Abstract
In the presented review, the past investigations have been complied and critically analyzed to highlight the influence of microscopic features on self-cleaning ability. In addition, challenges and research gaps that currently exist are discussed. This review concludes the current methods and processes to obtain self-cleaning ability using the surface features of textile fabrics manipulated with the help of the coatings and nanoparticles. However, no research was conducted to explore the self-cleaning potential of microscopic geometrical features of fabric at the woven structural level.
Keywords
The self-cleaning concept is one of the fastest-growing research areas in industries around the world.1,2 Due to significant changes, several self-cleaning materials have been developed in many industries, such as fabrics, furnishing materials, windows, glasses, outdoor construction materials, and solar cells. 3 Many scholars have published their findings on these materials in the academic field. According to publications, the self-cleaning concept can be defined as “the ability to clean the surface without any external source.” 2 Furthermore, It is considered as an environmental advantage that reduces cleaning effort and saves a considerable amount of resources (i.e., time and cost). 4
There are two main approaches used in developing self-cleaning ability. The first one is based on the hydrophobic surface approach. It is also known as the lotus effect, as shown in Figure 1(a).5 In this approach, the water drops roll off the surface and removes the dirt particles, as shown in Figure 1(b).6,7 The second is based on the hydrophilic surface approach, where is the water drops spread over the surface and form a layer of water without removing any dirt; as shown in Figure 1(c), both of the surfaces clean themselves with the use of water. 8 These approaches have been significantly used in various industries, such as textiles and medical textiles, athletic wear, military uniforms, and outdoor fabrics. 9
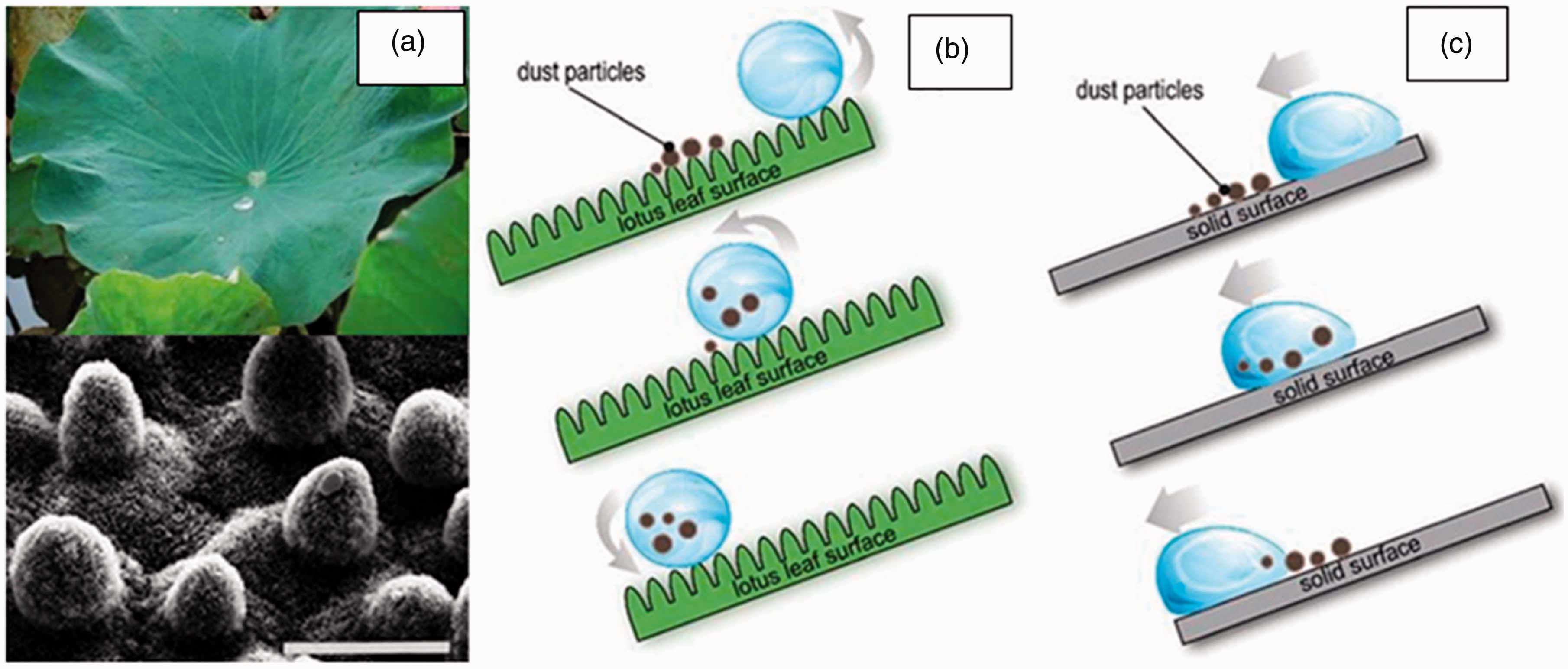
(a) A typical example of a superhydrophobic surface in nature: lotus leaf with self-cleaning properties and a scanning electron microscopy image of the surface structures of the lotus leaf. Schematic illustration of the “self-cleaning” concept: a water droplet rolling off on (b) a lotus leaf rough surface and (c) a water droplet on a hydrophilic solid surface. 7
Self-cleaning abilities in textile fabrics have been comprehensively investigated over the last decade. Most of these investigations have used surface roughness and low surface energy features to establish a self-cleaning mechanism. Extensive research articles and reviews have been published to describe these processes, along with their microscopic features. However, when these were reviewed with a critical eye, it was found that a comprehensive effort is still required to compile all of these previous researches, emphasizing how the microscopic features of textile fabrics can influence their self-cleaning ability. In the presented review, the past investigations have been complied and critically analyzed to highlight the influence of microscopic features on self-cleaning ability. In addition, challenges and research gaps that currently exist are also discussed. This review concludes that the current methods and processes to obtain self-cleaning ability use the surface features of textile fabrics manipulated with the help of coatings and nanoparticles (NPs). However, as yet no research has been conducted to explore the self-cleaning potential of the microscopic geometrical features at the fiber surface morphology level.
In this paper, the previous efforts relevant to create self-cleaning surfaces are compiled. Their key working elements are discussed. Later, a comprehensive discussion is provided on how the microscopic features of these surfaces can influence the self-cleaning ability. Finally, challenges in the existing processes and evaluations of their effectiveness are provided.
Conventional methods of self-cleaning
