Abstract
Objective
To assess the overall survival (OS) and cancer-specific survival (CSS) in patients with different primary locations of hypopharyngeal squamous cell carcinoma (HSCC) and to develop a predictive model incorporating key clinical and treatment variables (including systemic therapy) to serve as a prognostic reference for patients with locally advanced HSCC.
Methods
This retrospective cohort study extracted data for 1,591 patients with locally advanced HSCC from the Surveillance, Epidemiology, and End Results (SEER) database. X-tile software was used to determine optimal cutoff values for continuous variables. Kaplan–Meier analysis compared survival by primary site. Independent risk factors were identified using stepwise Cox regression, and a nomogram was constructed. Model performance was assessed using the concordance index (C-index), area under the receiver operating characteristic curve (AUC), calibration curves, and decision curve analysis (DCA).
Results
OS and CSS differed significantly by primary site. Stepwise Cox analysis identified 13 independent prognostic factors. The nomogram showed a C-index of 0.727 in both training and validation sets. The AUCs for 1-, 3-, and 5-year OS were 0.792, 0.791, 0.788 (training) and 0.780, 0.796, 0.789 (validation). Calibration curves indicated good agreement between predicted and observed outcomes. DCA demonstrated superior net benefit over TNM staging alone.
Conclusion
Prognosis in locally advanced HSCC varies by primary site. The developed nomogram provides moderate prognostic accuracy and may serve as a supplementary tool for risk stratification and clinical discussion, though external validation is warranted.
1. Introduction
Hypopharyngeal squamous cell carcinoma (HSCC) is an aggressive and metastatic malignant tumor. 1 The most common primary site is the pyriform fossa, followed by the posterior region of the cricoid cartilage and the posterior pharyngeal wall, with some cases occurring in the arytenoid epiglottic folds or multisite involvement.2,3 The common characteristics of hypopharyngeal carcinoma originating from different sites include hidden onset location, mild early symptoms, diagnosis at advanced stages, poor differentiation, and high propensity for regional and distant metastases factors that contribute to poor prognosis. Additionally, previous studies showed that the primary tumor location,4–7 tumor size human papillomavirus (HPV) infection status,8,9 postoperative tumor recurrence status with or without treatment with radiotherapy or chemotherapy,5,10 and presence of a second primary tumor are also independent risk factors affecting the prognosis. 11 Therefore, a decision-making tool that can be used to optimize the treatment strategy for patients with hypopharyngeal cancer and improve their prognosis through accurate prediction of survival rates is necessary.12,13 The epidemiological logic has been observed by the National Cancer Institute. The Surveillance, Epidemiological Sciences and End Results (SEER) database is an open-access database of cancer diagnosis, treatment, and survival data, which can provide examples for developing predictive models for tumors, particularly rare tumors.12,14 It should be noted that the SEER database lacks detailed treatment information, and we did not analyze the interaction between tumor site and treatment modality, which limits the ability of the model to guide personalized treatment plans directly.
Previous studies have demonstrated prognostic effects and prediction of hypopharyngeal carcinoma. Zhongyang et al. developed a nomogram for predicting cancer-specific mortality for patients with primary HSCC. 15 Meanwhile, Heng et al. constructed a prognostic column chart for long-term survival outcomes following surgery in patients with HSCC. 6 SEER database research on tumor features, therapy, and survival in patients with HSCC was also available. 16 However, patients with different primary locations may have variable prognoses, and the nomogram is used as a viable approach to predict the incidence of clinical events. 17 Currently, prognostic models based on different hypopharyngeal cancer primary sites are unavailable to accurately predict the long-term prognosis of patients with HSCC. To the best of our knowledge, this is the first study to systematically evaluate epidemiological characteristics, oncological information, treatment-related outcomes, and prognostic influences based on primary site using SEER data and to construct a novel nomogram for predicting OS.
2. Method
2.1. Patients
This study was conducted in accordance with the principles of the Declaration of Helsinki (1975), as revised in 2024. Ethical approval and informed consent were waived as the study utilized de-identified, publicly available data from the SEER database. All patient identifiers were removed prior to analysis. This study was reported in accordance with the STROBE guidelines (https://www.equator-network.org/guidelines/strobe/). 18
We obtained information from the SEER database plus, 17 registries (2000–2019), which accounts for approximately 28% of the US population. Baseline, tumor, and survival information for patients included in this study was included in the SEER database. Because the SEER database contains de-identified, publicly available data, ethical approval and informed consent were not required. Our institutional review board granted exemption for this study. Our study protocol strictly adhered to the SEER database guidelines.
