Abstract
Objective
Sepsis is a heterogeneous syndrome, and identifying its subphenotypes may enhance clinical management. Hypothermia may be an indicator of poor outcomes in septic patients.
Methods
This retrospective observational study included septic patients with hypothermia identified from the Medical Information Mart for Intensive Care (MIMIC)-IV database. Group-based trajectory modeling was used to identify temperature trajectory subphenotypes. Cox proportional hazards model, inverse probability of treatment weighting, and doubly robust estimation were used to explore the association between the subphenotypes and clinical outcomes.
Results
A total of 1816 patients were included in the analysis, and their 48-hour temperature trajectories were categorized into three classes. The Cox proportional hazard model revealed that compared with class 2, class 1 was associated with increased 7-day mortality (HR 1.90; 95% CI 1.18–3.07; P = 0.009), whereas class 3 was associated with reduced 28-day mortality (HR 0.81; 95% CI 0.66–1.00; P = 0.045). The doubly robust estimation methods yield similar results.
Conclusions
We identified three temperature trajectories from septic patients with hypothermia, with significant variability in clinical characteristics and outcomes. A better understanding of temperature trajectories may help with the early identification of deteriorating patients with “cold sepsis.”
Introduction
Sepsis is one of the leading causes of hospital mortality in the intensive care unit (ICU) and is characterized by a dysregulated systemic inflammatory and immune response to infection. 1 Despite decades of research, no therapies have consistently proven beneficial for septic patients overall. 2 One potential explanation lies in the heterogeneity of patient populations. Sepsis manifests as a heterogeneous condition with diverse infection sources, microbial etiologies, genetic factors, and comorbid conditions. Therefore, personalized treatment strategies are recommended to improve outcomes in septic patients. 3 Recently, many efforts have been dedicated to developing personalized treatment strategies for sepsis management.4,5 Previous studies have delineated several sepsis subphenotypes through genomics and transcriptomics approaches, revealing their significant implications for both therapeutic interventions and precision trial design.6,7 However, genotyping has not been adopted as a routine clinical tool, with current applications remaining predominantly in the research sphere.
One of the most clinically evident manifestations of heterogeneity in septic patients is the variability in thermoregulatory responses. While fever is traditionally considered a cardinal indicator of infection, a substantial proportion of patients manifest normothermic or hypothermic temperature profiles. Thermoregulatory patterns carry differential prognostic implications in clinical settings. The contrasting thermoregulatory responses of fever and hypothermia to infection may represent distinct pathophysiological mechanisms with potential adaptive benefits for the host organism. The febrile state is characterized by significantly elevated metabolic demands, energy consumption, and physiological costs. When the homeostatic challenge the patient experiences is severe, and the energy expenditure required to induce fever exceeds its benefits, hypothermia becomes evident, and the primary goals of the host shift toward energy conservation and tolerance. 8 Several studies have shown that hypothermia is associated with increased mortality in patients with sepsis.9,10 However, the pathophysiology and regulatory mechanisms of hypothermia in sepsis remain unclear and even more challenging to elucidate. Whether hypothermia reflects severe infection or is an indicator of worsening sepsis, and whether it constitutes a maladaptive and detrimental response, is still unknown.11,12 “Cold sepsis” is known to worsens. In a clinical setting, septic patients with hypothermia may experience fluctuations in body temperature over time. Although hypothermia independently predicts a poor prognosis, whether subsequent temperature trajectories are persistent hypothermia or convert to normothermia or hyperthermia remains unknown. Temperature transitions may have potential clinical implications. We hypothesized that using group-based trajectory modeling (GBTM), we could identify subphenotypes of hypothermic sepsis patients that are associated with distinct clinical outcomes. In this study, we aimed to explore how many hypothermic sepsis subphenotypes might exist and characterize their clinical and prognostic distinctions.