Previous studies examined the self-cleaning ability on textile fabric surface roughness and low surface energy by employing surface coatings with the use of different chemical solutions. These chemical solutions conventionally use nanotechnology to enhance the coatings for desirable surface roughness or low surface energy. Textile materials, such as cotton cellulose, silk, and wool, are useful for clothing properties due to their abundance, excellent moisture, and absorption. 10 These materials are technically called “hydrophilic” in nature. For hydrophilic coatings, the dirt and other contaminations chemically break down when exposed to sunlight, and this process is called “photocatalysis.” 11 The hydrophilic nature can be further modified to superhydrophobic by increasing their surface roughness with the use of NPs such as titanium dioxide (TiO2) and zinc oxide (ZnO). Those materials have been used to functionalize and oxidize the coloring ingredients in solutions and stains to have self-cleaning and other properties. Deposition (TiO2) can change the surface energy, the absorption of liquids, and the adhesion of contaminants, permitting their photolytic degradation. As surface morphology is essential, the surface energy and sorption characteristics also play a part. So, an optimized surface microstructure and surface chemistry can create superhydrophobicity.12,13 Different methods and materials were used to change the surface roughness and create self-cleaning ability in textile fabrics. This change can be obtained by creating hierarchical micro or nanostructures on the surface or by decreasing the surface energy with the help of low surface energy chemicals. 14
Dip-coating (sol–gel) method
The dip-coating method has a significant impact on the chemical finishing of textile materials. Researchers have paid much attention to the fabrication of superhydrophobic surfaces using this method. Simple dip-coating, self-cleaning, antibacterial, ultraviolet (UV) protection, and other finishes can be applied on textile surfaces. 15 In this method, the textile goes through two processes. Firstly, it is immersed in a solution that includes the needed chemicals (1. Dip), then padded through the pressure of 2 bar (2. Pad); this process is called immerse and distribute. Secondly, the treated textile enters the “drying” (3. Dry) and “curing” (4. Cure) stages at a fixed temperature; this process is called heat treatment, as shown in Figure 2. 16 Lee et al. 17 developed self-cleaning textiles (cotton and nylon) with carbon nanotube (CNT)-TiO2 and methyl methacrylate butyl acrylate (MMA/BA) suspension polymerization using the dip-coating process. Lin et al. 18 prepared a durable superhydrophobic and super-oleophobic cotton fabric with a dual-size hierarchical structure containing modified SiO2 NPs and fluoropolymer. The coated textile fabrics showed excellent self-cleaning performance. Yang et al. 19 modified cotton textile surfaces with anatase TiO2 sol and polydimethylsiloxane (PDMS) using a simple dip-coating method. As a result, a robust fluorine-free self-cleaning cleaning cotton textile was prepared with excellent durability, superhydrophobicity, and photocatalytic ability. Yang et al. 20 prepared a superhydrophobic and photocatalytic self-cleaning cotton fabric by a sol–gel method. In this method, the glacial acetic acid was catalyzed and then modified by heptadecafluoro-1,1,2,2-tetrahydrodecyl triethoxysilane (F-17) solution, which could provide superhydrophobicity owing to its low-energy hydrophobic groups (–CF2, –CF3). The achieved self-cleaning property was excellent toward solid pollutants, daily liquids, oil, and organic chemicals. Ahmad et al. 21 presented a visible-light-driven self-cleaning cotton fabric coated with dye-sensitized TiO2 nano-sol by the dip-coating (dip–pad–dry–cure) method. A phthalocyanine (PC) reactive dye (RB-25) was used for the photosensitization of the TiO2. The coated textiles demonstrated good photocatalytic and self-cleaning results under visible light. Qi et al. 22 produced nanocrystalline anatase coatings on a cotton fabric surface under ambient pressure. They used hydrolysis of TiO2 in an acidic aqueous solution and coated the fabric by the sol–gel (dip-pad–dry-cure) process. The TiO2-coated cotton textile showed significant photocatalytic self-cleaning properties. Zhu et al. 23 fabricated self-cleaning cotton fabrics by coating photocatalytic ZnO NPs on cotton surfaces, using a traditional dip-coating (dip–pad–dry–cure) coating process. The self-cleaning tests showed that the coated fabrics demonstrated good photocatalytic activity against methylene blue (MB) solution, good durability, and excellent UV blocking properties. As for wash fastness, it showed color removal after 10 washing cycles under light irradiation, as shown in Figure 3. These findings provide a potential model for the practical application of self-cleaning textiles. The sol–gel method is considered to be a high-quality, cost-efficient, low-thermal energy, and low-pressure procedure that can develop rough surfaces and fabricate superhydrophobic surfaces. It is often used to create the bond between NPs and a fabric. 1
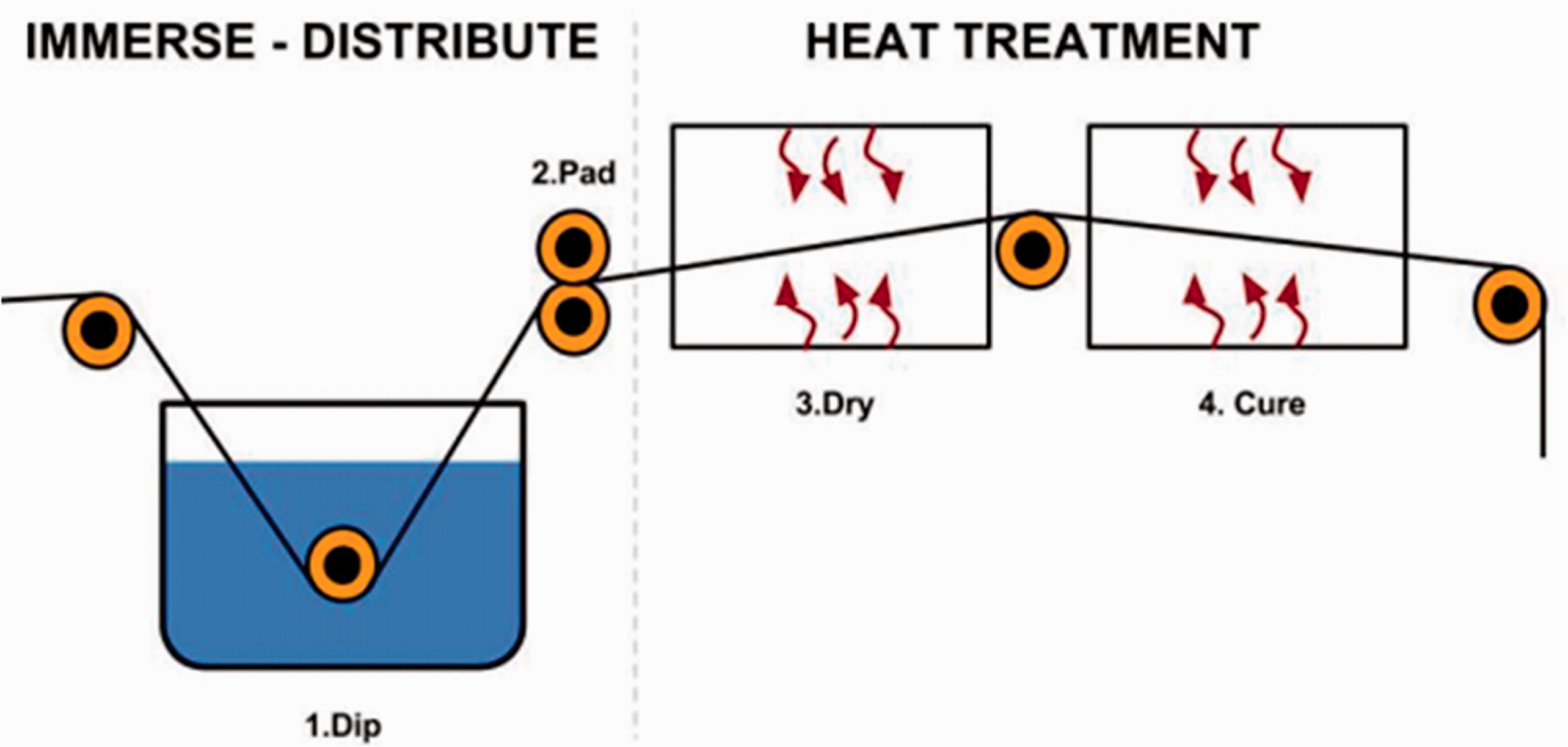
Illustration of the industrialized version of the dip–pad–dry–cure method used to apply the photo-responsive materials on the textile substrate. 16
