Abstract
Objectives:
This study aims to assess access to essential medicines used in the management of noncommunicable diseases through analysis of the availability, prices, and affordability of these essential medicines in Arba Minch town, Gamo Zone, Southern Ethiopia.
Methods:
A cross-sectional design was carried out using the World Health Organization/health action international methodology between 2 March and 2 May 2023, within public and private healthcare facilities located in Arba Minch town, Southern Ethiopia. The median price ratio served as a metric. Statistical tests like the Shapiro-Wilk and Kolmogorov-Smirnov were utilized to assess the normal distribution of price data. The Wilcoxon-Mann-Whitney U test was also employed to compare median buyer’s prices (patient prices) between public and private healthcare institutions. Treatment affordability was determined by estimating the number of days’ wages required by the lowest-paid government employee in Ethiopia to afford the prescribed medication regimen.
Results:
Among 23 health facilities surveyed, the pooled availability of essential medicine used in the management of noncommunicable diseases was 18.7% (range: 0%–30.1%), with the public and private facilities contributing 16.3% and 38.3%, respectively. The overall percentage of availability originator brand versions was 1.1% for overall health sectors, 0.6% for public sectors, and 1.2% for private sectors. The overall percent availability of lowest price generics was 36.2% (range: 0%–26.2%; public: 32.0%; private: 37.1%). Only seven lowest price generics satisfied the World Health Organization target of 80% and above. The overall median price of lowest price generic medicines in private was two times higher than in public sectors. The top five median price scorers were amlodipine, furosemide, insulin, beclomethasone, and salbutamol. The Mann-Whitney U test showed that 11.6% of lowest price generics medicines had a statistically significant median price disparity between the public and private sectors (p < 0.05). The overall percent of unaffordability was found to be 100.0%, (public: 70.4; private: 100.0%).
Conclusions:
This study revealed the limited availability and potential financial burdens on patients seeking essential noncommunicable disease medications. Limited availability suggests the need for better supply chain management and consistent stock availability. The price disparities and affordability challenges identified underscore the necessity for policy interventions such as price regulation and subsidized programs to ensure equitable access to essential noncommunicable disease medications in Arba Minch town, Southern Ethiopia.
Keywords
Background
Noncommunicable diseases (NCDs) represent a significant health burden globally, contributing to increased morbidity and mortality rates, particularly in low- and middle-income countries (LMICs).1,2 In Ethiopia, NCDs have emerged as a growing public health challenge, accounting for one-third of the population and presenting a considerable portion of the disease burden. 3 Access to essential medicines is a critical component of effective NCD management. 4 However, studies reveal difficulties in accessing these medicines in LMICs due to factors such as inadequate availability, high prices, and limited affordability.5,6
Accessing medicines within health systems involves dimensions such as availability and affordability. 7 Ensuring affordable, quality-assured essential medicines is pivotal, as it alleviates financial burdens, reduces suffering, curtails illness durations, and prevents unnecessary disabilities and deaths globally. 8 Nonetheless, approximately one-third of the world’s population lacks consistent access to essential medicines, leading to a cascade of preventable suffering.9,10 This figure escalates to over 40% in low-income countries and exceeds 50% in the poorest regions of Asia and Africa, despite legislative efforts supporting Universal Health Coverage (UHC) implementation in these nations. 11
National and subnational surveys using the World Health Organization/Health Action International (WHO/HAI) approach have revealed insufficient availability of medicines, particularly within the public sector, and inadequate affordability of medications meant to address various NCDs such as cardiovascular diseases (CVDs), diabetes, psychiatric disorders, asthma, and epilepsy.6,12–16 In 2007, Mendis et al. 15 evaluated the availability and affordability of medicines for four NCDs in six LMICs using a modified WHO/HAI technique. Their findings highlighted that generic availability in the public sector was notably low, not exceeding 7.5% in Bangladesh, Malawi, Nepal, and Pakistan. Furthermore, standard treatments often required a substantial portion of individuals’ incomes, equivalent to five or more days’ wages when purchased from the private sector. Other similar NCD surveys within Middle Eastern and sub-Saharan countries also demonstrated the challenges of access to NCD medicines.17,18 For example, Cameron et al. 19 reported suboptimal availability of medicines in both public and private sectors in developing nations, emphasizing that generics for NCDs were notably less accessible compared to generics for acute conditions.
