Abstract
The necessity of postmastectomy radiotherapy (PMRT) for patients whose initially positive lymph nodes become node-negative (ypN0) after neoadjuvant therapy (NAT) is uncertain. This study analyzed data from the Surveillance, Epidemiology, and End Results database to evaluate PMRT's effect on these patients. Women with unilateral breast cancer who achieved ypN0 status post-NAT from 2010 to 2019 were categorized into PMRT and non-PMRT groups. Propensity score matching (PSM) minimized confounding factors. Statistical tests and multivariate analysis identified survival prognostic factors, while Kaplan-Meier curves and forest plots assessed survival outcomes. The study involved 699 cases, with 458 receiving PMRT and 241 not. After matching, 194 patient pairs were examined. Multivariate analysis revealed stage III disease (hazard ratio: 2.06; 95% CI: 1.12-3.79, p = 0.02) and lack of PMRT (hazard ratio: 2.48; 95% CI: 1.31-4.62, p = 0.01) as independent survival risk factors. PMRT significantly improved overall survival (hazard ratio: 0.43; 95% CI: 0.26-0.72, p < 0.001), especially in patients with clinical node status cN+ (cN1, hazard ratio: 0.47; 95% CI: 0.25-0.88, p = 0.016; cN2-3 hazard ratio: 0.35; 95% CI: 0.15-0.86, p = 0.017, respectively), grade 3 tumors(hazard ratio: 0.47; 95% CI: 0.25-0.88, p = 0.016), stage III disease (hazard ratio: 0.47; 95% CI: 0.26-0.83, p = 0.007), and triplenegative breast cancer (hazard ratio: 0.15; 95% CI: 0.05-0.42, p < 0.001). However, it did not significantly benefit those with grade 1-2 tumors, stage II disease, HER2-positive, or hormone receptor-positive/HER2-negative tumors. The study suggests PMRT may not be necessary for these groups, particularly for HR-positive/HER2-negative grade 1-2 with stage II disease, due to limited short-term benefits.
Introduction
Breast cancer is the most common cancer among women, with incidence rates increasing annually. 1 Despite significant advancements in patient care and treatment, which have resulted in a 5-year survival rate of up to 90%, 2 several challenges remain unresolved.
Neoadjuvant therapies (NATs), including neoadjuvant chemotherapy (NAC), endocrine therapy, and targeted therapy, have become increasingly common for the treatment of breast cancer prior to surgery. Both the European Society for Medical Oncology and the National Comprehensive Cancer Network (NCCN) consistently recommend the implementation of NAC in patients with hormone receptor (HR)-positive, human epidermal growth factor-2 (HER2)-negative tumors larger than 5 cm or those involving more than three lymph nodes.3,4 Furthermore, a substantial proportion of patients diagnosed with triple-negative breast cancer is recommended to undergo NAC, while for HER2-positive patients, the administration of NAC in combination with targeted therapy is advised.3,4 Mamounas et al. conducted a combined analysis of the NSABP B-18 and B-27 trials to investigate predictive factors for locoregional recurrence (LRR) after NAC. They found that pathological response (e.g. pathologic complete response (pCR)) and axillary lymph node status had a significant impact on LRR risk. The study indicated that patients who achieved pCR and had negative axillary lymph nodes had a significantly reduced risk of LRR, suggesting the potential for reduced radiotherapy use in patients with favorable pathological responses. However, the study did not explore in depth the specific value of postoperative radiotherapy in node-negative (ypN0) patients. 5
Postmastectomy radiotherapy (PMRT) represents a critical therapeutic strategy for patients receiving NAT, as it has the potential to improve locoregional control and overall survival (OS). 6 Nonetheless, PMRT is associated with side effects that can adversely impact patients’ quality of life.7,8 Therefore, the selection of appropriate candidates for PMRT following NAT is of paramount importance. NCCN guidelines recommend that decisions related to administration of adjuvant radiotherapy for patients receiving NAT should be made based on maximal stage (i.e. clinical/anatomic stage, tumor characteristics) at diagnosis (before preoperative systemic therapy), and pathologic stage at definitive surgery (after preoperative systemic therapy). 4 However, some breast cancer radiotherapy guidelines lack explicit recommendations concerning the application of PMRT after NAT. 9 Despite previous studies on PMRT, there remains a lack of high-level evidence guiding its use in ypN0 patients after NAT. Our study aims to fill this gap by leveraging Surveillance, Epidemiology, and End Results (SEER) data to evaluate its impact on OS. Studies have demonstrated that approximately 20–40% of patients with clinically node-positive breast cancer who undergo NAC exhibit pathological ypN0 upon subsequent examination, and ypN0 has been identified as an independent prognostic marker indicative of favorable outcomes in breast cancer patients.10,11 However, the necessity of PMRT for patients who experience positive lymph node conversion to ypN0 following NAT remains uncertain.
