Abstract
Objective
This study evaluated the hepatotoxicity risk of anaplastic lymphoma kinase (ALK)/ROS1- tyrosine kinase inhibitors (TKIs) in non-small cell lung cancer (NSCLC) using the Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS).
Methods
A retrospective analysis of 19,611 FAERS reports related to ALK/ROS1-TKIs (2011–2025) identified 1495 hepatotoxicity cases. Disproportionality analysis (reporting odds ratio, proportional reporting ratio, empirical Bayes geometric mean) assessed six ALK-TKIs and three ROS1-TKIs. Temporal trends, clinical outcomes, and differences across TKI generations were examined.
Results
Second-generation ALK-TKIs (e.g. alectinib) showed the most favorable hepatic safety profile, with the lowest observed mortality (3.5%) and hospitalization (13.0%). First-generation TKIs (crizotinib, ceritinib) and newer agents (lorlatinib, entrectinib) were associated with higher fatality rates (12.0%–13.2%). Most hepatotoxic events (51.9%) occurred within one month; however, 8.37% developed after one year. Mechanistically, the lower risk with second-generation TKIs may reflect less CYP-dependent metabolism and greater target selectivity.
Conclusion
Analysis of FAERS reports indicates substantial heterogeneity in hepatotoxicity risk among ALK inhibitors. Second-generation agents showed lower mortality and hospitalization than first- and third-generation counterparts. Crizotinib, ceritinib, and lorlatinib were associated with notably high fatality rates, warranting continuous hepatic monitoring. ROS1 inhibitors (e.g. entrectinib) may exhibit distinct hepatotoxicity profiles. These findings should be interpreted cautiously given inherent reporting biases. Future priorities include prospective studies, biomarker-integrated risk stratification, mechanistic investigation, and AI-enhanced monitoring to optimize the risk–benefit profile of ALK/ROS1-TKIs.
Introduction
Lung cancer remains a major global health burden. According to the International Agency for Research on Cancer, lung cancer is the leading cause of cancer-related mortality worldwide. 1 With over two million new cases diagnosed annually, non-small cell lung cancer (NSCLC) accounts for approximately 80–85% of all lung cancer cases. 1 Major NSCLC subtypes include adenocarcinoma, squamous cell carcinoma, and large-cell neuroendocrine carcinoma. Over recent decades, NSCLC treatment has shifted from surgery, chemotherapy, and antiangiogenic therapy to immunotherapy and targeted therapies, which are now central to precision medicine. Notably, anaplastic lymphoma kinase (ALK) fusion–positive (ALK-rearranged) NSCLC represents a clinically important and therapeutically actionable molecular subtype with direct implications for treatment selection. It occurs in approximately 3–7% of NSCLC overall and 6.6–9.6% of lung adenocarcinomas. 2 ROS1 fusions are enriched in younger patients who are never or light smokers. 3 Structurally, ROS1 shares substantial sequence similarity with ALK, with 49% identity in the kinase domain and 77% identity at the ATP-binding site. High conservation within the kinase domain and ATP-binding site may partly explain the activity of many ALK inhibitors against ROS1, including inhibition of ROS1-positive cell proliferation and clinical responses in ROS1 fusion–positive NSCLC.4,5 To date, the U.S. Food and Drug Administration (FDA) has approved six ALK tyrosine-kinase inhibitors (ALK TKIs) for ALK-positive NSCLC and three ROS1-TKIs for ROS1-positive disease as standard therapies. Crizotinib, the first-in-class ALK kinase inhibitor, is also the inaugural first-generation TKI approved for dual targeting of both ALK and ROS1.6–8 Its antitumor effect results from inhibition of ALK and ROS1 kinase activity, which blocks oncogenic signaling and suppresses tumor-cell proliferation. Compared to chemotherapy, targeted therapies significantly improve outcomes in advanced ALK-positive NSCLC patients, regardless of prior chemotherapy exposure.9,10 However, the increased incidence of adverse effects—particularly severe hepatotoxicity associated with ALK-TKIs, which often leads to elevated mortality rates11,12—and the emergence of drug resistance have driven the development of second- and third-generation TKIs. Although hepatotoxicity has been reported in clinical trials for these agents,13–17 a comprehensive real-world comparison of liver safety profiles among NSCLC patients receiving ALK/ROS1-TKIs remains lacking. Real-world data serve as a critical complement to conventional clinical trial findings, offering insights into long-term safety and heterogeneous patient populations.