In numerous developing countries, limited financial means or access to information act as significant barriers, hindering the accessibility of essential medicines and contributing to elevated rates of morbidity and mortality.20,21 Apart from the scarcity of essential medicines, the high cost of medications presents a formidable challenge in delivering adequate health services. 22 Affordability concerns regarding medicines are particularly pronounced in LMICs, where as much as 90% of the population acquires medications through out-of-pocket payments, greatly affecting their ability to afford treatment and affecting disease outcomes.23,24 Ethiopia, among other developing nations, grapples with the severe consequences stemming from the lack of availability and affordability of medicines. 25 A systematic review conducted by Tewuhibo et al. 26 underscores that the average national availability of essential medicines in Ethiopia falls below the WHO’s recommended target.
Therefore, the objective of this study was to assess the access to a thorough analysis of availability, prices, and affordability of essential medicines used to address NCDs within selected healthcare facilities situated in Arba Minch town, Southern Ethiopia.
Methodology
Study area, period, and design
The study was carried out in public and private health institutions of Arba Minch town between 2 March and 2 May 2023. The town of Arab Minch can be found 505 km from Addis Ababa, the capital of Ethiopia. The estimates for 2021 indicated that the town had a total of 192,043 residents. 27 During the study, the town had 1 public general hospital (Arba Minch General Hospital), 1 primary hospital (Dilfana Primary Hospital), 2 health centers, 11 health posts, 33 private clinics, and 23 drug stores.
The study utilized a facility-based survey to determine the availability, patient price, and affordability of 43 essential medicines for managing NCDs. This study adopted the methodology (comprises study design, data collection, and analysis) prepared by the WHO/HAI or evaluating medicine prices, availability, and affordability. 28
Population and inclusion criteria
All public and private health facilities located in Arba Minch town were considered as a source population. The study included private drug outlets and outpatient outlets in public health facilities that satisfied the WHO/HAI guideline requirements. This study also included essential medicines used in the treatment of NCDs that are listed on the WHO/HAI guideline and Ethiopian Essential Medicine List (EML)-2020 and budget items available for sale to patients.14,29 Based on WHO/HAI recommendations, the availability and affordability of essential medicines for the NCDs were evaluated based on the top 10 morbidities, that could be treated without hospitalization, in the Arba Minch town. Nongovernmental health sectors, cost-free and/or programmed essential NCD medicines were excluded in the recent study.
Sample size determination and sampling procedure
Survey selection of areas and institutions
According to the WHO/HAI standard sampling approach, one survey area covering about 100,000–250,000 people, accessible within 1 day’s travel from the major town, large enough to adequately represent the survey region, and that includes the required number of health sectors could be studied. 28 The guideline also suggests determining the scope of the studies, such as if they are national or regional surveys. Regional surveys are recommended for countries that possess large geographical areas or populations. Thus, Arba Minch town was the survey region in this study. Considering the administrative divisions and health sectors in Arba Minch, this study selected four administrative subcities and two kebeles namely Secha, Sikella, Abaya, Nechsar, Limat, and Shara, respectively, making six survey areas. To select the medicine outlets for the study based on the WHO/HAI standard sampling technique, four public health facilities (such as 1 general hospital, 1 primary hospital, and 2 health centers) were selected. Then, 20 private drug outlets were selected, comprising five to near each of the included public health facilities.
Survey selection of chronic disease types
The recent (2022) burden of diseases in the town was obtained in Microsoft Excel from the Gamo Zone health office. The top 10 chronic diseases were grouped as CVDs, diabetes, chronic respiratory diseases (CRDs), and central nervous system disorders.
Survey selection of medicines
In this study, the most recent core EML of 2015 WHO/HAI, the 2020 Ethiopian EML, and the 2021 national standard treatment guideline (STG) were combined with the current situation of Arba Minch town, and a supplementary list was added based on consultation with experts (Supplemental file). Hence, an overall of 43 types of medicines were included in the study. Table 1 summarizes the generic name, strength, unit of measurement, manufacturer, and originator brands of each investigated medicine.
