Abstract
With increased use of immune checkpoint inhibitors (ICIs) for treating tumors, immune-related adverse events (IrAEs) have risen. Severe cases of anti-PD-1 therapy-induced IrAEs such as Stevens–Johnson syndrome (SJS)-like and toxic epidermal necrolysis (TEN)-like lesions are rare but fatal. We report two cases of patients with malignant tumors who developed oral and cutaneous severe IrAEs: one induced by a PD-1 inhibitor alone and the other by a combination of a PD-1 inhibitor and an antibody–drug conjugates (ADCs). Both of them developed severe lesions resembling erythema multiforme (EM) and SJS, accompanied by lichenoid reactions. Treatment with short-term low-dose oral glucocorticoids with topical glucocorticoids resulted in satisfactory outcomes. These case reports highlight rare and severe IrAEs induced by PD-1 inhibitors and ADCs and demonstrate how optimal outcomes were achieved for patients with severe IrAEs.
Keywords
Introduction
In recent years, humanized monoclonal programmed death-1 (PD-1) immune checkpoint inhibitors (ICIs) have been shown to enhance the cytotoxic activity of T lymphocytes against tumor cells and control tumor growth. 1 They have demonstrated outstanding clinical efficacy in various cancer types, including advanced cervical and bladder cancer. In the KEYNOTE-028 clinical study, pembrolizumab was used in advanced cervical cancer and exhibited promising efficacy. 2 Based on the results of the KEYNOTE-045 study, pembrolizumab received full approval from the Food and Drug Administration (FDA) for treating patients with locally advanced or metastatic uroepithelial cancer. 3 In addition to ICIs, antibody–drug conjugates (ADCs) are attracting continued attention. FDA-approved ADCs include enfortumab vedotin, targeting nectin-4, and sacituzumab govitecan, targeting Trop-2, and are intended to be used for uroepithelial carcinoma treatment.
However, while immunotherapy is expected to improve cancer outcomes, immune-related adverse events (IrAEs) frequently occur. They are believed to cause organ toxicity due to the loss of tolerance to autoantigens. 4 All organs may be affected, with the skin, gastrointestinal tract, endocrine system, lung, and kidney showing higher susceptibility.5–7 Oral IrAEs have been frequently reported to present as lichenoid reactions and vesiculobullous lesions, while serious adverse events include erythema multiforme (EM), pemphigoid, Stevens–Johnson syndrome (SJS), and toxic epidermal necrolysis (TEN). 8 Similarly, common ADC-related adverse events include skin reactions, ocular toxicity, gastrointestinal reactions, hematologic toxicity, neurotoxicity, hepatotoxicity, pulmonary toxicity, and metabolic abnormalities. Skin reactions include rash, itching, dry skin, and maculopapular rash, while severe skin reactions include dermatitis herpetiformis, exfoliative dermatitis, SJS, and TEN.9–11
Herein we report two patients: one with cervical cancer treated with tislelizumab, and the other with bladder cancer treated with pembrolizumab and enfortumab vedotin. Both patients presented with oral and cutaneous IrAEs. This report aims to highlight the rare presentation of concurrent severe oral and skin IrAEs and to describe their clinical course and management. We describe, in detail, the clinical presentation and management of these patients to provide a reference for managing patients with severe oral and skin IrAEs.
Case presentation
Case 1
A female patient in her late 50s presented to the Department of Oral Medicine, Peking University School and Hospital of Stomatology, Beijing, China in March 2024, with extensive oral mucosal erosion and rashes persisting for >1 month. She had been diagnosed with moderately differentiated cervical squamous cell carcinoma four months earlier and subsequently underwent total abdominal hysterectomy and bilateral salpingo-oophorectomy. Following the surgery, she was treated with paclitaxel and cisplatin chemotherapy, followed by the addition of tislelizumab based on oncologic evaluation. As part of the first treatment cycle, she was intravenously administered 240 mg paclitaxel and 40 mg cisplatin, 40 mg cisplatin, 30 mg cisplatin and 200 mg tislelizumab on the first three days. After an approximately three-month interval, the patient underwent the second treatment cycle, intravenously receiving 240 mg paclitaxel and 110 mg cisplatin, followed by 200 mg tislelizumab, on the first two days. Three days after starting the second treatment cycle, she developed extensive oral mucosal erosion and skin rashes. A dermatologist consequently prescribed amoxicillin (0.5 g per dose, three times daily) for 3 days, but her condition showed no significant improvement.
