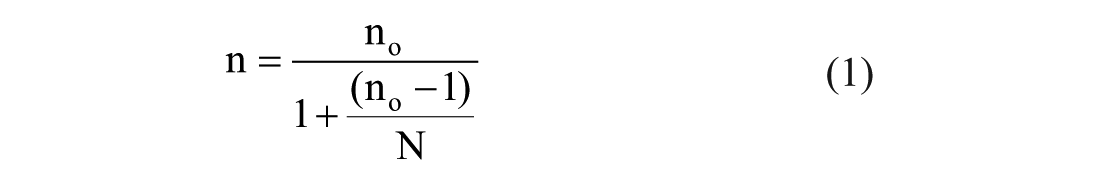Abstract
Flawed regulatory oversights restrict access to quality-assured medicines. Not more than 16% of African regulatory authorities have the capacity to execute standard regulatory functions; the challenges seem more pronounced in the Intergovernmental Authority on Development (IGAD) member states. Regional harmonization initiatives elsewhere demonstrated promising outcomes. However, progress in the IGAD Medicines Regulatory Harmonization Program has stalled calling for renewed commitment. This study explored the perspectives of pharmaceutical sector stakeholders in Ethiopia, and international experiences regarding the implementation of harmonized framework in the IGAD region. A survey-based descriptive design was sent to 582 professionals from key pharmaceutical sectors. Data analysis and reliability tests were conducted using SPSS version 26. An in-depth document review was carried out to inform the proposal of a harmonized regional framework. A total of 477 (82.0%) participants completed the survey. A large majority (83%) concurred the feasibility of regional harmonization and identified key benefits such as improved technical capacity for expedited regulatory services, enhanced access to quality medicines, and improved collaboration. Nevertheless, participants highlighted potential barriers such as instabilities, regulatory systems heterogeneity, and financial constraints. Benchmarking global and continental experiences, a centralized regulatory procedure comprising 6 milestones with an average target authorization timeline of 330 days along with phased prerequisite steps, is proposed as a feasible framework for IGAD. Participants largely favored the adoption of a harmonized regional regulatory framework similar to those implemented elsewhere in Africa. The success of IGAD’s harmonization efforts would be strengthened by establishing regional office with legal framework to ensure balanced contributions and benefits among member states, facilitating judicious listing of priority products, and coordinating phased implementation.
Keywords
Introduction
Medicines play indispensable roles in protecting health and socioeconomic development globally1,2 but they have inherent risks that require regulatory oversight to ensure public safety. Governments bear the responsibility for regulating healthcare products and technologies, which they typically assign to National Regulatory Authorities (NRAs).3,4 However, these authorities can sometimes fail to prevent products with design problems, quality defects or misleading information from reaching the market.5,6 At the same time, NRAs must facilitate access and equitable distribution of essential health interventions, particularly in resource constrained regions.7,8 The work of the NRAs is complicated by the fast pace of scientific advances, increasing complexities of healthcare technologies and evolving regulatory requirements.9,10
Many NRAs lack the resources and technical capacity required to execute standard regulatory functions effectively. Globally, an estimated 70% of NRAs operate within weak regulatory systems and fewer than 60 have achieved World Health Organization (WHO) Maturity Level 3 (ML 3).11,12 In Africa, only 9 (16%) of NRAs have reached ML3 level and appear to have the technical capacity to carry out core regulatory functions adequately. The challenges are more pronounced among the IGAD member states due to deep-rooted geopolitical and socioeconomic factors.13 -15 The Ethiopian Food and Drug Authority (EFDA) is the only NRA among the IGAD member states to achieve ML 3. 16
Many challenges confront for NRAs in sub-Saharan Africa, including insufficient system structure and resources, fragmented and complex regulatory procedures, lack of sustainable financing, shortage of skilled personnel, feeble law enforcement practices, and weak collaboration among countries.14,17 On the other hand, the complex pharmaceutical supply chain often permits the infiltration of substandard and falsified (SF) products and other illicit practices. Africa has been estimated to receive about 42% of global SF medical products with an estimated prevalence of 18.7%; close to half of online marketed medicines are reported to be SF.18 -20
Ongoing and future changes in the global health industry and market will continue to bring new challenges for regulators because of the increasing complexity and volume of products to be regulated. Such a dynamic industry must have responsive regulatory services but this is difficult to provide in resource constrained countries. Recently, African NRAs are pursuing regulatory harmonization initiatives that standardize technical expectations and reliance mechanisms that allow them to share review and oversight responsibilities.4,21 These initiatives can offer several benefits, including efficient resource utilization, cost-effective and enhanced regulatory oversight, enhanced access to essential products, better capacity to govern public health risks, predictable regulatory requirements for technological advancement, and concerted law enforcement.22 -25
The WHO championed the concept of harmonization in 1969 by establishing the “Certification Scheme on the Quality of Pharmaceutical Products” to ensure product safety in international trade. 26 Subsequently, several international and regional initiatives began to standardize technical expectations and then establish work-sharing and reliance mechanisms, typically under the aegis of formalized associations among countries. Early developments included the harmonized regulatory procedures of the European Community, which were developed to support a unified pharmaceutical market.27,28 These efforts were reinforced by the work of the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH).29,30 More recent examples of regional initiatives include the Association of Southeast Asian Nations (ASEAN) Pharmaceutical Regulatory Framework 31 ; the collaboration and harmonization initiative of the Gulf Co-operation Council (GCC) member states 32 ; the Pan American Network for Drug Regulatory Harmonization (PANDRH) 33 ; and the African Medicines Regulatory Harmonization (AMRH) initiative. 21
Launched in 2009, the AMRH initiative set the ambitious goal of establishing a single continental regulatory body, the African Medicine Agency, AMA.21,34 Harmonization activities subsequently cascaded into regional economic blocs, which initiated pilot projects involving joint GMP inspection, quality verification testing, dossier reviews, and regulatory enforcement. While the specific approaches varied across regions, most produced encouraging outcomes. For example, the East African Community (EAC) collaborative procedures reduced market authorization timelines by 40% to 60%, and the ZAZIBONA pilot project shortened the total dossier review and registration timelines to 8 months.6,21,35
The IGAD Medicines Regulatory Harmonization (IGAD-MRH) program was launched in 2017 to address the limited technical capacity and inadequate institutional systems that constrain the effectiveness of NRAs in most member states. However, progress toward implementing a harmonized and reliant regulatory system has been modest for several reasons. Some member states may be distracted by their participation in other harmonization and reliance initiatives. For example, Kenya and Uganda are active participants in the EAC pilot projects. In addition, legal and structural dissonance, insufficient resources, weak organizational performance, and various operational challenges remain significant barriers to success. Yet the extent to which these factors influence program implementation has not been systematically characterized. A clearer understanding of stakeholder experiences and perspectives could provide valuable insights for system design and organizational restructuring, alignment of legal and regulatory frameworks, and more effective sharing of resources. In this study, we explored the perspectives of a specific group of stakeholders, those working in the Ethiopia’s pharmaceutical sector, on the implementation of a harmonized regulatory system within the IGAD region.
