Abstract
Objective
It is widely accepted that elevated low-density lipoprotein cholesterol (LDL-C) levels are a major risk factor for coronary heart disease (CHD). However, even patients who have achieved LDL-C levels below the currently recommended targets may still have residual risk. Recently, Lipoprotein(a) [Lp(a)] has attracted significant attention as independent causal risk factor for CHD. Nonetheless, the role of Lp(a) in acute coronary syndrome (ACS) patients with type 2 diabetes mellitus (T2DM) who have undergone percutaneous coronary intervention (PCI) and achieved the LDL-C target (≤1.4 mmol/L) remains unclear.
Methods
This retrospective study enrolled 462 ACS patients with comorbid T2DM who underwent PCI, with a median follow-up duration of 27 months post-procedure. The primary endpoint was major adverse cardiovascular events (MACE), defined as all-cause death, recurrent acute myocardial infarction (AMI), ischemic stroke, or hospitalization due to recurrent angina. Based on MACE occurrences, patients were divided into MACE group and non-MACE group. Furthermore, patients were further divided into three groups according to their Lp(a) levels. Kaplan–Meier and Cox regression analyses were performed.
Results
Patients with MACE had more coronary artery lesions, and the plasma Lp(a) concentrations in the MACE group were significantly higher than in the non-MACE group. The incidence of MACE and recurrent AMI was higher in the Lp(a) ≥ 180 mg/dL group compared to the other two groups. Even after multivariate adjustment, Lp(a) ≥ 180 mg/dL remained closely associated with an increased risk of MACE (HR 2.82, 95% CI: 1.47–5.41, P = .002) and recurrent AMI (HR 3.71, 95% CI: 1.17–11.81, P = .026).
Conclusion
Elevated Lp(a) levels were strongly associated with poor prognosis in ACS patients with T2DM who underwent PCI.
Keywords
Introduction
Despite significant advancements in medical therapies in recent years, acute coronary syndrome (ACS) remains a prevalent condition with high morbidity and mortality rates. Many clinical guidelines recognize type 2 diabetes mellitus (T2DM) as a major risk factor for coronary heart disease (CHD), and CHD is a common comorbidity in patients with prediabetes and diabetes.1,2 In patients with T2DM, the occurrence of major adverse cardiovascular events (MACE) after percutaneous coronary intervention (PCI) is significantly higher than in non-T2DM patients. Therefore, long-term secondary prevention following PCI is crucial for T2DM patients. Dyslipidemia plays a key role in the pathogenesis of ACS, and clinical guidelines recommend achieving a low-density lipoprotein cholesterol (LDL-C) level of < 1.8 mmol/L or a ≥ 50% reduction in LDL-C from baseline for ACS patients. 3 In recent years, lipid management requirements for patients with a medical history of ACS or T2DM have become increasingly stringent. The 2019 European Society of Cardiology (ESC)/European Atherosclerosis Society (EAS) Guidelines for the management of dyslipidemias recommend a ≥ 50% reduction in LDL-C from baseline and an LDL-C target of <1.4 mmol/L. 4 However, even patients who achieve LDL-C levels below the currently recommended targets may still experience cardiovascular events. 5 Therefore, it is crucial to identify new potential risk factors.
Lipoprotein(a) [Lp(a)] is an LDL-like particle composed of an apolipoprotein(a) moiety covalently bound to apolipoprotein B-100. Numerous studies have demonstrated that Lp(a) plays a key role in the development of atherosclerosis and increases the incidence of ACS.6–8 A high Lp(a) level may be associated with poor prognosis after PCI. 9 Other research has shown a strong correlation between Lp(a) and diabetes. 10 Jin et al. demonstrated that Lp(a) levels are closely linked to the presence and severity of stable CHD in individuals with diabetes. 11 Thus, Lp(a) levels may serve as an additional lipid target to optimize cardiovascular risk management in patients with ACS and T2DM who have undergone PCI and achieved their LDL-C goals. Elevated Lp(a) is an independent risk factor for atherosclerotic cardiovascular disease, and emerging evidence suggests that reducing Lp(a) levels could further decrease the incidence of MACE in this high-risk population. 12 Thus, beyond LDL-C control, targeting Lp(a) may provide a complementary strategy to improve long-term outcomes. However, few studies have analyzed the clinical significance of Lp(a) in diabetic patients with ACS who have undergone PCI. Therefore, in this retrospective analysis, we evaluated the impact of Lp(a) on long-term outcomes in diabetic patients with ACS who underwent PCI.
