Abstract
Objective
In the intensive care unit (ICU), sepsis is a leading cause of mortality, and survivors frequently experience serious long-term sequelae. In addition to identifying risk variables influencing post-sepsis syndrome (PSS) and post-intensive care syndrome (PICS), this study sought to present comprehensive data on the three-month prognostic and functional outcomes of sepsis patients in the ICU—a demographic frequently overlooked in long-term recovery discourse.
Methods
A prospective cohort study was conducted in the intensive care units of Jining Medical University Affiliated Hospital between April 2024 and July 2025, enrolling patients diagnosed with sepsis or septic shock. Follow-up assessments were conducted one and three months post-discharge via telephone or outpatient visits. The frequency of each dimension in PICS and PSS was investigated, alongside patients’ health-related quality of life. Risk factors for the two syndromes were analyzed using multivariable logistic regression and stepwise multiple linear regression.
Results
Sepsis survivors included in the one- and three-month follow-ups totaled 147 and 132, respectively. PICS and PSS had overall respective prevalence rates of 93.2–98.6% and 61.4–77.6%. Multivariable analyses revealed that body mass index (BMI), red blood cell (RBC) count, partial pressure of oxygen (PO2), and globulin levels were independent predictors for PSS. For PICS severity, Glasgow Coma Scale (GCS) score, venous thromboembolism (VTE) risk score, bicarbonate (HCO3-) levels, and specific infection sites were identified as independent predictors. Sepsis survivors’ quality of life improves over the three months following discharge; however, PICS and PSS maintain a high prevalence, with a notable symptom overlap between the two syndromes. Clinicians must be mindful of specific in-hospital risk factors to tailor post-discharge care.
Conclusion
This study highlights the profound and persistent risks for ICU sepsis survivors. The results indicate that early multidisciplinary intervention is necessary, which may potentially reduce long-term sequelae and improve recovery trajectories.
Keywords
Introduction
Sepsis remains a formidable systemic inflammatory response triggered by infection, exacting a massive global toll. 1 Septic shock, its most severe form, is marked by severe cellular, metabolic, and circulatory abnormalities that significantly raise the chance of death. 2 Multiple organ failure and substantial mortality rates are observed in intensive care units (ICUs) patients with sepsis and septic shock. 3 Recent epidemiological analyses have demonstrated an increase in the prevalence of sepsis and septic shock. In 2017, there were approximately 49 million sepsis cases and 11 million sepsis-related deaths globally. 4 Furthermore, the advances in critical care medicine and sepsis care induced by the Surviving Sepsis Campaign guidelines have improved survival among patients with sepsis. 5 As sepsis survival has improved over time, the population of patients who live beyond the acute phase has expanded progressively.6,7 Many survivors have significant comorbidities that may be life limiting or significantly impair quality of life. 5 Hence, mapping the long-term trajectories of these survivors is not merely an academic exercise—it is a clinical imperative.
As more sepsis survivors return to the community, the burden of illness is shifting from acute care hospitals to chronic long-term care settings. 8 They could encounter restrictions and long-term consequences that significantly affect their health-related quality of life (HRQoL), as well as their physical, mental, and cognitive well-being. 9 This is often referred to as the “post-sepsis syndrome” (PSS). 10 Studies have shown that survivors of critical illness, including sepsis, have similarly poor quality of life. 11 Consequently, recent studies have investigated the prevalence of Post-intensive care syndrome (PICS) in sepsis survivors.12,13 PICS can bring a heavy burden to patients’ lives and damage their long-term prognosis.12,14 Both PICS and PSS involve cognitive, psychological, and physical impairments, resulting in significant overlap in clinical manifestations. However, it remains unclear whether these are truly distinct pathologies or merely contextual manifestations of a universal post-critical illness dysfunction.15,16 Therefore, we wish to explore their relationship. Furthermore, the existing literature addresses the cognitive, emotional, and physical impacts experienced by sepsis survivors. 17 There is a significant gap in the exploration of potential in-hospital risk factors associated with an altered PICS and PSS. Not enough research has been done on certain in-hospital factors that influence these long-term results.