Methodology
Research Design and Study Setting
A survey-based descriptive research design was used to explore the views of professionals from 3 major stakeholder groups: NRAs (including the Ethiopian Food and Drug Authority [EFDA] and the Ethiopian Agricultural Authority [EAA]); local pharmaceutical manufacturing companies; and pharmaceutical import firms. EFDA is mandated to regulate food and pharmaceutical products for human use, whereas EAA oversees veterinary medicines and feed. The import firms included in the study were responsible for importing human medicines and/or medical devices, which were based in Addis Ababa, and were registered with EFDA. Participants consisted of technical staff working across various directorates within EFDA and EAA, Chief Executive Officers of manufacturing companies and technical staff from core departments, and technical managers or regulatory affairs officers from pharmaceutical import companies.
Data Collection Methods
A structured self-administered questionnaire was used to collect the perspectives of professionals on the feasibility of harmonization of regulatory requirements and procedures within the IGAD region, potential opportunities, and implementation challenges. The survey instrument was initially developed by the first author based on an extensive literature review. It was subsequently reviewed and refined by the senior co-authors. Face validity was assessed by 2 experts with extensive experience in the regulatory sector, and the instrument was finalized after incorporating their feedback. In addition, an in-depth review of relevant literature, including continental and regional policies and strategies, progress reports, and peer-reviewed articles, was undertaken to inform the development of a proposed adaptable regional harmonization framework with metrics for evaluating performance.
Sample Size Determination and Sampling Procedures
The sample size for the survey was determined separately for the 3 subpopulations using single population proportion formula 36 with adjustment made for finite population (equation (1)). 37
Based on the total number of eligible technical staff (Ni) from each sector, a sample size of 582 (the sum of ni) was determined, derived from the calculated sample size (no) of 385 for each group at a 95% confidence level and a 0.05 margin of error. The overall sample was then proportionally allocated among the various departments within the manufacturing companies and regulatory authorities. Potential respondents with more than 1 year of work experience who were willing to participate in the study and available during the data collection period were randomly selected using a lottery method and subsequently approached.
Data Collection and Analysis
Data were collected between January and February 2023 by 3 research associates holding B. Pharm and MSc degrees who received training on the data collection tools under the supervision of the principal investigator. The data collection process was supported by leadership teams from the respective organizations. Participants from Branch Offices of the NRAs and import firms completed the survey via online form distributed by email, with regular reminders sent by the principal investigator. Daily data review and cleaning ensured that only complete surveys were analyzed. Data analysis was conducted using SPSS version 26; descriptive statistics using percentages and weighted averages were used to analyze ordinal data from Likert scale variables. Internal consistency of the data collection tool was tested (Cronbach Alpha values exceeding .8), indicating excellent reliability and data quality (Table 1). The range between highest value (5) and lowest value (1), and the interval between each response category of 0.8 were used for interpreting Likert Scale results.
Reliability Measures of Variables, Regional Regulatory Harmonization (N = 477).

Ethical Considerations and Confidentiality
Ethical clearance was obtained from Institutional Review Board of the Authors’ institute (CHS_IRB-086/19/SoP: 02/2021). Permissions were secured from the relevant institutions following official requests, and informed verbal consent was obtained from all study participants after they were briefed on the study’s purpose and expected significance. To ensure anonymity and confidentiality, and to mitigate social desirability bias, no personal identifiers were collected during data gathering or included in the analysis.
Reporting Guidelines
This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 38
Results
Sociodemographic Characteristics of Study Participants
Of the 582 individuals approached to participate, 477 (82.0%) completed and returned the survey. Most participants were men (74.6%) between 21 and 40 years of age (82.2%). Experts from the 3 key sectors included local pharmaceutical manufacturing (49.1%), regulatory affairs (36.1%), and pharmaceutical imports (14.9%; Table 2). Most professionals had backgrounds in pharmacy (43.3%) and engineering (16.8%), while those with regulatory affairs qualification accounted for only 0.8%, marking the limited pool of qualified regulators. Over half (56.0%) held a Bachelor’s degree and one-third (32.9%) had Master’s degree. In addition, about 44% reported having more than 10 years of work experience in their respective sectors.
Sociodemographic Characteristics of Respondents (N = 477).

