Abstract
Solitary fibrous tumor (SFT) generally arises from different soft tissues. It was previously known as hemangiopericytoma. Intracranial SFT is a distinct mesenchymal type of primary brain tumor with an intermediate biological potential. This report described the manifestation and management of a woman (in her late 60 years of age) with a rare extra-axial SFT of cerebellum. SFT has some neuroimaging characteristics that mimic meningioma. We have reported its distinct histopathological findings. Importantly, SFT demonstrates more aggressive local behavior and a significantly higher rate of recurrence than meningioma. In the present case, the skull bone was eroded and eventually penetrated with focal destruction by the tumor. We completely excised the tumor and subsequently reconstructed the skull bone with an artificial alloplastic titanium mesh. This uncommon case report emphasized the value of accurate SFT diagnosis and demonstrated outcomes achieved by complete surgical resection with bony reconstruction.
Introduction
Solitary fibrous tumor (SFT) consists of spindle cells in a collagen-rich tissue with specific immunohistochemical (IHC) traits. SFTs can be identified in the central nervous system (CNS) and various soft tissues. Prior large-scale studies of patients with SFT in the CNS showed the brain to be affected in majority of the cases. 1 Reported intracranial SFTs were mostly located in the tentorium (15.6%), followed by frontal convexity (14.8%), cerebellopontine angle (CPA) (9.6%), parietal lobe (8.1%), and non-CPA posterior fossa (7.4%). 1 To the best of our knowledge, SFT of cerebellum was rarely discussed.2–4 Cerebellar SFTs with extensive skull destruction which require reconstruction are exceedingly rare. Herein, we present a unique case of SFT arising from the cerebellar convexity.
