a higher level. Those documents are crucial for the availability of tracer medicines and should always be available when and where needed.
Concerning the quality of the logistics report, the finding of this study showed RRF completeness of 72.2%, RRF timeliness of 88.8%, and accuracy of 69.4%. This finding is comparable to the previous study done in East Wollega, except for RRF completeness. RRF completeness is less than the finding reported by the previous study. 28 The difference could be because of the absence of supportive supervision from a higher level, and a decrease in IPLS training, as KIs responded.
Poor LMIS data quality leads to distortion of logistics data and results in poor supply decisions. This, in turn, causes a shortage of tracer medicines. Thus, timely supportive supervision from a higher level helps the professionals to efficiently perform their logistics activities by equipping them with professional knowledge and skills. 27
Good pharmaceutical storage practice
Regarding storage conditions, this study showed that 25% of assessed public health facilities met the accepted storage condition. This result is similar to the study conducted in East Shewa, where 5 (25%) of the health centers had fulfilled good storage condition criteria. 20 However, the current finding is less than the study done in Gambella region, 30 and study done in Diredawa town. 26 KIs identified some of the reason behind the issues, for instance poor infrastructures and inadequate skilled human resources. This study also revealed that storeroom were not sufficient for 9 (75%) of health facilities, and fire safety equipment is not available and accessible for 10 (83.33%). Similar storage criteria were not fulfilled in previous studies done in East Shewa and West Wollega.20,30 Storage conditions can affect the safety and quality of pharmaceutical products in store. A well-organized warehouse will streamline its different activities; the storage area has to be dry, well ventilated, out of direct sunlight, well lit, complete with safety equipment, clean, of adequate space, and with standard pallets and shelves. 11
Strength
The study employed mixed-method design which increases the level of evidence. For qualitative study, professionals from multiple organizations were interviewed. Therefore, the challenges identified were seen from multiple perspectives.
Limitation
Since we didn’t include health posts, we cannot generalize this result for the lowest-stream supply chain. But they only manage some of the tracer drugs. Many health facilities lack accurate and complete records. Therefore, we couldn’t calculate some logistics performance indicators like wastage rates, excess inventory, below emergency order points, and near emergency order points. Since all health centers managing ART drugs were considered we didn’t calculate sample size.
Conclusion
From this study, we concluded that the availability of tracer medicines on the day of the visit, logistics record updating practices, and inventory accuracy rate were better than the majority of the previous studies. However, facility report quality and inventory record accuracy require improvements. In addition, the majority of the assessed health facilities didn’t fulfill acceptable storage conditions and use electronic LMIS. The main challenges identified were a shortage of supplies, budget constraints, insufficient infrastructure, lack of commitment from top-level management, unavailability of an automated recording and reporting system, weak capacity building practice, and weak supportive supervision and feedback report. Therefore, EPSS and the Southwest Shewa zonal health department should arrange appropriate training, supportive supervision, and work to improve the availability of medicines and budget. The assessed health facilities should improve their pharmaceutical storage condition and avail electronic logistics management information system.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241274041 – Supplemental material for Inventory Management Performance for Tracer Medicines in Public Health Facilities of Southwest Shewa Zone Oromia Region, Ethiopia: A mixed study
Supplemental material, sj-docx-1-smo-10.1177_20503121241274041 for Inventory Management Performance for Tracer Medicines in Public Health Facilities of Southwest Shewa Zone Oromia Region, Ethiopia: A mixed study by Alemayehu Gutesa, Tariku Jebena and Oliyad Kebede in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121241274041 – Supplemental material for Inventory Management Performance for Tracer Medicines in Public Health Facilities of Southwest Shewa Zone Oromia Region, Ethiopia: A mixed study
Supplemental material, sj-docx-2-smo-10.1177_20503121241274041 for Inventory Management Performance for Tracer Medicines in Public Health Facilities of Southwest Shewa Zone Oromia Region, Ethiopia: A mixed study by Alemayehu Gutesa, Tariku Jebena and Oliyad Kebede in SAGE Open Medicine
Supplemental Material
sj-docx-3-smo-10.1177_20503121241274041 – Supplemental material for Inventory Management Performance for Tracer Medicines in Public Health Facilities of Southwest Shewa Zone Oromia Region, Ethiopia: A mixed study
Supplemental material, sj-docx-3-smo-10.1177_20503121241274041 for Inventory Management Performance for Tracer Medicines in Public Health Facilities of Southwest Shewa Zone Oromia Region, Ethiopia: A mixed study by Alemayehu Gutesa, Tariku Jebena and Oliyad Kebede in SAGE Open Medicine
Footnotes
Acknowledgements
The authors would like to thank data collectors for their commitment and staffs in public health facilities of Southwest Shewa zone for providing relevant data. We are also grateful to Addis Ababa University for facilitating the research.
Authors’ contributions
AG involved in reviewing articles, data collection process, involved in data analysis, interpreted data, participated in the sequence alignment, and drafted the manuscript and communicated for publication. TJ and OK also reviewed articles, participated in the design of the study, and performed the statistical analysis, interpreted data, and participated in the sequence alignment and drafted the manuscript. All authors read and approved the final manuscript.
Availability of data and materials
The data sets generated and/or analyzed during the present study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Addis Ababa University covered data collection fee.
Ethics approval
Ethical approval for this study was obtained from Addis Ababa University Institutional Review Board (Ref no: ERB/SOP/262/13/2021).
Informed consent
Written informed consent was obtained from all subjects before the study.
Trial registration
Not applicable.
Consent for publication
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