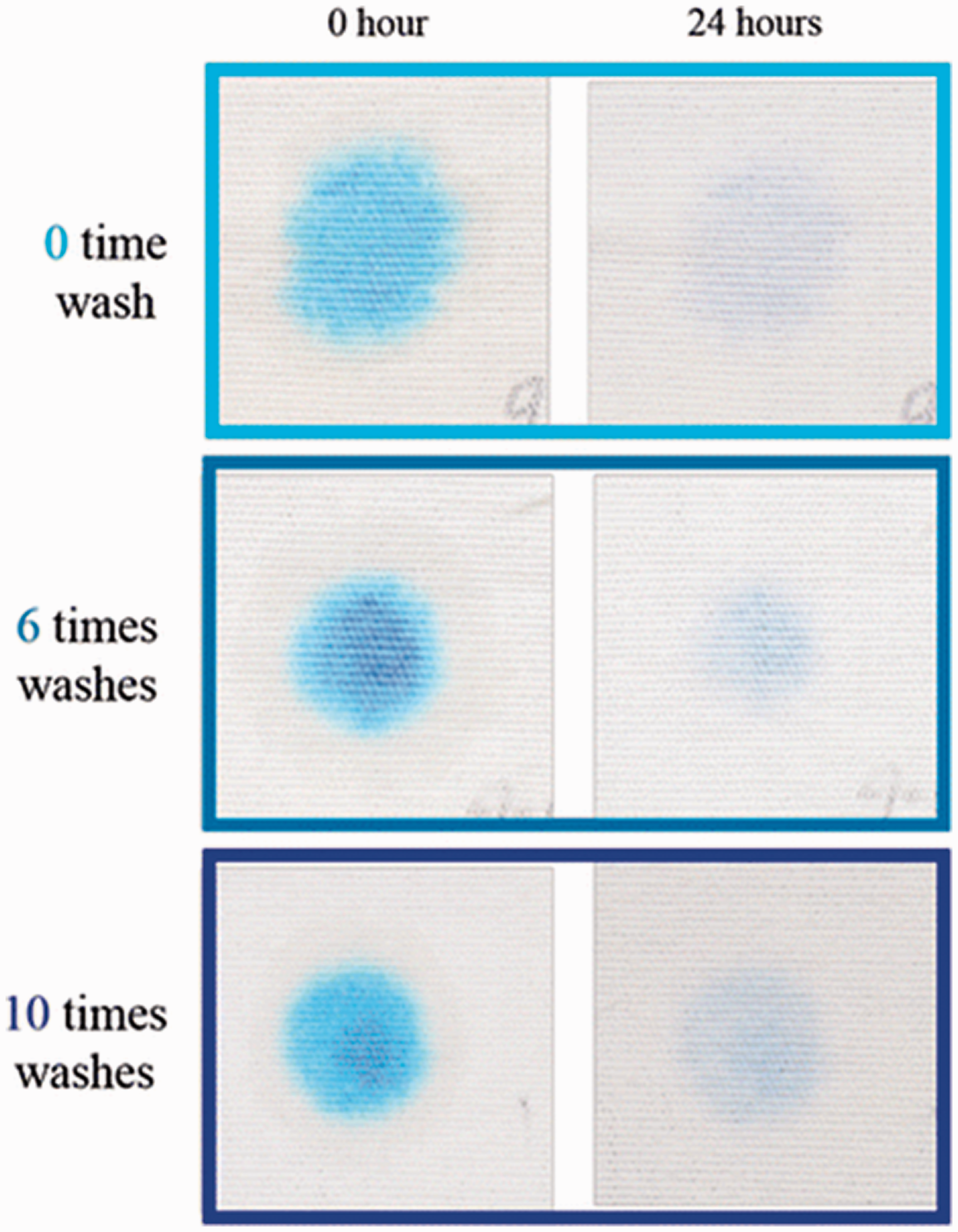
Wash fastness of ZnO nanoparticle-coated cotton fabrics, showing different wash times. 23
Spin coating method
The process contains three steps: deposition, spinning, and curing. The substrate is deposited on the turntable then spun up and off, while the evaporation stage occurs at the end. 24 Rilda et al. prepared self-cleaning cotton textile by interacting acrylic acid binder as a cross-linking agent followed by TiO2–SiO2 clusters using a spin-dip-coating machine. The qualitative measurement of the self-cleaning agent was done by observing the ability of the modified cotton textile (3 × 3 cm2) to degrade 75 μL of MB spot (15 ppm) under UV light. The quantitative measurement was done by checking the ability of the cotton textile to reduce 20 mL of 15 ppm MB spot in a 1 × 1 cm2 cotton textile. The coated samples with Ti:Si molar ratios of (1:1), (1:2) and (2:1) at different dipping durations showed different results, as shown in Figure 4. From the results of the MB degradation under UV irradiation, the best self-cleaning effect was shown by the cotton textile coated with TiO2–SiO2 with a Ti:Si molar ratio of (1:2). 25 Spin coating is generally used in the microelectronics industry, whereas it is less popular in the textile coating industry. 26 It is considered a low-cost method used to deposit thin films on flat substrates. 24
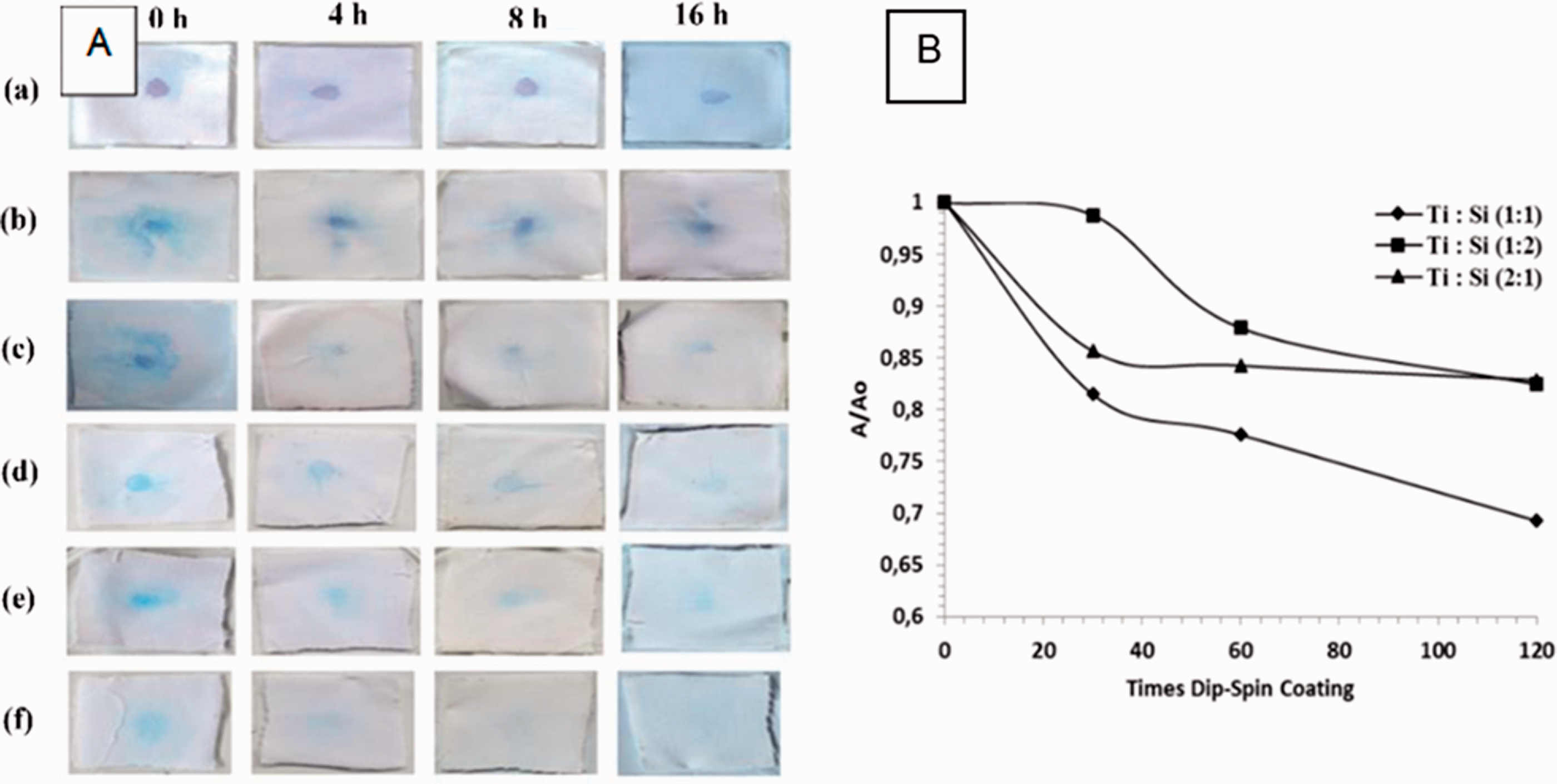
A. The degradation of a methylene blue spot in a cotton textile by dipping in acrylic acid for 24 h on (a) non-coated cotton, (b) cotton coated with TiO2, (c) cotton coated with SiO2, (d) cotton coated with TiO2–SiO2 (1:1), (e) cotton coated with TiO2–SiO2 (2:1), and (f) cotton coated with TiO2–SiO2 (1:2). B. The degradation pattern of methylene blue on cotton fibers coated with different Ti:Si molar ratios under UV irradiation at ʎmax = 661 nm. 25
Spray coating and electro-spraying methods
Yao et al. 27 modified a textile surface by fabricating a two-dimensional (2D) graphitic carbon nitride nanosheet (CNNS). The colloidal suspension of the CNNS was directly sprayed on the surface of the textile. These textiles obtained an excellent photocatalytic self-cleaning property. Roy et al. 28 developed a simple method with a sustainable superhydrophobic coating to produce self-cleaning–healing fabrics. The coating was created using cellulose nanofibers (CNFs) as the row material and spraying it on polyester and cotton fabrics. They became superhydrophobic with outstanding self-cleaning–healing ability. An alternative method was used by Shams-Ghahfarokhi et al., who successfully coated polyester fabrics with silica aerogel particles using a fluorocarbon finishing method via the electro-spraying process. They showed high insulation properties and high hydrophobicity with low dust-releasing behavior. The results implied that the aerogel particles improved the hydrophobic properties of the electro-sprayed samples. Images of the un-sprayed and electro-sprayed polyester fabrics with the aerogel/fluorocarbon mixture for 16 h together and the sessile water droplets on their surfaces are present in Figure 5. A dual-scale surface roughness provided the appropriate structure to achieve a hydrophobic surface. 14 Li et al. prepared a superhydrophobic and a robust superhydrophobic coated cotton woven fabric. A one-step single-solvent electro-spraying of polycaprolactone (PCL), which is considered to have excellent biocompatibility and degradation and low immunogenicity, along with honeycomb-like hierarchical structure microspheres (PCL-b-PTFOA) and abundant nanometer pores of (PCL-b-PEG400) microspheres were used. They were fabricated by solvent evaporation and copolymer diffusion-induced separation during the electro-spraying. The coated fabric surface exhibited excellent self-cleaning and laundering durability properties, and good washing fastness against mechanical abrasion without significantly reducing the water contact angle (WCA). The bonding stability of PCL-b-PTFOA(4h) microsphere-coated cotton fabric was measured by liner abrasion. As shown in Figure 6, the superhydrophobic property of coated fabric was decreased with the loss of microspheres. Figure 6(a) shows the coated fabric, and the results in Figure 6(b) show minor wear and tear in the scanning electron microscopy (SEM) morphology of the coated fabric, but the film was still able to repel water with a WCA of 150.6° after 10 abrasion cycles. However, once rubbed for 20 cycles, many microspheres were removed, part of the fibers were worn down, and the WCA decreased to 143.5° (Figure 6(c)). It was found that dyed water droplets maintained a spherical shape on the coated fabric surface with a WCA of 151.3 ± 0.5° after five tape adhesion cycles, as shown in Figure 6(d). 29 The spray coating and electro-spraying coating of block copolymers can provide a simple, facile, and promising technique for producing multifunctional textiles. It can be easily applied to a large surface area and does not involve complicated and costly processes.27,29