Physical examination revealed extensive hemorrhagic crusts with exudation on the upper and lower labial mucosa; extensive erosion of the buccal mucosa, dorsal tongue mucosa, and floor of the mouth; accompanied by white lichenoid lesions on the buccal mucosa and some scattered skin rashes (Figure 1(a)–(d)). According to the Common Terminology Criteria for Adverse Events (CTCAE) v5.0 and the World Health Organization (WHO) criterion, she was identified as having grade 3 IrAEs.12,13

Clinical course of case 1 (a woman in her late 50s with cervical cancer) showing severe oral and cutaneous immune-related adverse events (IrAEs) induced by PD-1 inhibitors. (a–d) Initial presentation of oral mucosal erosions and cutaneous rashes. (e–h) Significant improvement after treatment with oral and topical glucocorticoids. (i) Gantt chart summarizing the treatment timeline and IrAEs management.
A saliva sample culture was performed to detect and identify Candida and specific bacteria, with the results showing no fungal or bacterial infections. Consequently, the initial diagnosis of IrAEs prompted the administration of topical glucocorticoids. The patient was given 0.002% dexamethasone saline gargle thrice daily, but her symptoms showed no significant improvement after a week. Oral glucocorticoids were prescribed following consultation with the oncology department. Prednisolone acetate tablets (20 mg once daily) were prescribed for a week, along with chlorhexidine mouthrinse thrice daily to prevent infections. Two weeks later, her symptoms significantly improved. Subsequently, she was treated with prednisolone acetate tablets at a 25 mg dose once daily; the dose was gradually reduced by 5 mg every three days until the medicine was depleted. To accelerate healing, the concentration of dexamethasone saline gargle was increased to 0.005%. The patient was also prescribed a 3% sodium bicarbonate solution thrice daily to prevent fungal infections and an anesthetic ointment thrice daily for pain management. A follow-up visit three weeks later revealed complete healing of the labial and buccal mucosa, disappearance of the rash, and only residual dorsal tongue erosion (Figure 1(e)–(h)). The efficacy of oral glucocorticoids was significant. Nevertheless, subsequent immunotherapy was discontinued to avoid recurrence. The patient is now under follow-up. The patient's IrAEs management was outlined in a Gantt chart for clarity (Figure 1(i)).
Case 2
A male patient in his early 60s presented to Department of Oral Medicine, Peking University School and Hospital of Stomatology, Beijing, China in April 2024, with extensive oral mucosal erosion. He had been diagnosed with bladder cancer four years earlier and had undergone surgical treatment in 2020, with regular follow-up after surgery. After discovering lymph node metastases from bladder cancer in October 2023, he was started on a combination regimen of pembrolizumab (200 mg every 3 weeks) and enfortumab vedotin (1.25 mg/kg on days 1, 8, and 15 of a 28-day cycle). Following two cycles of the combination therapy, the patient developed extensive oral mucosal erosions causing severe pain and feeding difficulties, accompanied by an itchy sensation on the skin of the back and extremities. He received a single 40 mg intramuscular injection of glucocorticoids and was instructed to apply a topical glucocorticoid ointment three times for 3 days after consulting the dermatology department, which provided relief but not a cure. At the time of the visit, the patient was still undergoing immunotherapy.