As shown in Figure 1, the inclusion criteria were (a) screening of patients diagnosed with hypopharyngeal carcinoma from 2010 to 2015; (b) stage III and IV tumors in the AJCC 7th edition TNM staging system; (c) patients without distant metastases (M0); (c) patients without distant metastases; and (d) patients with pathologic type of squamous cell carcinoma. And we excluded patients (a) with uncertain survival duration and (b) with malignant tumors that were not the initial primary. Finally, we included 1,591 patients with stages III and IV first-primary carcinoma of squamous cells in the hypopharynx identified between 2010 and 2015. Additionally, all survival data were comprehensive, allowing for additional analysis of their relevance to the survival for patients with locally advanced hypopharyngeal cancer. Flow chart of the inclusion and exclusion criteria of this study.
2.2. Variables
We obtained data on sex, age, race, marital status, median household income, months from diagnosis to treatment, laterality, extent of disease, grade, stage, tumor grade, tumor size, lymph node size, tumor T-stage, tumor N-stage, regional nodes examined, positive regional nodes, lymph node metastasis, surgery, radiation therapy, chemotherapy, and systemic therapy (this variable was designated as the independent variable in the present study, and corresponding data were extracted from the “RX SUMM-SYSTEMIC SUR SEQ” code in the SEER database. This code is defined as: this data item records the sequencing of systemic therapy and surgical procedures given as part of first course of treatment. We dichotomized this variable into “Yes” and “No” categories for statistical analysis, with code 0 assigned to the “No” category and all other codes to the “Yes” category). Lymph node ratio (LNR) is defined as the ratio of pathologically positive lymph nodes to the total number of lymph nodes examined. (‘TNM staging’refers to the 7th edition of the AJCC Tumor-Node-Metastasis staging system, which classifies tumors based on tumor size (T), lymph node involvement (N), and distant metastasis (M). ‘Stage’ specifically refers to the clinical stage derived from TNM staging (e.g., Stage III, IVA, IVB in this study). ‘Staging’ is a general term referring to the process of determining the tumor’s extent using the TNM system or other classification criteria. In this study, all staging-related analyses were based on the 7th edition AJCC TNM staging system.)
To make subsequent analysis of statistics easier, the X-tile program (https://tissuearray.org/) was used to create groups with the best cutoff values for continuous variables. The subsequent group details were the following: age (≤66, 66–76, and ≥76 years), number of months from diagnosis to treatment (0, 1, and ≥2 month), tumor size (≤30, 31–56, and ≥57 mm), regional nodes examined, positive regional nodes (≤2, 3–5, and ≥7), and LNR (<0.05 0.05–0.18, and ≥0.18). In addition, the primary location of HSCC was categorized into six groups: (a) pyriform fossa; (b) posterior cricoid region; (c) aryepiglottic fold; (d) posterior pharyngeal wall; (e) multiple combined primary locations; and (f) hypopharynx, patients whose location was not otherwise specified (NOS). The treatments were categorized into four types based on the SEER code: surgery, radiation, chemotherapy, and systemic therapy.
2.3. Construction and validation of nomogram
Survival analysis of OS and cancer-specific survival (CSS) for different sites of HSCC was performed using Kaplan-Meier curves and log-rank test. We randomly divided 1,591 patients diagnosed with HSCC between 2010 and 2015 into the training (n = 1,114) and validation (n = 477) cohorts at a 7:3 ratio. Univariate Cox proportional hazards regression analysis was applied to the data of patients in the training cohort to screen for potential risk factors affecting OS. Subsequently, based on the Akaike.
Information Criterion (AIC), factors with statistical significance were incorporated into a stepwise Cox regression model to further identify independent prognostic indicators for the survival of patients with locally advanced HSCC. And we calculated the hazard ratios (HRs) and 95% confidence intervals (CIs) of each prognostic independent influencing factors. Then, a nomogram was created using significant variables from the stepwise Cox analysis. Data from the patients in the training group were used to calculate concordance index (C-index) and plot receiver operating characteristic (ROC) curves to evaluate clinical utility. Data from 477 patients in the validation group were used to confirm the predictive model’s discrimination and calibration.