The U.S. FDA Adverse Event Reporting System (FAERS), one of the world's largest pharmacovigilance databases, enables the detection of potential risk signals through post-marketing spontaneous reports of adverse events (AEs). 18 Previous studies leveraging FAERS have successfully identified TKI-associated cardiotoxicity 19 and interstitial lung disease signals,20,21 validating its utility in drug safety research. However, data mining focused on hepatotoxicity of ALK/ROS1-TKIs in NSCLC remains unexplored in FAERS. This study represents the first systematic investigation of hepatotoxicity signals for six ALK-TKIs (crizotinib, ceritinib, alectinib, brigatinib, lorlatinib, ensartinib) and three ROS1-TKIs (crizotinib, entrectinib, repotrectinib) using the FAERS database. Multiple signal detection methodologies—including the reporting odds ratio (ROR) and Bayesian confidence propagation neural network (BCPNN)—were employed to quantify risks. By analyzing temporal patterns, clinical outcomes, and inter-drug disparities of hepatotoxic events, this research aims to inform clinical decision-making, optimize liver function monitoring protocols, and stratify high-risk patients. Furthermore, it elucidates real-world characteristics and challenges associated with ALK/ROS1-TKI-induced hepatotoxicity.
Methods
Data sources
Quarterly AE reports were retrospectively retrieved from FAERS for first quarter (Q1) 2011 through Q1 2025. FAERS is a U.S. pharmacovigilance repository that documents drug-related AEs.22,23 Reports are submitted by manufacturers, healthcare professionals, and consumers, and are supplemented by postmarketing clinical-trial data from domestic and international sources. The database encompasses multiple structured categories: demographic and administrative information (DEMO), drug-specific details (DRUG), reported adverse events (REAC), patient outcomes (OUTC), reporting sources (RPSR), drug therapy initiation/cessation dates (THER), indications (INDI), and deleted cases. The investigated agents included crizotinib, ceritinib, alectinib, brigatinib, lorlatinib, ensartinib, entrectinib, and repotrectinib. Target drugs were designated as the primary suspect (PS) in all analyses. AEs were codified using the Medical Dictionary for Regulatory Activities (MedDRA) at the Preferred Term (PT) level. This study was conducted entirely in silico (i.e. a “dry lab” computational study) using the FAERS database and standard pharmacovigilance signal-detection algorithms, consistent with best practices for computer-based analyses. 24
Data processing
Data packages in ASCII format downloaded from the FAERS database were imported into R (version 4.4.2) for integration, cleaning, and standardization. Duplicate reports in the DEMO table were deduplicated following FDA-recommended deletion criteria, 25 and the primaryid field was used to map and deduplicate records across six additional tables (e.g. DRUG). Reports with misspelled drug names, duplicates, or unrelated AEs were excluded. AEs were standardized using PTs from the MedDRA, version 27.1 and categorized by System Organ Class (SOC). 26 Drug names were compiled from FDA-approved listings, including generic and brand names, to query the drugname and prod_ai fields in the DRUG table. Only entries with role_cod (drug role code) designated as “PS” were retained. Clinical features of patients experiencing AEs—including age, sex, reporter type, geographic origin, and outcomes—were extracted to comprehensively characterize the safety profiles of each drug. To identify NSCLC-related reports, cases with “non-small cell lung cancer” in the FAERS “indication” field were included, while those with ambiguous or unrelated indications were excluded. For hepatotoxicity analysis, the REAC dataset was standardized prior to drug-specific queries. A predefined lexicon of liver injury PTs derived from MedDRA was applied (Table S2). 27 A flowchart summarizing the analytical workflow is provided in Figure S1. All data integration, deduplication, and signal computations were performed in silico using scripted pipelines, without wet-lab experimentation.
