Abstract
Objective
To evaluate glucagon-like peptide-1 receptor agonist (GLP-1 RA)-specific associations with hair loss, characterize reported alopecia subtypes and discuss potential underlying mechanisms.
Methods
A systematic literature search was conducted across four databases (PubMed, Embase, Scopus, and Web of Science) according to PRISMA guidelines and registered in PROSPERO (CRD420261297384). Studies were included if they were primary articles assessing hair loss related to GLP-1 RA use.
Results
Of 133 studies identified, 24 met inclusion criteria. Among GLP-1 RAs, semaglutide and tirzepatide demonstrated the highest incidence rates of hair loss and more frequent signal detection in pharmacovigilance studies. Although infrequently classified overall, androgenetic alopecia and telogen effluvium were the predominant subtypes of hair loss reported. Tirzepatide, associated with the greatest magnitude of weight loss, was most frequently linked to telogen effluvium. Hair loss associated with semaglutide appeared to be dose-dependent, with doses < 2mg weekly rarely implicated while higher obesity-treatment doses were more commonly associated with hair loss. Females appeared to be disproportionately affected. Rapid weight loss emerged as a potential contributor, particularly for telogen effluvium. In contrast, fewer studies assessed hair loss with liraglutide, dulaglutide, lixisenatide and exenatide, and they typically exhibited lower reported risk when compared to semaglutide and tirzepatide.
Conclusions
Accumulating evidence from pharmacovigilance databases and clinical cohorts suggests an increased risk of hair loss with certain GLP-1 RAs, particularly semaglutide and tirzepatide. Further studies are needed to clarify the etiology of drug-induced weight loss, identify vulnerable populations, and establish causality and temporal relationship through large, prospective randomized trials.
Keywords
Introduction
The rapid increase in the utilization of glucagon-like peptide-1 receptor agonist (GLP-1 RAs) for treatment of obesity and diabetes has brought to attention adverse effects not prominent in early clinical trials. Originally developed for management of Type 2 diabetes (T2DM), GLP-1 RAs confer substantial clinical benefits, including glycemic control, weight loss, and cardiovascular risk reduction. These drugs have changed the weight loss scene causing patients to loss up to 15-20% of their body weight within a year. Hair loss/alopecia has arisen as an underexplored safety signal, emerging from data in pharmacovigilance studies and recent clinical trials for weight loss.1–3 Public interest in a potential association between GLP-1 RAs and hair loss has increased markedly from 2023 to 2025, particularly regarding subcutaneous semaglutide (Ozempic and Wegovy) and tirzepatide (Mounjaro and Zepbound). 4 The Food and Drug Administration approved GLP-1 RAs for obesity or T2DM include semaglutide, liraglutide, dulaglutide, exenatide, lixisenatide and the dual glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 RA, tirzepatide.
GLP-1 RAs have been associated with non-scarring hair loss, most commonly telogen effluvium (TE) and androgenetic alopecia (AGA),4–7 both of which involve alterations in hair follicle cycling. TE is a transient reversible hair loss characterized by diffuse, temporary hair shedding triggered by physiological or psychological stressors, where hair follicles are accelerated into telogen phase. 8 AGA is a hereditary patterned alopecia, characterized by progressive follicular miniaturization and distinct distribution patterns, including frontotemporal recession and vertex involvement in men and diffuse thinning in women. TE may accelerate or unmask the clinical presentation of underlying AGA. Given that hair loss is distressing for patients and may adversely affect treatment adherence and quality of life, a clearer understanding of the relationship between GLP-1 RAs and hair loss is warranted. We have systematically reviewed the current literature from clinical trials, pharmacovigilance and observational studies to explore the GLP-1 RA-specific evidence for hair loss, the type of hair loss caused, and whether it differs by drug indication.
Methods
Protocol and registration
A systematic review was undertaken as per Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) to assess the available literature on alopecia and GLP-1 RAs. 9 The study protocol has been registered in the PROSPERO database (Prospero ID: CRD420261297384).
Search strategies
A search was conducted in January 2026 across PubMed (MEDLINE), Scopus, Web of Science and Embase (Ovid) with the terms including: ‘hair loss,’ ‘alopecia,’ ‘GLP-1 agonist,’ ‘semaglutide,’ ‘tirzepatide,’ ‘liraglutide,’ ‘dulaglutide,’ ‘exenatide,’ and ‘lixisenatide.’ Package inserts and product monographs for GLP-1 RAs were also consulted, but not included in the systematic review.
Eligibility criteria
Studies were screened by via Covidence by one reviewer and if inclusion was questioned it was confirmed by an additional reviewer.
Inclusion criteria
Studies were included if reporting hair loss in patients using GLP-1 RAs regardless of indication. These included randomized clinical trials, retrospective analyses of pharmacovigilance databases and cohort studies.
Exclusion criteria
Reviews, meta-analyses and case reports were excluded. Studies were excluded if they did not specify number of participants with hair loss or the group with hair loss (whether or not they were using GLP-1 RAs).
Studies were grouped based upon type of GLP-1 RA indicated and discussed as such.