Physical examination revealed extensive erosion of the soft palate and bilateral ventral tongue covered with a yellow pseudomembrane and accompanied by yellowish exudate. Erosion was also evident on the inner mucosa of the lower lip, surrounded by white reticular lesions (Figure 2(a) and (b)). The itchy skin on the back and extremities had no visible lesions. According to the CTCAE v5.0 and the WHO criterion, this patient was also identified as having grade 3 IrAEs.12,13
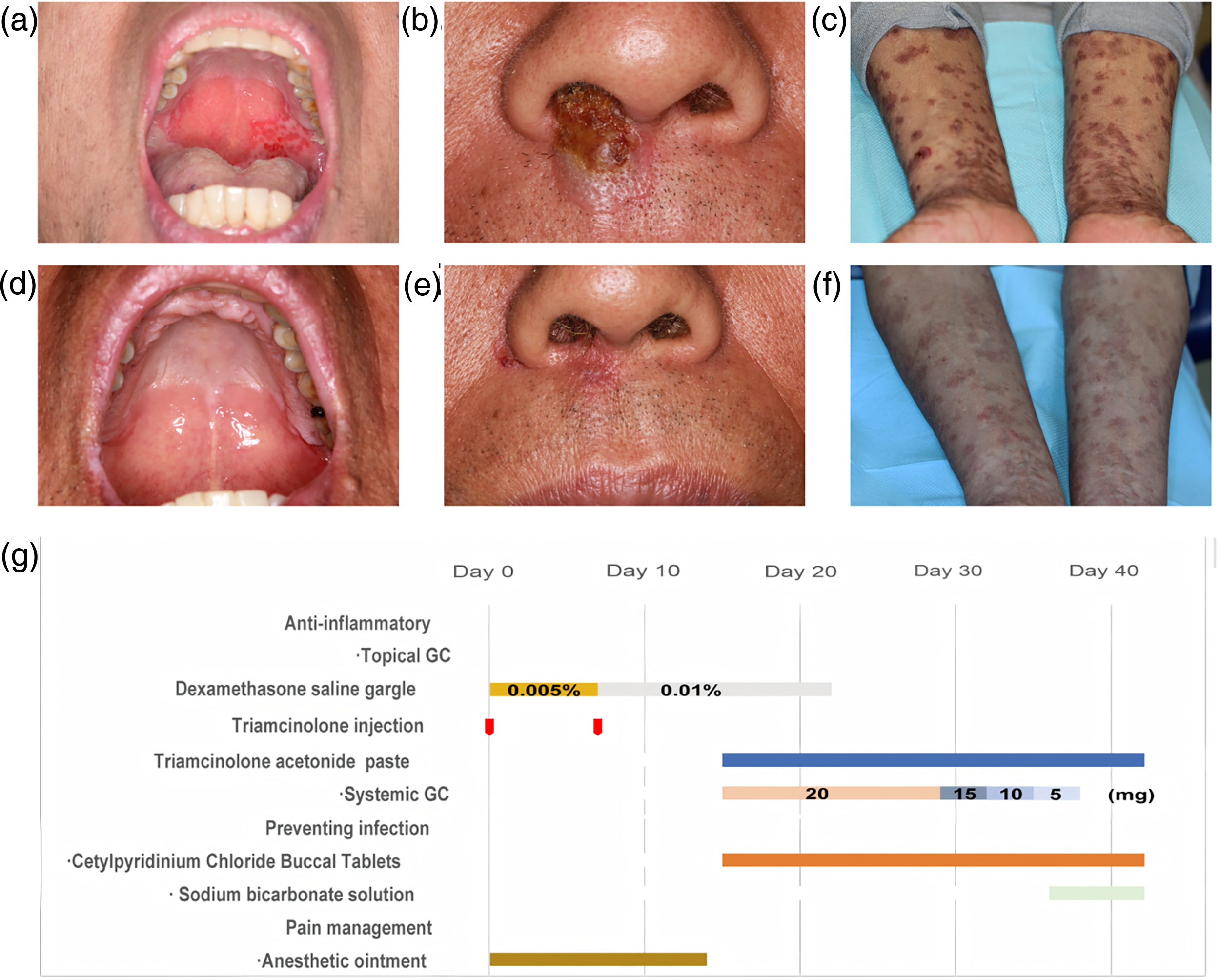
Clinical course of case 2 (a man in his early 60s with bladder cancer) presenting with severe oral mucosal erosions and SJS-like lesions induced by PD-1 inhibitors. (a–c) Initial presentation of oral lesions. (d–f) Almost complete resolution of mucosal and cutaneous lesions following systemic and topical glucocorticoid therapy. (g) Gantt chart illustrating the treatment timeline and management of IrAEs.