To address this critical gap, this study aims to investigate the prevalence and characteristics of PICS and PSS in survivors of sepsis and septic shock three months after hospital discharge, and to identify independent in-hospital predictors of these outcomes to inform strategies for improving long-term recovery.
Materials and methods
Study design
This prospective longitudinal cohort study was conducted in accordance with the Declaration of Helsinki (as revised in 2024), and its reporting follows the STROBE guidelines. 18 Ethical approval was obtained from the local ethics commission on 22 November 2023 (approval number: 2023-11-C032), and the study was registered prior to publication (Chinese Clinical Trial Registry, CTR2500112184). All potential participants or their legal next of kin were informed about the study using a consent form. They were assured that their free will to participate (or to permit participation on the patient’s behalf) would be respected, that the patients would remain anonymous, and that refusal or withdrawal during the study would not be disadvantageous.
Patients and setting
Patients were consecutively enrolled from the ICUs of Jining Medical University Affiliated Hospital between April 2024 and July 2025. Those with a confirmed diagnosis of sepsis or septic shock according to the Sepsis-3 criteria were enrolled within 24 hours of ICU admission. Sepsis was defined as life-threatening organ dysfunction caused by a dysregulated host response to infection, operationalized as an acute increase in the Sequential Organ Failure Assessment (SOFA) score of 2 points or more from baseline in the presence of suspected or documented infection. 1 Septic shock was defined as a subset of sepsis with persistent hypotension requiring vasopressor therapy to maintain mean arterial pressure ≥ 65 mmHg and having a serum lactate level > 2 mmol/L despite adequate fluid resuscitation.1,2
Written informed consent was obtained from the patient or their legal next of kin prior to any study procedures. In many cases, consent was obtained from the legal next of kin because the study subjects were in a state of critical illness, such as sepsis or septic shock, and lacked the decisional capacity to provide written informed consent themselves at the time of enrollment (due to sedation or sepsis-associated encephalopathy). When patients regained decisional capacity, they were approached to provide their own consent to continue participation, ensuring full respect for their autonomy. Patients were then followed up for three months after discharge.
The exclusion criteria were as follows: age under 18 years, end-stage disease (malignant tumor with metastases, AIDS, end-stage renal or liver disease), refusal to participate in the study, and incomplete or missing data. Incomplete or missing data was defined as: (1) missing baseline characteristics that could not be obtained from medical records (such patients were excluded at enrollment and are not included in the initial cohort); or (2) loss to follow-up due to incorrect contact information, refusal, or non-response at 1 or 3 months. All patient details have been de-identified to ensure anonymity. No personally identifiable information is disclosed in this manuscript.
Data collection
We recorded the following in-hospital characteristics of patients admitted to the ICU within the first 24 hours including: age, gender, comorbidities, infection sites, SOFA score, and laboratory indicators. These variables represent baseline characteristics measured within the first 24 hours of ICU admission. In addition, the following cumulative ICU data were documented throughout the entire ICU stay: duration of mechanical ventilation, sedative use, analgesic use, and ICU length of stay. Follow-up data were collected using the same questionnaire at two time points: one and three months after discharge. Follow-up assessments at post-discharge included only questionnaires, and no laboratory tests were repeated at these time points. Two experienced graduate nursing students conducted follow-up visits with patients either by phone or in the outpatient department. The mortality assessment included cumulative all-cause mortality at 1 and 3 months after discharge. Patients who died during hospitalization and those who were lost to follow-up after discharge were not included in this part of the analysis.