Results
Clinical evidence linking GLP-1 RAs to hair loss
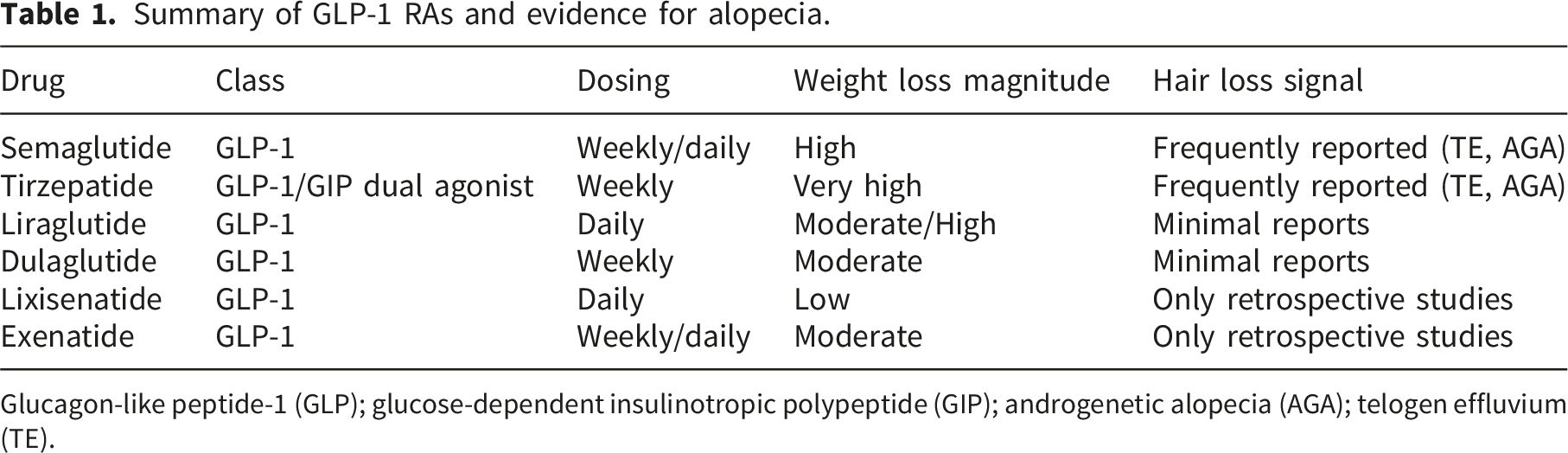
The literature search gathered 133 studies, 24 of which met inclusion criteria (Figure 1). Given the heterogeneity of study design and outcome reporting, results are discussed narratively focusing on the associations between hair loss and specific GLP-1 RAs, as well as the types of alopecia implicated. Evidence supporting the association between GLP-1 RA usage and hair loss remains limited for some individual GLP-1 RAs, particularly those with only retrospective data or small clinical trials assessing hair loss incidence (Table 1). Nevertheless, the broader literature supports an overall increased risk of hair loss with GLP-1 RA use, most consistently in the context of pharmacological weight loss, though cases have been reported in the context of T2DM and other conditions (Table 2).2–5,10 PRISMA chart. Summary of GLP-1 RAs and evidence for alopecia. Glucagon-like peptide-1 (GLP); glucose-dependent insulinotropic polypeptide (GIP); androgenetic alopecia (AGA); telogen effluvium (TE). Literature on GLP-1 RA and hair loss. Food and Drug Administration Adverse Events Reporting system (FAERS), Hair loss type not specified (NS), telogen effluvium (TE), androgenetic alopecia (AGA), alopecia areata (AA) subcutaneous (s.c.)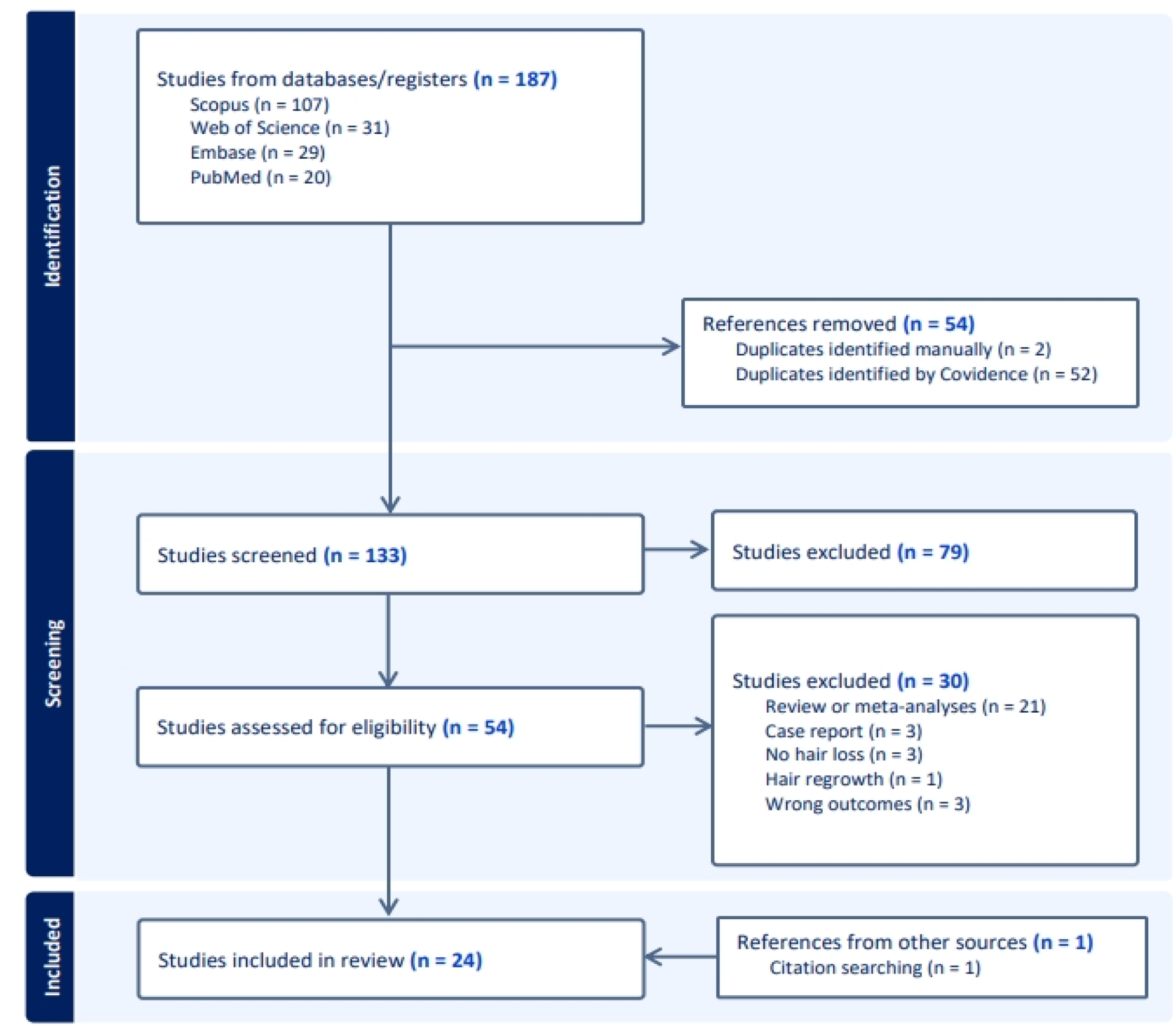
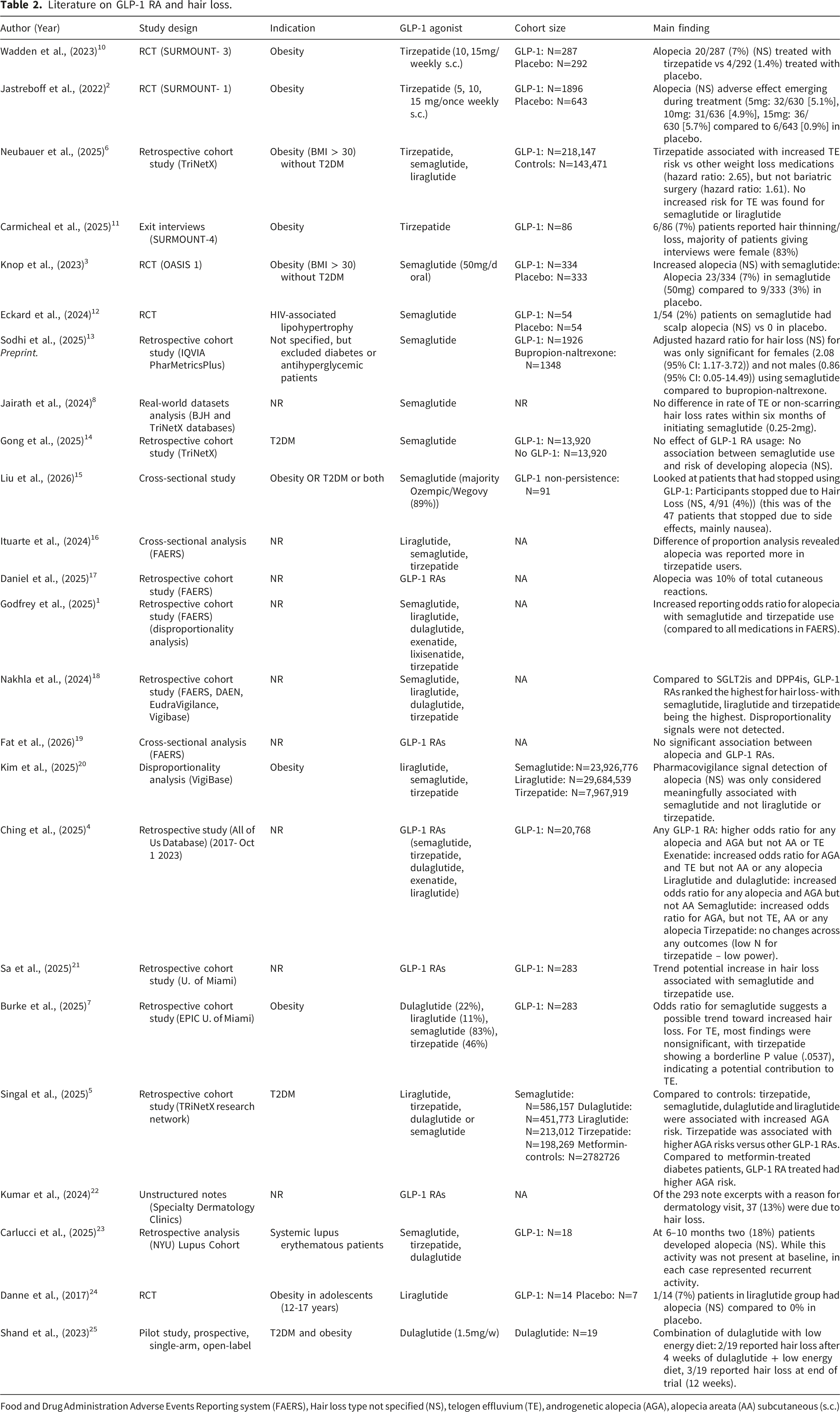
In keeping with the broader literature, a recent meta-analysis of randomized clinical trials reported an alopecia incidence rate of 6.0 per 1000 patient-years for GLP-1 RAs users vs 0.8 per 1000 patient-years in placebo groups, corresponding to a three-fold increased risk of alopecia (risk ratio 3.40 [95% CI 1.18-9.81], I2 = 67.77%, P = 0.02). 26 Consistent with this signal, pharmacovigilance studies most frequently implicate tirzepatide and semaglutide as being associate with an increased risk for hair loss, indicating potential drug specific effects (Table 1).1,5,20,27
The type of hair loss associated with GLP-1 RA use is often not reported. When specified, GLP-1 agonists have been primarily associated with non-scarring forms of alopecia, namely Telogen Effluvium (TE) and Androgenetic Alopecia (AGA), with little evidence to support an increase in alopecia areata (AA) or other hair loss disorders (Table 2).4–6 Notably, not all studies have