A saliva sample culture was performed to detect and identify Candida, ruling out the complication of a fungal infection. An initial diagnosis of IrAEs prompted the administration of topical glucocorticoids. The patient was given 0.005% dexamethasone saline gargle thrice daily, along with a single local injection of 1 mL triamcinolone (1 mL: 40 mg) and 1 mL lidocaine hydrochloride (5 mL: 100 mg) in bilateral ventral erosions of the tongue. In addition, an anesthetic ointment was applied thrice daily for pain management. At the seven-day follow-up, no significant improvement was observed, so the concentration of dexamethasone saline gargle was increased to 0.01%. Meanwhile, the patient underwent immunotherapy again. One week later, his condition worsened, manifesting as SJS-like lesions. Blisters were observed on the nasal and perinasal mucosa, extremities, and back, with significant hyperpigmentation occurring after blister rupture on the extremities and back, affecting >50% of the body surface area (Figure 2(c)). Due to the ineffectiveness of topical glucocorticoids, oral glucocorticoids were prescribed following consultation with the oncology department. Prednisone acetate tablets were administered at a 20 mg dose once daily for two weeks; the dose was gradually reduced by 5 mg every three days until the medicine was depleted. Simultaneously, the patient continued treatment with topical glucocorticoids, including triamcinolone acetonide dental paste and 0.005% dexamethasone solution (both three times daily). In addition, he was prescribed cetylpyridinium chloride buccal tablets (1 tablet per dose, three times daily) and 3% sodium bicarbonate solution (10 mL pre dose, used as a gargle three times daily) to prevent infections. After this treatment, his lips, nasal and perinasal mucosa, and oral mucosa lesions almost healed, with only a few scattered lesions on the limbs (Figure 2(d)–(f)). For residual lesions, treatment with topical glucocorticoids was continued. The efficacy of oral glucocorticoids was significant and did not affect his oncology treatment. However, during subsequent follow-ups, it was found that the patient still required immunotherapy. If IrAEs recur, the previously effective treatment regimen will be re-applied. The patient's IrAEs management was outlined in a Gantt chart for clarity (Figure 2(g)).
The reporting of this study conforms to CARE guidelines. 14 All patient details have been de-identified in accordance with ethical and privacy guidelines. We have obtained the patient's consent for treatment and publication.
Discussion
Monoclonal antibodies targeting PD-1 (anti-PD-1) inhibit the interaction between PD-1 and its ligands, enhancing the cytotoxic activity of T lymphocytes against tumor cells and boosting the T-cell immune response, thereby controlling tumor progression. Despite the promising efficacy of anti-PD-1 therapy, IrAEs frequently occur.1,4,15 The mechanisms of anti-PD-1-induced IrAEs remain to be elucidated. The PD-1/PD-L1 pathway blocks T-cell responses to autoantigens and plays an important role in autoimmunity. 15 In a PD-1 knockout mice model, PD-1 blockade was reported to affect regulatory T cell function and participate in autoantibody production. 16 With the use of anti-PD-1 monoclonal antibodies, this immune balance may be disrupted, leading to T cells attacking both normal and tumor cells, resulting in toxic side effects involving multiple organs.17–20 Among IrAEs, oral lichenoid lesions are the most common oral IrAEs. 8 They may be related to the infiltration of activated T cells in normal tissues, catalyzing a local cell-mediated immune response. 21
Both of our patients presented with extensive erosion and lichenoid reactions of the oral mucosa—an uncommon and severe manifestation of oral IrAEs. Previous reports have documented oral and dermatologic reactions to ICIs; however, very few have described such extensive and severe oral mucosal involvement in the context of combination regimens involving PD-1 inhibitors and ADCs or chemotherapy.8,22,23 This highlights the novelty and clinical significance of our cases.