Case presentation
The patient was a Taiwanese woman in the late 60 years age range, and suffered from insidious onset of dizziness, nausea, and poor appetite daily for several days. Due to persistent symptoms, the patient visited the Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, New Taipei City, Taiwan, and was admitted on January 2024. The patient provided signed consent to the treatment. Initial preoperative neuroimaging studies included a brain computed tomography scan (Figure 1(a)) which showed a left occipital skull defect destructed by a cerebellar tumor. Further gadolinium contrast enhanced magnetic resonance (MR) imaging scan (Figure 1(b)) exhibited a well-defined mixed solid and cystic extra-axial mass measuring approximately 4 cm in diameter above the left side of the cerebellum. The tumor was avidly enhanced with mass effect. It compressed and displaced the fourth ventricle. Diagnostic digital subtraction angiography showed one large tumor stain over left posterior fossa which was supplied by occipital artery (Figure 1(c)). Preoperative imaging characteristics of the lesion.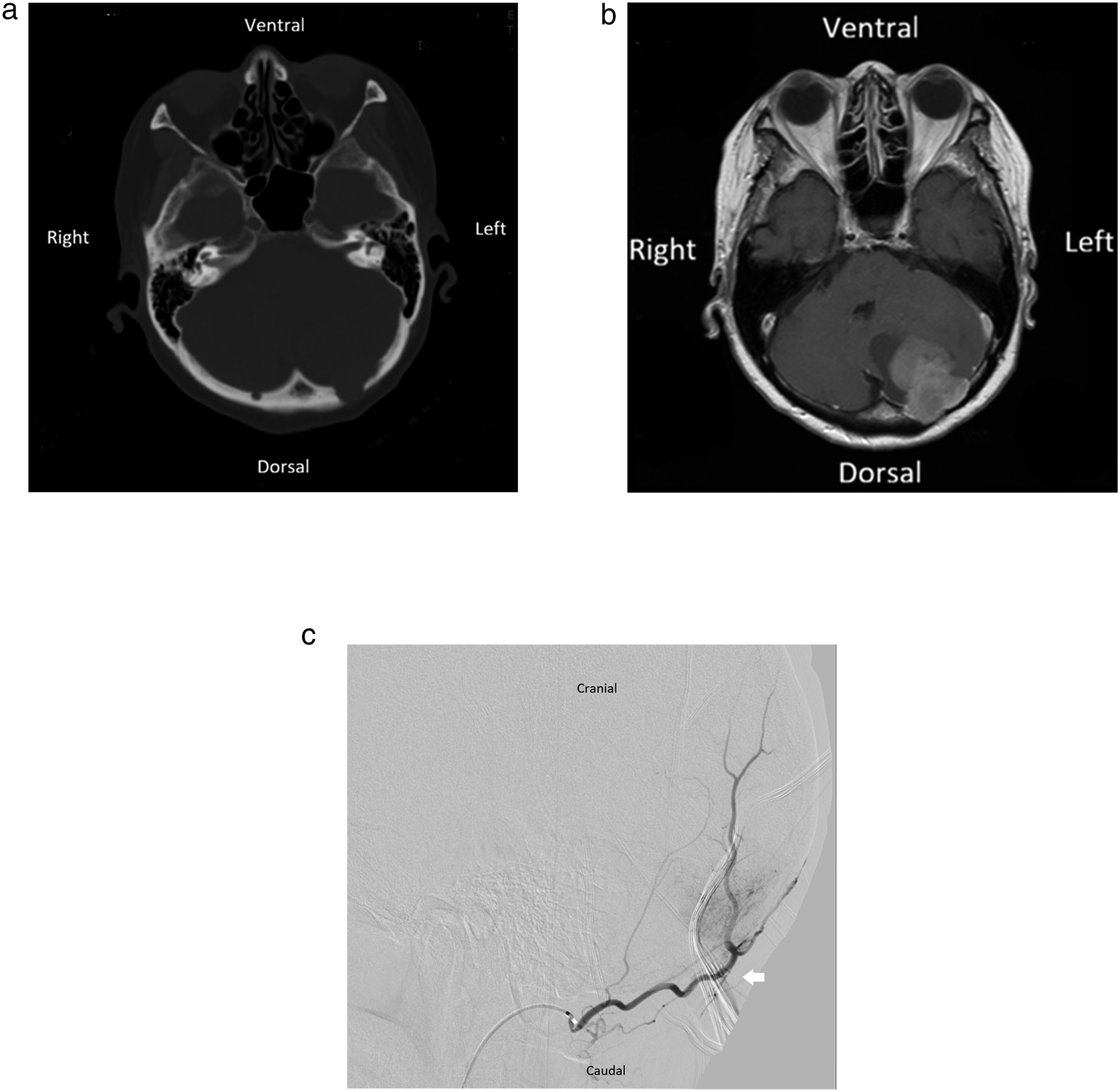
Before a formal brain surgery, the patient underwent a transarterial embolization (TAE) of the tumor using particle infusion (Embosphere® microspheres: 100-300um, 300-500um), as a measure to minimize and control possible blood loss from the surgical site. Postembolization left external carotid angiogram showed complete obliteration of blood supply to the tumor. A day after TAE, we performed a suboccipital craniotomy to remove the tumor. The procedure involved using a cavitron ultrasonic surgical aspirator, bipolar cautery, and suction to debulk the tumor. Then, microdissectors and microscissors were used carefully to separate and excise the tumor. We achieved