found an increased risk of hair loss with GLP-1 use, particularly retrospective analyses. This inconsistency may reflect limited event capture, underreporting, or lack of specification regarding both the GLP-1 RA used and the hair loss phenotype.14,19,28 Further, pharmacovigilance studies have yielded conflicting results, with some studies identifying no safety signal and others demonstrating increased reporting odds ratios for various GLP-1 RAs. These discrepancies may reflect methodological differences, including variation in comparator groups (e.g., controlled to another drug (metformin, DPP4i) or non-GLP-1 RA users), differences in inclusion criteria and propensity score matching (such as adjustment for body mass index and HbA1c), inconsistent outcome definitions (alopecia broadly versus specific subtypes), and whether GLP-1 RAs were analyzed collectively or independently.1,5,6,14,19
Evidence of alopecia by GLP-1 RA subtype
Semaglutide
Among GLP-1 RA, semaglutide has often been implicated in an increased risk of alopecia, with signals observed in randomized controlled trials and retrospective cohort studies (Table 2).1,3,12,20 Data regarding the type of hair loss associated with semaglutide remain limited; however, several pharmacovigilance studies report an increased risk for AGA, but not TE or AA.4–6
Variability in reported associations may be partially explained by dose-dependent effects, with effects not observed at lower doses used for T2DM (0.25-2 mg),14,28 but increasing risk of alopecia with higher doses more often prescribed for obesity. For example, in trials with high-dose oral semaglutide (50mg/d) for weight loss, alopecia was reported in 7% of treated patients compared with 3% of controls. 3 Consistent with this, Tran et al., (2024) found that increased alopecia risk was observed only with 50mg/day oral semaglutide (23/334 [6.9%]), but not 2.4mg weekly subcutaneous semaglutide (1/407 [0.2%]). 29
Large product trials of subcutaneous semaglutide (Wegovy; 2.4mg weekly; N=2116) nevertheless demonstrated a modestly increased incidence of hair loss compared with placebo (3%
Tirzepatide