Methods
Source of data and participants
We conducted a retrospective observational cohort study using the Medical Information Mart for Intensive Care (MIMIC)-IV version 2.2 (v2.2), which consisted de-identified, comprehensive, and high-granularity clinical data of patients admitted to the Beth Israel Deaconess Medical Center in Boston, MA, USA, from 2008 to 2019. 13 One of the authors obtained access to the database and subsequently extracted study-related information (certification number: 7574829). As the study was an analysis of a third-party anonymized, the requirement for informed consenting access was waived. The reporting of our study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 14
Critically ill adult patients who met the Sepsis 3.0 criteria were included, 15 which were documented or suspected infection plus an acute increase of ≥ 2 Sequential Organ Failure Assessment (SOFA) points. Hypothermic septic patients were defined as those whose first body temperature measurement in the ICU was less than 36 °C, on the basis of previously used thresholds.10,16 To control possible inaccuracies in body temperature documentation within the database system, extreme temperature values (<30 °C or >42 °C) were substituted with medians. Patients who had a length of ICU stay < 48 hours were excluded. For patients who had multiple hospital admissions, only the first ICU admission was analyzed. We also excluded patients with fewer than three temperature records within the first 48 hours after ICU admission were also excluded, as the GBTM requires sufficient longitudinal measurements. Additionally, patients treated with therapeutic hypothermia (e.g. targeted temperature management) were also excluded.
Variable extraction
Three data types were extracted from the MIMIC-IV using Structured Query Language (SQL) by PostgreSQL software (version 10.17): baseline characteristics, serial clinical observations, and outcome data. The following data were collected on the first day of ICU admission: age, gender, ethnicity, comorbidities, laboratory results (including white blood cell counts, platelet counts, and hemoglobin level), Sequential Organ Failure Assessment (SOFA) score, Simplified Acute Physiology Score II (SAPS II) score, and therapeutic intervention (use of ventilation, renal replacement therapy [RRT], vasopressors, antibiotic, antipyretic, and rewarming intervention). The severity score was calculated within the first 24 hours after the ICU admission. If a variable was recorded more than once in the first 24 hours, the value related to the greatest severity of illness was used. The temperature data from hours 0 to 48 were split into 4-hour blocks of time. If a patient had multiple temperature measurements within a 4-hour period, the earliest temperature was extracted. The details of the missing values were presented in Supplemental Figure S1. Variables with more than 10% missing values were not included in the analysis, and the remaining missing data were addressed using the single imputation method. 17
The primary endpoints were 7-day mortality and 28-day mortality. Mechanical ventilation- and vasopressor-free days within 28 days after ICU admission were considered secondary outcomes.
Statistical analysis
Variables are expressed as the mean (standard deviation) or median [interquartile range (IQR)] for continuous variables when appropriate and as the frequency (percentage) for categorical variables. Comparisons between different profiles were performed using chi-square tests or Fisher's exact test for categorical variables. For continuous variables, the Mann-Whitney U-test or the Kruskal-Wallis test was applied, depending on the number of groups.
We employed group-based trajectory modeling to identify subphenotypes based on temperature trajectories within the first 48 hours after ICU admission. GBTM represents a specialized finite mixture modeling approach designed to classify heterogeneous populations into homogeneous subgroups that exhibit distinct longitudinal patterns of a target variable. 18 Participants were classified into trajectory groups depending on their posterior probabilities, and the model parameters were estimated using maximum likelihood estimation. We selected the optimal number of trajectories on the basis of the following statistical criteria: (a) Bayesian information criteria (BIC); (b) the average posterior probability (AvePP) for each trajectory group exceeds 0.7; (c) the odds of correct classification (OCC) based on the posterior probabilities of group membership exceeding 5.0; (d) each trajectory subgroup size exceeding 1% of the study population; and (e) model parsimony and clinical interpretability.
Four sequential Cox proportional hazard models with increasing covariates were constructed to explore the association between temperature trajectories and mortality outcomes. Model I represented a univariate analysis without adjusting for any covariates. Model II was adjusted for age and gender. Model III was further adjusted for SAPS II scores in addition to the covariates included in Model 2. In Model IV, covariates were selected on the basis of clinical expertise and showed a univariate relationship with outcome (P value < 0.05). Schoenfeld residuals were used to check the proportional hazards assumption. The variance inflation factor (VIF) was calculated to assess multicollinearity. A VIF value of 5 or greater indicates multicollinearity.
A doubly robust estimation method was employed to assess the association between temperature trajectories and patients’ primary outcomes. We applied inverse probability of treatment weighting (IPTW) using the estimated propensity scores to generate a weighted cohort. 19 Standardized mean differences (SMDs) were compared pre- and post-IPTW to evaluate covariate balance improvement across trajectory classes. Univariate Cox regression was conducted on the weighted cohort, and the IPTW model was further adjusted for the remaining imbalanced variables between trajectory groups (with SMDs > 0.1), thereby achieving a double robust analysis.