List of essential medicines used for NCDs included in the evaluation of availability, price, and affordability among healthcare sectors in Arba Minch town, Ethiopia, 2023.

CVD: cardiovascular disease; CRD: chronic respiratory disease; DM: diabetes mellitus; CNS: central nervous system; DDD: defined daily dose.
Data collection and quality control
A standardized data collecting tool produced by WHO/HAI, with required modifications, was utilized to gather data from both public and private health sectors (Supplemental file). The data were collected by five graduating class undergraduate pharmacy students. The data regarding the price and availability of essential medicines were collected from each selected drug retail outlet. Data on the prices and availability of included medicines were gathered from each selected medicine drug outlet.
The medicines’ availability and their most recent prices on the day of data collection were recorded by reviewing the stock cards in each health facility. A storeman or someone in charge at the medicine outlet was contacted for medicines that did not have stock records. Availability was verified by observation, and price data were crosschecked by asking a storeman or responsible individual in the drugstore/pharmacy. Prior to the survey, data collectors received 2 days of training on the basic approach to reduce collector bias on data quality. The pretest was then performed on 10% of the sample size, which consisted of two health institutions, one public and one private. Based on the results of the pretest, necessary changes were made.
Study variables
The outcome variables were the availability, prices, and affordability of selected core medicines used in the management of NCDs. As independent variables, the type of sector (private and public), the daily wage of the lowermost paid government employee, type of medicines (originated brands (OBs) and lower cost generics (LPGs)), and kinds of chronic diseases were included.
Data processing and analysis
The Excel® WHO/HAI Medicine Prices Workbook was used to enter and evaluate the collected data. The findings were then summarized and illustrated in tables and graphs. Measuring the availability of medicines involved calculating their percent availability, their mean percent (%) availability within a therapeutic group of medicines, and their variances among product types (e.g., LPGs vs OBs) and health sectors (public vs private). XLSTAT® version 2023 was used for further statistical analysis. The Shapiro-Wilk and Kolmogorov-Smirnov tests were used to verify the normal distribution of the price data. Furthermore, the Wilcoxon-Mann-Whitney U statistical test was performed to compare the median buyer’s price (patient prices) across public and private health institutions. Medicine prices were estimated as the median price for each medicine in local currency (Ethiopian Birr (ETB)).
Treatment affordability was calculated based on the patient’s pocket payment on medicine for 30 days’ supply and the daily wage of the lowest-paid government employee. Ethiopian 2021-STG and WHO/HAI manual were used to estimate the number of units of defined daily dose, for tablets/capsules/vials, required for a monthly treatment of NCDs.14,30 The daily wage of the lowest-paid government employee (36.75 ETB) was used to measure local affordability and the number of days’ wages required to purchase 30 day’s supply. Thus, the status of affordability was also calculated for the public, private, and overall sectors to ease comparisons. The following formula was used to determine affordability (measured in terms of daily wages):

According to WHO, access to essential medicine is achieved when the given medicine is available and affordable for the patient. 31 Thus, in a given survey region or country, a medicine is considered accessible if it is present in more than 80% of premises and costs no more than 1 day’s wage to purchase standard treatments. “The threshold for availability is 80%, no matter whether a medicine is affordable or not. But, a medicine to be accessible, it must meet both the criteria of availability and affordability.”
Ethics approval and consent to participate
The study was approved by Arba Minch University, College of Medicine and Health Sciences, Department of Pharmacy Research and Publication Committee (AMU-CMHS-DPRPC) with a reference number Phar 20/15. Following the ethical approval, official letters of permission were drafted and disseminated to the administrators of the Gamo Zone health office and all healthcare facilities surveyed, and permission was obtained to proceed with the study. The purpose of the study was subsequently explained to the participants, confidentiality was guaranteed, and written consent was obtained before the commencement of data collection.
Results
Availability
Among 23 health facilities surveyed, the pooled availability of essential medicine for the management of NCDs was 18.7% (range: 0%–30.1%). The availability did not exceed 80% for any therapeutic group across the healthcare facilities as shown in Figure 1. Availability of medicines varied from as low as 0.0% of (ipratropium and captopril) to 47.8% of (glibenclamide and metformin) across health facilities (Table 2).