Statistical analysis
Quantitative and qualitative analyses were performed on clinical characteristics of ALK/ROS1-TKI-associated hepatotoxicity cases retrieved from the FAERS database. Signal detection was conducted using established disproportionality metrics: the proportional reporting ratio (PRR), ROR, BCPNN, and Multi-item Gamma Poisson Shrinker (MGPS).28–32 MGPS employed an empirical Bayesian model to generate adjusted observed-to-expected ratios, reducing false-positive ROR inflation. RORs were calculated using 2 × 2 contingency tables incorporating drug-event co-occurrences, drug-only reports, event-only reports, and unrelated cases. In this study, we applied a case–noncase reference framework within FAERS to construct 2 × 2 contingency tables with cells defined as: a = target drug + hepatotoxicity terms; b = target drug + non-hepatotoxicity terms; c = other drugs + hepatotoxicity terms; and d = other drugs + non-hepatotoxicity terms. The distribution of the same event among all other drugs served as the background control. All analyses were performed after FDA-recommended de-duplication and data cleaning. In the primary analysis, we preferentially retained reports in which the drug was designated the PS and, when possible, restricted the indication to NSCLC to reduce confounding. Signal detection thresholds were defined as follows. Signal thresholds were defined as ROR [95% confidence interval (CI) lower bound >1 with ≥3 total reports] or information component (IC)-based metrics (IC > 0 and IC–2SD > 0). 33 A positive PRR required PRR ≥ 2, χ2 ≥ 4, and n ≥ 3. For the BCPNN, IC_025 > 0 was considered positive; for the MGPS, a positive signal required an empirical Bayes geometric mean (EBGM) lower bound EB05 > 1. To mitigate small-sample inflation and sparse cells, we prioritized conservative lower-bound indices (IC_025 and EB05) and interpreted signals based on concordance across methods. Accuracy was evaluated in three ways: (i) agreement across algorithms; (ii) benchmarking