This study focused on patients diagnosed with clinically node-positive breast cancer who demonstrated pathologically negative nodes after NAT. Utilizing data from the SEER database, the study examined the relationship between clinicopathological characteristics and prognostic factors. Additionally, it investigated whether patients within this category experienced a significant survival benefit from PMRT.
Materials and methods
Data acquisition and patient selection
The cases included in this study were from the SEER database. The analysis included women diagnosed with primary unilateral breast cancer between 2010 and 2019 who had no prior history of cancer. The patients who achieved nodal downstaging to ypN0 following NAT were included. Patients who underwent breast-conserving surgery and did not undergo mastectomy procedures were excluded from the analysis. Furthermore, cases involving pathological tissue types such as sarcoma, carcinosarcoma, or pseudo-sarcoma-like carcinoma, as well as those with incomplete basic information or pathological features of the tumor, were excluded. Patients with an unknown radiotherapy status were also excluded from the analysis.
We used SEER*Stat software (version 8.4.0.1) to generate a comprehensive case listing including the following variables: age at diagnosis, race, marital status, tumor site, tumor location, pathological type, degree of tumor differentiation, surgical modality, clinical stage, T stage, N stage, estrogen receptor (ER) status, progesterone receptor (PR) status, human epidermal growth factor receptor-2 (HER-2) status, molecular subtyping, history of NAC, history of radiation therapy, follow-up duration, and survival status. The follow-up period extended until November 2022, with a median follow-up duration of 49 months. The primary endpoint of this study was to determine OS, defined as the duration from the pathological diagnosis of breast cancer to the date of death.
Statistical methods
The data was analyzed using R-studio statistical software and SPSS 26. A 1:1 propensity score matching (PSM) was conducted with a caliper value of 0.02 to mitigate the effects of data bias and confounding variables. Categorical variables were analyzed using the chi-square test or Fisher's exact test. Multivariate analysis was conducted to identify independent prognostic factors influencing survival. PSM (1:1 caliper = 0.02) was selected over other methods (e.g. inverse probability weighting) to directly balance covariates between groups, as it reduces selection bias in observational studies. Missing variables were addressed using multiple imputation with predictive mean matching. Survival outcomes were assessed using the Kaplan–Meier survival curve, and subgroup analyses were presented as a forest plot. A p-value of less than 0.05 was considered statistically significant.
Results
A total of 699 breast cancer cases were included in this study, with 458 cases in the PMRT group and 241 cases in the non-PMRT group, based on the specified inclusion and exclusion criteria. The flowchart is shown in Figure 1.

Inclusion case screening flowchart.
Comprehensive demographic, clinicopathological, treatment, and survival data were obtained from all participants. The demographic and clinicopathological characteristics of the eligible patients are presented in Table 1. Analysis revealed that the decision to undergo PMRT was independent of variables such as age, race, marital status, tumor location, tumor position, histological type, degree of tumor differentiation, HR and HER2 status, and molecular subtyping of breast cancer. However, a statistically significant association was observed between the decision to undergo PMRT and clinical stage, T stage, and N stage (p < 0.05) (Table 1). The selection of PMRT was more common among patients with higher T staging, lymph node metastasis. Kaplan–Meier curves indicated that PMRT did not achieve statistical significance (p = 0.152) (Figure 2).