2.4. Statistical analysis
We used R version 4.1.2 software for statistical analysis, and the regplot package was used to construct nomogram. Univariate and stepwise Cox regression analyses were used to identify independent factors on overall survival time. Additionally, Kaplan–Meier curves and log-rank test were used to compare prognostic differences in patients. Unknown categories were retained as separate levels in regression models to avoid bias from complete case exclusion. Statistical significance was determined using two-sided P < 0.05.
3. Results
3.1. Demographic characteristics
Baseline characteristics in patients with hypopharyngeal squamous cell carcinoma (HSCC) at different sites.

Chemotherapy was administered to most patients (76.2%) with locally advanced HSCC; 226 patients (14.2%) were administered both surgical and radiation therapy; 1108 patients (69.6%) underwent radiation therapy alone, but not surgery, and most patients (73.2%) did not receive systemic therapy. The primary cause of death was hypopharyngeal carcinoma (81.9%), followed by heart disease (4.1%), lung and bronchial illnesses (1.8%), and cerebrovascular disorders (0.9%).
3.2. Survival analysis of tumors at different sites
Notably, survival analysis showed OS and CSS were statistically different in locally advanced HSCC at different sites. Patients with locally advanced HSCC originating from the aryepiglottic fold and pyriform sinus showed a relatively good prognosis, whereas those whose primary lesions originated from the posterior wall of the hypopharynx and the overlapping lesion had poorer prognosis (Figure 2). Compared with hypopharyngeal cancer patients whose primary lesions were located in the pyriform sinus, patients with primary lesions in the postcricoid region had no significant difference in OS (Figure 2(a)), but their CSS was significantly shorter (Figure 2(b)). Kaplan-Meier curves of (a) Overall survival and (b) Cancer-specific survival based on different primary sites.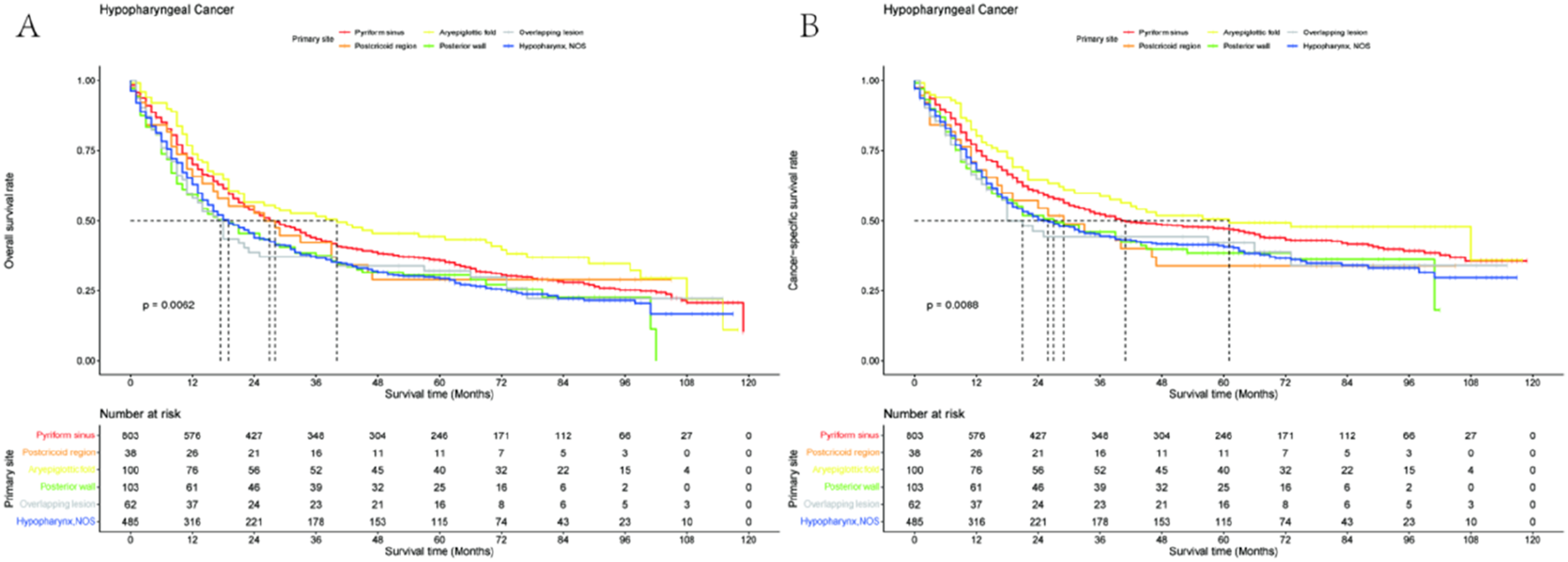
3.3. Predictor screening and nomogram construction
The demographic, tumor, and treatment information for patients with hypopharyngeal squamous cell carcinoma (HSCC) in training and validation cohorts.