against established hepatotoxicity evidence from prior literature and product labeling to assess face validity; and (iii) sensitivity analyses, with results compared with the primary analysis for consistency. PRR and ROR demonstrated robust performance in identifying drug-event risk associations. Analyses were performed in R (v4.4.2), with statistical significance set at P < 0.05.
Results
Descriptive analysis
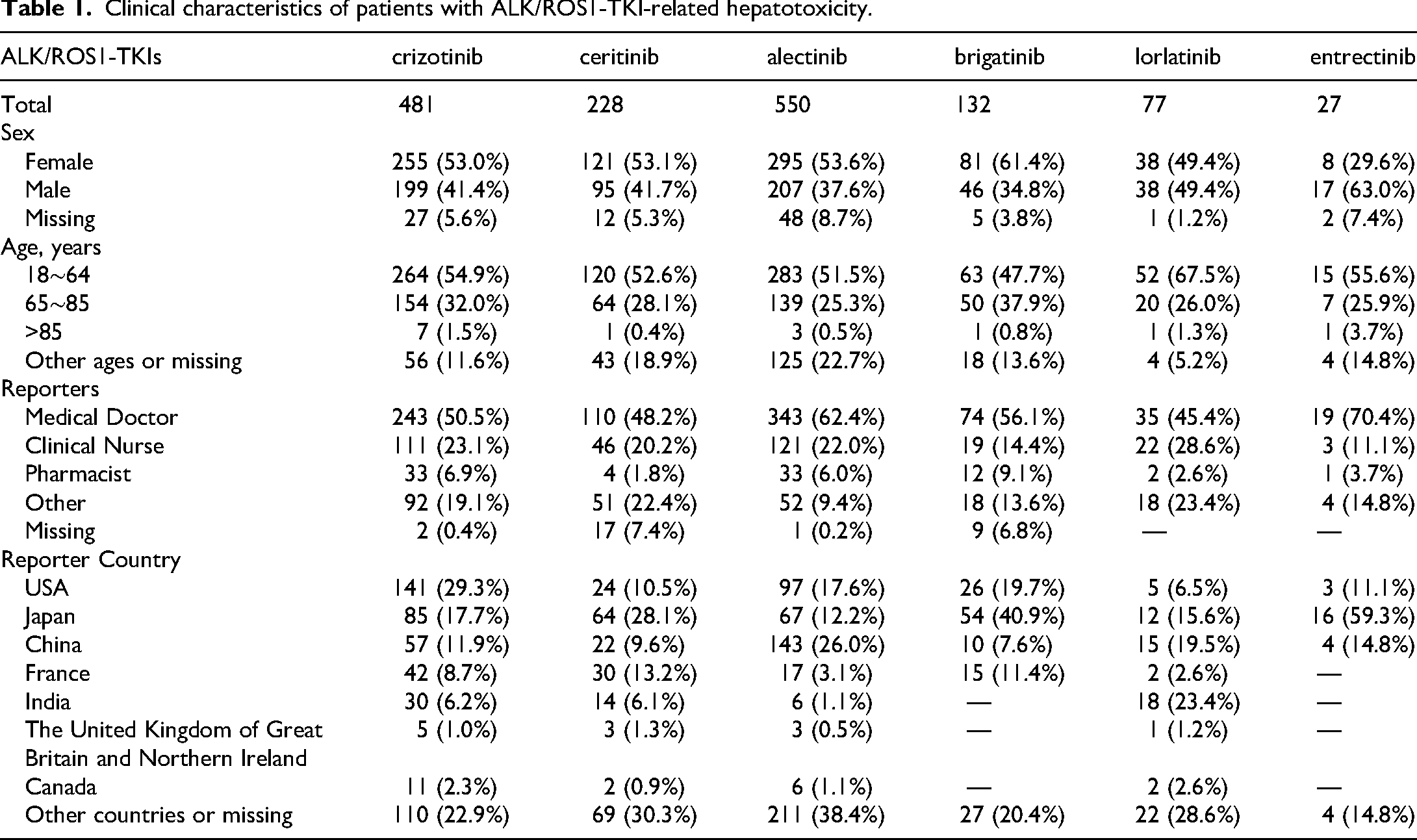
Following deduplication in accordance with FDA recommendations, the FAERS database yielded 19,611 AE reports associated with ALK/ROS1 TKI therapy in NSCLC patients from Q1 2011 to Q1 2025. Of these, 1495 reports of hepatotoxicity post-ALK/ROS1-TKI treatment were identified for further analysis (Figure S1). Drug-specific hepatotoxicity signals were distributed as follows: 481 cases (7.67%) among 6270 reports listing crizotinib as the PS, 550 cases (8.66%) among 6349 alectinib reports, 228 cases (13.28%) among 1716 ceritinib reports, 132 cases (5.91%) among 2232 brigatinib reports, 77 cases (3.80%) among 2027 lorlatinib reports, and 27 cases (3.28%) among 822 entrectinib reports. No hepatotoxicity signals were detected for ensartinib or repotrectinib. Demographic characteristics of the cohort are detailed in Table 1. Among the 1495 hepatotoxicity cases, males accounted for 53.38%, females 40.27%, and patients with unknown/missing age data 6.35%. The 18–64 age group exhibited the highest incidence of ALK/ROS1-TKI-associated hepatotoxicity, representing 49.97% of reported cases. Clinicians constituted the majority of reporters, while pharmacists submitted only 6.9% of cases, highlighting potential profession-based discrepancies in severity assessment (e.g. clinicians prioritized laboratory abnormalities, while nurses focused on symptomatic manifestations). Geographically, reports originated predominantly from the United States (20.47%) and Japan (19.93%).
Clinical characteristics of patients with ALK/ROS1-TKI-related hepatotoxicity.