R software (version 4.1.2) and Stata (version 17) were used for the statistical analyses. A two-tailed P-value < 0.05 was considered statistically significant.
Results
Baseline characteristics
A total of 1817 patients fulfilled the inclusion criteria for the analysis (Figure 1). There were 20,211 body temperature measurements collected (Supplemental Table S1). The absolute values of the BIC decreased substantially from the two-class to the three-class model but plateaued between in the three-class and four-class models. The entropy for the three-class model was 0.866, indicating good classification accuracy. Balancing these fit indices with clinical interpretability and distinct subgroup characteristics, the three-class models were selected as the optimal choices (Supplemental Table S2). The trajectory of the change in temperature of the three-class model is shown in Figure 2. Class 1 accounted for 3.08%, in which the initial temperature was very low and slowly resolved. Class 2 accounted for 45.81%, in which the initial temperature was moderately low and gradually resolved. Class 3 comprised the largest sample size, accounting for 51.10% of the total population, with transient, mild temperature reductions. The clinical characteristics of all three classes are presented in Tables 1 and 2. The median age of the whole patient was 71.40 years old, with the majority being male (55.18%) and white (68.06%). The proportion of emergency admissions was 79.07%. In comparison to patients in the other classes, the patients in class 3 exhibited milder disease severity, fewer comorbidities, and lower rates of RRT use.

The flowchart of the study patients.

Temperature trajectory of septic patients with hypothermia. The shaded area indicates the 95% confidence interval for each trajectory.
Baseline characteristics among the three classes.

CHF: congestive heart failure; CKD: chronic kidney disease; SOFA: Sequential Organ Failure Assessment; SAPSII: Simplified Acute Physiology Score II; WBC: white blood cell; RRT: renal replacement therapy.
Difference of clinical outcomes across the three classes.

ICU: intensive care unit.
Class 1 showed the highest 7-day and 28-day mortality rates (41.07% and 50.00%, respectively), and class 3 showed the lowest mortality rates (9.81% and 19.50%, respectively). Class 1 had the highest duration of mechanical ventilation and vasopressor use (ventilation-free days on day 28 of 11.97 and vasopressor-free days on day 28 of 12.35). No significant differences were observed in ICU length of stay (LOS) or total hospital LOS among the classes.
Temperature trajectories and the primary outcome
Kaplan–Meier (KM) survival curves are shown in Figure 3. The landmark test revealed significant differences in mortality rates among the three classes over different periods. Within the first 7 days, class 1 had the highest mortality rate, after which the mortality curve shifted, whereas class 3 had the lowest mortality rate beyond 7 days.

Kaplan–Meier survival curves of the temperature trajectories.
The findings of the Cox proportional hazards model are presented in Table 3. The PH assumption was not rejected, as indicated by a global Schoenfeld test P-value of 0.3948, with all individual covariates also meeting the requirement (Supplemental Figure S2). Multicollinearity was not present in the remaining covariates (Supplemental Table S3). Compared with class 2, class 1 was associated with an increased 7-day mortality (HR 1.90; 95% CI 1.18–3.07; P = 0.009), whereas class 3 was associated with a reduced 28-day mortality (HR 0.81; 95% CI 0.66–1.00; P = 0.045) after adjusting for most confounders (Supplemental Tables S4 and S5).
HRs for mortality across the three classes.