complete removal of the tumor in the operating room. The patient recovered well after surgery with no neurological deficits, and was discharged after the relevant evaluations were completed. MR imaging scan after surgery revealed a fluidic cavity in the left cerebellum without evidence of residual tumor. No spinal seeding was detected in the subsequent checkup. Since occipital bone was destructed by the highly invasive tumor (Figure 2(a)), we reconstructed and replaced the skull with a new artificial alloplastic titanium mesh after intraoperative re-approximation of the dura (Figure 2(b)). Postoperative clinic follow-up for 18 months to date showed no evidence of tumor recurrence. This patient remained asymptomatic in neurological function and returned to our hospital outpatient clinic every 3 months without any clinical symptoms related to the tumor. The patient’s consciousness level was clear and she did not show any neurological deficits or functional disabilities during follow-up. Intraoperative gross photograph of the surgical excision procedure.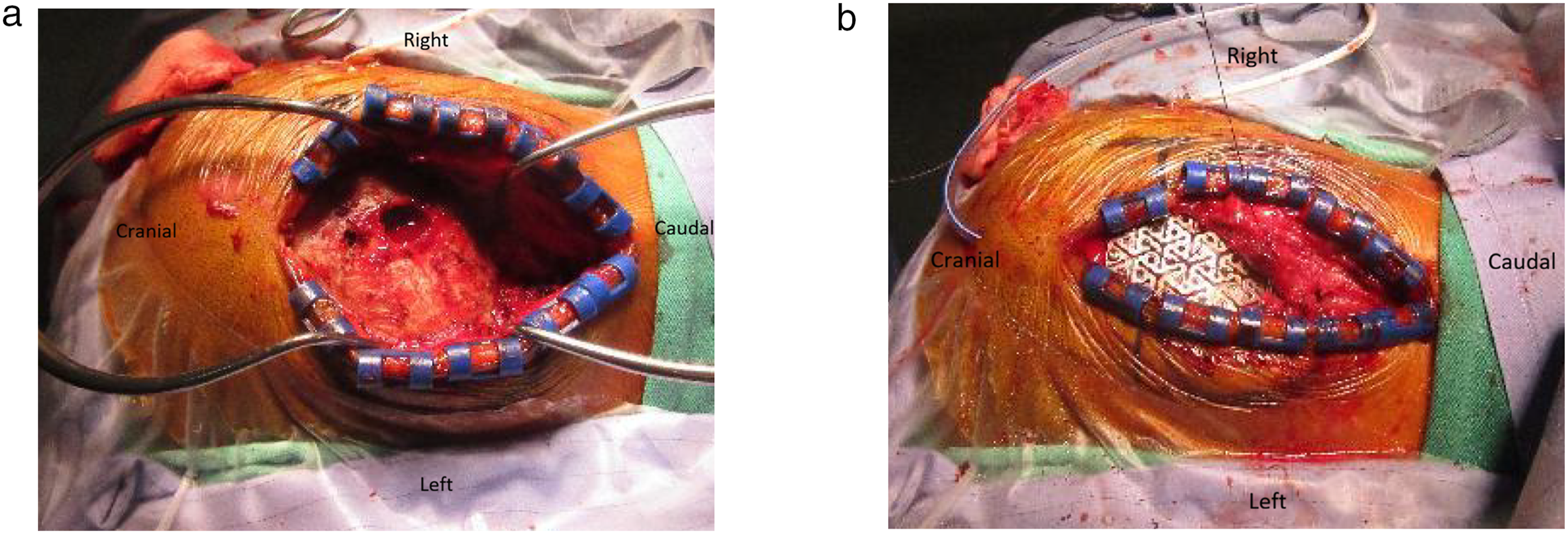
Histopathological characteristics observed using hematoxylin and eosin staining illustrated a bland spindle cell tumor without nuclear atypia and mitosis less than 1 per 10 high-power fields (HPFs) microscopically. Staghorn vasculature was occasionally present and detected (Figure 3(a)). The tumor cells were negative for epithelial membrane antigen (EMA) (Figure 3(b)) and S-100 protein (Figure 3(c)) in IHC stain. Notably, the tumor cells were positive for signal transducer and activator of transcription (STAT)-6 (Figure 3(d)). The summarized histopathological characteristics and immunological profile matched those typical for grade I SFT. The reporting of this study conformed to CAse REport (CARE) guidelines.