The ability of each variable to predict overall survival (OS) in the training cohort using the Univariate Cox proportional hazards regression analysis.

Further analysis incorporating these variables into stepwise Cox hazard regression model showed that independent prognostic influences of the OS of patients with locally advanced HSCC were age, race, marital status, median household income, primary site, laterality, tumor T and N stage, tumor size, lymph node size, positive regional nodes, surgery or radiation therapy, and chemotherapy (Figure 3). The AIC value of the model was 10289.79, and the C-index in the training and validation groups was 0.727 (se = 0.009) and 0.727 (se = 0.016), respectively. Forest plot of factors predicting OS in stepwise Cox proportional risk regression model.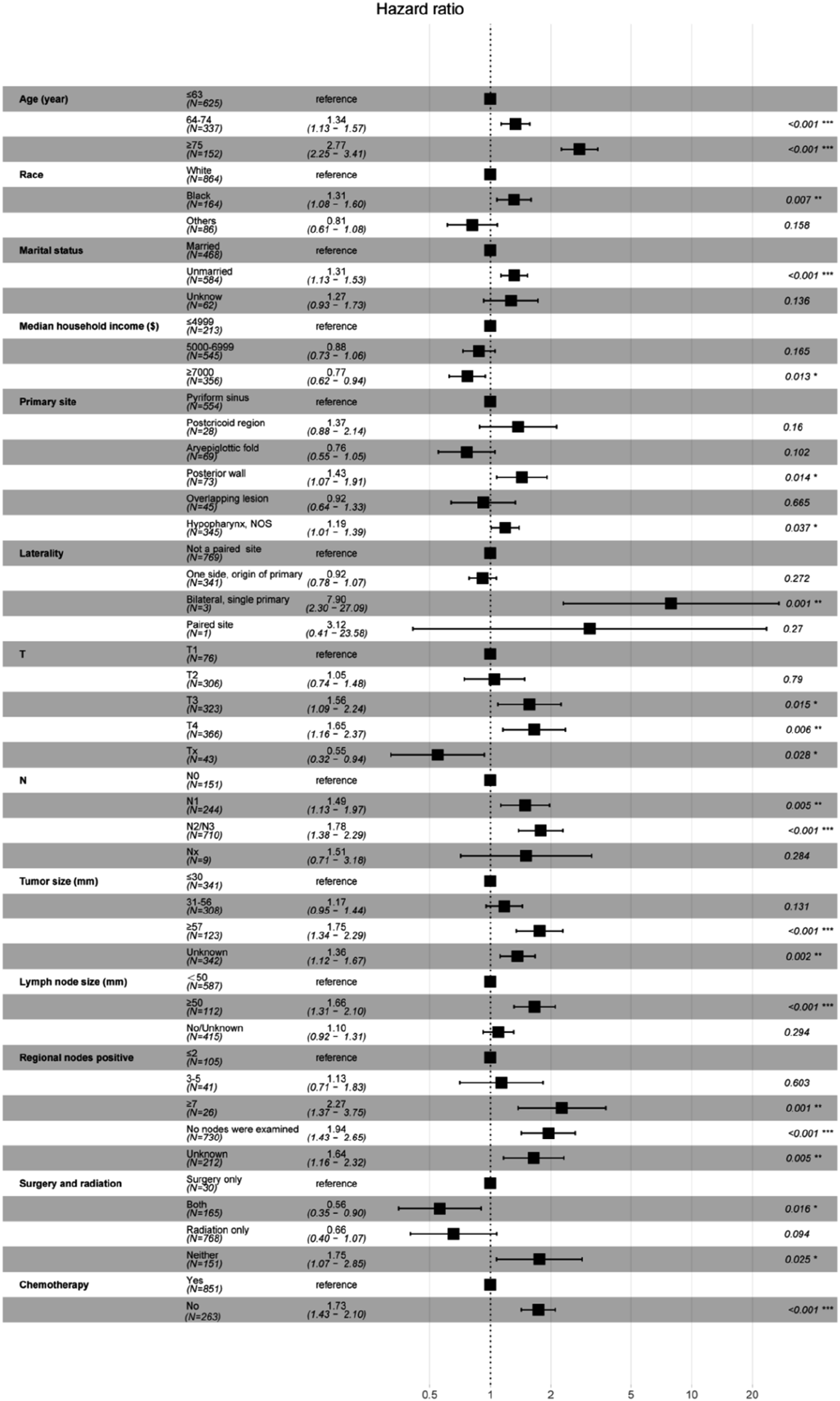
A nomogram (Figure 4) for predicting OS in patients with locally advanced HSCC was created using independent risk factors from stepwise Cox analysis. A risk score was assigned to each inclusion variable. Subsequently, the patient’s total score (range 250-450) was calculated. Finally, the obtained overall risk score was used to calculate 1-, 3-, and 5-year survival probabilities. Nomogram for predicting 1-, 3- and 5-year OS probabilities in patients with hypopharyngeal cancer of different primary sites.