Disproportionality analysis of hepatotoxicity signals
Disproportionality analysis revealed significant hepatotoxicity signals across ALK/ROS1-TKIs. Alectinib had the highest number of hepatotoxicity reports (N = 550), while entrectinib had the fewest (N = 27). Crizotinib demonstrated a ROR of 1.55 (95% CI, 1.43–1.68) based on 481 reports, ceritinib showed an ROR of 2.19 (95% CI, 1.95–2.46) from 228 reports, and alectinib exhibited the strongest signal (ROR = 2.43, 95% CI, 2.25–2.63) with 550 reports. Disproportionality metrics for all agents—including ROR, PRR, IC, and EBGM—are comprehensively summarized in Table 2.
Association between ALK/ROS1-TKIs and hepatotoxicity.

95% CI: 95% confidence interval; χ2: chi-squared; IC: information component; ROR: reporting odds ratio; PRR: proportional reporting ratio; EBGM: empirical Bayesian geometric mean.
Sensitivity analysis
In sensitivity analyses excluding concomitant medications, drug-event pairs classified as positive in the primary analysis remained positive across ROR, PRR, IC, and EBGM in the PS monotherapy dataset, with concordant directionality and greater magnitudes than in analyses including co-medication (see Supplemental Table S3). Effect sizes increased for several PTs, and cross-method concordance was unchanged. These findings indicate that the detected signals are robust and not dependent on the composition of concomitant therapies, consistent with recent FAERS studies showing similarly robust results after excluding combination treatment.34,35
Analysis of hepatotoxicity onset timing
As illustrated in Figure 1, we analyzed the temporal distribution of ALK/ROS1-TKI-associated hepatotoxicity cases. After deduplication, 765 reports with precise onset dates were included. The median time to hepatotoxicity onset was 29 days. Overall, 51.9% of cases (397/765) occurred within the first month after treatment initiation, whereas reporting frequency was lowest during months 2–3. Additionally, 8.37% of cases occurred more than one year after treatment initiation. These findings support vigilant monitoring for hepatotoxicity throughout the treatment course, extending beyond the initial months of ALK/ROS1-TKI therapy.

Time to onset of ALK/ROS1-TKI-related hepatotoxicity.
Analysis of hepatotoxicity-related outcomes
As detailed in Table 3, we analyzed 1910 hepatotoxicity outcomes from non-unique cases (multiple outcomes per patient permitted). Significant heterogeneity in hepatotoxicity severity was observed across ALK/ROS1-TKIs. First-generation agents crizotinib (12.0%, 80/668) and ceritinib (13.1%, 43/329) exhibited the highest mortality rates, while second-generation inhibitors alectinib (3.5%) and brigatinib (4.0%) demonstrated the lowest lethality. Notably, lorlatinib, despite limited sample size (N = 106), showed a mortality rate of 13.2%, warranting heightened vigilance. Hospitalization rates were highest with entrectinib (35.3%), followed by brigatinib (31.8%) and lorlatinib (26.4%). Alectinib displayed optimal safety profiles, with the lowest hospitalization (13.0%) and life-threatening event rates (1.0%). Disabling outcomes were rare (<2.0% across all agents), with crizotinib (1.0%) and alectinib (0.7%) showing minimal risks. Data completeness analysis revealed substantial missing outcomes for alectinib (47.2%), potentially attributable to its recent approval and reporting delays, whereas other agents had <18% missing data (lorlatinib: 0.9%). While multiple imputation addressed missingness, alectinib's high missingness may introduce residual bias. These findings suggest that second-generation ALK inhibitors (e.g. alectinib, brigatinib) may confer advantages in reducing hepatotoxicity-related mortality and hospitalization, while first-generation agents (crizotinib, ceritinib) and select newer TKIs (lorlatinib, entrectinib) require cautious monitoring for severe hepatic outcomes. Prospective studies are warranted to validate these signals.
Outcomes due to ALK/ROS1-TKI-related hepatotoxicity.