5
Histopathological findings of the lesion. Microscopically, it is consistent with a solitary fibrous tumor.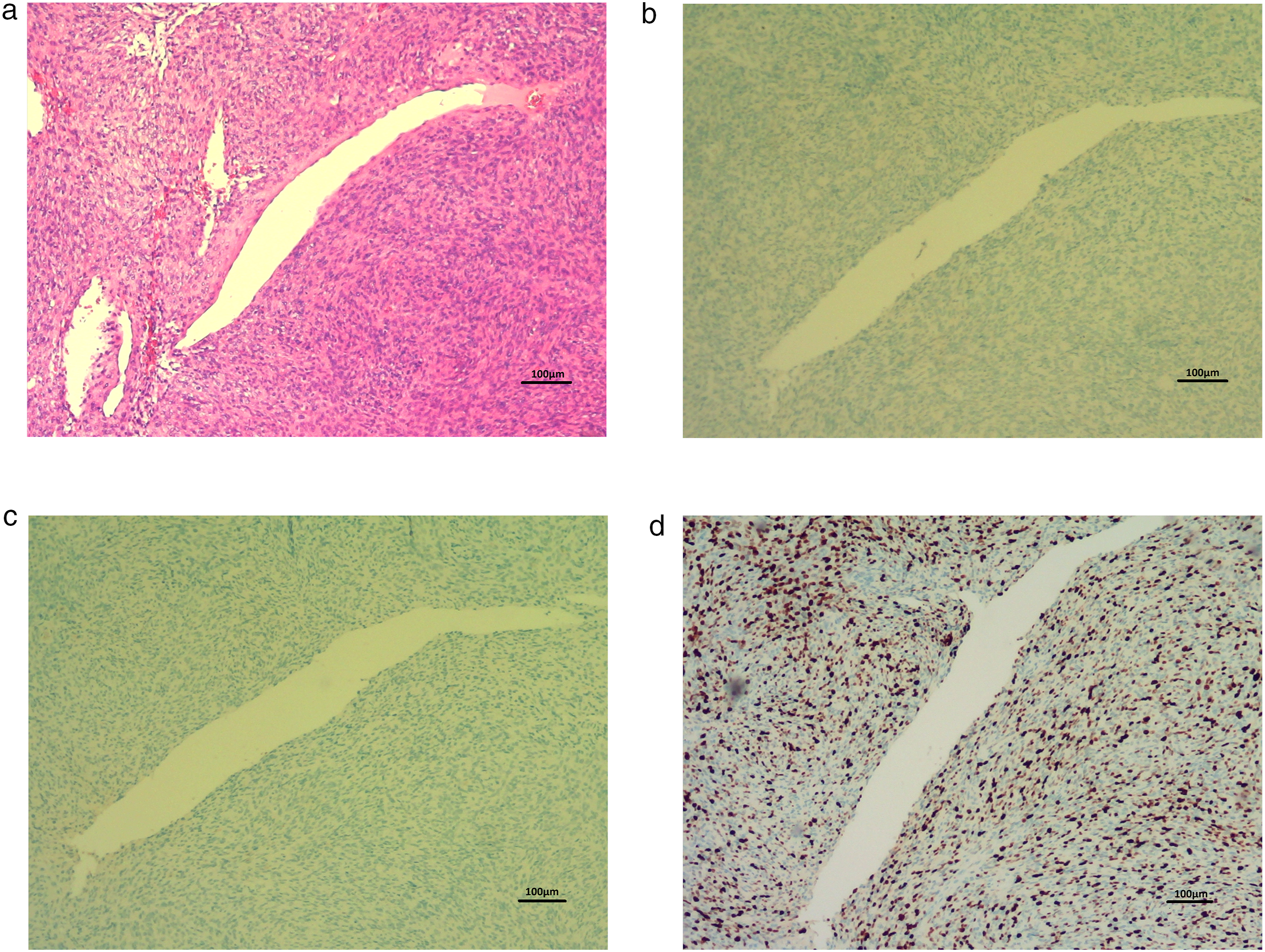
Discussion
SFT in CNS was first described in 1996 by Carneiro and colleagues. 1 SFT, formerly known as hemangiopericytoma (HPC), is a scarce intracranial extra-axial tumor of mesenchymal origin, and account for <1% of all primary intracranial tumors. 1 SFT and meningioma have some similar neuroimaging features. Therefore, accurate differential diagnosis between these tumors is clinically challenging which leads to different prognosis and follow-up management. SFT is usually more adverse than meningioma. It particularly results in local recurrence and metastasis, and has a high incidence rate of 20%. 6 Intracranial SFT symptoms differ based on its location and include headache, dizziness, muscle weakness, gait disturbance, hearing impairment, or cognitive disorders due to elevated intracranial pressure. SFT can erode and penetrate the skull to cause conditions such as hyperostosis or destruction. 7
While SFT and HPC were once considered distinct entities, the current classification integrates these into a single category given the NAB2/STAT-6 gene fusion indicating the overlapping or identical characteristics. 8 Consequently, SFT is immunohistochemically strongly positive for STAT-6 nuclear expression. 8 SFT is classified by a three-tiered grading system. 8 As per the fifth edition of the World Health Organization (WHO) grading system for CNS SFT, grade I tumors are characterized by fewer than 5 mitotic figures per 10 HPFs, grade II by 5 or more mitotic figures per 10 HPFs, and grade III by both high mitotic activity and tumor necrosis. Further, recent studies indicated that the extent of tumor necrosis indicates the prognosis and classification of tumor grading. 8 A recent article demonstrated that various prognostic factors of CNS tumors, such as extent of surgery, Ki-67 index, and necrosis areas are important because of the association with recurrence and malignant transformation. 9 Demicco et al. 10 proposed a novel risk stratification scheme for SFTs incorporating patient age, tumor size, mitotic activity, and necrosis to assess the metastatic risk and malignant potential. However, they also showed that the risk score was not validated for prediction of survival outcomes. In our present case, microscopic sections showed bland spindle cell tumor without nuclear atypia and mitosis in less than 1 per 10 HPFs. Occasional staghorn vasculature was observed. IHC staining was positive for STAT-6. Additionally, it was negative for EMA which excluded meningioma and S-100 protein which excluded schwannoma. However, EMA could be 0-29% IHC positive in meningeal SFTs. 11 CD34 immunohistochemistry can be used to validate a diagnosis of SFT because tumor cells typically show diffuse CD34 positivity which can help to distinguish SFT from meningioma and other spindle cell neoplasms. 11 Based on the above specific histopathological findings, the final diagnosis in the present case was consistent with SFT grade I.