Kaplan–Meier (K–M) curves before propensity score matching (PSM).
Demographic and clinical characteristics in breast cancer patients before PSM.

Note: PMRT: postmastectomy radiotherapy; HR: hormone receptor; HER2: human epidermal growth factor-2; PSM: propensity score matching.
An asterisk (*) denotes statistical significance.
After performing PSM on the baseline data of the two groups and excluding 311 patients without corresponding propensity score, a total of 194 matched pairs of patients were obtained. The p-values for all covariates after matching exceeded 0.1, indicating successful PSM (Table 2).
Demographic and clinical characteristics in breast cancer patients after PSM.

Note: PMRT: postoperative radiotherapy; HR: hormone receptor; HER2: human epidermal growth factor-2; PSM: propensity score matching.
We next performed univariate and multivariate analysis to identify the risk factors for OS (Table 3). Univariate analysis revealed that unmarried status (hazard ratio: 1.68; 95% confidence interval [CI]: 1.03–2.75, p = 0.03), stage III disease (hazard ratio: 2.15; 95% CI: 1.17–3.95, p = 0.01) and the absence of PMRT (hazard ratio: 2.36; 95% CI: 1.23–4.54, p = 0.009) were risk factors and multivariate analysis identified stage III disease (hazard ratio: 2.06; 95% CI: 1.12–3.79, p = 0.02) and the absence of PMRT (hazard ratio: 2.48; 95% CI: 1.31–4.62, p = 0.01) as independent risk factors for breast cancer patients in the study.
Univariate and multivariate analysis comparing overall survival among selected variables in breast cancer patients after PSM.

Note: 95% CI: 95% confidence interval; HR: hormone receptor; HER2: human epidermal growth factor-2; PMRT: postmastectomy radiation therapy; reference: control group, within-group HR = 1; NA: represents any values that are “not available” or “missing”; PSM: propensity score matching.
We subsequently conducted a survival analysis on these patient cohorts and found that PMRT significantly improved OS (hazard ratio: 0.43; 95% CI: 0.26–0.72, p < 0.001) (Figure 3). The improvement in OS with PMRT was observed across most subgroups (Figure 4). Specifically, PMRT did not improve OS in patients with grade 1 and grade 2 tumors (hazard ratio: 0.44; 95% CI: 0.16–1.2, p = 0.11); however, a significant improvement in OS was observed in patients with grade 3 tumors (hazard ratio: 0.47; 95% CI: 0.25–0.88, p = 0.016) (Figure 5). Patients with stage II disease did not derive benefit from PMRT (hazard ratio: 0.33; 95% CI: 0.1–1.08, p = 0.067), whereas those with stage III disease did (hazard ratio: 0.47; 95% CI: 0.26–0.83, p = 0.007) (Figure 5). Additionally, patients with cN1 and cN2–3 breast cancer both experienced benefits from PMRT (hazard ratio: 0.47, 95% CI: 0.25–0.88, p = 0.016; hazard ratio: 0.35; 95% CI: 0.15–0.86, p = 0.017, respectively) (Figure 5). Furthermore, a subgroup analysis was conducted for HER2-positive tumors, hazard ratio-positive HER2-negative tumors, and triple-negative breast cancer. The results indicated that patients with triple-negative breast cancer significantly benefited from PMRT (hazard ratio: 0.15; 95% CI: 0.05–0.42, p < 0.001), whereas patients with other molecular subtypes did not exhibit a similar benefit. For example, patients with grade 1–2 tumors, stage II disease, HER2-positive tumors, and hazard ratio-positive/HER2-negative tumors did not show a statistically significant improvement in OS with PMRT.