Feasibility of Regulatory Harmonization in IGAD
Asked about the feasibility of harmonizing regulatory requirements and procedures in the IGAD region, most participants (83%) agreed or strongly agreed that regional harmonization is viable, while a small proportion (1.5%) expressed their strong disagreement (Figure 1).

Stakeholders’ perspectives on the feasibility of regulatory harmonization in IGAD (N = 477).
The participants were also asked to rate the benefits and importance of regional regulatory harmonization across 17 parameters (Table 3). Improving regional and international collaboration, enhancing access and affordability of essential medicines, and strengthening the technical capacity of NRAs were the most frequently cited benefits, rated as extremely or very important (WA: 4.31, 4.28, and 4.23, respectively). In contrast, reduction in product registration and market authorization costs, as well as overall regulatory system expenses in individual countries, were reported to have lower levels of importance (WA: 3.75 and 3.81, respectively).
Participants’ Rating on the Importance of Major Benefits of Regional Regulatory Harmonization in IGAD (N = 477).
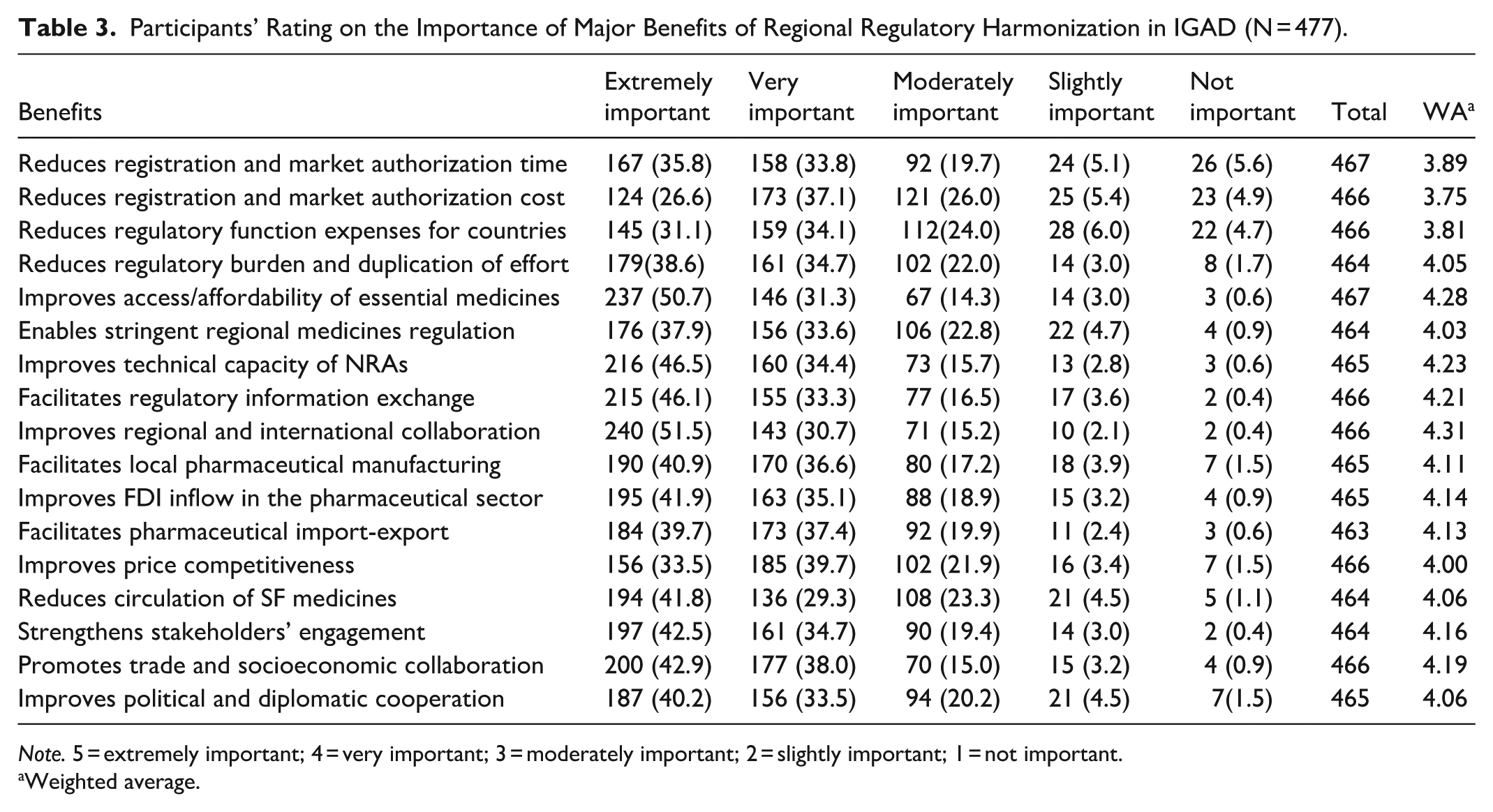
Note. 5 = extremely important; 4 = very important; 3 = moderately important; 2 = slightly important; 1 = not important.
Weighted average.
Challenges and Opportunities for Regulatory Harmonization in IGAD
Participants were asked to rate the level of influence of 9 potential factors that could impact the process of regulatory harmonization in IGAD. All the listed factors were perceived as having high impact. Internal and regional instabilities (70.4%), heterogeneity in regulatory systems (69.2%), and financial constraints (68.0%) were most frequently rated as having remarkably high or extremely high impact (WA: 34.0; 3.9; 3.9, respectively; Table 4). Fears of losing national sovereignty and shortage of a qualified workforce were perceived to have comparatively lower impact, with WA values of 3.44 and 3.56, respectively, although half or more of the respondents still rated their impact as extremely or remarkably high.
Major Challenges to Regional Regulatory Harmonization in IGAD and Their Level of Impact (N, %; N = 477).