Percentage availability of NCD medicines by therapeutic group in Arba Minch Town, Ethiopia, 2023.
Disaggregated figures on availability of NCD medicines in public and private health facilities in Arba Minch town, 2023.

OB: originator brand; LPG: lowest priced generic; AP: any product; SD: standard deviation; SD: The range always starts from 0.0% to SD-values.
AP: represent the combined quantities of both LPGs and OB version of products/signaling the total aggregated amount.
Concerning the therapeutic groups, higher availability was observed among medicines used for CVDs (32.0%) followed by DM (25.2%), respectively. In terms of types of health sectors, the overall availability was higher in private sectors (38.3%; range: 0%–33.2%) than in public sectors (16.3%; range: 0%–3.8%).
Concerning the specific versions, the percent availability of the OB version of surveyed medicines is 1.1% for overall health sectors, 0.6% for public sectors, and 1.2% for private sectors. During the study, only one public sector had one OB version of carbamazepine. In contrast, eight private drug outlets had OB versions of Furosemide (40 mg cap and injection 10 mg/ml (n = 3), glibenclamide 5 mg tab (n = 3), metformin 500 mg tab (n = 1), and salbutamol inhaler 100 µg/dose (n = 1; Table 2). In terms of the LPG version of products, the overall percent availability of the LPG version was 36.2% (range: 0%–26.2%). Thus, amitriptyline, amlodipine, enalapril, hydrochlorothiazide, nifedipine, glibenclamide, and metformin have satisfied the WHO’s target for essential NCD medicines (⩾80%) in this version. In the public sector, the overall availability of LPG versions across all medicine was 32.0% (range: 0%–32.1%), where enalapril, hydrochlorothiazide, nifedipine, and glibenclamide capsules/tablets were available in more than 75% of surveyed public sectors. In the private sector, the overall availability of LPGs across all medicines was 37.1% (range: 0%–33.2%). More than 75% of private drug outlets had LPG versions in capsules/tablets dosage forms for amlodipine, amitriptyline, enalapril, hydrochlorothiazide, nifedipine, glibenclamide, and metformin and salbutamol syrup during the study.
In terms of therapeutic categories, medicines used in the treatment of diabetes mellitus (DM; 44.2%) and CRD (34.6%) had higher availability. Among the therapeutic groups, LPG medicines used to treat DM (60.0%) and CVDs (51.7%) exhibited relatively higher availability (Figure 1).
Medicine prices
The overall median price of LPG medicines in private settings was two times higher than in public sectors. Specifically, the private sector’s median prices of LPG versions were three times higher than that of the public sector for medicines such as acetylsalicylic acid, amlodipine, atenolol, nifedipine, propranolol, spironolactone, salbutamol, and amitriptyline. According to the Mann-Whitney U test, 11.6% (n = 5) of these medicines had a statistically significant median price disparity between the public and private sectors (p < 0.05; Table 3).
The median prices of LPG version of NCD medicines among health facilities in Arba Minch town, 2023. (Prices in ETB/dosage form unit (cap/tab, g, ml, or vial)).

Source: The skewed price distribution of two independent data sets (public and private sectors) were tasted with Wilcoxon–Mann–Whitney U.
LPG: lowest priced generic; NCD: noncommunicable disease; ETB: Ethiopian Birr; CVD: cardiovascular disease; CRD: chronic respiratory disease; DM: diabetes mellitus.
Looking at the therapeutic groups such as the CVD medicines, Furosemide injection possessed the highest median price across public and private settings, accounting for 10 and 15 ETB, respectively (Table 3). On the other hand, amlodipine and acetylsalicylic acid had the lowest median price in the respective sectors, with 0.6 and 1.5 ETB, respectively. Insulin (NPH) and metformin had the highest median price scores among antidiabetic medicines, with 179.5 and 11.65 ETB in the private and public sectors, respectively. In the group of machines used in CRD, beclomethasone (214 ETB) and salbutamol inhaler (300 ETB) had the highest median prices in the respective health sectors. Amitriptyline had the lowest median prices in the CNS medicine category across all health sectors, while the score decreased from 0.83 to 0.59 ETB in the public and private sectors, respectively.