To evaluate the superiority of our nomogram over the traditional AJCC TNM staging system, we separately calculated the C-index of TNM staging for predicting OS. The C-index of TNM staging was 0.562 (95% CI: 0.551–0.573) in the training cohort and 0.546 (95% CI: 0.533–0.559) in the validation cohort, which were significantly lower than the C-index of our nomogram (0.727). This result indicates that our nomogram exhibits higher prognostic accuracy than the AJCC TNM staging system.
3.4. Validation and model performance
We evaluated the clinical utility of the nomogram versus the AJCC TNM staging system using receiver operating characteristic (ROC) curve analysis. The areas under the ROC curve (AUCs) of nomogram for predicting 1-, 3-, and 5-year OS for locally advanced HSCC in the training and testing group were 0.792, 0.791, and 0.788, respectively, and 0.780, 0.796, and 0.789, respectively (Figure 5). ROC curves were constructed for the TNM staging system to predict overall survival (OS). As shown in Supplemental Figure 1, consistent with this, our model demonstrated superior predictive performance for 1-, 3-, and 5-year OS relative to the conventional TNM staging system. Specifically, the areas under the ROC curves (AUCs) for predicting 1-, 3-, and 5-year OS in patients with locally advanced head and neck squamous cell carcinoma (HSCC) were 0.563, 0.608, and 0.591 in the training cohort, and 0.573, 0.657, and 0.681 in the validation cohort, respectively. These findings confirm that our nomogram provides incremental clinical value beyond the conventional TNM staging system. The predictive model of the OS probability at 1, 3 and 5 years in the training and test cohorts was highly predictive. ROC curves used to predict OS probabilities at 1-, 3-, and 5-year points in (a) The training cohort and (b) The validation cohort.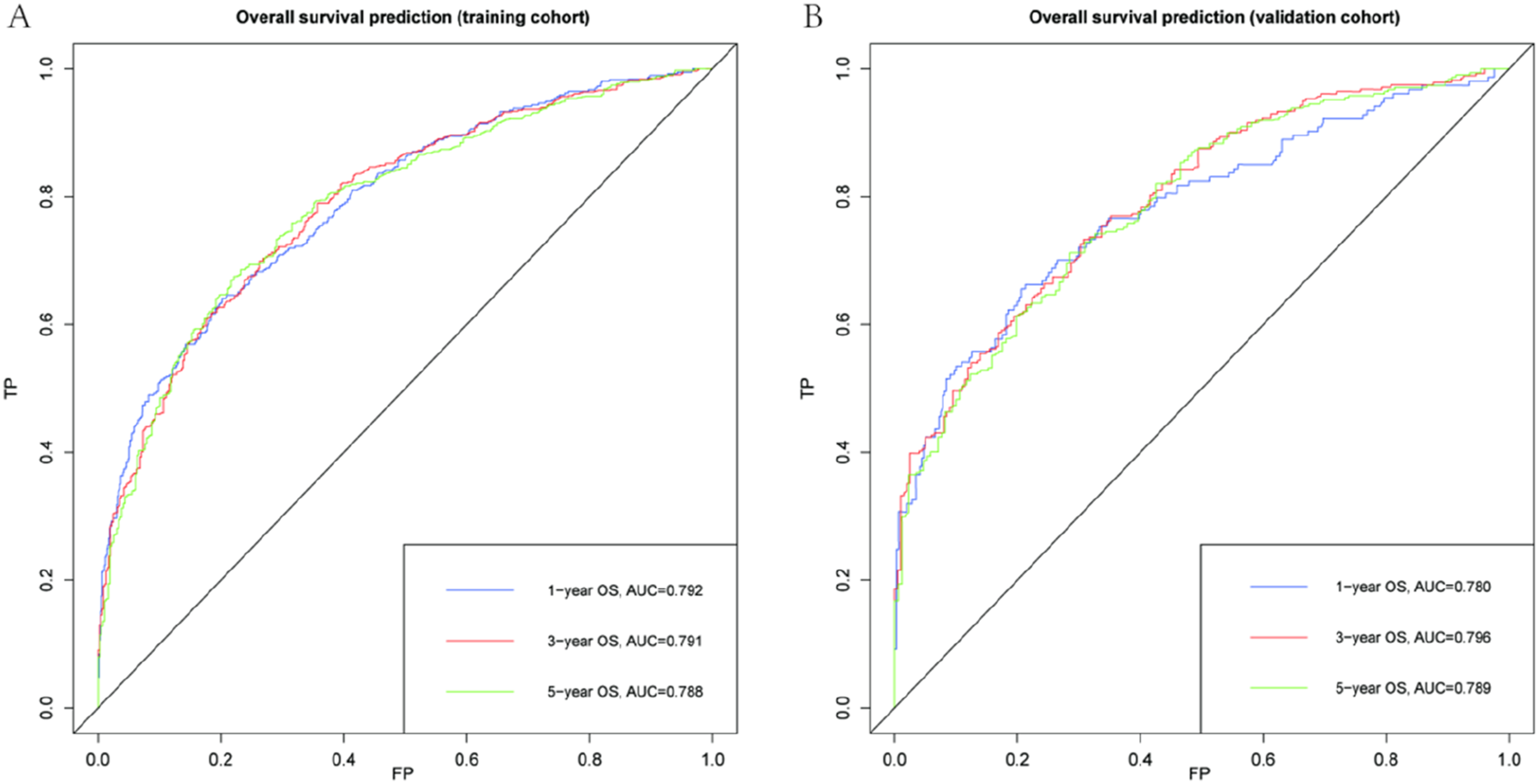
The calibration plots of the 1-, 3-, and 5-year OS predicted by the nomograms in the training and test set showed the best agreement between predictions and observations (Figure 6). Calibration plots of predicted 1-, 3-, and 5-year OS probabilities between column–line plots and actual observations in (a–c) The training cohort and (d–f) Validation cohort.
4. Discussion
Most hypopharyngeal carcinomas are poorly differentiated squamous cell carcinomas with inconspicuous sites and atypical manifestations, making early detection challenging. The submucosal tissues of the pharynx lack natural biological barriers, which easily leads to submucosal infiltration and cervical lymph node metastases. 19 Metastases to the cervical lymph nodes, as well as distant organ metastasis, are found in most patients during the initial consultation; therefore, patients are mostly in advanced clinical stage during consultation, and the prognosis is very poor. Currently, most studies in China are based on single-center clinical data, and multicenter large-sample hypopharyngeal cancer case data and prognostic statistics are lacking. Moreover, the insufficient number of hypopharyngeal cancer cases has led to difficulty in accurately evaluating the clinical prognosis of patients and their influencing factors in many studies. However, this study used the SEER database and had a bigger sample number, which substantially corrected this limitation. To determine cancer prognosis, TNM staging, a significant tumor staging system, is crucial. 20 Nevertheless, patients with the same TNM stage may have varying prognoses. 21 The hypopharynx has abundant lymphoid tissues and is prone to mucosal infiltrative metastasis. Based only on the TNM stage, predicting the outcome of locally advanced HSCC is difficult. 22