Methods
Study participants and endpoints
This retrospective study consecutively enrolled patients diagnosed with ACS and T2DM (age ≥18 years, complete lipid data) who received primary PCI treatment at the Cardiology Department of Qingdao University Affiliated Hospital from January 2020 to December 2023. This study was conducted in accordance with the ethical principles of the Declaration of Helsinki (2024 revision). ACS was defined as an ST-segment elevation myocardial infarction (STEMI)/left bundle branch block myocardial infarction, a non-ST-segment elevation myocardial infarction (NSTEMI) or unstable angina. Unstable angina is defined as a non-ST-elevation acute coronary syndrome that falls between stable angina and acute myocardial infarction, characterized by worsening anginal symptoms but without elevation of myocardial injury biomarkers. After excluding 368 patients, 462 patients were enrolled in this study. The exclusion criteria included patients (1) with LDL-C > 1.4 mmol/L and an LDL-C reduction of <50% from baseline at the 1-month follow-up; (2) those with severe liver or renal impairment; and (3) those with cancer, stroke, or valvular heart disease. Among the 368 excluded cases, 281 patients did not meet LDL-C criteria, 36 had severe liver impairment, 19 had severe renal impairment, 12 were diagnosed with cancer, 14 had a history of stroke, and 6 presented with valvular heart disease (Figure 1).

Flow diagram of patients enrollment and grouping.
All patients received either 180 mg ticagrelor or 300 mg clopidogrel, along with 300 mg of aspirin, before PCI. Unfractionated heparin was used for peri-procedural anticoagulation. Coronary angiography and PCI were performed by experienced physicians, and a stenosis of more than 75% was the indication for stent placement. After PCI, all patients received chronic antithrombotic therapy (100 mg of aspirin and 75 mg of clopidogrel or 180 mg of ticagrelor) along with statin treatment (20 mg of atorvastatin or 10 mg of rosuvastatin) for at least 12 months. Patients were followed for a median of 27 months. The primary endpoint was MACE, defined as all-cause death, recurrent acute myocardial infarction (AMI), ischemic stroke, or hospitalization due to recurrent angina attacks. Recurrent AMI is defined as a new myocardial infarction event occurring more than 28 days after the initial AMI. Ischemic stroke is defined as a clinical syndrome caused by focal cerebral ischemia lasting more than 5 minutes due to stenosis or occlusion of brain-supplying arteries, resulting in persistent neurological deficits (>24 hours), while excluding cerebral hemorrhage or other non-vascular etiologies. The patients were divided into two groups based on MACE occurrences: the MACE group and the non-MACE group. Additionally, we further categorized the patients into three groups according to their Lp(a) levels. The study was approved by the Ethics Committee of The Affiliated Hospital of Qingdao University (approval number: QYFY WZLL 28730, Qingdao, China; approval date: February 1, 2024) and the reporting of this study conforms to STROBE guidelines. 13 All the patients’ details have been de-identified.
Demographic and laboratory data
We recorded the clinical data of all subjects upon their arrival at the hospital, including sex, age, and the presence of hypertension and diabetes, as well as their history of smoking and alcohol consumption. Hypertension was diagnosed as a systolic blood pressure > 140 mmHg and/or diastolic blood pressure > 90 mmHg, or if the patient was using antihypertensive agents. T2DM was defined as current treatment for diabetes, a previous diagnosis of type 2 diabetes or a fasting plasma glucose concentration ≥ 126 mg/dL. All patients underwent fasting blood withdrawal before PCI. Lp(a), apolipoprotein B (ApoB), Apolipoprotein A-1 (ApoA1), total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), LDL-C, creatinine (Cr), uric acid (UA), and glycated hemoglobin (GHbA1) were analyzed using a Siemens Advia 2400 automatic biochemical analyzer in the biochemical laboratory of The Affiliated Hospital of Qingdao University.