Kaplan–Meier (K–M) curves after propensity score matching (PSM).
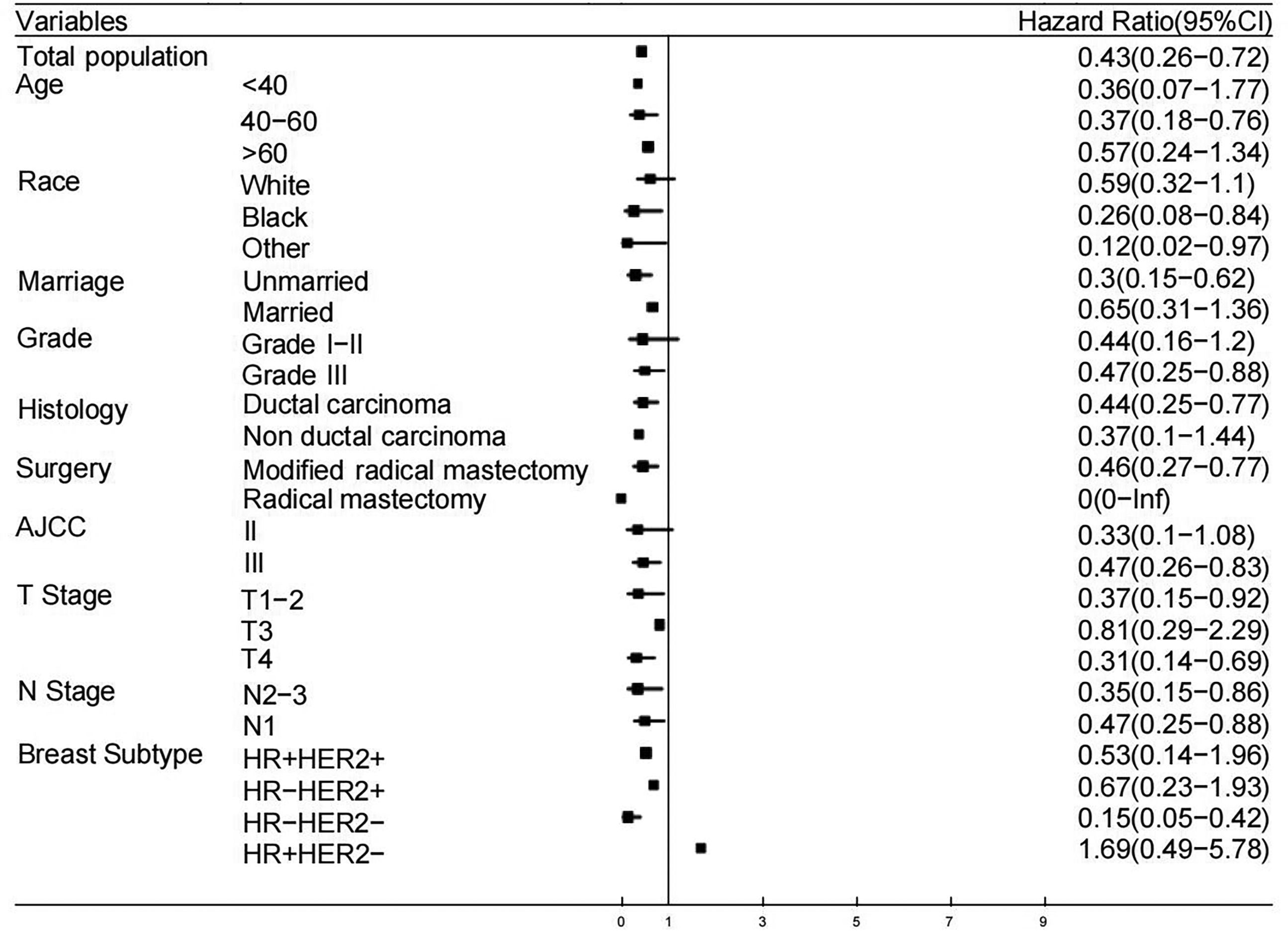
Forest plot of the effect of postmastectomy radiotherapy (PMRT) on overall survival (OS) improvement.