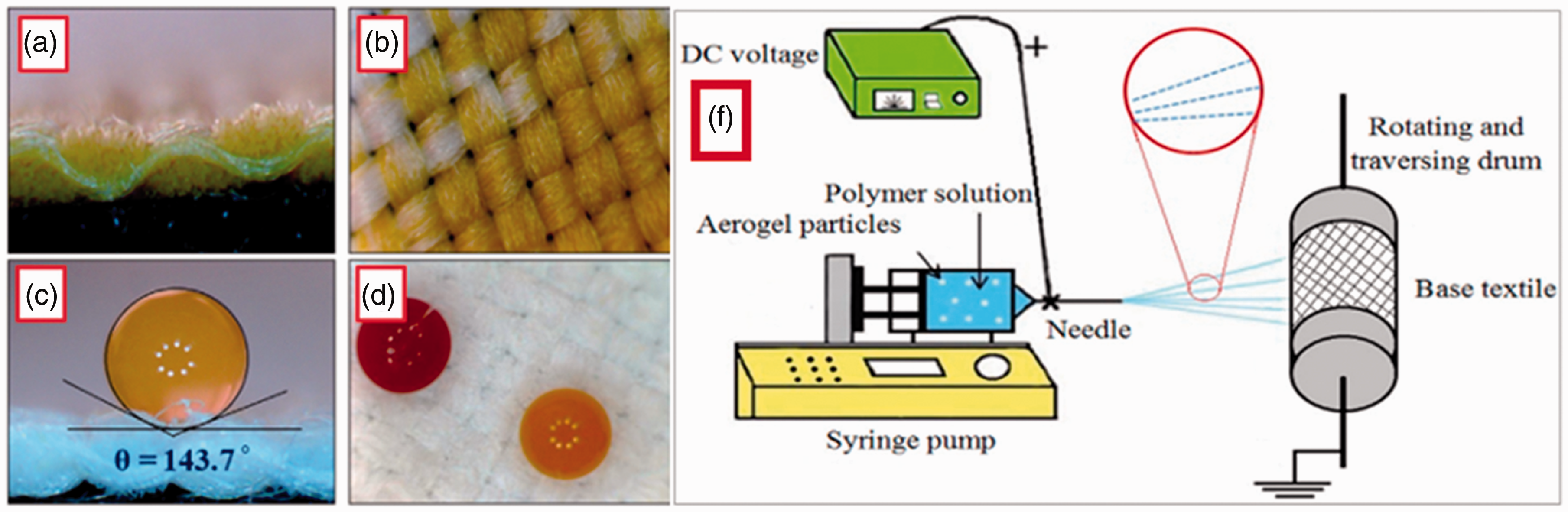
Sessile water droplets on the (a) and (b) un-sprayed and (c) and (d) electro-sprayed polyester fabrics with the aerogel/fluorocarbon mixture for 16 h. (e) Schematic design for the basic setup of the electro-spraying device used in this study. 14
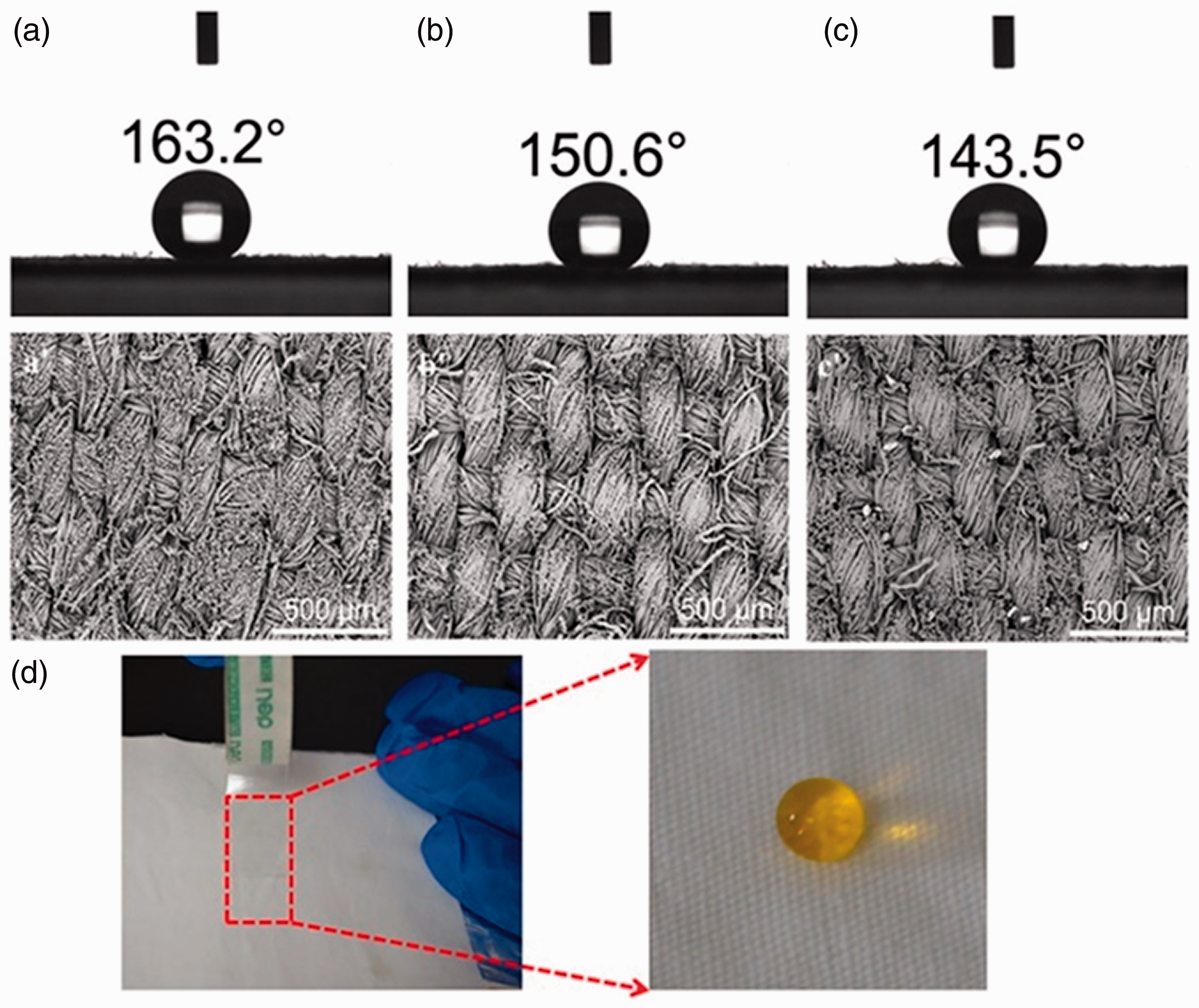
The abrasion and mechanical durability of the honeycomb-like hierarchical structure microspheres (PCL-b-PTFOA) microsphere-coated cotton fabric: (a)–(c) sandpaper abrasion tests; (d) tape adhesion tests. 29
Electrospinning method
Electrospinning is a method that produces a nano-scale fiber in both laboratories and industrially. 30 The basic principles of the electrospinning process consist of three main components: a feeding system, a high-voltage power supply, and a grounded collector. 30 Bedford and Steckl 31 created photocatalytic self-cleaning textile fibers by using coaxial electrospinning. They used cellulose acetate for the core phase and dispersion of nanocrystalline TiO2, as shown in Figure 7. Electrospinning is a promising technique to create superhydrophobic self-cleaning surfaces, which obtains the basic surface morphology due to a comprehensive set of parameters that effectively control the resulting web roughness. On the other hand, a manufacturing process that includes various steps is usually more expensive than a single-step process. The primary issue is the low production rate of single nozzle electrospinning. Therefore, needleless electrospinning was studied in several research studies with a higher production rate, and was used for mass-scale production of electro-spun webs. 32
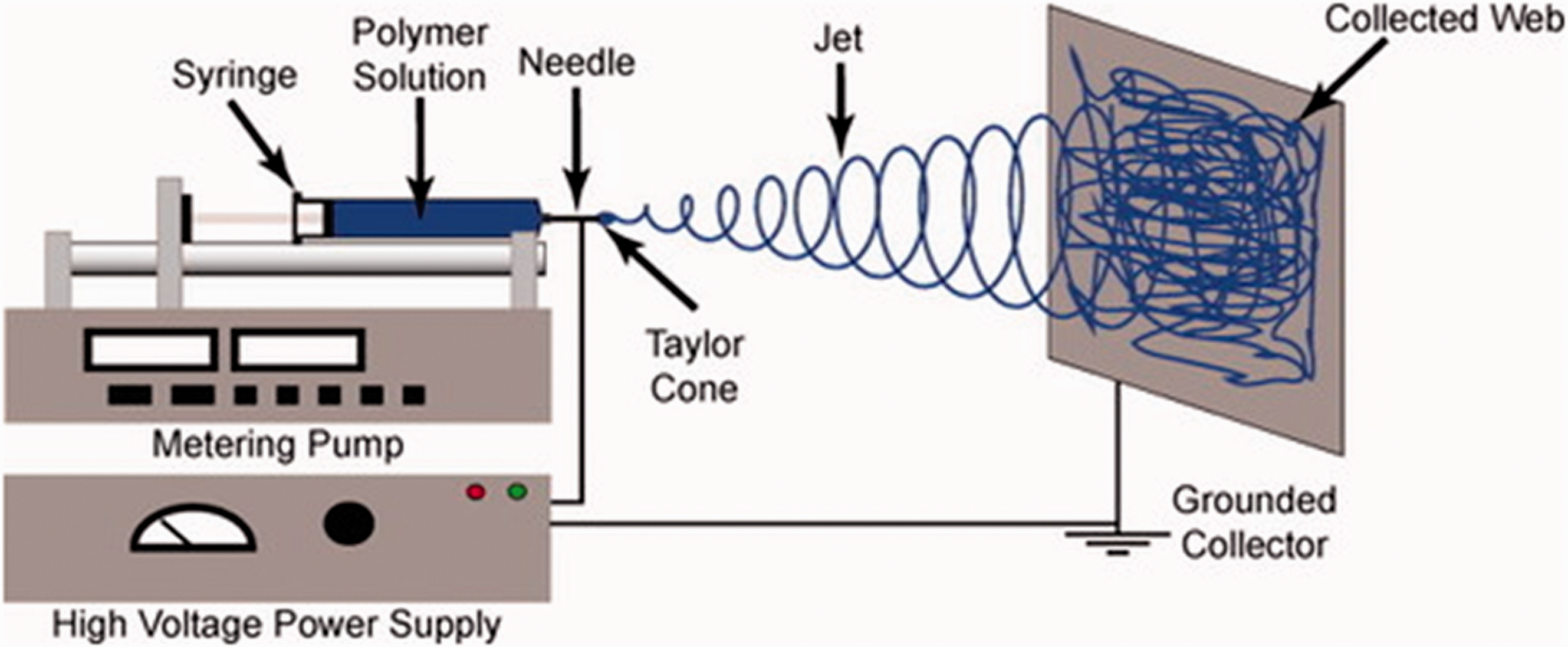
Schematic of the basic setup for electrospinning. 30
Chemical etching and chemical vapor deposition methods
Chemical etching and chemical vapor deposition (CVD) methods are used with polymers to fabricate functional surfaces with different geometries. They make the surface exhibit hydrophilic and hydrophobic properties.11,33 Xue et al. 34 successfully created washable and wear-resistant superhydrophobic polyester resistant polyethylene terephthalate (PET) using chemical etching combined with PDMS. The coated textile obtained a self-cleaning property. Zheng et al. 35 developed a superhydrophobic and self-cleaning polyester fabric from a mixture of methyl trichlorosilane (MTS) and dimethyl dichlorvos silane (DDS) solution at the micro- and nano-scale. It was grown and fabricated with a layer of polydimethylsiloxane (PDMS) nanofilaments onto textile fibers. Faride et al. 33 coated a self-cleaning superhydrophobic and super-oleophilic commercial textile with an effective oil–water separation. The micro–nanometer scale roughness was achieved by a single fabrication step using a handmade CVD device, as presented in Figure 8.
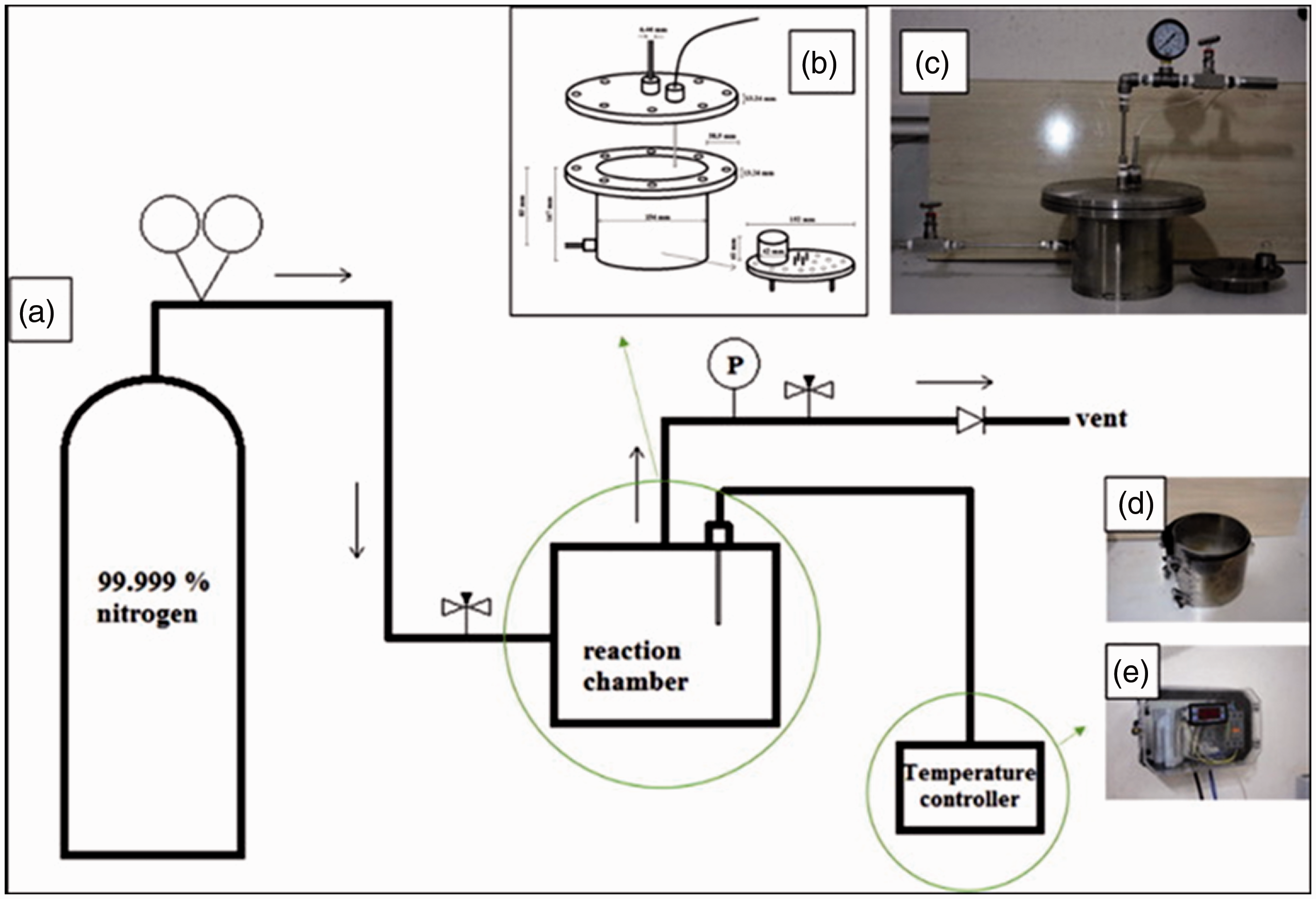
(a) Schematic diagram of the textile coating setup. (b) Schematic and dimensions of the chemical vapor deposition (CVD) reaction chamber. (c) Picture of the CVD reaction chamber. (d) Heating element. (e) Temperature controller. 33
Hydrothermal treatment method
Hydrothermal treatment (HTT) is considered an environmentally friendly technology. It is a basic technique that can effectively control the geomorphology of NPs, nanowires, and mesoporous inorganics. 36 Li et al. 37 developed a facile one-pot hydrothermal reaction method to create micro/nanostructure flower-like TiO2 particles modified with fluoroalkyl silanes on the cotton fabric surface. The as-prepared TiO2 cotton exhibited superhydrophobicity, self-cleaning properties, laundering durability, and oil/water separation. Huang et al. 38 prepared a composite TiO2 cotton fabric by a simple self-assembling coating process by soaking in a mixed methanolic solution of hydrolyzed puorodecyltriethoxysilane (PTES) and F-17 solution. They created marigold flower-like hierarchical TiO2 particles through a one-pot hydrothermal reaction on the surface. The superhydrophobic F-17/TiO2-coated fabrics showed excellent mechanical and environmental stability, good anti-UV ability, effective self-cleaning, and oil–water separation. Tan et al. 39 formed two kinds of self-cleaning cotton fabrics coated with TiO2 NPs by dipping the fabrics in the TiO2 sols by peptizing and hydrothermal methods. The two methods were successful and showed high photocatalytic activity and good self-cleaning performance.