In this study, the top five median price scorers were amlodipine, furosemide, insulin, beclomethasone, and salbutamol across the surveyed health facilities.
Affordability
The majority of the included medicines appeared to be unaffordable, with prices exceeding 1 day’s wage in both private and public institutions. The percentages of unaffordable NCD medicine were 70.4% and 100% in the public and private sectors, respectively, with 100% of them being unaffordable when both health sectors were pooled.
The overall affordability estimation also indicated that the top five unaffordable medicines were beclomethasone inhaler, salbutamol inhaler and salbutamol syrup, insulin (NPH) vial, and insulin (soluble/natural) vial, which required 1404.49, 1639.49, and 489.40, 48.18, and 46.03 days wage of the lowest-paid government employee, respectively. Similarly, beclomethasone inhaler, salbutamol inhaler, insulin (NPH) vial, insulin (soluble/natural) vial, and carbamazepine were the top five unaffordable medications in public sectors, with 1388.10, 1038.34, 48.32, 45.09, and 18.35-day’s wage, respectively. In the private sector, salbutamol inhaler took the lead owning 1639.49 day’s wage followed by salbutamol syrup, furosemide injection, propranolol, and metformin, with day’s wages of 49.4, 12.24, 11.42, and 9.50, respectively.
On the other hand, some sort of medicines such as acetylsalicylic, amlodipine, atenolol, hydrochlorothiazide, spironolactone, glibenclamide, and prednisolone took less than 1 day’s wage for the lowest-paid government employee in public facilities (Figure 2).
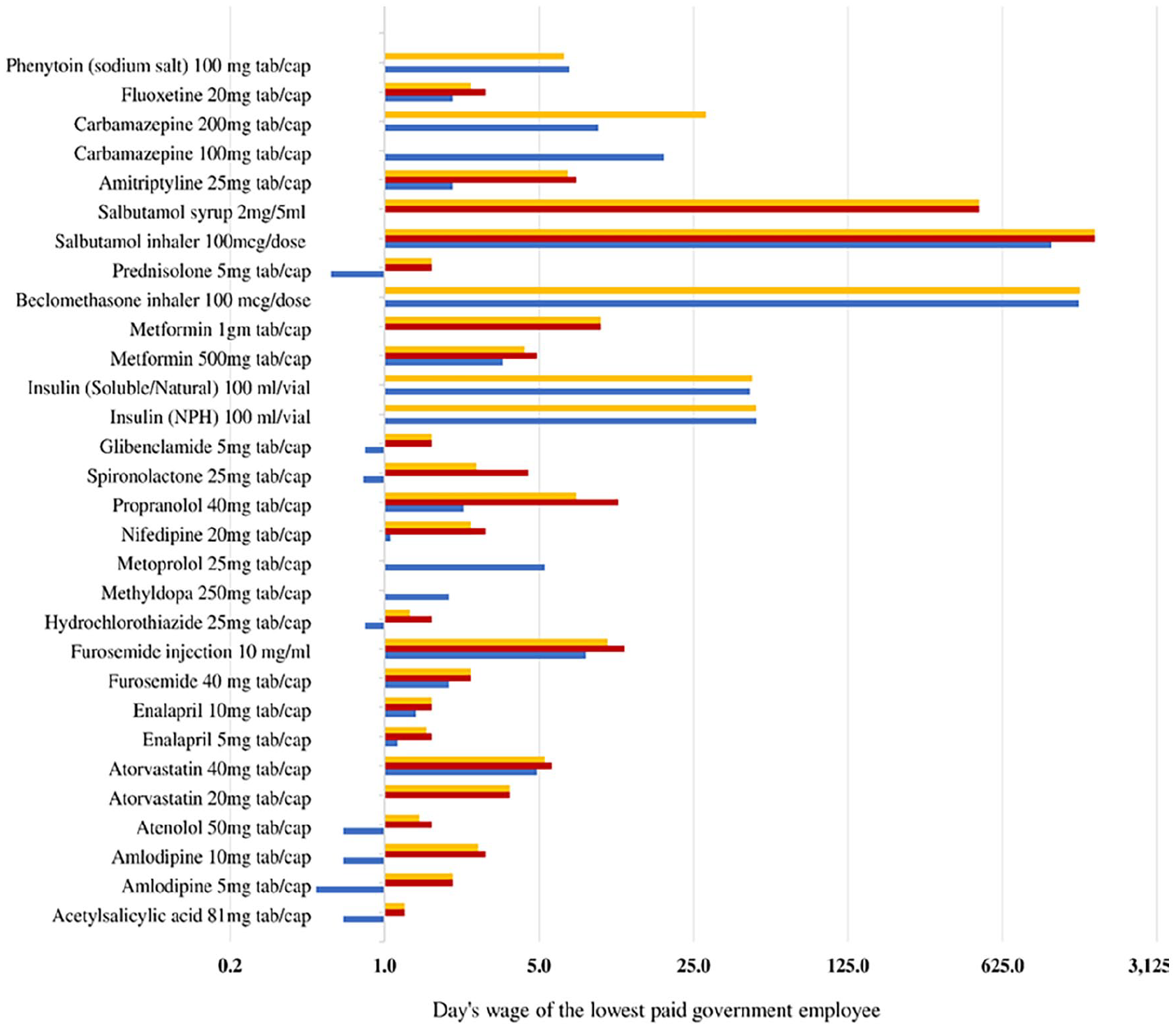
Affordability of NCD medicines in Arba Minch Town, Ethiopia, 2023. The number of day’s wages required to purchase a 30 days’ supply in the health sectors was estimated from the daily wage of the lowest-paid government employee (36.75 ETB).
Achieving WHO’s target for accessibility
In this finding, the percentage of surveyed medicines with ⩾80% availability and required less than 1 day’s wage of the minimum paid government worker to purchase medicines were uncommon across all health sectors and therapeutic groups (Table 2 and Figure 2). Thus, the LBG version of glibenclamide was the only medicine that met the target of both availability and affordability target in the public sectors. In the private sector, the percentage of medicines used to treat NCDs that met the WHO target appeared to be low (0.00%; this remains true when combining both sectors).