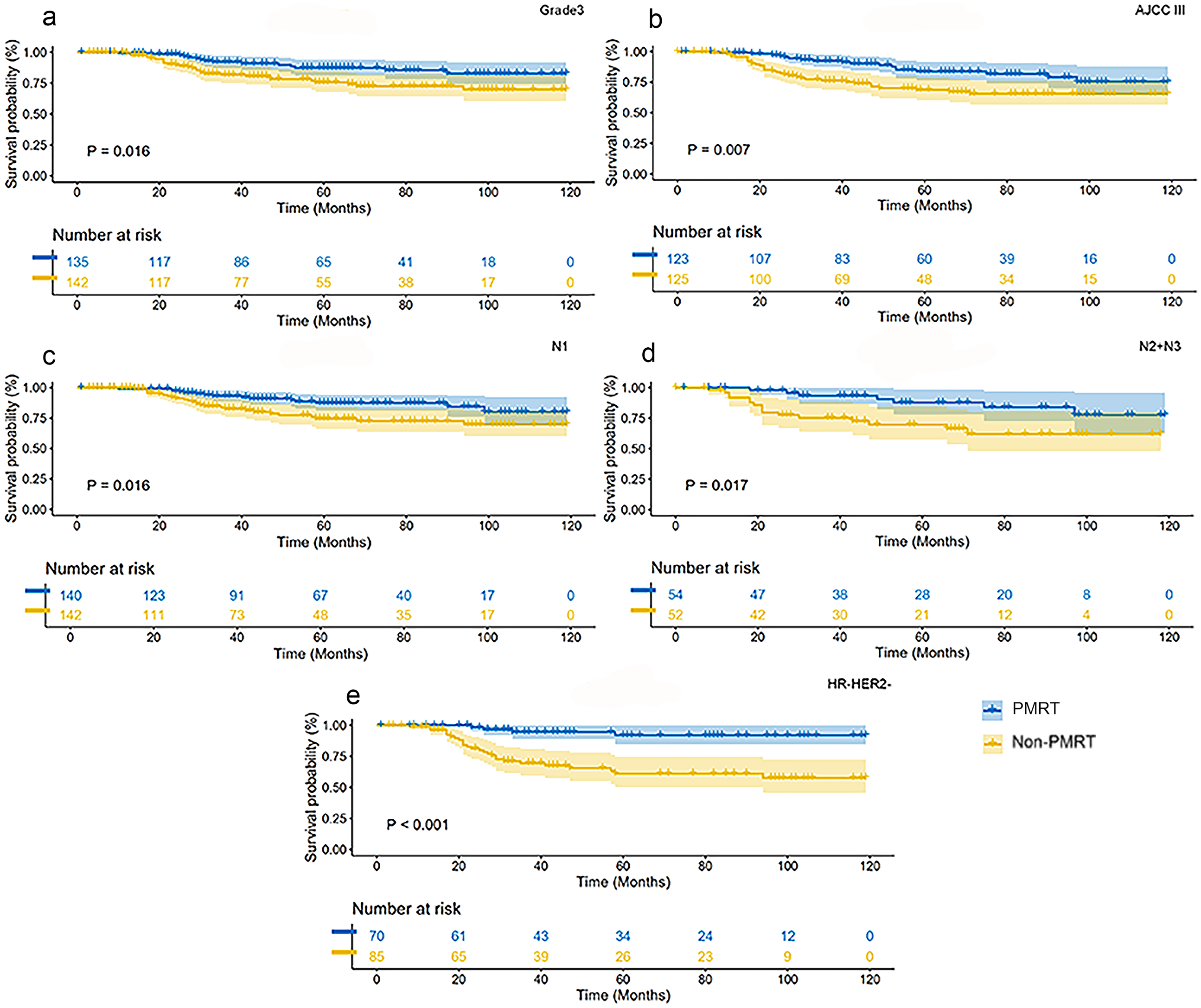
Kaplan–Meier survival curves of postmastectomy radiotherapy (PMRT) and non-PMRT groups in patients with grade 3, AJCC stage III, N1, N+, and hormone receptor (HR)–human epidermal growth factor-2 (HER2) status after propensity score matching (PSM); the blue curve represents PMRT, and the yellow curve represents non-PMRT.