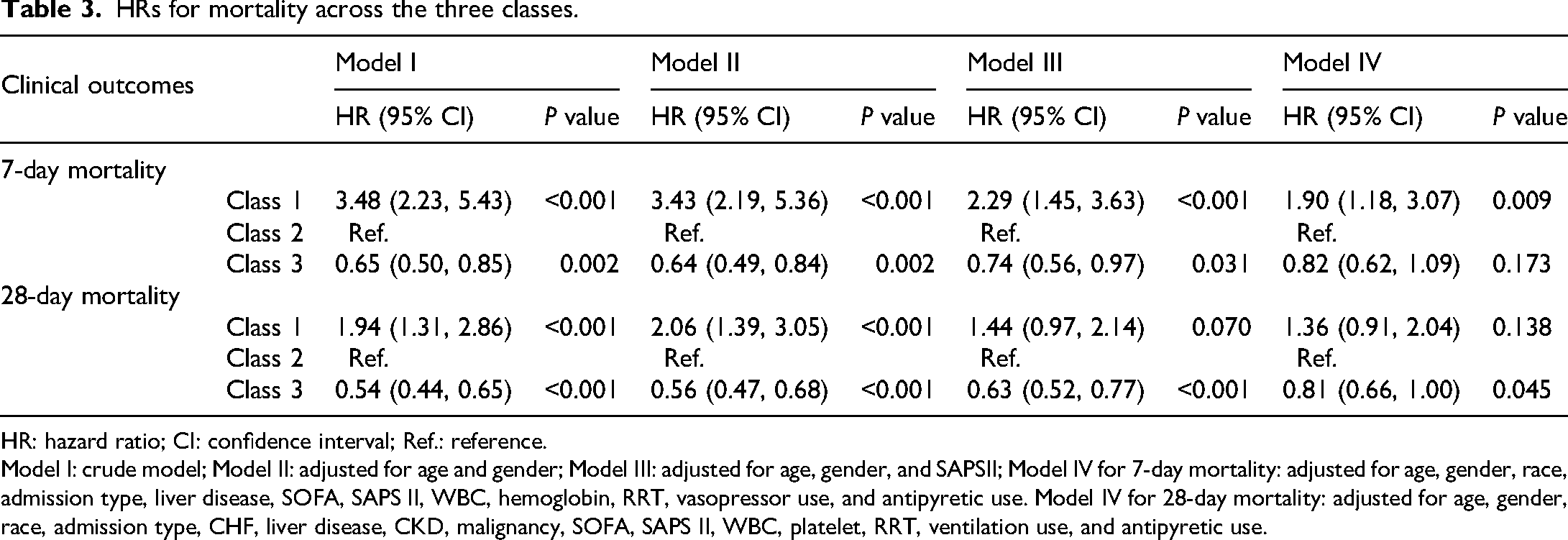
HR: hazard ratio; CI: confidence interval; Ref.: reference.
Model I: crude model; Model II: adjusted for age and gender; Model III: adjusted for age, gender, and SAPSII; Model IV for 7-day mortality: adjusted for age, gender, race, admission type, liver disease, SOFA, SAPS II, WBC, hemoglobin, RRT, vasopressor use, and antipyretic use. Model IV for 28-day mortality: adjusted for age, gender, race, admission type, CHF, liver disease, CKD, malignancy, SOFA, SAPS II, WBC, platelet, RRT, ventilation use, and antipyretic use.
Double robust analysis
The results of the double robust analysis are shown in Table 4. The SMDs of the original dataset and the IPTW dataset are provided in Supplemental Figure S3. Overall, compared with the original dataset, the IPTW dataset demonstrated reduced SMDs. Within the doubly robust estimation paradigm, a regression model was used to address the imbalanced covariates in the weighted cohort. The findings of the doubly robust analysis were consistent with the results from the primary cohort. Specifically, compared with class 2, class 1 showed an elevated risk of 7-day mortality, while class 3 exhibited a reduced risk of 28-day mortality. Conversely, class 3 was not significantly associated with 7-day mortality, and class 1 was not significantly associated with 28-day mortality.
Primary outcome analysis with different models.

HR: hazard ratio; IPTW: inverse probability of treatment weighting; CI: confidence interval; Ref.: reference.
Unbalanced covariates: Sofa, CKD, and diabetes.
Discussion
In this study, we developed a novel approach to identify subphenotypes in septic patients with hypothermia. Using group-based trajectory modeling, we identified three classes of patients that exhibited distinct temperature trajectories within 48 hours after ICU admission. We found significant clinical characteristics and outcomes differences between these classes. Compared to patients whose initial temperature was moderately low and gradually resolved (class 2), patients with very low initial temperature and slowly resolved (class 1) exhibited a significantly increased risk of 7-day mortality, whereas patients with transient, mild temperature reductions (class 3) had a reduced risk of 28-day mortality.