Discussion
To our knowledge, this is the first systematic evaluation of hepatotoxicity risk among ALK/ROS1-TKIs in patients with NSCLC using the FAERS database. We observed substantial heterogeneity in hepatotoxicity incidence and clinical outcomes across TKI generations. Second-generation agents (e.g. alectinib) showed a more favorable safety profile, with lower mortality (3.5%) and hospitalization (13.0%) than first-generation TKIs (crizotinib, ceritinib) and selected newer agents (lorlatinib, entrectinib). These findings are consistent with previous studies,36,37 which attribute the improved hepatic safety of second-generation ALK-TKIs to optimized molecular design and less CYP-dependent metabolism that may reduce hepatic oxidative stress. In real-world settings, rates of severe hepatotoxic events were higher than those reported in clinical trials. 15 This discrepancy likely stems from stringent trial enrollment criteria (e.g. exclusion of patients with hepatic impairment) and the broader comorbidity profiles and polypharmacy patterns observed in real-world populations. These results support the need for robust postmarketing surveillance to fully characterize drug safety, particularly for therapies with narrow therapeutic indices.
A predominance of AE reports among female patients was observed (Table S1), consistent with established sex-based differences in drug-related toxicities across therapeutic classes.38,39 This pattern may reflect hormone-mediated modulation of drug transporters, receptors, and drug-metabolizing enzymes.
In FAERS, we observed a relatively strong hepatotoxicity signal for alectinib (ROR = 2.43), indicating the need for heightened liver-function monitoring in real-world practice. However, when clinical severity outcomes were compared (Table 3), mortality (3.5%) and hospitalization (13.0%) associated with alectinib were lower than those for crizotinib (12.0% and 26.9%, respectively) and lorlatinib (13.2% and 26.4%, respectively), suggesting a more favorable profile for severe outcomes. Furthermore, updated safety data from the ALEX trial 40 reported alanine aminotransferase (ALT) elevations in 17.1% and aspartate aminotransferase (AST) elevations in 15.8% of patients receiving alectinib, with grade 3–5 ALT and AST elevations of 4.6% and 5.3%, respectively. Although transaminase elevation was the most common AE leading to treatment discontinuation, discontinuation rates were lower with alectinib than with crizotinib (e.g. due to ALT elevation: 1.3% vs 6.0%; due to AST elevation: 1.3% vs 4.0%; due to pneumonitis/interstitial lung disease: 0.7% vs 3.3%). These observations underscore that the “strength” of an ROR signal does not correspond one-to-one with toxicity grade, clinical severity, or the risk of treatment discontinuation.
ROR reflects the degree of disproportionality in spontaneous reporting for a given drug–event pair and is influenced by factors such as the size of the exposed population, duration of therapy, intensity of laboratory surveillance, labeling or media warnings, and reporting behavior. It is not equivalent to a true incidence rate and does not discriminate toxicity grades or clinical severity. Accordingly, a high ROR identifies a signal warranting attention but cannot, by itself, adjudicate comparative clinical safety. An additional limitation is that FAERS does not include Criteria for Adverse Events (CTCAE) grading, which precludes direct comparisons of grade 3–4 severe hepatotoxicity; therefore, we used severe outcomes (death, hospitalization, life-threatening events) as proxy indicators of real-world severity.
Mechanistically, ceritinib (second-generation ALK-TKI) is primarily metabolized by CYP3A enzymes, whereas alectinib exhibits reduced CYP dependency, favoring non-enzymatic pathways such as glucuronidation. This metabolic divergence likely mitigates hepatic damage by limiting the generation of reactive metabolites. Second-generation TKIs demonstrate superior target specificity, with alectinib showing 10-fold higher selectivity for ALK over off-target kinases (e.g. ROS1, IGF-1R), thereby reducing hepatocyte stress from off-target effects. Enhanced central nervous system penetration in this class may lower systemic drug exposure, particularly hepatic concentrations. 41 Emerging evidence suggests second-generation agents may attenuate oxidative stress via ferroptosis-related pathways, including NQO1-mediated reduction of lipid peroxides.42,43 Notably, third-generation lorlatinib exhibited mortality rates (13.2%) comparable to first-generation TKIs, potentially attributable to increased systemic exposure due to enhanced blood-brain barrier penetration,44,45 underscoring the need for dose optimization to prevent concentration-dependent hepatotoxicity. Temporal analysis aligned with TKI pharmacokinetics: 51.9% of hepatotoxicity cases occurred within the first treatment month, reflecting early metabolic adaptation phases. Notably, 8.37% of hepatotoxic events occurred after one year of treatment, challenging the conventional paradigm that TKI-induced hepatotoxicity predominantly manifests during early treatment phases.