Tirzepatide is a highly effective weight loss drug, typically producing greater reductions in body weight than semaglutide in clinical trials. 31 In individuals with obesity but without T2DM, clinical trials report body weight reductions of 16 to 21% with tirzepatide (10 or 15mg subcutaneous/weekly) compared with placebo.2,10 In multiple clinical trials evaluating tirzepatide for weight loss, alopecia was reported in 5.4 % [119/2183] of treated participants versus 0.9% [22/2221] receiving placebo.2,10 Alopecia was recorded through systematic assessment; however, time to onset was not reported.2,10 The risk of hair loss appears to be comparable across the studied doses (5, 10 and 15 mg subcutaneous/weekly). 2
Tirzepatide is particularly associated with TE,6,7 more so than other GLP-1 RAs, which may reflect its rapid weight loss effects.6,31 Notably, even the lowest tirzepatide dose (5mg weekly) induces weight loss comparable to that observed with high-dose semaglutide (50mg oral/day and 2.4mg subcutaneous/week).2,3 Importantly, the risk of TE with tirzepatide does not differ substantially from that following bariatric surgery, 6 a well-established trigger of acute and delayed hair loss due to rapid weight reduction. 32 Tirzepatide has also been associated with an increased AGA risk, with higher reported risk compared with semaglutide, dulaglutide and liraglutide. 5 Similar to semaglutide, females using tirzepatide appear to be at higher risk for alopecia (7.1% in females vs 0.5% in males in Zepbound product trials). 33
Liraglutide
Evidence supporting an increased risk of hair loss with liraglutide use remains limited and is largely derived from retrospective cohort studies, many of which report no association between liraglutide exposure and alopecia.1,5,6,20 One retrospective study using All of US database found increased odds of any alopecia and AGA, but not AA, among liraglutide users. 4 A clinical trial for obesity in adolescents suggested a possible association with alopecia reported in 1 of 21 treated participants (7%) compared to none in placebo group; however, the small sample size precludes definitive conclusions. 24 Product trials of liraglutide (Victoza) in combination with metformin or insulin detemir (Levemir) reported hair loss at 1.3% and 1.8%, respectively, compared to 0.2% in controls. 34
Dulaglutide