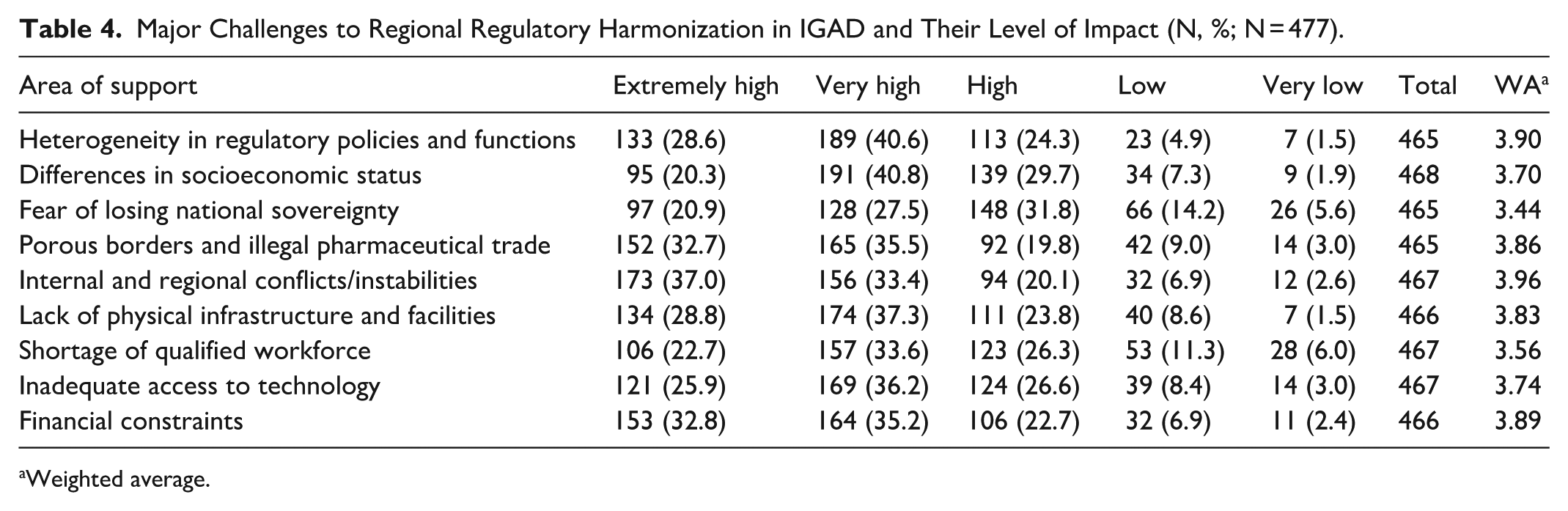
Weighted average.
When asked about potential opportunities for regulatory harmonization, the participants similarly ranked all of the listed options (Figure 2). The most commonly noted opportunities were the already established diplomatic and economic cooperation among member countries (56.8%), experiences in cross-border control (53.5%), and the potential for international support and collaboration (52.2%). The existence of strong regulatory system in some member states (44.7%) and the recent African Continental Free Trade Area Agreement (47.0%) were less frequently identified as opportunities.

Potential opportunities for regional regulatory harmonization in IGAD (N = 477).
Regulatory Harmonization Procedures for IGAD
The development of the IGAD medicine regulatory harmonization program has been slow, but could profit from a phased-approach that prioritizes the implementation of selected core procedures and functions. To this end, participants were presented with 9 areas of activity and asked to suggest the order of priority for implementation by assigning each to a first, second, or third phase. The 3 areas most commonly assigned to the first phase, compared with the third phase were: harmonization of regulatory policies and instruments (80.5% vs 2.3%); standardization of technical requirements (80.1% vs 0.4%); and joint facility inspections (67.9% vs 6.1%). In contrast, joint post-marketing surveillance, border control, and collaborative regulatory law enforcement were most frequently suggested for third-phase implementation (35.8%, 25.2%, and 30.2% respectively; Figure 3).

Prioritization of regulatory functions toward harmonization in IGAD (N = 477).
Suggested Harmonization Framework and Performance Metrics for IGAD
Findings from the survey and literature review suggest that a harmonization framework is an adaptable option for the resource constrained IGAD region. However, there is a considerable disparity in the system structures and technical capacities of NRAs across member states (as referenced in the annexed Supplemental File, S1 Table). The comprehensive literature review allowed comparison of the challenges for and best experiences within IGAD and those observed in other regions, particularly African regional economic blocs to inform the proposed centralized regulatory harmonization framework (Figure 4) which also incorporates performance monitoring metrics (annexed Supplemental File, S2 Table). Under this framework, a regional secretariat would be established to coordinate, implement, monitor, and communicate activities undertaken by participating agencies. The model assumes that IGAD, similar to other regional blocs, will primarily handle generic and biosimilar products with established records of successful market entry in other countries/jurisdictions. Evidence from other pilot programs in Africa was referenced to justify a reasonable average target of 330 days (11 months) for product registration and market authorization. This timeline may be extended for more complex products or repeated query cycles. A limit on the number of query cycles may be imposed, and applications that exceed the allowable timeframe could be removed from active review and advised for resubmission.

Suggested centralized regulatory harmonization framework for IGAD.
The suggested performance evaluation metrics were aligned with milestones corresponding to core regulatory activities. The first milestone involves receipt and recording of applications at the primary information desk. The second milestone concerns validation of submissions prior to entry into the review queue to ensure that they meet administrative and technical requirements. The third milestone is the completion of scientific and technical evaluation by internal and external experts, including the full query management process. The fourth milestone marks the final decision by the central secretariat to approve or reject the submission. The fifth milestone involves applicants submitting market authorization requests to respective NRAs. The sixth milestone is the granting of registration and marketing authorization by member states. In cases where an applicant holds a valid GMP certificate issued by a well-resourced NRA from within the IGAD region or by other stringent regulatory authorities (SRAs), the designated inspection team may review the existing inspection reports and decide accordingly.