Statistical analysis
All databases were analyzed using SPSS 22.0 or GraphPad Prism 9. Continuous variables are presented as mean ± SD for normally distributed data or as median with interquartile range (Q1, Q3) for non-normally distributed data. To compare symmetric and asymmetric continuous variables between two groups, independent-samples t-test (two-tailed) and Mann–Whitney U-test were applied, respectively. Categorical variables are expressed as numbers (percentages) and were compared using Chi-square tests or Fisher's exact test. Kaplan–Meier analysis was performed to compare event-free survival among groups, with differences assessed using the log-rank test. Cox proportional hazards regression models were employed to identify the association between Lp(a) levels and the risk of MACE. In this study, Lp(a) ≥ 180 mg/dL was used as the dependent variable in multiscale Cox analysis to evaluate whether Lp(a) ≥ 180 mg/dL affects MACE and recurrent AMI. Accordingly, multiscale Cox analysis were performed without adjustment (Model I), after adjusting for demographic characteristics (such as sex, BMI, and age) (Model II), and after adjusting for traditional coronary risk factors (TG, TC, LDL-C, HDL-C, Scr, and GHbA1c) (Model III). A two-tailed p-value of <0.05 was considered statistically significant.
Result
Demographic characteristics
Based on the occurrence of MACE, patients were divided into the MACE group and the non-MACE group. As shown in Table 1, patients with MACE had more coronary artery lesions, including multiple vascular lesions. Additionally, plasma Lp(a) concentrations in the MACE group were significantly higher than those in the non-MACE group. There were no significant differences in age, BMI, the proportion of hypertension, smoking status, type of ACS, or medication at discharge between the two groups. Furthermore, the levels of creatinine (Scr), triglycerides (TG), total cholesterol (TC), LDL-C, HDL-C, and the ApoB/ApoA1 ratio did not differ among the groups.
Characteristics of patients.

BMI: body mass index, UA: unstable angina, NSTEMI: non-ST-elevation myocardial infarction, STEMI: ST-elevation myocardial infarction, Scr: serum creatinine, UA: uric acid, TG: triglycerides, TC: total cholesterol, LDL-C: low-density lipoprotein cholesterol, HDL-C: high-density lipoprotein cholesterol, GHbA1: glycated hemoglobin, Lp(a): Lipoprotein(a), ApoB: apolipoprotein B, ApoA1: apolipoprotein A1, ACEI: angiotensin-converting enzyme inhibitor, ARB: angiotensin II receptor blocker.
Major adverse cardiovascular events
During the median follow-up of 27 months, 63 patients experienced MACE (1 died, 25 had reinfarction, 4 suffered a stroke, 18 were hospitalized for unstable angina pectoris (UAP), and 15 were hospitalized for heart failure (HF)). As shown in Figure 2, the Kaplan–Meier survival curves for MACE indicated that the Lp(a) ≥ 180 mg/dL group had a significantly higher incidence of MACE than the other two groups (P = .005). Similarly, the incidence of recurrent AMI in the Lp(a) ≥ 180 mg/dL group was higher than in the other groups (P = .023). However, the prevalence of adverse events (stroke, HF, UAP, and all-cause deaths) did not differ among the three groups.

The Kaplan–Meier survival curves analysis of MACE (A) and recurrent AMI (B).
In multiscale Cox analysis (Table 2), Lp(a) ≥ 180 mg/dL and a per 1-standard deviation (1-SD) increase in ln[Lp(a)] were closely associated with an increased risk of MACE (for Lp(a) ≥ 180 mg/dL, unadjusted HR 2.65, 95% CI: 1.49–4.93, P = .002; for each 1-SD increase in ln[Lp(a)], unadjusted HR 1.60, 95% CI: 1.27–2.03, P < .01) and recurrent AMI (for Lp(a) ≥ 180 mg/dL, unadjusted HR 3.72, 95% CI: 1.23–11.32, P = .021; for each 1-SD increase in ln[Lp(a)], unadjusted HR 1.69, 95% CI: 1.16–2.45, P = .006). After adjusting for gender, BMI, age, and hypertension, Lp(a) ≥ 180 mg/dL and a per 1-SD increase in ln[Lp(a)] remained independently associated with the occurrence of MACE and recurrent AMI (Figure 3). Similar results were observed in Model II, which included further adjustments for baseline levels of TG, TC, LDL-C, HDL-C, Scr, and GHbA1. Moreover, after additional adjustments for medication use in Model III, the results indicated that patients in the Lp(a) ≥ 180 mg/dL group had a 2.82-fold increased risk of MACE (95% CI: 1.47–5.41, P = .002) and a 3.71-fold increased risk of recurrent AMI (95% CI: 1.17–11.81, P = .026). Additionally, each 1-SD increase in ln[Lp(a)] was associated with a 67% increased risk of MACE (95% CI: 1.29–2.17, P < .01) and an 87% increased risk of recurrent AMI (95% CI: 1.20–2.91, P = .005).