Layer-by-layer self-assembly method
The layer-by-layer (LBL) assembly method is an efficient, facile, and multipurpose strategy in coating substrates with multilayers of different structures, properties, and functions for many applications, such as dipping, immobilization, spinning, spraying, and electrodeposition. Hua et al. 40 fabricated superhydrophobic, flame-retardant, and conductive cotton fabrics. They applied LBL assembly of polyethyleneimine, ammonium polyphosphate, CNTs, and post-treatment with PDMS at the end. The developed fabrics showed excellent superhydrophobic stability toward acid, alkali, organic solvent, UV irradiation, scratching, and long-time laundering. It provided an efficient layer on the cotton fabric surface to prevent the fiber from damage when it is burned, as shown in Figure 9. Uğur et al. 41 coated a denim fabric with TiO2 NPs using the LBL deposition process as a continuous method. The coated denim fabrics showed self-cleaning properties, especially with the increase of NP concentration. The LBL self-assembly methods provide the simplicity of process and equipment and the availability of materials and solutions, along with the flexibility of application to substances of different shapes and sizes. They are suitable for most surfaces, and the control over the required thickness is stabilized. 42
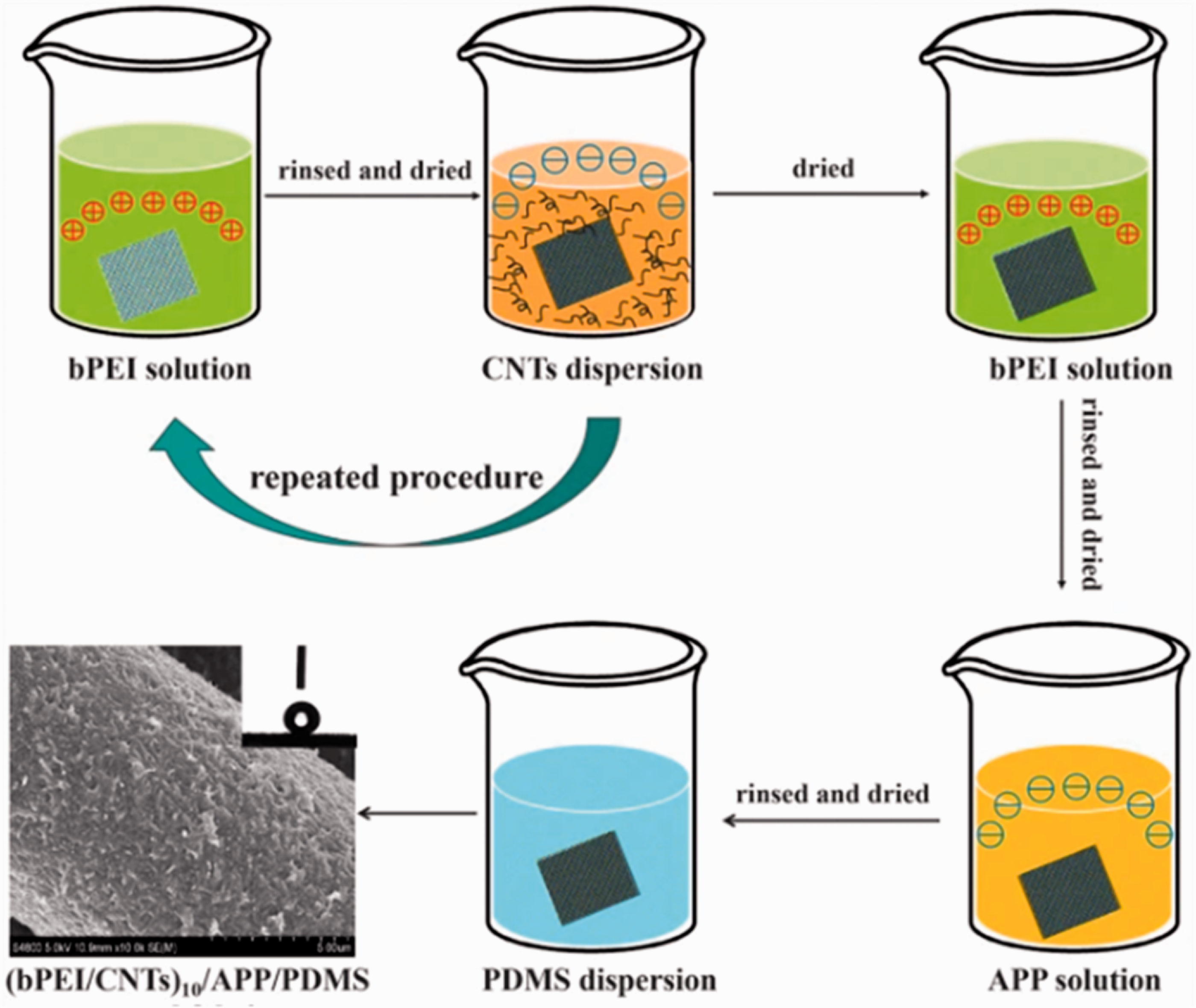
Schematic illustration of the fabrication of multifunctional superhydrophobic fabric. 40 bPEI: sequential assembly of poly(ethylenimine); APP: ammonium polyphosphate; CNTs: carbon nanotubes; PDMS: post-treatment with poly(dimethylsiloxane).
Wet chemical synthesis method
The wet chemical synthesis method controls the structure of the produced nanostructures, NPs, nanowires, and mesoporous inorganics. This method has been commonly used to fabricate superhydrophobic surfaces.43,44 Komeily-Nia et al.45 coated multifunctional nylon fabric to form finely dispersed CuO/MnO2 core/shell nanostructures on the surface via a wet chemical (in situ). Moridi Mahdieh et al. 46 coated a polyester/cellulose fabric by a facile single-step (in situ) synthesis method for Ag–TiO2 NPs without adding any chemical agents. The fabric was immersed in a suspension containing TiO2 NPs/AgNO3 and then squeezed. The coated fabric showed good self-cleaning. Panda et al. 47 coated cotton fabric by using two silane trichloro octadecyl silane (OTS) and pentafluorophenyl triethoxysilane (FAS) with a wet chemical immersion technique. The coated cotton fabric showed excellent self-cleaning and oil–water separation properties, making it suitable for industrial applications. Jiang et al. 48 treated cotton fabric with two kinds of nano TiO2–polyacrylate hybrid dispersions. They were synthesized by in situ suspension and solution immersion, respectively, to fix the nano TiO2 on fabrics. It is an efficient way for immobilizing nano TiO2 on self-cleaning cotton fabrics successfully and a facile, cost-effective single-step method for superhydrophobic coating fabrication. 48
The use of nanotechnology
The potential of nanotechnology innovation in the textile industry is promising. The use of nanotechnology in the textile industry has increased rapidly.30,49 Nanocoating is a relatively new technique in the textile industry and is in the research and development phase. Therefore, nanotechnology has opened enormous possibilities in textile finishing techniques by making chemical finishing more controllable, durable, and significantly enhanced. To generate superhydrophobic surfaces on textiles, which replicate the nanostructure of the lotus leaf and show signs of self-cleaning properties, 49 Anca et al. 50 decorated a cotton fabric surface with synthesized TiO2 using the sol–gel approach. The TiO2 fabrics became hydrophobic and exhibited a self-cleaning character successfully. Montazer and Pakdel 51 treated raw wool fabric and oxidized it by nano TiO2 powder in an ultrasonic bath. They used butane tetracarboxylic acid and sodium hypophosphite as a catalyst to link this article to the wool surface. The self-cleaning property was achieved, and the increase in the concentration of nano TiO2 in the bath can help the property to improve. Galkina et al. 52 produced modified cotton fibers with good self-cleaning properties. A nanocrystalline sol TiO2 was obtained, and the aspects of cross-linking of TiO2 NPs and 1,2,3,4 butane tetracarboxylic acid as a spacer were used. Shabana et al. 53 presented a new class of self-cleaning textiles with meso-tetra(4-carboxyphenyl) porphyrin (TCPP) followed by hydrophilization with trimethoxy octadecyl silane (OTMS). They used a simple post-adsorption method that contained a stepwise deposition of anatase TiO2. The prepared cotton fabrics showed successful superhydrophobic and visible-light photocatalytic activities along with self-cleaning ability. Similarly, they coated another cotton fabric surface with thin films of TCPP with different metal centers (MTCPP, M = Fe, Co, and ZnO) in combination with anatase TiO2. The MTCPP/TiO2-coated cotton fabrics developed a significant photocatalytic activity. 54 Xu et al. 55 combined photoactive TiO2 and superhydrophobic SiO2 to make TiO2–SiO2 composite particles with fluorine-free superhydrophobic cotton fabrics. The fabric showed superhydrophobic and photocatalytic properties along with excellent self-cleaning performance. Pakdel et al. 56 applied TiO2 and TiO2–SiO2 nanocomposites on wool fabrics through a low-temperature sol–gel method. Titanium tetraisopropoxide (TTIP) and tetraethylorthosilicate (TEOS) were employed as precursors. The self-cleaning function and hydrophilicity of wool fabrics were successfully prepared. Shengnan et al. 57 fabricated self-cleaning cotton fabric with superhydrophobicity and high amphiphilicity by building micro–nano structures with low surface energy. The TiO2 and SiO2 NPs blended by hydrolytic (heptadecafluoro-1,1,2,2-tetrahydrodecyl triethoxysilane) trimethoxysilane (F-17) were modified and co-deposited onto the cotton fabric. They used two different coating methods: (1) suction filtration for immobilizing the modified TiO2/SiO2 onto cotton fabric based on swollen cellulose action by N-methyl morpholine-N-oxide (NMMO) solution; (2) adhesion by spraying waterborne polyurethane (WPU) before coating. The accomplished self-cleaning fabric achieved excellent water-repellency and oil-repellency. It is considered a beneficial tool to improve the performance of textile production and provide new or enhanced properties. In contrast, the cost of NPs in mass production is expensive. It impacts the environment and human health due to the uncontrolled release of NPs and the whole ecosystem is widely covered under the state of “nanotoxicology.” 49
The development of self-cleaning textiles with additional properties
Superhydrophobic surfaces can be modified by improving specific properties through surface functionalization and combining new materials and technologies. 58 Currently, the development of textile finishes makes it possible to produce self-cleaning textiles with many properties, such as antimicrobial, UV protection, laundering durability, and water/oil repellency.