Discussion
Adjuvant radiotherapy is an important therapeutic scheme for breast cancer, which can significantly reduce LRR and improve survival. 4 However, high-quality evidence to guide PMRT decisions in the patients following NAT, especially those with ypN0 status, remains lacking. Here, we presented a comprehensive analysis of the impact of PMRT on OS in patients treated with NAT who achieve ypN0 status, utilizing data from the SEER database.
To mitigate selection bias, PSM was employed, resulting in more accurately matched baseline characteristics between the PMRT and non-PMRT groups. Multivariate Cox regression analysis identified the absence of PMRT as independent prognostic factor for poor survival. Concurrently, our survival analysis of the patients in this study demonstrated that PMRT was associated with improved OS. This positive association was observed across most subgroups, including the patients with N1 disease. For patients with N2 and N3 disease, the majority of studies corroborate the benefit of PMRT.12,13 Consistent with our findings, Rusthoven et al. evaluated the impact of PMRT on women with cT1–3 cN1 M0 breast cancer treated with NAC using data from the National Cancer Database and demonstrated that PMRT was independently associated with improved OS among ypN0 (hazard ratio: 0.729, 95% CI: 0.566–0.939, p = 0.015). 14 Another retrospective study suggested that PMRT improved LRR-free survival (a 5-year rate of 94.7% vs. 72.9%), distant metastasis-free survival (a 5-year rate of 92.8% vs. 75%), and disease-free survival (a 5-year rate of 92.9% vs. 62.5%) in the subgroup of 53 patients who achieved ypN0 status. 15 Conversely, several other studies have reported conflicting results, suggesting that PMRT may not be necessary for ypN0 breast cancer patients.16–19 Possible reasons for this discrepancy in findings may stem from variations in the sample sizes of the study populations. While awaiting prospective results, the present analysis supports the application of PMRT in patients with ypN0 status.
Our analyses identified stage III as an independent prognostic factor for patients who initially presented with clinically positive nodes and subsequently converted to ypN0. Subgroup analysis further revealed that patients with stage III significantly benefited from PMRT, whereas those with stage II did not. The findings aligned with previous research indicating that PMRT improved local control even in patients with stage III disease who achieve a pCR to NAC.12,20,21 A prospective registry study (RAPCHEM, BOOG 2010-03) included patients with clinical stage T1-2N1 (stage II) breast cancer who underwent primary chemotherapy followed by breast and axillary surgery, conducted between January 1, 2011 and January 1, 2015. The 5-year LRR rate was 2.1% (0.9–4.3) in the ypN0 group, supporting the oncological safety of de-escalating locoregional radiotherapy based on LRR risk. 22 However, two other retrospective studies demonstrated that PMRT might not be necessary for ypN0 patients after NAC, irrespective of clinical stage.10,19 It is important to note that the patients included in these studies were treated between 1990 and 2009, which could influence the results. Based on the aforementioned findings and those from previous studies, we recommend that stage III patients with ypN0 status after NAT should receive PMRT.