Clinical and pathological features of reported solitary fibrous tumor of cerebellum.
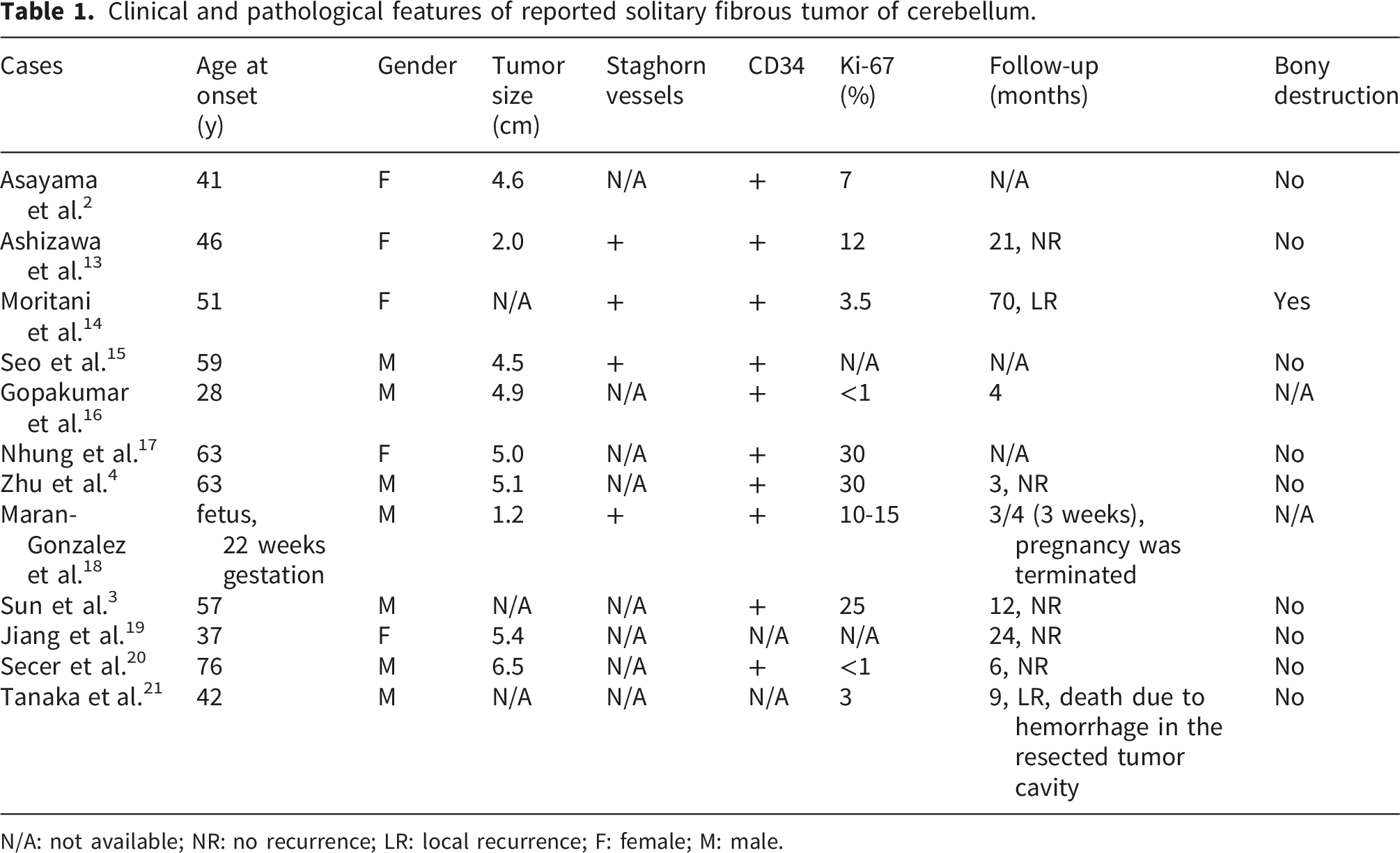
N/A: not available; NR: no recurrence; LR: local recurrence; F: female; M: male.