Temperature is one of the critical clinical indicators in the host's response to infection. In recent decades, studies have indicated that temperature abnormalities provide important prognostic information for patients with infections. Some studies have shown that hypothermia occurring in the context of infection is related to increased mortality, whereas fever is related to decreased mortality.9,16,20 Sepsis is considered a highly heterogeneous disease, with significant differences in the type of organism, source of infection, inflammatory response, and pathophysiological mechanisms between patients.21,22 This heterogeneity complicates the implementation of a standardized treatment regimen for all patients. Recent evidence has shown that identifying biologically relevant subgroups of a disease may facilitate personalized therapeutic interventions. Through latent profile analysis, Zhang et al. 23 identified four subphenotypes of sepsis, which demonstrated different clinical outcomes and fluid resuscitation responsiveness. Yang and colleagues 24 identified three longitudinal PaO2/FiO2 subgroups of patients with ARDS with distinct clinical characteristics and outcomes, reflecting the dynamic complexity of ARDS. Additionally, compared with the static Berlin criteria, the longitudinal PaO2/FiO2 subgroups had a greater ability to predict prognosis and response to PEEP. Bhavani and colleagues investigated subtypes of sepsis by using temperature within the first 72 hours. 25 This study identified four subgroups of sepsis: “hyperthermic, slow resolvers,” “hyperthermic, fast resolvers,” “normothermic,” and “hypothermic,” with the “hypothermic” group having the highest mortality. Similarly, Yehya et al. 26 reported the same four temperature subtypes of pediatric sepsis, with the “hypothermic” group presenting lower levels of pro- and anti-inflammatory cytokines. In prior studies, septic patients with hypothermia were categorized together without considering subgroups on the basis of variations in temperature and differences in patient outcomes. In the present study, three longitudinal temperature subphenotypes in hypothermic septic patients with different clinical characteristics and outcomes were identified, capturing the potential complexity of hypothermic status.
Previous studies have shown that hypothermia is significantly associated with adverse outcomes in patients with sepsis. Our findings indicated that, although initially classified as having a poor prognosis due to hypothermia, patients who experience changes in body temperature may provide important insights for understanding hypothermia. In a retrospective study, Thomas-Rüddel et al. 27 reported that 28-day mortality decreased with increasing body temperature over short time intervals in septic patients. However, the elevated mortality risk related to hypothermia was less pronounced after adjusting for confounding variables. With respect to gynecological surgery, Wang et al. 28 reported that the duration of hypothermia upon admission to the postanesthesia care unit (PACU) or ICU is associated with an increased risk of postoperative complications. In the present study, we identified three subclasses of sepsis patients exhibiting varying degrees of initial hypothermia and subsequently different recovery patterns associated with different clinical outcomes. Overall, mild hypothermia and a shorter duration of hypothermia were associated with reduced mortality, whereas severe hypothermia and a prolonged duration of hypothermia were associated with increased short-term mortality.
Our results confirmed the heterogeneity among hypothermic septic patients, concurrently suggesting that distinct classes may possess different adaptive biological values. In a retrospective study, Han et al. 29 categorized hypothermic septic patients into three groups on the basis of delta temperature and reported that a body temperature increase of 1 °C or more after the initial 6 hours was associated with improved 28-day mortality. Furthermore, we employed the GBTM method to fully utilize the data from each time point to describe the evolution trend of hypothermia. A critical consideration in our analysis is the interpretation of the identified subphenotypes, particularly class 1, which comprises a small proportion of the cohort (3.08%). While this group exhibited the lowest baseline temperature, our rationale for retaining it as a distinct class lies in its temporal dynamics rather than its intercept alone. Class 1 was associated with higher severity scores, extended duration of organ support, and increased 7-day mortality, potentially reflecting a sepsis phenotype characterized by multiple organ dysfunction. In contrast, class 3, characterized by mild initial hypothermia and a shorter duration of hypothermia, was associated with improved clinical outcomes. With respect to patients in class 2, although there was an association with a reduction in 7-day mortality compared to class 1, no significant difference was observed in 28-day mortality. We caution that while initial temperature is a dominant factor, the evolution of temperature over the first 48 hours provides additional prognostic value beyond the initial measurement. Therefore, clinicians should pay close attention to temperature trajectories in hypothermic patients and develop a comprehensive plan of treatment.
In a study of the genomic profiling of blood leukocytes, Harmon et al. 11 found that hypothermic septic patients exhibit a unique gene expression profile compared to septic patients presented with fever, characterized by largely similar but also significantly altered leukocyte transcriptomes. These signaling pathways may be related to the modulation of body temperature, disease severity, and early immunosuppression. Additionally, prior basic research has shown that the formation and clearance of neutrophil extracellular traps (NETs) are enhanced at 40 °C, whereas temperatures of 35 °C and 42 °C attenuate this response. 30 These findings underscore the critical importance of identifying and managing hypothermic phenotypes. Future large trials are needed to validate these associations.