Antimicrobial properties
Karimi et al. 59 coated cotton fabric with graphene oxide and the composite of TiO2 with a simple dipping coating method. The treated cotton showed excellent self-cleaning along with excellent antimicrobial activity. Scacchetti et al. 60 prepared self-cleaning, antimicrobial cotton fabrics by a finishing process that combines microcapsules of phase change materials (MPCMs) and TiO2 NPs. Liu et al. 61 dyed cotton fabrics with TiO2 by adding cross-linking agents using the dip-coating process. The prepared fabrics showed excellent self-cleaning properties and UV light fastness, and improved antimicrobial activity. Pakdel et al. 62 coated wool fabrics by the functionality of TiO2 NP sol, TiO2/SiO2, TiO2/metal, and TiO2/metal/SiO2. They were modified by incorporating three types of noble metals (Ag, Au, and Pt) and silica under visible light through a low-temperature sol–gel method. The coated wool fabrics showed an enhanced visible-light-induced self-cleaning property and, photocatalytic and antimicrobial activities. Hebeish et al. 63 coated antimicrobial and self-cleaning cotton textiles with TiO2 nanowires and TiO2 nanowire doped (Ag-PVP) nanocomposites. An alkaline solid medium by the hydrothermal method was used and TiO2 NPs as a precursor. Based on the above results, coated cotton fabrics could be promising for possible medical and industrial applications. Sundaresan et al. 64 successfully treated cotton fabrics with nano TiO2 combined with an acrylic binder. The layer was added on the cotton fabric surface by using the dip-coating method. It provided antimicrobial activity, UV protection, and self-cleaning ability.
UV protection properties
Abidi et al. 65 modified cotton fabric by titania nano-sols prepared through the sol–gel process with tetrabutylorthotitanate [Ti (OC4H9)4] as the active ingredient. The photocatalytic properties of titania nano solid treatment on the fabric surface imparted self-cleaning properties to the cotton fabric and showed excellent UV protection. Wang et al. 66 coated a cotton textile with ZnO–SiO2 nanorods to maintain superhydrophobic and UV blocking properties. The treated cotton textile showed an excellent UV blocking property with UV protection and durable superhydrophobicity under UV irradiation. This multifunctional cotton textile will have potential applications in various areas, such as medical, military, and biological industrial fields. Qi et al. 67 treated polyester fibers with low-temperature plasma coated with anatase TiO2 prepared by an aqueous sol–gel process. The coated polyester showed significant improvement in self-cleaning performance, adhesion, and excellent UV protection. Ahmed and Kan 68 coated pristine cotton fabric with Reactive Blue 21 (RB-21)/(TiO2) sol by the sol–gel method. It showed an excellent UV protection and photocatalytic self-cleaning performance. Mishra and Butola 69 coated cotton fabric with TiO2 by using the sol–gel technique and a precursor through three different process routes (pad–dry–cure, pad–dry–hydrothermal, and pad–dry–solvothermal) to provide a stable coating and improved wash durability, UV protection, and self-cleaning property successfully.
Water/oil separation properties
Jin et al. 72 coated a superhydrophobic cotton textile by a cost-effective and straightforward dip-coating route using ZnO sol and mercapto silanes. Hydrophobic treated 3-mercaptopyl trimethoxysilane (MTS) and dodecafluoroheptyl methacrylate (DFMA) with thiol–ene click chemistry reaction was included. The prepared cotton superhydrophobic showed excellent self-cleaning and oil–water separation properties along with acid resistance. Jiang et al. 71 fabricated robust superhydrophobic fabric by modifying the surface with anatase TiO2 sol and mercapto silanes followed by hydrophilization with perfluorooctyl methacrylate (PFOMA) and the thiol–ene click reaction. The fabric showed great self-cleaning, along with photocatalytic degradation and resistance to intense acidic or alkaline environments. It also demonstrated excellent wettability behavior and promising application in oil–water separation.
Laundering and rubbing durability
Liu et al. 72 presented self-cleaning cotton by immobilizing TiO2 NPs onto the surface with co-graft polymerization of 2-hydroxyethyl acrylate (HEA). It showed improved hydrophilicity and photocatalyzed self-cleaning ability, well-retained after 30 accelerated laundering circles. Ashraf et al. 73 grew ZnO seed into ZnO nanorods, then placed it on a polyester fabric surface and finalized it using the hydrothermal method. The functionalized fabric showed self-cleaning under UV light, excellent washing fastness, and good rubbing durability.
Previous studies examined the influence of microscopic features on textile fabrics at two different levels: the surface coating level with nanotechnology and the microscopic features at the fabric woven structural level. This research review seeks to identify the microscopic features and their influence on the self-cleaning ability of textile fabrics. The features mainly focused on are roughness, porosity, and wettability. A detailed discussion of their influence from the existing literature is provided in the following sections.
The influence of microscopic features on textile fabric self-cleaning ability
Microscopic features at the self-cleaning coating level
Combining the previous literature outcomes involved self-cleaning and included the other microscopic features of fabrics, which can positively affect self-cleaning ability. Textile fabrics consist of fibers with complex mechanical structures constructed by two multiscale structures: the microscale in a yarn structure and the macroscale in a fabric structure. To make a superhydrophobic surface, we need to make the surface hydrophobic by creating appropriate roughness, porosity, and wettability behavior. They have become the critical point of comprehensive and intensive research activity because of the steadily growing interest in technical textiles and composites.43,74,75 The surface of the textile fabrics is not flat, and its geometrical roughness is considerable and plays a significant role in the use of the fabric. The surface roughness amplifies the water-repellency of hydrophobic materials. In addition, the surface roughness and surface energy are two significant factors affecting the wettability, as shown in Figure 10.
37
The porosity is an important parameter, which is the ratio of air volume to the total volume of a surface. Therefore, the total porosity of woven fabrics usually comprises two types of porosity: the microporosity caused by the void spaces among fibers in yarns and the macroporosity resulting from the void spaces among yarns.