Predictors for recurrent MACE (A) and recurrent AMI (B).
Cox proportional-hazards model of risk of development of MACE and recurrent AMI.
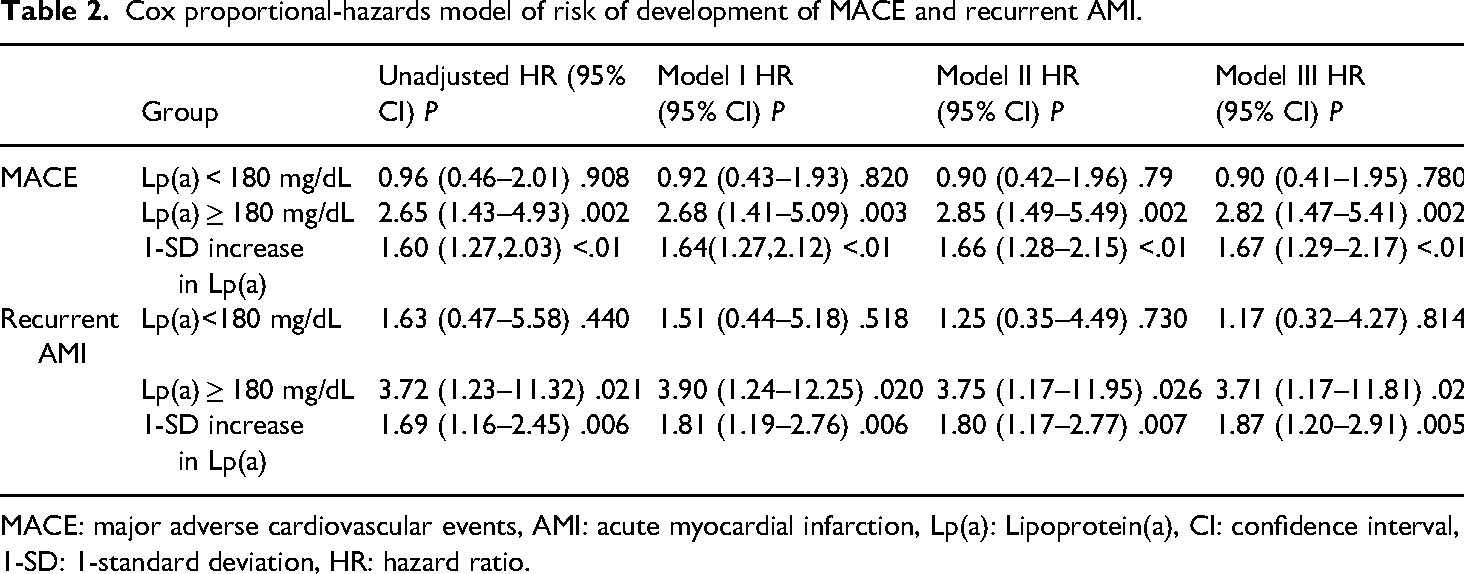
MACE: major adverse cardiovascular events, AMI: acute myocardial infarction, Lp(a): Lipoprotein(a), CI: confidence interval, 1-SD: 1-standard deviation, HR: hazard ratio.
Discussion
As an independent risk factor for CHD, T2DM is a common comorbidity in patients with ACS. In patients with ACS and coexisting T2DM, persistent chronic hyperglycemia damages the blood vessel walls and endothelial cells. The action of various inflammatory cytokines accelerates the formation of atherosclerotic plaques, ultimately leading to the occurrence and progression of CHD and other atherosclerotic lesions. Insulin resistance plays a key role in the development of T2DM and can also result in abnormal lipid metabolism. It is well established that hyperlipidemia also serves as an independent risk factor for ACS, and the coexistence of these two conditions further deteriorates the prognosis for ACS patients. Therefore, there is a more stringent lipid-lowering target for patients at high risk. 14 As we all know, LDL-C is an important risk factor for the occurrence and progression of ACS. Recent guidelines have explicitly recommended target LDL-C values. However, several studies indicate that despite effective control of various risk factors, including LDL-C, blood pressure, and glucose levels, patients who survive ACS remain at increased risk of recurrent MACE, especially those with T2DM.15,16 Therefore, it is essential to consider other components to improve the prognosis for these patients.