Outcome measures
The primary outcomes were: the prevalence of PICS and the prevalence of PSS among survivors of sepsis. PICS was measured using the Healthy Aging Brain Care Monitor Self-Report (HABC-M SR), a self-report measure that covers the cognitive, physical, and psychological domains. Higher total scores indicate higher levels of PICS. 19 The HABC-M SR Chinese version, utilized in this study, encompasses 19 items, with 7 items allocated to the cognitive domain, 6 items to the physical domain, and 6 items to the psychological domain. The total scale ranges from 1 to 57, with scores of 21, 18, and 18 indicating the worst possible outcome in the three domains, respectively. In line with the previous research, 20 the presence of PICS at each follow-up was defined as a HABC-M SR total score greater than 0 points. Additionally, to investigate factors associated with the severity of PICS symptoms, the continuous HABC-M SR total score was also used as an outcome in linear regression analyses.
The clinical diagnostic methods for PSS and PICS are similar. 21 Universal scales are usually used to assess whether patients have experienced any new or worsening functional impairments in cognition, physiology or psychology in order to operationalise the presence of PSS for research purposes. The existence of any of the following circumstances was considered to be the occurrence of PSS, 22 based on instruments widely validated in the context of long-term outcomes after sepsis and critical illness: (1) physical dysfunction, 23 Barthel index (BI) score < 60, the BI has been validated for functional assessment in critically ill patients at ICU discharge; (2) cognitive impairment, 24 as indicated by a Mini-Mental State Examination (MMSE) score < 24; (3) mental dysfunction, 24 Hospital Anxiety and Depression Scale (HADS) ≥ 8 points. The MMSE and HADS are well-established instruments widely used in sepsis survivor cohorts to assess cognitive and psychological outcomes. 24 HRQoL was evaluated using the Short Form-36 (SF-36) measure. 25
Sample size calculation
Based on the number of independent variables in the multiple regression analysis, the adjusted sample size required was 210 patients.
Statistical methods
Normality was tested using the Shapiro-Wilk test. For normally distributed data, means with standard deviations (SD) were used; otherwise, medians with interquartile range (IQR) were reported, and compared using the independent samples t-test (for normally distributed data) or the Mann-Whitney U test (for non-normally distributed data). Categorical variables were expressed as numbers and percentages, and compared using the Chi-square test or Fisher’s exact test. The 95% confidence interval of the proportion index was calculated using the Wilson method. A stepwise multiple linear regression analysis was employed to introduce variables with a
Result
Participants’ characteristics
From April 2024 to July 2025, a total of 228 patients who were diagnosed with sepsis or septic shock upon admission were screened. A total of 26 patients were excluded, with the specific reasons including: age less than 18 years (n = 3), expected to be transferred out of the ICU within 24 hours (n = 9), having severe underlying conditions (n = 8), and declined to participate (n = 6). A total of 202 patients were enrolled in the final cohort (Figure 1). Among them, 17 patients (8.4%) died in the hospital, and 185 patients (91.6%) were discharged alive. One month after discharge, 6 patients (3.2%) were lost to follow-up and 32 patients died. Based on this, the cumulative mortality rate at 1 month was 17.9% (32/179; 95% CI, 12.6%–24.2%). At 3 months after discharge, 9 patients (4.9%) were lost to follow-up, and 44 patients died. The cumulative mortality rate at 3 months was 25.0% (44/176, 95% CI: 18.8%–32.1%). The characteristics of the participants are summarized in Table 1, which also presents the baseline characteristics of survivors at the 1-month and 3-month follow-up assessments. Flowchart of the follow-up process for patients. Baseline characteristics of all enrolled patients and survivors at 1-month and. Unless otherwise specified, all data represent baseline characteristics measured within 24 hours of ICU admission, as detailed in the Data Collection section. Abbreviations: BMI: Body Mass Index; APACHE II: Acute Physiology and Chronic Health Evaluation II; SOFA: Sequential Organ Failure Assessment; GCS: Glasgow Coma Scale; VTE: Venous Thromboembolism; RBC: Red Blood Cell; WBC: White Blood Cell; Hb: Hemoglobin; RDW-SD: Red Cell Distribution Width-Standard Deviation; RDW-CV: Red Cell Distribution Width-Coefficient of Variation; PO2, Partial Pressure of Oxygen; CRP: C-Reactive Protein; PCT: Procalcitonin; ALB: Albumin; GLB: Globulin; TBil: Total Bilirubin; Lac: Lactate; HCO3-: Bicarbonate; BE: Base Excess; AG: Anion Gap; IL-6: Interleukin-6; cTnT: Cardiac Troponin T.