76
The chemical properties and surface microstructure control the wettability of a solid surface with a liquid. It is determent by the interfacial energy among the solid, liquid, and vapor phases. Wetting is the term used to describe the study of how a drop of liquid spreads out on a solid surface, and it is divided into two different systems: total wetting and partial wetting.77,78 The contact angles (CAs) between a liquid with known surface tension and a solid are measured to evaluate the surface energy. Superhydrophobic surfaces should exhibit CAs with water greater than 150
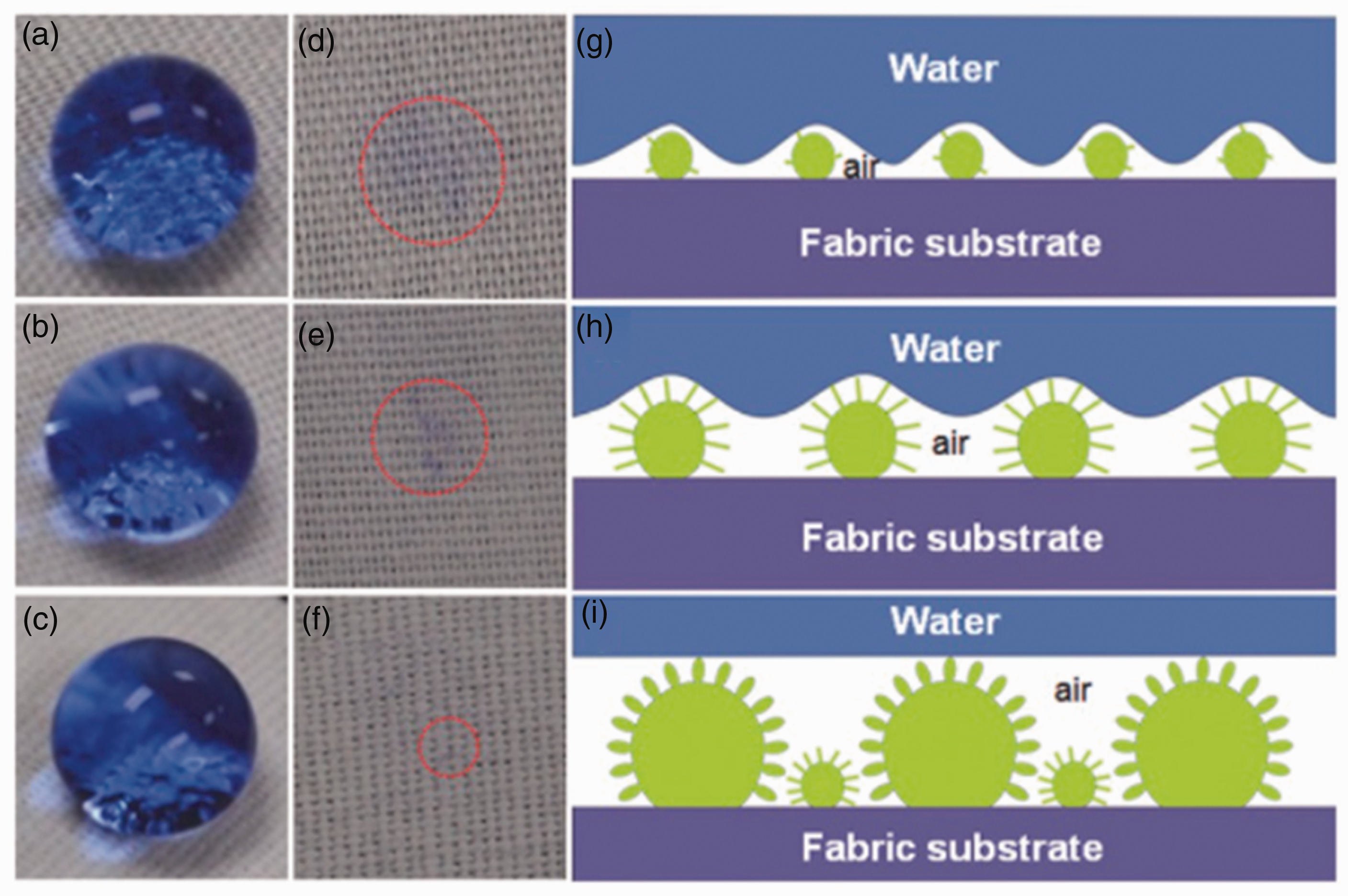
Optical photographs of water droplets dyed with methyl blue on superhydrophobic TiO2@cotton fabric surfaces with different adhesion (a)–(c) and the surface traces after the water droplet left the cotton fabrics (d)–(f). Schematic illustration of three types of solid/liquid/air three-phase contact models with adhesive forces ranging from high to low: (g) “area” contact model with high adhesion; (h) “line” contact model with medium adhesion; (i) “point” contact model with low adhesion. 37
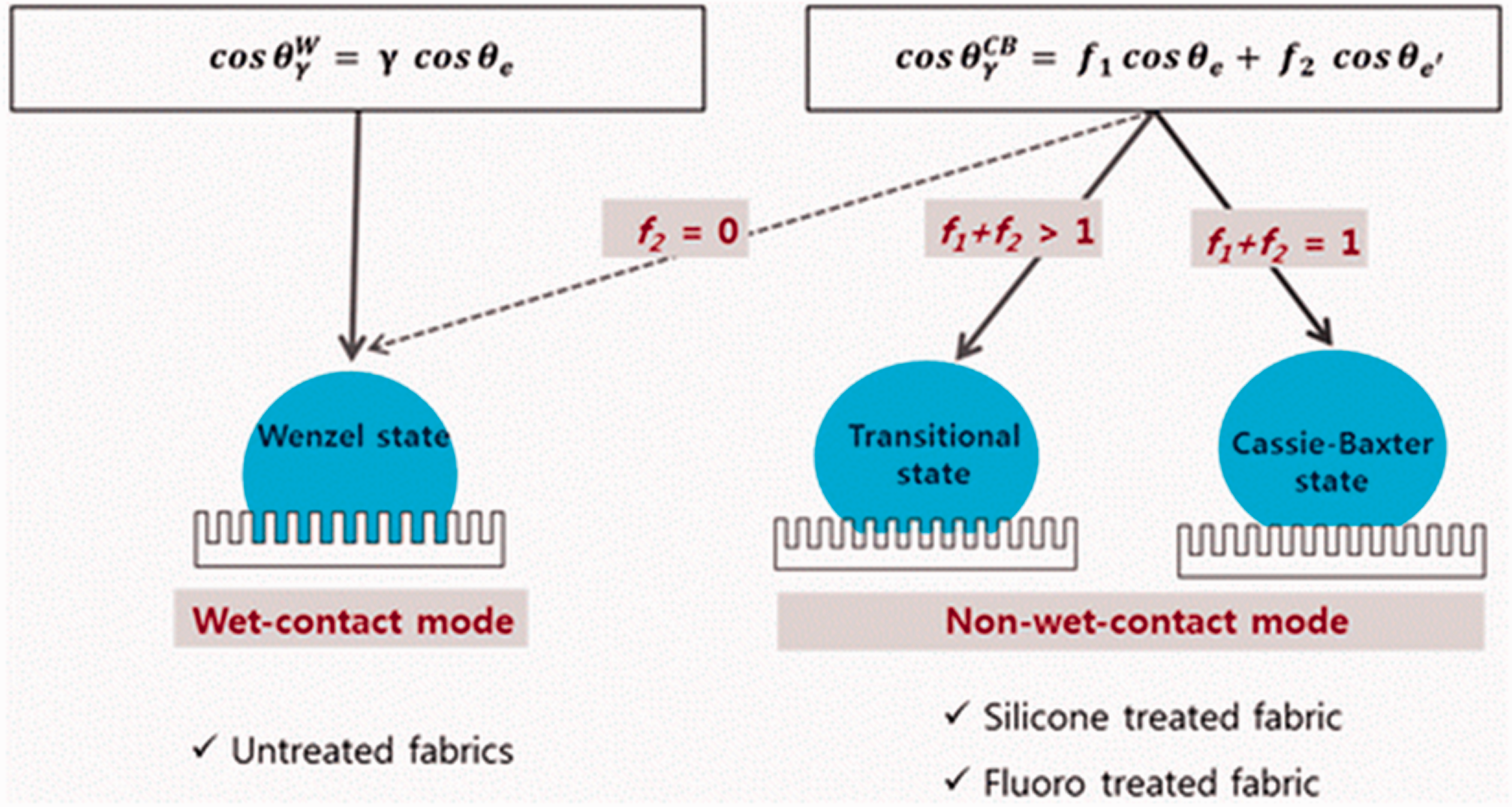
Wetting characteristic with models applied. 81
Moreover, as woven fabrics essentially possesses multiscale void spaces (porosity) and roughness due to the yarn and fabric structure, it is essential to consider them to create the liquid–vapor interface so that the area of the liquid contacting the solid can be reduced. 88 For that, surface roughening is required to reduce the spreading of a liquid on a solid surface, while the wetting behavior controls the surface energy and the geometric structure of the surface. 34
Microscopic features at the woven structural level
Weaving is the process of joining two upright sets of yarns, called the warp and the weft, to form a fabric. During the weaving process, warp yarns are fixed to the loom while weft yarns are inserted crossways. Depending on the pattern in which warps are raised, fabrics with very different forms and mechanical properties were produced. 89 The geometrical structures of fibers, yarns, and fabrics are considered important to magnify the energy of a surface. Thus, many researchers have focused on the morphology of fiber surfaces. 90 Nagamadhu et al. 91 studied the dynamic mechanical properties of the three different woven structures of sisal fabric, as shown in Figure 12, weft rib (WR) and Plain 1–Plain 2 (P1-P2). The difference between P1 and P2 is grams per square meter (GSM): P1 is heavier than P2. The influence of textile physical properties, such as grams per unit area, yarn diameter, yarn spacing/fabric density, and woven pattern on the thermo-mechanical properties of reinforced epoxy composites was investigated. The thermo-mechanical properties were improved three to four times by changing the woven pattern, as observed in the case of the WR composite woven pattern being better than P1-P2. 91 Li et al. 92 used three kinds of fabrics woven with familiar textures: plain, twill, and honeycomb weaves with identical warp and weft density, as shown in Figure 13. They investigated the effect of the structural factors of woven fabrics on the acoustic absorption property. The results showed that there is no significant connection between fabric sound absorption performance and surface density, although porosity, pore size, and pore shape were essential factors affecting the acoustic properties. Liu et al. formed woven, knitted, or nonwoven through weaving technology, affecting the electrical and electrochemical properties.93 Therefore, the effects of textile structures, including porosity and pore configuration, were studied as well. The porosity and pore configuration of the fabrics had an essential effect on the electrochemical properties. The woven fabric exhibited the best electrochemical performance compared with the other two fabrics, 93 as the porosity and pore configuration can affect the self-cleaning ability. Jaouani et al. 94 measured woven fabric structural and material parameters that affect dielectric properties and fabric surface roughness. The results were obtained by using 12 different fabric structures to measure their roughness. Moreover, it was noticed that the highest roughness values were on the plain weave and the lowest ones were on the sateen weave samples. Huan et al. 95 studied the influence of woven fabric structural and material parameters on dielectric properties. The yarn counts, fabric weaves, and fiber contents were the critical factors for different dielectric properties. The dielectric constants were associated with fabric yarn density, fabrics with high density, and fibers with high permittivity. Asayesh et al. 96 produced five different weave patterns (plain, hopsack 2/2, twill 2/2, twill 3/1, warp rib 2/2) with three different nominal weft densities and examined the effect of fabric structural parameters on the compressional properties. The results indicate that the compressibility parameters were affected by the fabric weft density and weave pattern. The increasing weft density decreases the dissipated compression energy and the fabric’s compressibility. Lim et al. 88 developed a fabric with different weave structures (plain-woven fabric, 2/2 basket, and 2/1 twill) and studied the wetting behavior of fabrics based on the geometry of woven fabrics and obtained an improvement of the liquid-resistant/repellent surface. The results indicated that the CAs of the fabrics were the highest for plain-woven fabric, lower in 2/2 basket, and the lowest in 2/1 twill, as shown in Figure 14. These results are in good agreement with experiments on synthetic woven fabrics. Similarly, they created void spaces at multiple scales (i.e., macro, micro, and nano) to increase the energy surface that the liquid–vapor interface requires. They used different void spaces in the weft direction to obtain an oil-resistant or repellent surface. The created models implied that the oil resistance fabric increases as the void space increases. 97

(a) Schematic of woven types prepared at OM Textile Industries. (b) Three types (two plain and one weft rib) of the sisal woven pattern. 91

Patterns of plain, twill, and honeycomb weave simulated with TexGen. 93
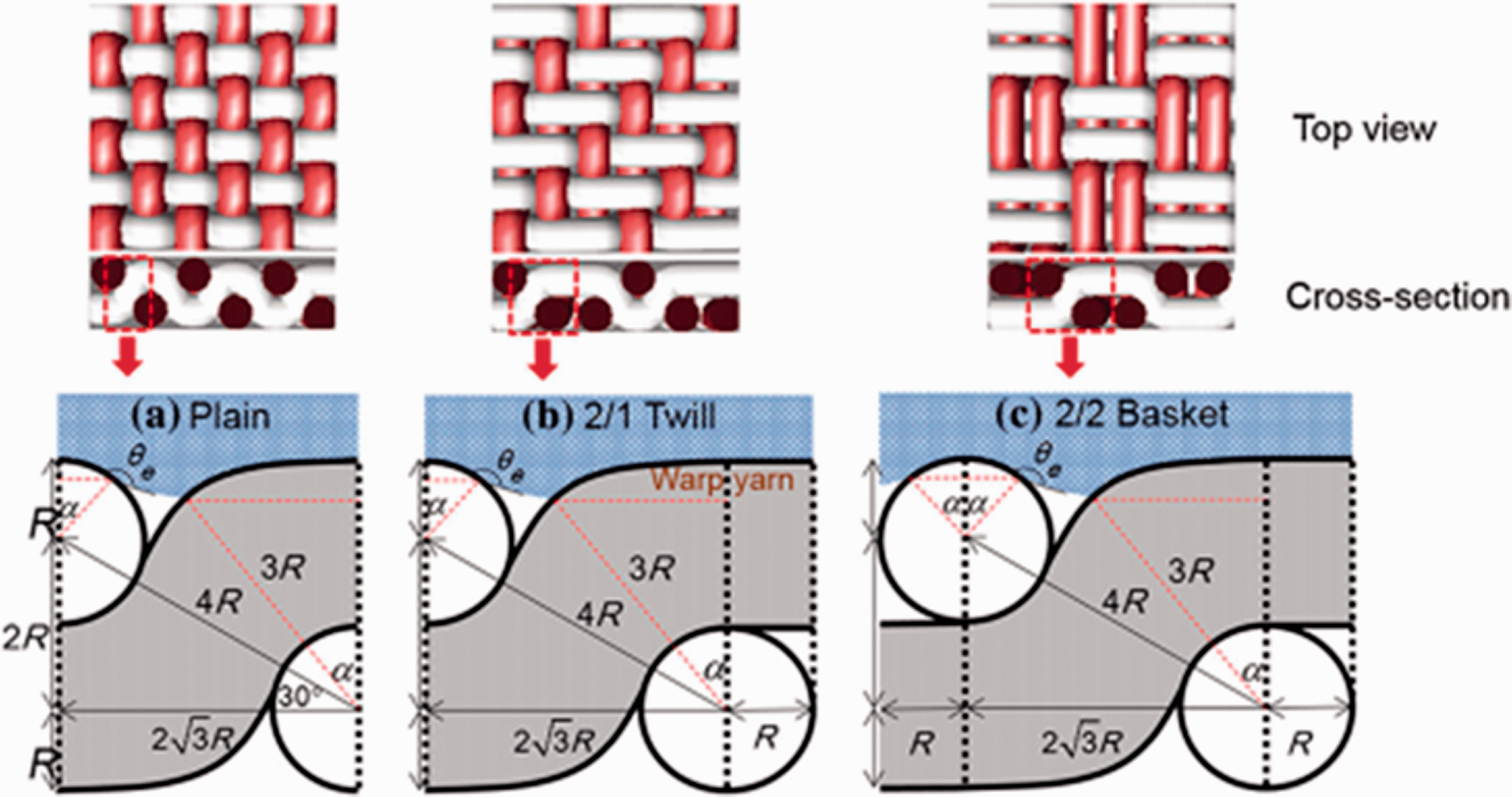
Three-dimensional images of three woven fabrics and geometries of the unit structure with liquid depositions: (a) plain; (b) 2/1 twill; (c) 2/2 basket. 88
Researchers have examined the woven level by focusing on the strength and durability along with other properties. They used microscopic features to increase the mechanical strength of the fabric. However, they did not apply it to change the microscopic features at the woven level to make a self-cleaning ability. Changing the shape of the surface to get self-cleaning properties has been done in other areas, such as polymers. Researchers changed the geometry of the microscopic features of the top specimen surface to create a self-cleaning ability. 98
The previous efforts on self-cleaning processes were critically analyzed as a particular focus is given to those efforts, which describes the influence of fabrics' inherent microscopic features on their self-cleaning ability. The associated challenges to obtain a self-cleaning surface, in general, are provided below.
Challenges in the existing processes for making self-cleaning textile fabric and their effectiveness