Discussion
The approval and oversight of quality medicines is a critical responsibility and can be especially challenging for regulatory agencies with immature structures and limited access to technical expertise. These difficulties were formally acknowledged in 2014, when the World Health Assembly adopted a resolution on regulatory system strengthening. 39 Since then, a growing wave of global initiatives has not only introduced harmonized standards and systems but also encouraged countries to build on these harmonized approaches through reliance and work-sharing mechanisms, enabling 2 or more regulatory authorities to collaborate and reduce their administrative burden. Reliance is the process by which 1 NRA fully or partially depends on the regulatory assessments conducted by another trusted NRA to inform its own decision-making, while still retaining responsibility and accountability for the final decision. 40 Reliance enables more efficient use of regulatory resources and supports timely, high-quality regulatory functions. When supported by harmonization initiatives, regulatory reliance has been successfully implemented across regions with diverse economic conditions.40 -42
Not surprisingly, African countries have attempted to introduce harmonization and collaboration at many levels. Their efforts have been reinforced by introduction of the African Union Model Law on Medical Products Regulation, 43 which has now been ratified by several countries. 44 Its adoption has encouraged the formation of several regional consortia, often organized around geographical proximity or economic cooperation agreements. Some of these groups have already improved regulatory efficiency. For example, the South African Health Products Regulatory Authority (SAHPRA) achieved more than a twofold reduction in marketing authorization timelines by adopting the WHO-recommended abridged regulatory review procedure. 45
Similarly, the EAC-MRH joint assessment and inspection program reduced the median review and approval time by half between 2015 and 2023. 46 The WHO collaborative registration procedure using SRAs pathways has likewise improved performance in Africa and the Caribbean. 47 Given these successes, it is therefore unsurprising that IGAD member states are eager to develop harmonization and reliance programs. What is surprising, however, is the slow progress toward implementation. A central aim of this study was therefore to determine whether stakeholders place insufficient value on such a program or whether the barriers to implementation are simply too high.
The study explored insights from major beneficiaries including manufacturers, importers, and regulators in Ethiopia. The creators of the African Union Model Law on Medical Products Regulation underscored the importance of addressing stakeholders’ concerns and incorporating their perspectives in harmonization efforts.44,48 Stakeholders are uniquely positioned to evaluate the “black box” of a specialized and opaque system, and articulate their perspectives. Their qualifications and experiences enable study participants to reflect on the potential values, the benefits as well as the limitations of a harmonized IGAD system and challenges it must overcome. Their feedback yielded at least 3 important messages relevant to the direction of IGAD’s regulatory initiative.
First, most participants strongly agreed that a harmonized program is valuable for IGAD, given the disparities among member states in regulatory structure and technical capacity. Participants emphasized the potential of regional harmonization to improve access to quality-assured medicines, counter illicit cross-border practices, strengthen regulatory information exchange, and foster competitive business environment.
Despite its strategic proximity to global trade routes, the IGAD region faces the highest levels of poverty in sub-Saharan Africa, exacerbated by subsistence farming and pastoralism, weak technology infrastructure, fragmented manufacturing and trade systems, limited access to essential healthcare products, recurrent internal and cross border conflicts, poor interstate collaboration, and illicit cross border trade. 49 Regulatory resources are severely limited. Harmonization therefore presents an opportunity not only to streamline transactional activities such as dossier review, but also to contribute to broader socioeconomic objectives such as inclusive regional growth, competitive markets, expanded domestic and foreign investment, and optimal use of scarce resources.
Second, respondents identified numerous challenges likely to slow implementation. Developing a transnational regulatory system is inherently difficult, and it must overcome the early-phase obstacles. In IGAD, the regulatory systems vary widely. Legal frameworks, system structures, technical capacity, and sociopolitical contexts differ substantially, and in several member states, these conditions are unpredictable.50,51 Participants of this study rated all the 9 listed challenges as having major implications for harmonization. Heterogeneity in system structure and regulatory policies, internal and regional instabilities, financial constraints, cross border illicit practices, and concerns of losing national sovereignty among member states were most frequently cited as critical implications. The findings mirror reports elsewhere, where misaligned policies, varying national priorities and competitive commercial interests, lack of common understanding on quality and safety standards, resource constraints, and nationalist concern hinder regional harmonization.9,13,52,53 As indicated by the findings, the limited pool of qualified regulatory workforce may also compromise the quality and efficiency of implementation; therefore, tailored short-term and long-term training mechanisms need to be strategically developed. Assessment of the EAC regulatory harmonization program identified lack of a binding regional office, inconsistent regulatory practices, varied technical standards and labeling requirements, absence of centralized submission and tracking system, and lack of a unified fee structure as major obstacles. 46 Comparable challenges such as capacity disparities, protracted decision-making, and complexities related to Halal certification were documented for the ASEAN framework. 54 Nevertheless, IGAD-MRH program can leverage decades-long political and socioeconomic integration efforts in the region and draw technical support from continental and global initiatives to strengthen its systems.