Although ROS1 shares homology with ALK, approved ROS1 inhibitors display distinct target selectivity and pharmacokinetic properties. For instance, entrectinib not only inhibits ROS1 but also effectively suppresses TRKA/B/C, 46 whereas repotrectinib utilizes a unique macrocyclic structure and binding mode to circumvent resistance mutations. 47 These differences in target engagement and off-target effects likely contribute to divergent safety profiles (including hepatotoxicity risk) warranting prospective multicenter studies to assess their hepatic risks independently. Moreover, ROS1 inhibitors exhibit varying metabolic pathways: entrectinib is predominantly metabolized by CYP3A4, 46 unlike some ALK inhibitors (e.g. alectinib), which rely minimally on CYP-mediated metabolism. Differences in metabolic activation mechanisms and the propensity to generate reactive metabolites are key factors influencing idiosyncratic liver injury risk. Our stratified analysis reveals notable differences: compared to the limited data available for ROS1 inhibitors, ALK inhibitors—particularly first-generation agents—typically exhibit stronger hepatotoxicity signals and higher mortality rates. Although entrectinib demonstrates a lower mortality rate, its hospitalization rate is significantly elevated. While the relatively small number of reported ROS1 inhibitor cases precludes definitive conclusions regarding class-wide differences, these observed variations underscore that the safety profiles of ALK inhibitors cannot be assumed to mirror those of ROS1 inhibitors.
Through systematic comparative analysis of label warnings and clinical monitoring parameters for ALK/ROS1-TKIs, this study revealed marked heterogeneity in hepatotoxicity risk profiles across agents. Leveraging pharmacovigilance data from the FAERS, our findings provide evidence-based guidance for tailoring individualized therapeutic regimens. The study further substantiates that implementing dynamic risk assessment algorithms, combined with therapeutic drug monitoring and hepatic function stratification strategies, enables precise modulation of benefit-risk ratios in targeted cancer therapy. This biomarker-informed risk stratification framework establishes a novel paradigm for optimizing longitudinal management in advanced NSCLC patients. 37
This study has several limitations: First, the voluntary reporting nature of the FAERS database may introduce underreporting bias, potentially underestimating the true incidence of hepatotoxicity. Second, FAERS does not include CTCAE grading, precluding direct comparison of grade 3–4 severe hepatotoxicity. Third, high rates of missing data for certain agents—particularly ROS1-TKIs—may compromise interpretation. The markedly lower hepatotoxicity reports for ROS1-TKIs compared to ALK-TKIs (Table S2) likely reflect the rare incidence of ROS1 fusions in NSCLC (1%–2%) and limited real-world data availability due to fewer approved agents (e.g. repotrectinib) and recent market entry (data cutoff: Q1 2025). The limited sample size for ROS1-TKIs reduces statistical power in signal detection, precluding robust conclusions akin to those for ALK-TKIs. Future efforts should extend data collection periods and integrate post-marketing surveillance data for newer ROS1-TKIs to refine safety profiles. Fourth, Pooling ALK/ROS1-TKIs was intended to improve the efficiency of signal detection; it should not be interpreted as treating patients with ALK rearrangements and those with ROS1 rearrangements as a single clinical population. In particular, given the temporal shift in crizotinib's labeled use over the study period (initially for ALK and now primarily for ROS1), we analyzed signals at both the individual-drug level and the target-class level to mitigate potential confounding. Nonetheless, residual heterogeneity may persist. Prospective multicenter studies are warranted to validate these pharmacovigilance signals, coupled with mechanistic investigations of novel serological biomarkers (e.g. extracellular vesicle-derived miRNAs or ferroptosis-related proteins) for early hepatotoxicity prediction.