Several factors clearly influence the prognosis of hypopharyngeal carcinoma. Although previous studies used nomograms to predict OS of hypopharyngeal cancer, race, tumor size, age, T stage, N stage, initial location, radiation, and chemotherapy were shown as separate risk factors for OS in patients with locally advanced hypopharyngeal cancer. 23 Additionally, locally advanced hypopharyngeal malignancies have separate risk variables for OS related to the original location, radiation, and chemotherapy. 24 However, no model can predict OS in patients with hypopharyngeal carcinoma from different primary sites. In our study, the outlook of individuals with locally advanced HSCC varies considerably depending on the primary locations. Patients with HSCC of the posterior cricoid region and posterior pharyngeal wall, which are relatively rare, had a poorer survival prognosis, which could be attributed to their different tumor origins compared with those with other primary sites, and that the location of the tumor was inconspicuous, and incomplete clearance of the foci of the lesion was not performed, and patients already manifested with lymph node metastases at diagnosis. 25 Hypopharyngeal cancer of the posterior pharyngeal wall is prone to early lymphoid distant metastases, the effect of radiotherapy is poor, and complete resection of the tumor is difficult because of the inconspicuous site, resulting in a poorer prognosis. 26 The AUC values of 0.780-0.796 in this study are consistent with the discriminative power of similar prognostic models for head and neck cancers, 23 indicating good predictive performance that meets the study’s objective of constructing a clinically useful prognostic model. In summary, although studies on hypopharyngeal cancer are limited, the prognosis of hypopharyngeal cancer varies among different primary sites. This study analyzed the impact of location on the long-term outlook of hypopharyngeal carcinoma, which improves our comprehension of the illness and provides a basis for the management of patients with locally advanced hypopharyngeal cancer in different locations.
The Cox proportionality risk model was used to determine the stand-alone risk factors included in the study variables. Patients who were older, unmarried, whose primary tumor originated from the posterior pharyngeal wall, bilateral, single primary, tumor stage T3/4, tumor stage N1/2/3, larger tumor, more lymph node metastases, and who never underwent surgery, radiotherapy, or chemotherapy had a worse prognosis.
A recent study reported that the prognosis of elderly patients with HSCC was worse as their age increases. 