Third, respondents highlighted areas that might be given priority if a phased-implementation becomes necessary. Alignment of regulatory policies, standardization of technical requirements, and joint inspections were highest-ranked first-phase priorities followed by system-level restructuring, and joint implementation of quality control, market authorization and post-marketing surveillance.13,20 Several regions, namely, Asia, Latin America, and Africa have successfully implemented similar phased approaches.46,47,55,56
For the IGAD region, phased implementation pilot projects for joint execution of core regulatory functions could prevent premature skepticism from member states due to initial challenges in policy alignment, resource mobilization and service optimization. This approach could also reduce inefficiencies that often arise when overly ambitious, long-term plans lack early practical grounding. Still, IGAD cannot operate in isolation; it must align with continental AMRH efforts and AMA’s emerging systems. Careful consideration and consensus-building among member NRAs will be crucial to avoid “reinventing the wheel.”57 -60
To improve equitable access to essential healthcare products, AMA, and regional harmonization programs perform complementary functions while operating at distinct structural levels and focus areas. After careful consideration of the challenges and technical feasibility, the AMRH program has prioritized clinical trials oversight, scientific review, GMP inspections, and post-market-surveillance for high-risk product categories such as APIs, vaccines, new chemical entities, and advanced diagnostics for continental implementation through AMA. In contrast, dossier assessment, product registration, GMP inspections, and post-marketing surveillance of most generic products and biosimilars, as well as regulation of multi-country clinical trials, are considered more suitable for centralized regional procedures.
Although the IGAD pharmaceutical sector is growing, it remains under-resourced, heavily import-dependent, price-sensitive, and minimally competitive. If effectively implemented, a centralized model could improve access to essential medicines, strengthen governance and technical capacity, facilitate cross-border pharmaceutical trade, and support industrial development. Respondents also identified socioeconomic and diplomatic cooperation, existing regional and international partnerships, potential for pooled resources, and the region’s dire need for inclusive growth as enabling conditions. Similar stakeholder support has been noted in Southern African Development Community (SADC) region. 61
Recognizing the rapidly evolving global dynamics in drug development, clinical trials, manufacturing and pharmaceutical trade, and the limited regulatory capacity in many low- and middle-income countries, major institutions such as WHO and the National Academy of Medicine have long advocated for the harmonization of regulatory requirements.26,53 Harmonization ultimately benefits not only manufacturers, regulators and importers, but also the public through enhanced access to safe and effective medicines. Harmonization creates unified standards, and streamlined risk-based oversight across the product lifecycle.
Previous studies show that harmonization reduces administrative burdens on NRAs, expands manufacturers’ market access by eliminating duplicative submissions, fosters innovation through predictable regulatory environment, and strengthens supply chain security with real-time data management systems and tracking technologies.35,53,62 Positive gains have been reported across multiple regions with illustrated promising outcomes.6,28,63 Accelerated approvals have been documented in ASEAN, 54 ECOWAS, 56 and EAC. 46 Greater than twofold reduction in marketing authorization time was reported by the SAHPRA’s adoption of abridged review reduced timelines by more than half. 45 Likewise, WHO’s collaborative registration using SRA reliance improved performance in Africa and the Caribbean. 47 The EAC-NRH joint program cut the median review times by half, 46 and has been praised by manufacturers for reducing duplicative submissions. This study also identified that review delays often stem from administrative bottlenecks, notably excessive query cycles. A centralized submission system with real-time tracking could reduce these cycles from 4 to 2.
While Africa is progressing toward establishing the continental medicine agency, AMA, the IGAD-MRH program remains limited to small-scale joint dossier assessments and GMP inspections. Benchmarks from the comprehensive review of international and continental harmonization initiatives informed a centralized procedure as an adaptable framework for the IGAD region. IGAD can accelerate progress by adopting lessons from other regions and transitioning to such a centralized model. This framework can resolve reported critical challenge by similar regional initiatives: lack of a central office with binding legal framework. Doing so would address major challenges that have hindered other African regional initiative, the absence of a binding central office. Pilot initiatives in ECOWAS, ZaZiBoNa, and EAC, while commendable haven’t realized their full potential due to operational misalignments, inconsistent commitments, and fragmented fee structures.35,58,59 International experience, particularly from EMA, and similar models in Asia and Latin America demonstrates the feasibility and benefits of centralized regulation. 64 A centralized IGAD-MRH system could coordinate and jointly execute core regulatory functions, particularly for priority products.
Market authorization timelines are essential performance indicators for NRAs but vary due to system structure, technical capacity, product complexity, and available experience. Based on regional and international benchmarking and the realities of IGAD, a target timeline of 330 days is proposed for generic medicines and biosimilars under a centralized procedure. For comparison, global experience reported varied timelines: (i) EMA timelines range from 254 to 434 days depending on product complexity 64 ; (ii) GCC timelines range 360 to 540 days, with a 60 days fast-track scheme for products registered in at least 2 member states57,65; (iii) ECOWAS joint review averages 316 days, including final national authorization60,66; and (iv) ZaZiBoNa and EAC issue authorizations in about 360 and 405 days, respectively, including the final national decision period (90 days).58,59 The IGAD target timeline takes into accounts: (i) the establishment of a central office with binding legal authority; (ii) availability of benchmarking experiences; (iii) existing national legal frameworks and technical capacity; and (iv) feasibility of phased implementation.
The urgency for continental and regional harmonization in Africa is emphasized due to public health and economic imperatives. Sustainable implementation of this proposed framework entails IGAD member states undertake fast-track alignment of regulatory policies, careful delimitation of products for the centralized procedure, and mobilization of financial and technical resources for pooled utilization along with the establishment of an accountable regional office. Socioeconomic, technological, and political interests of all member states need to be addressed in abreast with balanced consideration of their contribution and regional targets.
Study participants highlighted concerns over the potential erosion of national sovereignty as a barrier to expedited implementation. The final approval by NRAs is therefore often seen as a bottleneck, necessitating careful prior deliberation and consensus. This step ensures that member states retain sovereign decision-making authority, particularly during the initial phases. However, it also leads to duplication of efforts, longer approval timelines, higher costs, and inconsistent requirements. Mitigation strategies include inclusive stakeholder engagement, equitable benefit-sharing frameworks, agreed binding systems for reliance and joint approval procedures, harmonized timelines, shared digital platforms for real-time information exchange, and targeted capacity-building programs for NRAs.