Evidence supporting an association between dulaglutide use and alopecia is sparse. Most studies report no increased risk, with only one retrospective study finding increased odds ratio for any alopecia and AGA, but not AA with dulaglutide use. 4 In a retrospective cohort study, dulaglutide, along with liraglutide had the lowest AGA risk compared to tirzepatide and semaglutide. 5 In a small pilot study combining dulaglutide with a low-energy diet, hair loss was reported by 2/19 participants after 12 weeks, despite being absent at baseline. 25 Overall, further research, particularly controlled clinical trials, is necessary to better define the relationship between dulaglutide and hair loss.
Exenatide
To date, no clinical trials have reported an association between exenatide use and alopecia. As it is not as commonly used as some of the other GLP-1 agonists, populations may be more limited. Retrospective cohort studies using the Food and Drug Administration Adverse Event Reporting System (FAERS) have found no increased reporting odds with exenatide, 1 whereas analyses of All of US database identified increased odds of AGA and TE but not any alopecia classification. 4 These discordant findings highlight the limited and inconsistent nature of the current evidence base.
Discussion
Proposed biological mechanisms
Weight loss and nutritional insufficiency
Hair loss reported with GLP-1 RAs is likely multifactorial, with acute metabolic perturbations, particularly rapid weight loss, caloric restriction and hormonal shifts, the most likely contributors (Figure 2). GLP-1 RA use is associated with micronutrient deficiencies including vitamin D, iron deficiency, and insufficient dietary calcium and protein.
35
Substantial caloric restriction and rapid adipose loss can precipitate TE through transient nutritional deficiencies (e.g., iron, zinc, protein, or micronutrients) and systemic metabolic stress, a pattern well described after bariatric surgery and severe dietary restriction. Following bariatric surgery, alopecia may present as an acute process occurring within three months, typically TE-related, or as a chronic process with onset six months or more after surgery, often associated with nutritional deficiencies.
36
TE itself may occur in both acute and chronic forms. Acute TE typically emerges after some physical or psychological trigger with diffuse hair shedding lasting less than six months, whereas chronic TE persists beyond six months.
8
Chronic TE is thought to arise from repeated or persistent triggers such as ongoing caloric insufficiency or micronutrient defciency.