Primary outcome
Comparison of PICS, PSS and health-related quality of life between 1 and 3 months after discharge follow-up among sepsis survivors.
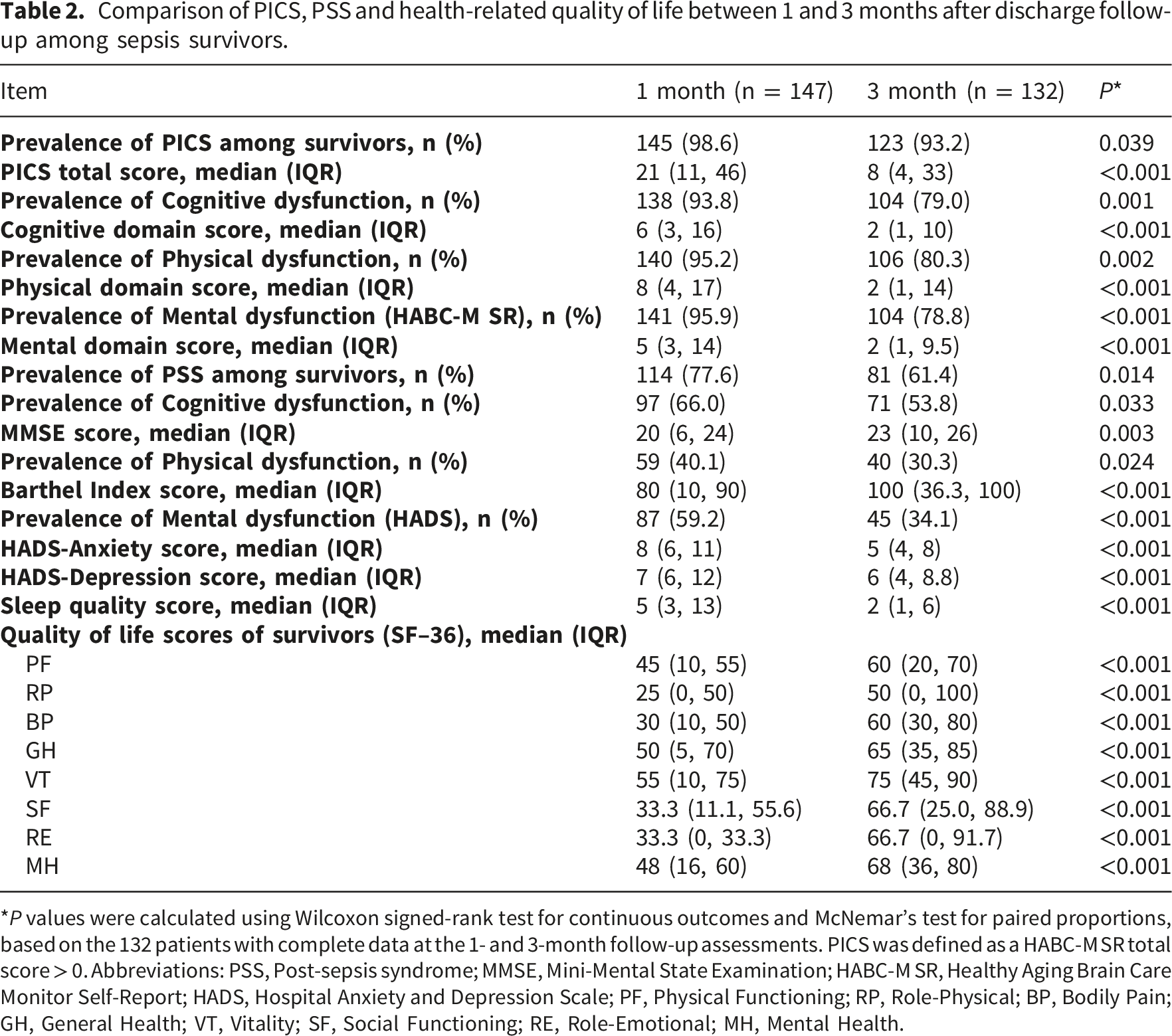
*

Outcomes of sepsis survivors 1- and 3-month after discharge. (a), the prevalence post-sepsis syndrome (PSS) at the first-month follow-up. (b), the prevalence post-sepsis syndrome (PSS) at the third-month follow-up. (c), Median (IQR) HABC-M SR Chinese version score for PICS and its three domains among the 1- and 3-month follow-ups. (d), Median (IQR) SF-36 and its eight domains among the 1- and 3-month follow-ups.