To address the limitations of pharmacovigilance databases and advance clinical translation, future research should focus on three key domains. First, prospective multicenter studies incorporating baseline liver function, pharmacogenomic profiling (e.g. CYP3A4/UGT1A1 polymorphisms), and dynamic multi-omics monitoring are critical to validate FAERS-derived hepatotoxicity signals while controlling for confounders like comedications and preexisting liver disease. Second, mechanistic investigations using hepatocyte organoids and animal models should elucidate TKI-specific impacts on mitochondrial dysfunction (assessed via oxygen consumption rate), ferroptosis pathways (NQO1/GPX4 activity), and oxidative stress biomarkers (e.g. lipid peroxidation products), complemented by serum/exosome-based biomarker discovery (miR-122, cytokeratin-18) for early injury detection. Third, longitudinal cohorts must refine risk stratification algorithms integrating Child-Pugh scores, early ALT/AST trends, and pharmacogenetic data to enable AI-driven surveillance systems, while PK/PD-guided therapeutic drug monitoring—particularly for CYP3A4 ultrarapid metabolizers or hypoalbuminemia patients—could optimize dosing strategies and mitigate late-onset hepatotoxicity (8.37% incidence beyond 1 year; Table S2). These interdisciplinary approaches, synergizing real-world evidence with experimental validation, will enhance precision management of TKI-associated hepatic AEs.
Conclusion
Analysis of FAERS reports revealed substantial heterogeneity in hepatotoxicity risk among ALK and ROS1 inhibitors. Second-generation ALK-TKIs showed lower observed mortality and hospitalization than first- and third-generation agents. Crizotinib, ceritinib, and lorlatinib were associated with higher mortality, supporting vigilant liver function monitoring during initiation and throughout long-term follow-up. ROS1 inhibitors—particularly entrectinib—may exhibit distinct patterns characterized by a higher hospitalization rate and warrant separate evaluation. These findings should be interpreted cautiously given reporting bias, confounding from concomitant medications, and potential underreporting, especially for ROS1 TKIs. Prospective multicenter studies are needed to validate these signals; integration of biomarker panels, mechanistic studies of metabolic pathways, and AI-enabled monitoring may improve early detection and refine dynamic risk stratification to optimize the benefit–risk profile in patients with NSCLC.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251387821 - Supplemental material for Hepatotoxicity of ALK/ROS1 tyrosine kinase inhibitors in non-small cell lung cancer patients: A pharmacovigilance study based on signal mining and analysis of the FDA adverse event reporting system database
Supplemental material, sj-docx-1-sci-10.1177_00368504251387821 for Hepatotoxicity of ALK/ROS1 tyrosine kinase inhibitors in non-small cell lung cancer patients: A pharmacovigilance study based on signal mining and analysis of the FDA adverse event reporting system database by Weixing Zhao, Xiaoni Jin, Yuan Li, Huiyuan Peng and Jun Jiang in Science Progress
Footnotes
Abbreviations
Acknowledgements
The authors used an AI-based language tool (for translation and polishing) to improve the English expression of this manuscript. The authors reviewed and edited the content as needed and take full responsibility for all aspects of the work.
Ethics approval and consent to participate
Because this study was an observational study using global open database (FAERS) with anonymized information, not involving treatment intervention or collection of human samples, informed consent was exempted.
Author contributions
Weixing Zhao: writing—original draft, writing—review & editing, methodology, conceptualization, formal analysis; Xiaoni Jin: writing—original draft, methodology, data curation, visualization; Yuan Li: writing—review& editing, visualization; Huiyuan Peng: writing—review & editing, supervision; Jun Jiang: writing—review & editing, supervision, conceptualization.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Agency of Qinghai Province under Grant 2022-ZJ-719.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets analyzed in the current study are derived from publicly available databases and can be accessed via their respective official websites. Further inquiries should be directed to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