Since the plasma level of Lp(a) is primarily determined by the LPA gene, it is minimally influenced by diet, exercise, or environmental factors. 17 Elevated Lp(a)] in T2DM patients promotes MACE through multifaceted mechanisms: (1) Lp(a) induces endothelial cells to release adhesion molecules (VCAM-1, ICAM-1) and pro-inflammatory chemokines (MCP-1), thereby amplifying inflammatory responses 18 ; (2) Lp(a) exhibits antifibrinolytic properties and inactivates tissue factor pathway inhibitor (TFPI), thereby promoting a prothrombotic state 19 ; (3) Lp(a) aggravates insulin resistance (IR) via oxidative stress mechanisms, and reciprocally, hyperinsulinemia upregulates the hepatic synthesis of Lp(a). 20 Recent studies have demonstrated that elevated levels of Lp(a) are an independent risk factor for CHD, and the association between Lp(a) and cardiovascular outcomes in patients with CHD has also been confirmed.21,22 The Copenhagen City Heart Study demonstrated that compared to patients with Lp(a) levels below 5 mg/dL, those with Lp(a) levels between 30 and 76 mg/dL had a 1.6-fold increased risk of incident AMI. This risk rose to 1.90 for individuals with Lp(a) levels between 77 and 117 mg/dL and increased further to 2.60 for individuals with Lp(a) concentrations above 117 mg/dL. 22 However, these studies primarily involved patients who did not have CHD at baseline. Schwartz et al. recruited 969 patients with recent ACS who were receiving statin therapy, and they found that Lp(a) concentration was not associated with adverse cardiovascular events (CVEs). 23 Since the patients in this study were derived from the dal-Outcomes randomized clinical trial, the findings may be influenced by the cholesteryl ester transfer protein inhibitor dalcetrapib. Additionally, Ren et al. have pointed out that plasma Lp(a) levels are related to the prevalence of MACE in patients who underwent PCI. 24 Study results demonstrated that occurrence of major adverse cardiovascular events (MACE) and recurrent myocardial infarction (MI) increased with the Lp(a) increasing in patients with LDL-C ≤ 1.8 mmol/L at 1-month follow-up. Furthermore, it has been shown that Lp(a) is independently associated with the severity of CHD in individuals with T2DM. Saeed et al. reported that elevated Lp(a) levels are predictive of CHD risk in apparently healthy Caucasian individuals with T2DM or pre-T2DM. 25 The study conducted by Jin et al. further suggested that high Lp(a) levels are associated with a significantly higher risk of subsequent CVEs in individuals with pre-T2DM and T2DM. Patients with both Lp(a) levels greater than 50 mg/dL and T2DM had a 3.47-fold higher risk of CVEs. 26
In this study, the baseline Lp(a) concentration was significantly higher in the MACE group compared to the non-MACE group. Further subgroup analysis indicated that plasma Lp(a) levels remained independently associated with the prevalence of MACE and recurrent AMI in patients with T2DM who achieved the LDL-C target after statin therapy. Thus, Lp(a) level monitoring is warranted in T2DM patients after PCI. Emerging evidence shows PCSK9 inhibitors lower Lp(a), correlating with better prognosis.27,28
The study also has some limitations. Firstly, it was a retrospective, small-sample, single-center study, which means that some confounding factors are inevitable. Secondly, Lp(a) was measured only at baseline, and subsequent Lp(a) levels may also hold clinical significance. Thirdly, while newer drugs like sacubitril/valsartan and vericiguat could influence MACE outcomes, data on these medications were not available for analysis in our study due to institutional constraints.
Conclusion
In summary, the findings of this study suggest that Lp(a) ≥ 180 mg/dL may serve as an independent risk factor for MACE and recurrent AMI in T2DM patients after PCI. Therefore, Lp(a) could potentially become a crucial biomarker for risk assessment in post-PCI patients and provide a foundation for future targeted intervention strategies in individuals with elevated Lp(a) levels.
Footnotes
Abbreviations
Acknowledgments
None.
Ethical approval
This study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. The study was approved by the Ethics Committee of The Affiliated Hospital of Qingdao University (approval number: QYFY WZLL 28730, Qingdao, China).
Author contributions
Zeyu Jiang: methodology, writing—original draft, and writing—review and editing. Shimiao Ruan: writing—original draft and data curation. Qingqing Duan: formal analysis. Wenzhong Zhang: conceptualization and supervision. Shuhan Pan: writing—review and editing, supervision, and project administration.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets utilized and/or examined in this study can be obtained from the corresponding author upon a reasonable request.