22 Our study’s multivariate evaluation came to an analogous conclusion, with a risk ratio of 1.947 (95% CI, 1.503–2.523, P <0.001) for longer OS in patients <65 years, which might be due to elderly patients having more illnesses, lower life expectancy, and are treated more cautiously. Zheng et al. showed that the prognosis worsens as the T-stage becomes more advanced in patients with HSCC. 27 We also found that tumor size T-stage as an independent risk factor for OS in patients with hypopharyngeal carcinoma impacts prognosis. Currently, the treatment of hypopharyngeal cancer is multidisciplinary, multimodal, and comprehensive based on surgery and despite previous studies suggesting that radical one-stage surgery (PS) may provide better survival outcomes for patients with HSCC.28,29 Although previous studies have shown that PS may provide better survival outcomes for patients with HSCC, our study showed that surgery alone is far from ideal, and patients who receive radiation alone for locally advanced hypopharyngeal carcinoma have a poor prognosis. Our study suggests that concurrent chemoradiotherapy is widely accepted as the main treatment modality for HSCC, 30 which may improve OS by decreasing the number of distant metastases of late-stage tumors. A combination of radiation therapy and chemotherapy enhances patient survival compared with radiation therapy alone. 31 Chemotherapy has been shown to improve patient survival when combined with radiation therapy, contrasted with radiation therapy alone. Moreover, patients with head and neck squamous cell cancer can tolerate chemotherapy well. 32 In patients with locally advanced hypopharyngeal cancer, OS, local control, and metastasis-free survival after induction of chemotherapy were not inferior to those after surgery, 33 indicating the importance of comprehensive treatment, which was consistent with our findings. Furthermore, earlier research found a substantial association between lymph node clearance and LNR for surgery and the prognosis of patients with HSCC. 34 However, in this study, lymph node dissection status and lymph node ratio (LNR) were not identified as independent prognostic factors for locally advanced hypopharyngeal cancer. This result is likely associated with incomplete data: 1,343 patients (84.4%) were categorized as having “no lymph nodes examined” or unknown LNR status, and this data limitation reduced the accuracy of assessing patients’ overall survival (OS) using LNR. Both metastatic lymph node dissection and lymph node ratio were strongly linked with the outcome of patients with HSCC. The number of positive metastatic lymphatic nodes is a stand-alone risk factor for patient OS, with the prognosis being worse as the number becomes higher, which was consistent with a previous study. 35 Meanwhile, our study showed that marital status and financial position were associated with OS in patients with HSCC (Figure 4), demonstrating the influence of financial level, social standing, and marital emotional support on the prognosis to some extent.36,37
In conclusion, the construction of a column–line diagram specialized for hypopharyngeal carcinoma of various primary sites is crucial, allowing the assessment of survival, clinical evaluation, and treatment methods of patients with HSCC. 12 The prediction model’s calibration curve and C-index showed high accuracy and dependability. Our nomogram demonstrated a higher C-index (0.727) compared to the AJCC 7th edition TNM staging system (0.653), suggesting improved prognostic discrimination when incorporating primary site, demographic, and treatment-related variables. Furthermore, we used AUC to measure the prediction model’s discriminative power, which proved the clinical practicality of this novel column–line diagram based on the AUC results.