Risk factors for PSS
Multivariable logistic regression analysis for PSS at 3 months.

All laboratory indicators were measured within the first 24 hours of ICU admission. No laboratory tests were performed during the post-discharge follow-up assessments. Abbreviations: PSS, Post-sepsis syndrome; BMI, Body Mass Index; RBC, Red Blood Cell; GLB, Globulin; BE, Base Excess; GCS, Glasgow Coma Scale; PO2, Partial Pressure of Oxygen; HCO3-, Bicarbonate.
Risk factors for PICS
Multiple linear regression analysis of factors independently associated with HABC-M SR score at 3-month follow-up.

Variables were selected using a stepwise regression method. Initial candidates included all factors with
Discussion
The mortality rate three months after sepsis in this study was similar to rates reported in previous literature. The in-hospital, 30-day, and 90-day mortality rates for patients with sepsis or septic shock were 8.4%, 17.9%, and 25.0%, respectively. Previous epidemiological studies have reported that the 90-day mortality rate for patients with sepsis was between 17.9% and 31.6%.26,27 These findings suggest that the long-term prognosis after sepsis is not favorable. In addition to survival and discharge, we should pay more attention to patients’ quality of life after discharge.
In this prospective observational study, we found that 92.5% of sepsis survivors experienced symptoms of PICS three months after discharge, with 60.9% meeting the criteria for PSS at the same time point, highlighting the substantial and persistent burden faced by this population. The prevalence of PICS declined from 98.6% at 1 month to 92.5% at 3 months (
In the multivariable logistic analysis for PSS, higher baseline partial pressure of oxygen (PO2), red blood cell (RBC) count, and globulin (GLB) levels were independently associated with increased odds of PSS at 3 months. These associations may reflect underlying disease severity rather than direct causal effects. Research has found that high-oxygen exposure can trigger inflammatory responses and oxidative damage in the lungs.
30
Elevated PO2 may indicate greater respiratory support requirements in patients with more severe lung injury. Elevated RBC may reflect hemoconcentration related to fluid resuscitation or underlying chronic conditions, impairing microcirculatory flow and tissue perfusion,
31
while higher globulin levels indicate a pro-inflammatory phenotype or chronic inflammation that predisposes to prolonged recovery.
32
Intriguingly, we observed that baseline inflammatory markers—specifically interleukin-6 (IL-6), C-reactive protein (CRP), and PCT—tended to be higher in survivors than in non-survivors. While hyperinflammation is conventionally equated with severity, this paradoxical trend aligns with the emerging concept of “immunoparalysis” in sepsis.