Successful adoption of a centralized IGAD framework will require: (i) alignment of legal frameworks and system structures through objective assessments; (ii) establishment of a regional office equipped with a robust real-time Regulatory Information Management System (RIMS); (iii) clear designation of priority product categories for mandatory centralized review; (iv) implementation of short-term and long-term capacity-building programs for regulatory personnel; and (v) progressive benefit-risk analyses to guide system refinement. Balancing the socioeconomic, political, and technological disparities among member states is essential to achieving equitable contributions and fair distribution of benefits.
It is important to note that countries are transitioning from fragmented national procedures, with varying levels of technical capacity, to a unified regional framework. As highlighted by participants, several factors may obstruct the implementation process. The feasibility of the proposed framework depends on breaking down of complex, and long-term initiatives into manageable pilot projects. Political commitment through transparent and pragmatic negotiations, signed treaties, allocation of requisite financial and technical resources, harmonization of technical requirements and targeted capacity-building programs are essential prerequisites. Undertaking joint pilot programs such as GMP inspections and dossier assessments, and leveraging existing reliance procedures can serve as preliminary gateways for full implementation. A sequence of activities, potentially spanning 1 to 2 years, is recommended as a logical testing model prior to full-scale rollout (Figure 5). These activities include: (i) mobilizing critical inputs (legal tools, financial, and technical infrastructure); (ii) undertaking shared and agreed-upon activities (staff training, technical standardization, development of digital platforms, and piloting of selected projects); (iii) measuring initial success indicators (adoption of technical standards, trained staff, interoperability of digital platforms, efficiency preliminary functions); and (iv) strategizing interventions to address identified challenges and deficiencies.
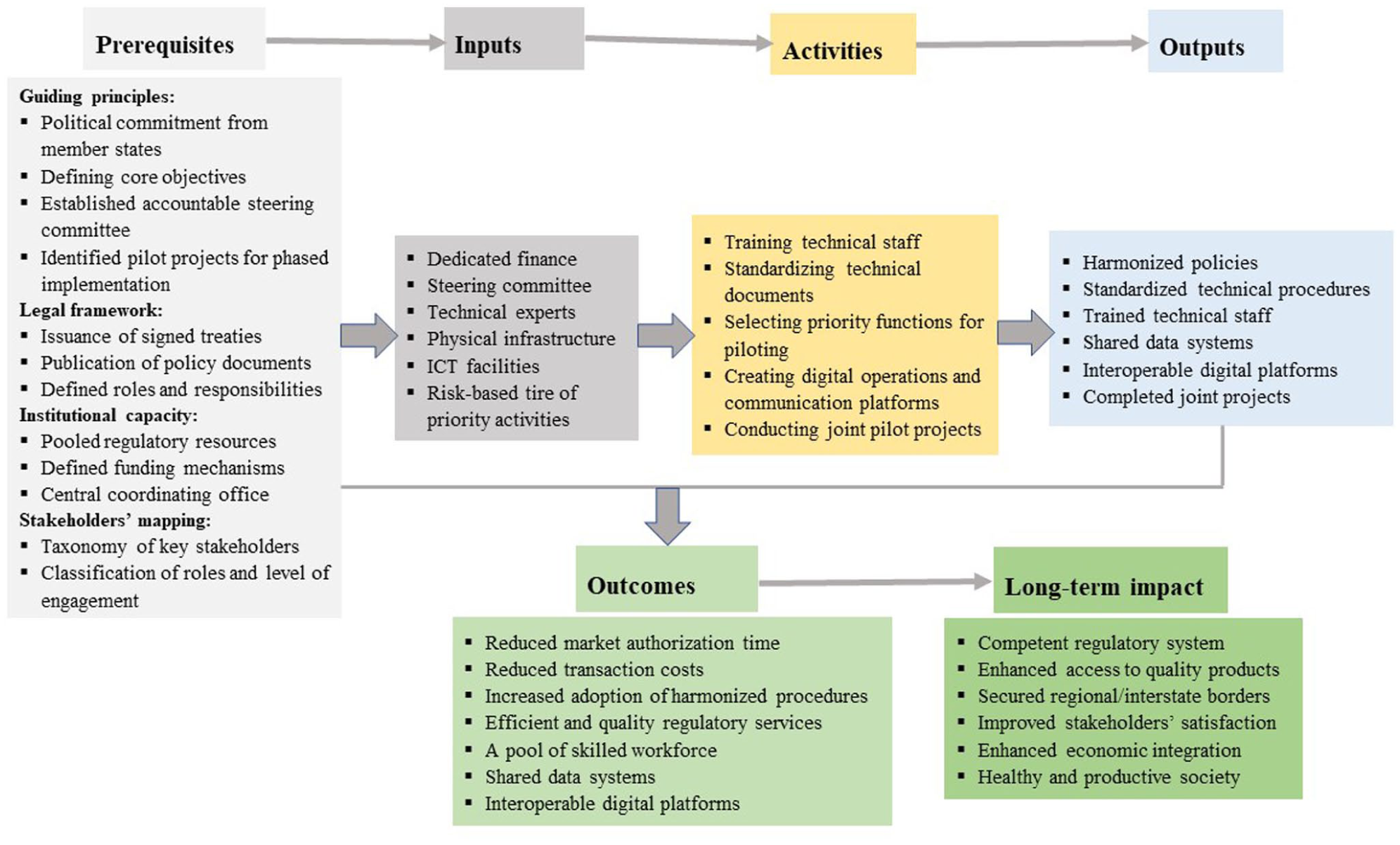
Implementation of the logical framework for the proposed centralized regional regulatory procedure.