Discussion
The WHO has set a target for nations to achieve an 80% availability of affordable, safe, and quality essential medicines and basic health technologies by 2025, aligning with Sustainable Development Goal Three and UHC.7,32 However, LMICs struggle to meet this target, with research consistently indicating inadequate availability and affordability of essential medicines. This study specifically investigates the prices, availability, and affordability of essential medicines for NCDs to assess Ethiopia’s progress in meeting these goals.
The findings reveal a concerning scarcity of OB essential medicines for NCDs, potentially creating financial barriers for patients requiring these more expensive branded medications. While some medicines meet the WHO’s availability target, the majority, especially for chronic conditions like CVDs and DM, fall short of this benchmark. Supporting evidence from Pakistan highlights the low overall availability of essential NCD medicines in both original brand and low-priced generic forms across public and private sectors. 33 Similarly, a study in Nepal demonstrates higher availability of essential NCD medicines in the private sector compared to the public sector. 34 Conversely, South Sudan struggles with poor availability across all sectors. 35 Moreover, a significant proportion of patients in LMICs face limited access to generic versions of essential medicines for various NCDs.6,14,36–42
In this study, the availability of LPG appears slightly higher in both public (32.0%) and private (37.1%) sectors, indicating better accessibility to more affordable generic versions. However, overall availability of essential NCD medicines remains low at 18.7%. Consistent findings from Nepal also highlight the low availability of essential NCD medicines in public healthcare sectors, indicating systemic issues that require broader policy interventions to improve access to these medications.15,43–45 Challenges such as inadequate government funding, inaccurate forecasting, procurement issues, regulatory hurdles, and price mechanisms affect medicine availability in these sectors.