A major limitation of this study is that the nomogram was only internally validated using a split-sample approach, and no external validation was performed. External validation using independent datasets from different regions or institutions is needed to further confirm the generalizability and clinical applicability of the model. We plan to collaborate with other centers to collect relevant data for external validation in future research. It should be noted that SEER lacks granular treatment details (e.g., radiation dose, chemotherapy regimens), and our model does not analyze interactions between tumor site and specific treatments; thus, its utility is primarily prognostic rather than prescriptive. Another limitation is that the SEER database lacks information on potential confounding variables such as smoking status, HPV infection status, comorbidities, and treatment intensity. These unmeasured variables may affect the survival outcomes of patients and confound the observed survival differences between different tumor subsites. Future studies with more comprehensive data collection are needed to address this issue.
Because the occurrence of hypopharyngeal cancer is uncommon among head and neck cancers, few studies have focused on the clinical traits and outlook of locally advanced hypopharyngeal cancer, despite the poor prognosis. However, the following limitations exist: (1) Selection bias was inevitable and may restrict the utility of our prediction model because all research data were derived from retrospective examination of the SEER database; (2) the SEER database lacks precise clinical information, such as surgery techniques, radiation regimens, and chemotherapy schedules, which may indicate additional unidentified factors; (3) patients with locally advanced hypopharyngeal cancer might have an improved prognosis than the patients in this research because surgical techniques, 38 immunotherapy, 39 and incorporation of other modalities also improved. External confirmation is also essential to prove its clinical performance.
Despite the good internal validation results, independent external validation is still needed to confirm the model’s generalizability. Our research team is currently establishing collaborations with multiple regional medical centers to collect an independent dataset of locally advanced laryngeal squamous cell carcinoma for external validation, and the relevant findings will be reported in subsequent research.
5. Conclusion
In conclusion, the SEER database, which features a large sample size, accurate pathologic diagnosis, and thorough follow-up, served as the foundation for this investigation. Our research showed that the prognosis of locally advanced hypopharyngeal carcinomas arising from different anatomical locations varied. In addition, age, race, marital status, median household income, primary site, laterality, tumor T and N stages, tumor size, lymph node size, positive regional nodes, surgery or radiation therapy, and chemotherapy were independent factors influencing OS in patients with locally advanced hypopharyngeal carcinoma. Based on these criteria, we constructed a column–line diagram that can potentially be a prediction model capable of effectively predicting the prognosis of patients with locally advanced hypopharyngeal carcinoma in various sites. This model can serve as a prognostic assessment tool to help healthcare practitioners better evaluate the prognosis of patients with locally advanced hypopharyngeal carcinoma from different primary sites, providing a reference for clinical management.
Supplemental material
Supplemental material - Comparison of survival and establishment of prognostic models for patients with locally advanced squamous cell carcinoma of the hypopharynx at different sites: A retrospective study based on the SEER database
Supplemental material for Comparison of survival and establishment of prognostic models for patients with locally advanced squamous cell carcinoma of the hypopharynx at different sites: A retrospective study based on the SEER database by Han Zhu, Shengteng Shao, Ming Yang, Hua Zhang in Science Progress
Footnotes
Author note
Acknowledgements
The authors thank the SEER program for providing the data. English language editing was performed by a professional editing service.
Ethical considerations
The information in the SEER database is public and anonymous, ethical clearance and informed consent are not required.
Author contributions
H. Zhang, H. Zhu and S.T. Shao contributed to the study conception and design. Data analysis were performed by S.T. Shao, H. Zhu and M. Yang. The first draft of the manuscript was written by H. Zhu and S.T. Shao. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data from the SEER database is free and publicly available and the original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