33
Higher BMI was associated with significantly lower odds of PSS (OR 0.864, 95% CI 0.767–0.974,
In the multivariable analysis for PICS severity, lower GCS score, higher VTE score, higher HCO3- level, pulmonary infection, and infection at other sites were independently associated with higher HABC-M SR scores. Lower GCS score emerged as the strongest predictor (standardized β = -0.311,
At the beginning of the twenty-first century, the first accounts of post-sepsis syndrome were documented, detailing persistent physical, mental, cognitive, and medical abnormalities that followed severe sepsis. 10 At present, a growing body of research on sepsis survivorship focuses on PICS.12,13 Rather than viewing PICS and PSS as distinct syndromes, our findings suggest they may represent two manifestations of a common underlying condition—post-critical illness dysfunction—differing primarily in the population studied rather than in clinical presentation. 15 Our data reveal a striking co-occurrence of symptoms, with 92.5% of survivors affected by PICS and 60.9% by PSS at 3 months, highlighting the multi-dimensional and overlapping nature of these impairments. These data underscore the substantial and persistent burden faced by this population. Rather than viewing them as isolated entities, the co-occurrence of PICS and PSS embodies the multi-dimensional nature of post-sepsis recovery, necessitating integrated clinical surveillance and supportive care tailored to these overlapping impairments. Consequently, future research should focus on developing integrated assessment tools and intervention protocols that simultaneously address the cognitive, physical, and psychological domains common to both syndromes.16,41
Several limitations were acknowledged. First, the study was single-centered, which would have limited how broadly our results could be applied. Furthermore, 34.7% of participants (n = 70) died during the follow-up period or were lost to follow-up, which could introduce non-response bias and distort our findings. The high cumulative mortality rate observed at 3 months (25.0%) represents a significant competing risk in this population. Because PICS and PSS can only be assessed in living patients, our findings are subject to survivor bias. The observed survival bias—where non-survivors exhibited paradoxically lower inflammatory markers—highlights the complexity of host responses; our findings thus reflect the burden specifically among those capable of mounting an initial immune response. Consequently, the reported prevalence likely represents the survivor burden of sepsis rather than the total impact on the entire initial cohort. Second, it is possible that the instruments used to evaluate PICS differ from those employed in previous research. Third, the three-month follow-up time is rather brief and is unable to cover all PICS symptoms. Thus, a long-term longitudinal study with multiple follow-ups will be needed. Future analyses involving a multicenter cohort with a broader participant demographic are planned.
Conclusion
In this prospective study, we analyzed PICS and PSS conditions and related quality of life dimensions in sepsis survivors and found significant overlap between the two syndromes. Furthermore, we identified risk factors associated with the severity of these syndromes in order to provide a framework for future research and facilitate better recovery outcomes for sepsis survivors.
Supplemental material
Supplemental material - Analysis of the prevalence and risk factors of post-intensive-care syndrome and post-sepsis syndrome in survivors of sepsis
Supplemental material for Analysis of the prevalence and risk factors of post-intensive-care syndrome and post-sepsis syndrome in survivors of sepsis by Yuanqing Li, Anhao Liu, Xuehui Zhang, Ningkang Lv, Tianqi Chen, Cuiping Hao and Dongmei Wu in Science Progress.
Footnotes
Ethical considerations
This study adhered to the Declaration of Helsinki, was approved by the local ethics committee on 22 November 2023 (approval number: 2023-11-C032), and registered at the Chinese Clinical Trial Registry (CTR2500112184).
Consent to participate
Written informed consent was obtained from all participants or their legal next of kin prior to their participation in the study.
Consent for publication
Written informed consent was obtained from all participants or their legal next of kin prior to their participation in the study. A copy of the signed consent form is available for review upon request.
Author contributions
LYQ, LAH and LNK contributed to data collection and manuscript drafting. LYQ,ZXH and CTQ participated in data analysis, interpretation, and critical revisions of the manuscript. WDM and WCP reviewed and approved the final version of the manuscript. All authors have reviewed and agreed to publish this final draft.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key R&D Program of Jining City, Shandong Province, China (No. 2023YXNS180, No. 2023YXNS107) and the Wu Jieping Medical Foundation (No.320.6750.2024-07-4). The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript, and no additional financial support will be provided.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