Moreover, ADCs such as enfortumab vedotin are designed to selectively target tumor cells by linking cytotoxic agents to monoclonal antibodies. While this approach enhances anti-tumor efficacy, it may also lead to off-target toxicities.22,23 Common adverse reactions to ADCs include skin reactions, ocular toxicity, gastrointestinal reactions, hematologic toxicity, neurotoxicity, hepatotoxicity, pulmonary toxicity, and metabolic abnormalities, similar to those associated with anti-PD-1 therapy. In Case 2, the use of both pembrolizumab and enfortumab vedotin complicates causality determination. It remains unclear whether the observed severe IrAEs were induced by pembrolizumab alone or by a synergistic toxicity of both agents. This is supported by Lacouture et al., who reported SJS-like cutaneous toxicities associated with enfortumab vedotin. 24
Similarly, Case 1 raises the issue of drug attribution. The patient received tislelizumab alongside paclitaxel and cisplatin. Paclitaxel and cisplatin, particularly the former, are known to induce dermatologic and mucosal adverse events. 25 While the temporal relationship between the second dose of tislelizumab and the onset of IrAEs supports immune-related causality, the absence of specific allergy testing (including skin prick, intradermal, and dermal patch tests), in vitro tests (such as biomarker detection or basophil stimulation test), or drug provocation tests limits our ability to definitively exclude chemotherapy-induced reactions.26,27 As reported by Sibaud et al., taxanes may provoke ulcerative reactions of the mucosa. 28
In both cases, IrAEs presented within three months of treatment initiation, consistent with known timelines. 29 Lesions were graded as CTCAE v5.0 grade 3 (severe), and WHO grade 3, warranting systemic intervention. Although systemic corticosteroids may blunt anti-tumor immune responses,8,30 we followed the American Society of Clinical Oncology (ASCO), the National Comprehensive Cancer Network (NCCN), and the European Society for Medical Oncology (ESMO) guideline-based recommendations, employing short-term low-dose systemic and topical corticosteroids. 12 Both patients responded well, with significant lesion resolution. Our approach underscores the importance of early, individualized intervention for high-grade IrAEs.
Notably, the initial management in Case 1 included empirical amoxicillin, which may not be ideal given the risk of confounding drug-induced eruptions. In both cases, systemic corticosteroids were introduced after initial conservative measures failed to achieve sufficient improvement. This stepwise escalation, though cautious, may have contributed to lesion persistence. These cases highlight the practical challenges in distinguishing early IrAEs from other etiologies and underscore the importance of timely recognition and guideline-informed escalation in similar clinical scenarios.
Alternative diagnoses such as infection or paraneoplastic syndromes were considered. Bacterial and fungal cultures were negative, and no systemic signs of infection or neoplasm-related mucocutaneous syndrome were observed. However, the lack of histopathologic confirmation or immunopathological profiling (e.g. immune complex deposition, T cell phenotype) limits mechanistic interpretation.
Given the limited sample size of this case-based study, we were unable to identify specific predictive or risk factors for severe IrAEs. However, this remains a critical unmet need in the clinical management of patients receiving PD-1 inhibitors and ADCs. Larger-scale studies are warranted to confirm whether the observed clinical patterns and treatment outcomes can be generalized to broader patient populations undergoing ICI and ADC therapy. Future studies should focus on identifying predictive markers for high-grade IrAEs, clarifying drug-specific risk profiles in combination regimens, and exploring long-term effects of corticosteroid use on tumor outcomes. The potential for glucocorticoids to impair immunotherapy efficacy remains a concern, and neither patient was followed long enough to draw conclusions on long-term cancer control.
Conclusion
In conclusion, we report two rare and severe cases of oral and cutaneous IrAEs triggered by combination therapies involving PD-1 inhibitors and either ADCs or chemotherapeutic agents. These cases illustrate the diagnostic challenges, highlight the need for cautious causality attribution, and demonstrate that early, guideline-concordant immunosuppressive therapy can lead to favorable outcomes. Our findings emphasize the importance of interdisciplinary management and raise awareness of atypical and high-grade IrAEs in the era of combination immunotherapy. A summary of our proposed management approach is presented in Figure 3.

Management strategies for severe oral and cutaneous IrAEs based on the two cases presented.
Footnotes
Acknowledgements
We would like to thank the native English speaking scientists of Elixigen Company (Huntington Beach, California) for editing our manuscript.
Ethics approval
Approval by the Institutional Review Board is not required for individual case reports.
Consent to participate
Written informed consent to participate in treatment was obtained from the patients prior to the start of clinical care.
Author contributions
HH and PZ conceived and designed the study. FY acquired, analyzed and interpreted the data. FY and PZ drafted and revised the manuscript, and confirm the authenticity of all the raw data. All authors read and approved the final version of the manuscript for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China [Grant Nos. 2023YFC3605603, 2022YFC2402905 and 2024YFB4607800] and the Program of the New Clinical Techniques of Peking University School and Hospital of Stomatology (PKUSSNCT-24A10).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
The data generated in the present study may be requested from the corresponding author.
Patient consent for publication
Written informed consent was obtained from the patients for the case information and images to be published in the present case reports.