8
Proposed GLP-1 RAs effects associated with hair loss. GLP-1 RAs lead to rapid weight loss, appetite suppression and improved glycemic control. Telogen effluvium may be induced through GLP-1 RAs rapid weight reductions or related nutritional deficiencies. Telogen effluvium may unmask underlying androgenetic alopecia. Secondary drug effects, such as hormonal fluctuations leading to transient changes in androgen levels, may also contribute to androgenetic alopecia in susceptible individuals. GLP-1 RA, glucagon-like peptide-1 receptor agonist.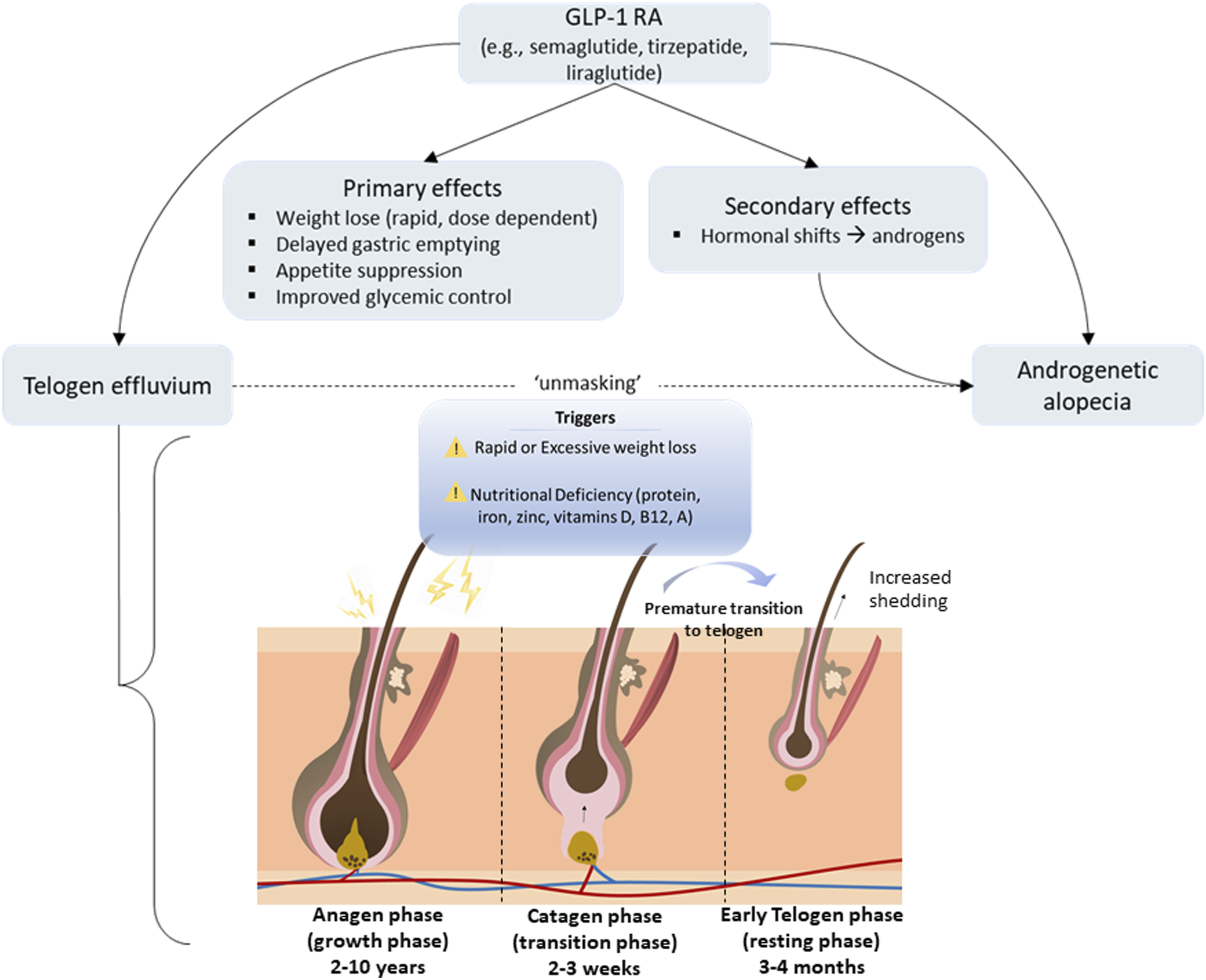
The magnitude and rate of weight loss associated with TE overlap with those commonly achieved using the most effective GLP-1 RAs implemented for weight loss. TE related to weight loss has been reported at mean percent weight reductions of 15%, and at rates of 3.54±2.85 kg/month.2,37 With lower rates of weight loss associated with TE in women than men, indicating they may be more vulnerable to TE even with a lower rate of weight loss.37,38 Consistent with this, tirzepatide, an agent associated with greater and more rapid weight loss, has higher reported rates of TE compared with other anti-obesity medications, but rates comparable to bariatric surgery, suggesting that weight loss dynamics, rather than the drug class alone drive risk. 6 Further supporting weight loss as a key upstream factor, alopecia incidence associated with semaglutide (Wegovy) increases among individuals experiencing the greatest weight reductions. 30 It is thought that the caloric restriction which may result in insufficient energy to support the hair matrix may be the cause of TE rather than the weight loss itself. 37 Rapid weight loss may also shift the bodies’ metabolism causing decreased thyroid output, changes in growth hormone and insulin-growth factor-1 release, and alterations in insulin and leptin levels.
Several pharmacovigilance studies report a low but elevated risk for AGA among users of GLP-1 RAs, particularly tirzepatide and semaglutide, compared to other anti-obesity drugs (e.g. metformin).4,5 Emerging real-world cohort data supports the idea that apparent AGA associated with GLP-1 RA use may, in part, reflect TE induced by rapid weight loss, which can unmask previously subclinical patterned hair loss rather than direct follicular toxicity of the drugs themselves.39,40 Beyond this unmasking effect, additional downstream hormonal or systemic effects may contribute to dysregulation of hair follicle cycling (Figure 2).