Despite the significant accomplishments that have been made for creating self-cleaning textile fabric, there are complexities and challenges. From Li et al.’s point of view, 37 the connections between the textile and chemical reagents' primary mechanism during the fabrication process are difficult to handle. Most fabrication methods hold only for laboratory use and are not suitable for large-scale industrial production. The mechanical durability of water-repellent fabric suffers from poor mechanical stability and durability during laundering. Although many researches have worked successfully in the laboratory, they failed when used in real-time products. “Peeling off” of the coatings from the surface is a significant concern that must be addressed while developing self-cleaning. The coating should be deposited onto the surface to avoid any air gaps. The behavior of the water droplets on the coated surface is an area that should be focused on as well. The produced coating should not allow the water droplets to assemble on the surface, but should just roll off. 99 Dalawai et al. 1 claim that the drawbacks of having a self-cleaning property were poor mechanical strength and lack of ability to withhold the surface property under severe environmental changes. Also, the use of hazardous chemicals and high-cost treatment that takes a longer time could affect the usage of the self-cleaning surface property in many applications. Li et al. 97 state that the major challenge of having flexible self-cleaning was the loss of the property due to the number of usages or after several washing cycles. Padmanabhan and John 100 addressed some critical challenges and concerns in developing self-cleaning surfaces and coatings; the primary issue is based on the surface's mechanical durability and chemical stability after the coating. The exposure to detergents and outdoor temperature changes leads to the separation of the material from the surface or distortion to the used material, which increasingly declined the coatings' performance. Taking a long time to decompose stains on the cloth, the mechanism of high decomposition power affects the fibers and the durability of the cloth due to repeated laundering cycles. For Roy et al., 28 for superhydrophobic self-cleaning textiles surfaces the low robustness of the coating limits applications; when being mechanically or chemically damaged, they lose their hydrophobic property, and the performances will eventually decline. The main problem with the previous researches is that they achieved the superhydrophobic surfaces on fabrics with the help of fluorinated compounds and various organic/inorganic NPs, which are harmful to the environment and human skin. Moreover, with the increase of environmental pollution and climate changes, the demand for efficient and sustainable material sources has grown enormously. As a result, sustainability in superhydrophobic surfaces has become a priority and a significant challenge to researchers.
On the other hand, the effectiveness of having the self-cleaning ability has many benefits in various industries. They are reducing cleaning efforts that can improve environmental protection, making the textiles last longer by improving aging behavior and extending surface purity. Heavy laundry bills can be reduced by lowering the number of washing cycles, as the new “easy-washable” textile is very economical and satisfies the market demand.12,101 Self-cleaning fabrics have environmentally friendly properties as they successfully reduce cleaning efforts, preserve a considerable amount of water and energy, and save time and money laundering.2,102,103 Considering the limitations associated with surface coatings, such as surface peel off, chemical affinity with washing material, and so on, it is worth evaluating whether we can obtain the self-cleaning ability by using the geometrical microscopic features at the woven structural level, as ultimately this ability can be obtained by changing the surface roughness and surface energy level.104,105
Conclusion
In this review, all the previous efforts to create self-cleaning textiles by creating proper surface roughness or decreasing the surface energy using low surface energy chemicals or nanotechnology are compiled. Their key working elements have been discussed in detail. The development of different methods and techniques in recent years, such as the dip-coating (sol–gel) method, spin coating, spray coating and electro-spraying, chemical etching, CVD, HTT, electrospinning, LBL self-assembly, and wet chemical synthesis, is reviewed. The preparation of these methods in the existing literature aimed to develop self-cleaning textiles with the help of coatings. They maintain different chemicals and processes; some are affordable and easy to handle, while others are complicated, expensive, and must be used in a laboratory only due to chemical usage. Several chemical solutions were used along with nanotechnology to enhance the coatings for desirable surface roughness or lower surface energy, as the nano-layer of anatase TiO2 and other NPs are successfully applied on the textile fabric surface or the fiber itself.
These coatings create a different surface on the fabric, and hence the changes in the microscopic features of this surface control the self-cleaning ability. The use of these organic solvents and chemicals can be a cause of environmental health hazards. Therefore, the development of user-friendly and cost-effective methods to structure self-cleaning surfaces can be for inspiration to excellent ideas. Instead of using an additional coating, the microscopic features of the fabric itself can also influence the surface roughness and low surface energy and provide self-cleaning ability at the woven structural level. Although the influence of the microscopic features at the fabric woven structural level has been studied in the past, a comprehensive effort is still required to evaluate the actual quantitative influence on the self-cleaning fabric phenomenon.
Researchers examined the woven level by focusing on the strength and durability along with other properties. They used microscopic features to increase the mechanical strength of the fabric. However, they did not change the microscopic features at the woven level to evaluate the self-cleaning ability. Changing the shape of the surface to get self-cleaning properties has been done in other areas, such as polymers, where the geometry of the microscopic features of the top of the specimen surface were changed to create the self-cleaning ability. However, the same can be used to employ the same concept of geometrical changes and obtain self-cleaning textile fabric surfaces.
Key microscopic features, such as surface roughness, porosity, and wettability of a textile fabric, have still not been comprehensively investigated for their influence on fabric self-cleaning ability. Significantly, the interdependencies between these features with overall fabric geometry at the woven level have not been explored quantitatively. Qualitative observations have been made mainly in the past literature. However, the mass production of fabrics with self-cleaning ability requires extensive empirical studies. These studies must involve parametric analysis on varying the values of the microscopic features and their quantitative influence on the desired self-cleaning feature. So, more exciting applications in self-cleaning surfaces need to be focused upon intensely in the coming years to use the surface property for more comprehensive industrial applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