Typically, SFT exhibits iso-to slightly higher intensity compared to gray matter on T1-weighted MR imaging and iso-intensity on T2-weighted MR imaging. In contrast, meningioma tends to appear hypo-to iso-intense compared to gray matter on T1-weighted MR imaging and iso-to hyper-intense on T2-weighted MR imaging. Additionally, pathognomonic neuroimaging features of SFT include less frequent calcification, narrow attachment base, absence of dural tail, and frequent presence of “flow void” sign on MR imaging. Nevertheless, differentiating SFT and meningioma is challenging because the above-mentioned neuroimaging characteristics do not provide reliable means to accurately differentiate these tumors. MR spectroscopy can be used for accurate diagnosis of SFT. Specifically, SFT shows elevated ratio of choline to myo-inositol when compared to meningioma. 22
Preoperative TAE is used as an adjunctive strategy in SFT management. Hanak et al. 23 demonstrated that TAE prior to surgery significantly reduced intraoperative blood loss in patients with intracranial SFT. Yammine et al. 24 reported that preoperative TAE was associated with improved surgical outcomes because necrotic softening facilitated surgical resection. As for treatment of intracranial SFTs, a wide surgical resection with preservation of neurologic function is essential. Gross total resection during surgery is a prognostic factor linked to both recurrence-free and overall survival. 25 In contrast, subtotal resection is associated with higher incidence of local recurrence, metastasis, and decreased survival. 25 Subtotal resection followed by adjuvant radiotherapy reduces the risk of local recurrence. 25 However, it cannot prevent further metastasis.25,26 To date, evidence for specific treatment modality for SFT with skull invasion is inadequate. The extent of skull invasion by SFT may not always correspond directly to its histopathological grade. Bony invasion has been reported in intracranial SFTs, which might indicate locally aggressive behavior, and increase surgical complexity. However, established prognostic factors for recurrence primarily are WHO grade, extent of resection, and adjuvant radiotherapy. 27 Future studies are warranted to provide more details regarding treatment modalities for SFT with skull invasion.
The main limitation of this study is that the follow-up duration was 18 months, which is relatively short for a tumor with known risk of late recurrence and metastasis. Although short-term outcome of this case is favorable, continuous long-term monitoring should not be overlooked due to the potential risk of recurrence.
Conclusions
Histopathological examination is essential to confirm the final diagnosis of SFT. Complete surgical resection of SFT should be of priority. Adjuvant therapies need to be identified. We suggest long-term follow-up for SFT tumor with bony invasion.
Footnotes
Acknowledgements
We appreciate support from the Core Laboratory of the Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation.
Consent for publication
Written informed consent for publication has been obtained from the patient. Approval from the institutional review board of Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation was obtained for the study on October 15, 2025 (Protocol No.: 14-IRB141). We have de-identified all patient details.
Author contributions
Li-Wei Hong, Chung-Tai Yue, and Woon-Man Kung designed the concept and analyzed the data. Li-Wei Hong and Chung-Tai Yue searched the literature. Li-Wei Hong wrote the original draft. Woon-Man Kung supervised, revised, and edited the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article: No funding was received.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors have no conflicts of interest to declare.
Data Availability Statement
All clinical and other information is available in the patient’s medical record in the Division of Neurosurgery, Department of Surgery, Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation.