Additionally, it was observed that PMRT did not confer a survival benefit in certain subgroups, such as those with HER2-positive tumors and HR-positive/HER2-negative tumors. Most previous studies on PMRT were conducted during an earlier treatment era, which did not incorporate molecular analysis.10,12 Several studies have since underscored the impact of molecular subtypes on treatment responses, including adjuvant radiotherapy.23,24 However, only a limited number of studies have investigated the influence of breast cancer subtypes on PMRT following NAT. A retrospective study, which reviewed 682 patients with clinical stage II–III breast cancer who underwent NAC and mastectomy from 2013 to 2017, demonstrated that PMRT was associated with improved LRR-free survival in patients with triple-negative breast cancer (p = 0.03). 25 However, a study from a Korean cohort reported that PMRT conferred no additional survival benefits for any molecular subtype in patients who achieved ypN0 status following NAC and mastectomy. 26 Our findings indicated that patients with grade 1 and grade 2 tumors, as well as those with non-ductal invasive tumors, experience less pronounced short-term benefits from PMRT. Further prospective studies are warranted to validate these observations.
The NRG Oncology/NSABP B-51/RTOG 1304 study is a pivotal prospective phase III randomized controlled trial designed to evaluate whether breast cancer patients who achieve nodal clearance (ypN0) after NAC require comprehensive postoperative regional radiotherapy. 27 Its significance lies in being the first study to provide high-quality evidence on the feasibility of radiotherapy de-escalation after NAC. If the results confirm that radiotherapy can be safely omitted, it will significantly reduce treatment-related toxicity, improve patients’ quality of life, and offer critical guidance for clinical practice. 28
This study had several limitations. First, the database reports OS without including other survival outcomes such as local or distant recurrence. Second, the database lacks information regarding systemic therapy, including the selection of chemotherapy or targeted regimens and the duration of treatment. Third, selection bias cannot be entirely eliminated in this retrospective study. Fourth, a notable limitation of this study is the relatively short median follow-up period of 49 months, which may be insufficient to fully capture the long-term survival benefits, especially in HR-positive patients. Lastly, due to the ethnic diversity of the SEER database, our findings may not be generalizable to regions characterized by high levels of ethnic homogeneity. Our study is limited by the absence of recurrence data and systemic therapy details, which may confound survival outcomes. Additionally, the retrospective design and short median follow-up (49 months) necessitate cautious interpretation. Future randomized trials (e.g. NRG Oncology/NSABP B-51) are needed to validate our findings and explore long-term outcomes. Our study highlights the potential benefit of PMRT in high-risk ypN0 patients but suggests a more selective approach for low-risk groups. Future studies with recurrence data and prospective validation are needed to refine treatment guidelines.
Conclusions
This analysis identified the prognostic value of PMRT in patients who underwent NAT and achieved ypN0 status. The majority of subgroups exhibited improved OS with the administration of PMRT. However, patients with favorable prognostic characteristics (such as grade 1–2 tumors, stage II disease, HER2-positive, and HR-positive/HER2-negative tumors) may derive limited short-term benefit from PMRT. These findings warrant further validation in prospective studies with longer follow-up periods. These findings suggest that PMRT may be unnecessary for these specific patient cohorts. Further studies with longer follow-up are needed to confirm these observations.
Footnotes
Author contributions
Xiaoyu Wang, Xiaolin Zhao, and Pian Cheng: conception and design of the analysis. Xiaoyu Wang, Xiaomeng Zou, Weike Zhang, and Jie Liu: collection of the data. Pian Cheng, Xiaomeng Zou, Weike Zhang, and Jie Liu: contribution of the data and analysis of tools. Xiaoyu Wang, Xiaolin Zhao, and Weike Zhang: perform of the analysis. Xiaoyu Wang, Xiaomeng Zou, and Jie Liu: conception of the manuscript, reviewing and editing of the manuscript, and final approval of the version to be published.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Shandong Province (ZR2020MH210) and the Jinan Medical and Health Science and Technology Development Project (202303110816).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The data in this research were obtained from the SEER database following approved guidelines. The information on patients had been studied by the US Department of Health and HumanServices. The data are publicly available and deidentified after permission. We confirm that the research was performed in accordance with the principles stated in the Declaration of Helsinki. No personally identifying information was used in the study, which eliminated the requirement for Institutional Review Board approval or informed patient consent.