Our study had several limitations. First, although this study included patients whose ICU stay of no < 48 hours, we may have excluded critically ill septic patients who died within 48 hours. Future studies with shorter time intervals are needed to validate these findings. Second, as the database spanned over a decade, changes in management strategies for septic patients during the study period, including the use of vasoactive drugs and mechanical ventilation, may have affected the practical relevance of the results. Nevertheless, the single-center design of the study reduced the potential for bias introduced by variability in institutional protocols, as patient management was relatively uniform within the single ICU environment. Third, the retrospective design of the study made it difficult to establish causal relationships. Key treatments such as CRRT and high-dose vasopressors are strongly associated with both hypothermia and mortality. Disentangling whether hypothermia is a direct driver, a passive marker, or a contributor within a vicious cycle requires future studies with granular temporal data and causal methods (e.g. marginal structural models) capable of addressing time-dependent confounding and mediation. Meanwhile, our models cannot distinguish whether rewarming per se confers a survival benefit or whether patients resistant to rewarming represent a biologically severe subgroup with irreversible homeostatic failure. Future research is warranted to clarify whether active temperature management alters prognosis. Specifically, future studies could employ advanced causal inference methodologies—such as target trial emulation, marginal structural modeling, or g-methods—to vigorously investigate the potential therapeutic efficacy of rewarming interventions in patients with “cold sepsis.” Fourth, we were unable to strictly differentiate between endogenous hypothermia (secondary to sepsis pathophysiology) and exogenous hypothermia (environmental exposure) due to the lack of pre-hospital data in the EHR. Future well-designed prospective trials are needed to confirm these results. Fifth, Severe trauma may induce hypothermia. However, the retrospective design made it difficult to precisely differentiate the diagnosis of severe multiple trauma or to ascertain whether hypothermia was present at the trauma scene or developed later due to infection. Finally, our multivariable analysis included both SOFA and SAPS II scores as covariates. Although these scales serve different purposes—SAPS II for global mortality risk prediction and SOFA for organ dysfunction assessment—they share overlapping variables (such as renal function, neurological status, and bilirubin levels). While VIF analysis ruled out significant multicollinearity, the inclusion of both scores may have introduced potential overadjustment. To address this and ensure the stability of our findings, a doubly robust model was employed.
Conclusion
This study revealed three temperature trajectories of septic patients with hypothermia, which presented different clinical characteristics and outcomes. GBTM-based serial temperature evaluation provides a framework for prognosticating distinct subphenotypes of critical illness. These findings need to be validated in prospective studies.
Supplemental Material
sj-docx-1-sci-10.1177_00368504261424390 - Supplemental material for Temperature trajectory in septic patients with hypothermia: When the ice begins to melt—A retrospective cohort study
Supplemental material, sj-docx-1-sci-10.1177_00368504261424390 for Temperature trajectory in septic patients with hypothermia: When the ice begins to melt—A retrospective cohort study by Chang Xu, Keying Ding, Xiaoyang Zhou, Zhaojun Xu, Bixin Chen and Hua Wang in Science Progress
Footnotes
Acknowledgements
Not applicable.
Ethical considerations
This study was conducted in accordance with the Helsinki Declaration of 1975 as revised in 2024. The creation of the deidentified MIMIC database was approved by the Massachusetts Institute of Technology (Protocol No. 0403000206) and the institutional review boards of the Beth Israel Deaconess Medical Center (Protocol No. 2001-P-001699/14). To access this database, one of the authors (Chang Xu) obtained the necessary certification (certification number: 7574829).
Consent to participate
Individual patient consent was not required due to the retrospective and observational nature of the study within this anonymized database.
Consent for publication
Not applicable.
Author contributions
Chang Xu: conceptualization, data curation, formal analysis, methodology, and writing–original draft; Keying Ding: methodology, validation, software, and writing–review and editing; Xiaoyang Zhou: validation, software, formal analysis, writing–review and editing; Zhaojun Xu: supervision, validation, and writing–review and editing; Bixin Chen: methodology, validation, and writing–review and editing; Wang Hua: conceptualization, methodology, supervision, and writing–review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the key discipline of Ningbo No. 2 Hospital (grant number 2023-Y06).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