Hormonal shifts
One proposed mechanism involves transient GLP-1 RA-associated increases in circulating bioavailable testosterone, 41 which may promote hair follicle miniaturization, a hallmark process in AGA. 31 Hormonal changes may reflect recalibration as insulin levels drop as limited evidence implicates GLP-1 RAs in having any direct effects on androgen production (total and free testosterone, DHEAS, etc.). Furthermore, by improving insulin resistance, they can reduce total androgen levels in the long term as exhibited in women with polycystic ovary syndrome. 42 These hormonal shifts may also be different between sexes and in those with hormone related comorbidities. Taken together, the side effects of hair shedding, skin changes and menstrual cycle changes early in GLP-1 RA treatment are more likely due to rapid weight loss and deficits in calorie or protein intake, rather than excess androgens or other hormonal shifts.
Some evidence suggests that females are more susceptible to GLP-1-induced hair loss,13,33 although the underlying factors driving these higher rates in females is not entirely clear. Further, it is unclear whether there are differences between pre-and post-menopausal females. It should be noted that women are generally more likely to notice hair loss and seek medical attention for it, which may especially affect reporting rate in pharmacovigilance databases. 43 However, reporting bias alone may not fully explain sex differences, and biological mechanisms may also contribute. A meta-analysis of randomized control trials found that females using GLP-1 RAs loss more weight than males. 44 Greater weight loss in females, along with a higher likelihood of concurrent caloric restriction when on GLP-1 RAs, may contribute to the higher rates of hair loss reported in females. Additionally, female hair follicles may be more sensitive to hormonal shifts. For example, GLP-1 RA-induced metabolic shifts may alter estrogen signaling which play a crucial role in hair follicle biology. 45
Metabolic cycling
GLP-1 also contributes to peripheral metabolic and circadian clock signaling, which play an important role in regulating the hair follicle cycle. 46 Alterations in GLP-1 activity induced by GLP-1 RA use could theoretically influence hair follicle cycling, however, direct evidence supporting this mechanism is currently lacking. 47 Collectively, these alterations may precipitate or exacerbate AGA in genetically or hormonally susceptible individuals.
Direct effects of GLP-1 RAs on hair follicle
Of yet there are no studies in humans to suggest that GLP-1 RAs have direct effects on hair follicles. GLP-1 receptors have been identified in the skin of newborn mice, particularly in regions surrounding hair follicles, where GLP-1 was found to activate mitogen-activated protein kinase (MAPK) pathways within skin-derived cells. 48 The presence of these receptors in the skin and hair follicles, although unclear whether true of humans also, suggests a possible effect of GLP-1 RAs within the hair follicle. Evidence indicates GLP-1 RAs in the activation of Wnt/β-catenin and IGF-1 signaling in other tissues,49,50 which if activating these pathways in hair follicles would in addition to activation of MAPK signaling promote hair growth rather than hair loss. GLP-1 receptor activation may also have anti-inflammatory effects and increase vascularity within the skin, with most research in this area focused on assessing liraglutide effects within the skin. 51 Further research is needed to clarify whether GLP-1 RAs exert direct effects within the hair follicle; current evidence remains limited but suggests a potential role in promoting hair growth rather than hair loss.
Timing and clinical pattern of hair loss
The temporal relationship between initiation of GLP-1 RA use and onset of hair loss is unclear, with minimal studies reporting this information. Ching et al., (2025) report the mean duration from GLP-1 RA initiation to new cases of alopecia being 1.75 years. 4 However, a shorter timeline of 1.5-3 months after starting treatment or increasing dosing would be expected if the hair loss is TE related to weight loss. 37 Delayed-onset hair loss may involve mechanisms distinct from those driving acute drug-induced hair loss. The relationship between onset of hair loss following treatment initiation and associated weight loss patterns as well as the type of hair loss caused with these varying factors needs to be clarified.
Clinical utility
From a clinical perspective, patients who are initiating or receiving GLP-1 RAs, particularly subcutaneous semaglutide and tirzepatide, should be counseled regarding the potential for diffuse, non-scarring hair loss, especially if they have a personal or family history of alopecia. Patients can be reassured that there is no strong evidence of permeant follicular damage associated with GLP-1 RA use, but reported cases most commonly resemble TE, which is typically self-limited and reversible. Shedding may occur with a delay of approximately 2-3 months after beginning treatment.
If hair loss occurs, initial clinical triage should distinguish between TE or AGA. TE is suggested by diffuse, generalized shedding, a positive hair-pull test and absence of patterned thinning. If shedding persists or if the patient has experienced rapid weight loss or restrictive caloric intake, a basic laboratory workup (i.e.,complete blood count, ferritin, thyroid stimulating hormone, vitamin D) may be considered to rule out other causes of hair loss or identify nutritional deficiencies.
Referral to dermatology is warranted if hair loss: • persists beyond 6-9 months • demonstrates patterned hair loss or rapid miniaturization • is associated with scalp itch or inflammation, suggests scarring alopecia • causes significant diagnostic uncertainty
In cases of severe shedding with significant distress, rapid excessive weight loss, or confirmed nutritional deficiency, dose adjustment or temporary hold of treatment may be considered on an individualized basis.23,29 Proactive patient education may help mitigate distress, support treatment adherence, and guide timely dermatologic evaluation when hair loss occurs.