Limitations of the Study
This study gathered insights from key stakeholders and synthesized regional and international experiences through an extensive literature review. However, the inherent limitation of descriptive study design, lack of rigorous content validation on the survey instrument, and the single-country focus limit the generalizability of findings across the IGAD region. Comparative, multi-country assessments, including qualitative inquiries, would provide a more comprehensive understanding.
Conclusion
Many African NRAs lack sufficient capacity to execute core functions and respond effectively to evolving industry needs, thereby limiting access to essential medicines and health technologies. Consequently, regulatory harmonization has emerged a strategic solution across multiple regions. African economic blocs have adopted AMRH initiatives with encouraging results; evaluations recommend establishing centralized regional frameworks. The IGAD-MRH program should therefore build on existing experiences and transition to a centralized procedure aligned with the continental AMRH program progress.
Based on benchmarked experiences, a target market authorization timeline of 330 days, with flexibility for complicated products, is proposed for the suggested centralized IGAD-MRH framework. Intercountry reliance, parallel decision making, and mutual recognition mechanisms can be incorporated. Sustainable implementation of the centralized framework necessitates the establishment of an accountable regional office supported by binding legal framework and equipped with real-time tracking and communication platforms. Transition efforts should emphasize reliance on NRAs, as better-resourced systems with strengthened technical capacity to enhance the transition. Additionally, transparent deliberations to balance committed contributions and inclusive benefits among member states, effective engagement of key stakeholders, and the formation of a critical mass of technically competent workforce can collectively drive sustainable implementation.
Although a centralized framework can accommodate all product types, prioritizing high-risk and essential products will facilitate a pragmatic phased implementation. As the AMA continues to evolve, IGAD must learn from regional and global experiences and proactively address early-phase challenges to realize a functional, efficient, and impactful regulatory system.
Supplemental Material
sj-docx-1-inq-10.1177_00469580261442037 – Supplemental material for Ethiopian Pharmaceutical Sector Stakeholder Perspectives on Regulatory Harmonization in IGAD: A Descriptive Study
Supplemental material, sj-docx-1-inq-10.1177_00469580261442037 for Ethiopian Pharmaceutical Sector Stakeholder Perspectives on Regulatory Harmonization in IGAD: A Descriptive Study by Tesfa Marew, Anteneh Belete, Frances J. Richmond and Tsige Gebre-Mariam in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-2-inq-10.1177_00469580261442037 – Supplemental material for Ethiopian Pharmaceutical Sector Stakeholder Perspectives on Regulatory Harmonization in IGAD: A Descriptive Study
Supplemental material, sj-docx-2-inq-10.1177_00469580261442037 for Ethiopian Pharmaceutical Sector Stakeholder Perspectives on Regulatory Harmonization in IGAD: A Descriptive Study by Tesfa Marew, Anteneh Belete, Frances J. Richmond and Tsige Gebre-Mariam in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-docx-4-inq-10.1177_00469580261442037 – Supplemental material for Ethiopian Pharmaceutical Sector Stakeholder Perspectives on Regulatory Harmonization in IGAD: A Descriptive Study
Supplemental material, sj-docx-4-inq-10.1177_00469580261442037 for Ethiopian Pharmaceutical Sector Stakeholder Perspectives on Regulatory Harmonization in IGAD: A Descriptive Study by Tesfa Marew, Anteneh Belete, Frances J. Richmond and Tsige Gebre-Mariam in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Supplemental Material
sj-pdf-3-inq-10.1177_00469580261442037 – Supplemental material for Ethiopian Pharmaceutical Sector Stakeholder Perspectives on Regulatory Harmonization in IGAD: A Descriptive Study
Supplemental material, sj-pdf-3-inq-10.1177_00469580261442037 for Ethiopian Pharmaceutical Sector Stakeholder Perspectives on Regulatory Harmonization in IGAD: A Descriptive Study by Tesfa Marew, Anteneh Belete, Frances J. Richmond and Tsige Gebre-Mariam in INQUIRY: The Journal of Health Care Organization, Provision, and Financing
Footnotes
Acknowledgements
TM gratefully acknowledges Addis Ababa University for sponsoring his PhD studies, and the Center for Innovative Drug Development and Therapeutic Trials for Africa (CDT-Africa), and Armauer Hansen Research Institute (AHRI) for partial funding. The Authors extend their sincere appreciation to the Ethiopian Food and Drug Authority (EFDA), the Ethiopian Agricultural Authority (EAA), the participating local pharmaceutical manufacturing companies, and pharmaceutical import firms as well as all study participants, for their valuable contributions. Ms. Blen Berhanu is duly acknowledged for her assistance in the SPSS analysis.
Ethical Considerations
Ethical clearance was obtained from the Institutional Review Board of the College of Health Sciences, Addis Ababa University (CHS_IRB-086/19/SoP:02/2021).
Consent to Participate
Informed verbal consents were obtained from all study participants prior to data collection. To maintain confidentiality, no personal identifiers were used during data analysis and reporting of the findings.
Consent for Publication
Not applicable.
Author Contributions
TM: Conceptualization, Methodology, Data collection, Data curation, Formal analysis, and writing the original draft. TGM: Conceptualization, Methodology, Visualization, Review & Editing, Supervision, and Project administration. AB: Conceptualization, Methodology, Review & Editing, and Supervision. FJR: Conceptualization, Visualization, Review & Editing, and Supervision.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The first author received a small amount of fund from the Center for Innovative Drug Development and Therapeutic Trials for Africa (CDT-Africa), College of Health Science, Addis Ababa University, and Armauer Hansen Research Institute (AHRI) that could cover the cost of data collection.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.