The disparity in median medicine prices between the public and private sectors is evident in this study, with a significant proportion priced higher than the international reference price, especially in the private sector. This aligns with global trends where originator brand prices tend to surpass those of other forms.37,46 Patient prices in the private sector consistently surpass those in the public sector across various studies.15,37,47 Similar to Pakistan, where prices were notably higher in the private sector compared to public procurement, this study emphasizes the prevalence of higher-priced medicines in the private sector. 19 These findings align with those reported for many LMICs where medicines in the private sector are substantially pricier than the international reference price. 33
The study reveals that a majority of surveyed essential medicines are unaffordable, often requiring several days’ wages to purchase, particularly in the private sector. Crucial medicines for NCD management, including inhalers, insulin, and certain cardiovascular drugs, are highly unaffordable for the lowest-paid government employees in both public and private sectors. Contrasting observations from Jordan indicate better availability of lowest-priced generics in both public and private sectors for chronic diseases, but affordability remains an issue particularly in private settings. 48 Similarly, in LMICs such as Malaysia, generic versions of essential medicines are generally affordable in the public sector. 49 Conversely, more positive reports of better availability and affordability of generic versions come from country-specific studies in Rwanda and Nepal.34,50
This study has some limitations. The study focuses specifically on selected healthcare facilities in Arba Minch town, Southern Ethiopia. This limited scope might not represent the broader situation across the entire country or other regions, affecting the generalizability of the findings to the entire Ethiopian context. The study might suffer from sampling bias as it concentrates on a specific geographic area. This might not provide a comprehensive view of the diverse situations across different regions or urban/rural settings within Ethiopia. The study primarily focuses on the availability, prices, and affordability of essential medicines for NCDs. Other important factors influencing access to medicines, such as quality assessment, patient perspectives, or healthcare provider behavior, are not thoroughly explored, limiting the holistic understanding of the issue.
Conclusions
The study underscores a concerning scarcity of essential medicines for NCDs, especially the OB version of medications. This shortage might create financial barriers for patients requiring these costlier branded drugs. Despite some essential medicines meeting the WHO’s availability targets, the majority, particularly those for chronic conditions like CVDs and DM, fall short.
In addition, medicines remain largely unaffordable, requiring several days’ wages to purchase, particularly in the private sector. The study highlights a significant price disparity between the public and private sectors, with many medicines priced higher than the international reference price, especially in the private sector. Patient prices in the private sector consistently surpass those in the public sector, reflecting a broader trend observed in various countries. The consistent findings across multiple studies underscore the necessity for comprehensive policy interventions to enhance access to essential medicines for NCDs. Addressing issues related to prices, availability, and affordability should be a priority within healthcare policy frameworks.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241266318 – Supplemental material for Access to essential medicines used in the management of noncommunicable diseases in Southern Ethiopia
Supplemental material, sj-docx-1-smo-10.1177_20503121241266318 for Access to essential medicines used in the management of noncommunicable diseases in Southern Ethiopia by Getahun Asmamaw, Tekalign Shimelis, Dinksew Tewuhibo, Teshome Bitew and Wondim Ayenew in SAGE Open Medicine
Supplemental Material
sj-xlsx-2-smo-10.1177_20503121241266318 – Supplemental material for Access to essential medicines used in the management of noncommunicable diseases in Southern Ethiopia
Supplemental material, sj-xlsx-2-smo-10.1177_20503121241266318 for Access to essential medicines used in the management of noncommunicable diseases in Southern Ethiopia by Getahun Asmamaw, Tekalign Shimelis, Dinksew Tewuhibo, Teshome Bitew and Wondim Ayenew in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to thank Arba Minch University, College of Medicine and Health Science, for their collaboration in reviewing the proposal and providing ethical clearance. We would like to extend our gratitude to the Gamo Zone Health Office for their excellent participation in offering the essential dataset. We are also grateful to the data collectors and study participants (including health facilities) who provided the data for this study.
Author contributions
Conceptualization: Getahun Asmamaw. Data collection: Tekalign Shimelis. Data Analysis: Getahun Asmamaw, Tekalign Shimelis, Dinksew Tewuhibo, Teshome Bitew, and Wondim Ayenew. Software: Getahun Asmamaw. Validation: Getahun Asmamaw, Tekalign Shimelis, Dinksew Tewuhibo, Teshome Bitew, and Wondim Ayenew. Writing the original draft: Getahun Asmamaw and Wondim Ayenew. Writing review and editing the final manuscript: Getahun Asmamaw, Tekalign Shimelis, Dinksew Tewuhibo, Teshome Bitew, and Wondim Ayenew.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from the Ethics Review Committee of the department of Pharmacy, Arba Minch University (Approval Number: Phar 20/15).
Informed consent
Written informed consent was obtained from all subjects before the study.
Consent for publication
Not applicable.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