Treatments for GLP-1 RA-induced hair loss
Treatment approaches depend on the type of hair loss. TE is typically reversible with improvements generally observed within 3-6 months following stabilization of weight. 37 Mitigation of underlying triggers is central to management and may prevent recurrent shedding episodes. Preventive strategies include avoiding dramatic weight loss and severe caloric restriction and ensuring proper nutritional intake, particularly adequate protein, iron, zinc, vitamin D, B12 and A.8,37 Gradual dose escalation of GLP-1 RAs may be considered to minimize rapid weight changes. Nutritional optimization and vitamin supplementation have been moderately successful in different TE cohorts,52,53 specifically with gastroparesis patients where hair loss incidence is high (46%) and individuals often have multiple nutritional deficiencies. 53 When the pattern of hair loss is consistent with AGA, standard AGA therapies such as topical minoxidil may be appropriate.
GLP-1 RAs and hair regrowth
Although limited, some literature indicates that GLP-1 RAs may promote hair growth in some cases. 54 By improving metabolic parameters and chronobiologic signaling, GLP-1RAs may support hair regrowth over the long-term, particularly in hair loss associated with metabolic syndrome, insulin resistance or polycystic ovarian syndrome. 54
For example, central centrifugal cicatricial alopecia pathogenesis is associated with metabolic dysfunction, and treatment with GLP-1 RAs has been associated with improvement of alopecia in 42% of cases. 55 Similarly, increased hair density has been reported alongside metabolic improvements following tirzepatide treatment for insulin resistance. 56 These effects are hypothesized to result from metabolic improvements that reduce scalp inflammation, as well as enhance insulin sensitivity, which may improve scalp blood flow and thereby support hair regrowth. 55
Limitations of current evidence
Several important limitations should be considered when interpreting the current evidence linking GLP-1 RAs to hair loss. Although some GLP-1 RA clinical trials have assessed hair loss events, this outcome is frequently not prespecified or systematically assessed, raising the possibility of underreporting. 26 Conversely, increasing public awareness of hair loss as a potential adverse effect in recent years may contribute to reporting bias, particularly in pharmacovigilance databases, where patients and clinicians may be more inclined to attribute hair shedding to GLP-1 RA therapy. Substantial heterogeneity exists across available studies. A recent meta-analysis reported an I2 statistic of 67.7%, suggesting moderate-to-high variability between studies. This heterogeneity likely reflects differences GLP-1 RAs, dosing regimens, population characteristics and study inclusion/exclusion criteria.
Importantly, the direct role of GLP-1 RAs in hair loss remains difficult to disentangle from downstream metabolic effects, particularly weight loss and associated nutritional deficiencies. Future studies should account for percent weight loss, rate of weight reduction and objective nutritional markers to clarify whether observed hair loss is a direct pharmacologic effect or a secondary metabolic consequence. Additionally, baseline history of hair loss, genetic predisposition for hair loss and concomitant medications are not consistently adjusted or controlled for. Further, caution should be taken when interpreting pharmacovigilance signals as they reflect disproportional reporting rather than true incidence and do not establish causality.
Reporting quality further limits interpretation. Many studies do not specify the type of hair loss, timing of onset relative to drug initiation, severity, or outcomes following drug discontinuation. Dermatology confirmation is rarely obtained. Understanding the onset and type of hair loss is instrumental in determining underlying cause, and managing hair loss. In addition, data remains sparse in adolescents as well as different racial or ethnic groups, limiting generalizability. Although several reports suggest higher incidence of GLP-1 RA-associated hair loss in females,5,33 it is unclear whether this reflects a true biological susceptibility or is driven by differential reporting patterns, as hair loss may be more readily noticed and reported by women than by men.
Conclusion
Although the reported incidence of non-scarring hair loss, particularly TE and AGA, varies widely across studies, the consistent observation of increased hair-loss risk across multiple cohorts, clinical trials and pharmacovigilance databases supports a likely association with GLP-1 RA use. The higher risk of hair loss observed with GLP-1 RAs that induce rapid weight loss, and among individuals who obtain a higher percentage weight reduction suggests an association between GLP-RA-induced hair loss and weight loss, likely influenced by both rate and magnitude of weight loss. Future studies should look to determine if metabolic stress, such as weight loss underlies the association between GLP-1 RA use and hair loss and if so what strategies could help to mitigate this (e.g. nutritional support). As GLP-1 RAs are increasingly prescribed for both approved and off-label metabolic indications, clinician awareness of this potential adverse effect is important for informed consent, early recognition and management.
Footnotes
Author contributions
Conceptualization, AKG; Supervision, AKG; Visualization, EMT; Writing – Original Draft Preparation, EMT; Writing – Review and Editing, AKG, PM and VE. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors AKG, EMT and VE declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article. PM reports research grants for conducting clinical trials from Amgen, Sun Pharmaceuticals, Eli Lilly, Incyte, and Pfizer; has served on the board of directors and medical advisory board for the American Hair Research Society, National Alopecia Areata Foundation, and Scarring Alopecia Foundation; and is a consultant for NeoGenesis.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
