Abstract
Introduction
Ischemia-reperfusion (IR) injury is a major concern that frequently occurs during vascular surgeries. Hydrogen-rich saline (HRS) solution exhibits antioxidant and anti-inflammatory properties. This study aimed to examine the effects of HRS applied before ischemia in the lungs of rats using a lower extremity IR model.
Material and Methods
After approval was obtained from the ethics committee, 18 male Wistar albino rats weighing 250–280 g were randomly divided into three groups: control (C), IR and IR-HRS. In the IR and IR-HRS groups, an atraumatic microvascular clamp was used to clamp the infrarenal abdominal aorta, and skeletal muscle ischemia was induced. After 120 min, the clamp was removed, and reperfusion was achieved for 120 min. In the IR-HRS group, HRS was administered intraperitoneally 30 min before the procedure. Lung tissue samples were examined under a light microscope and stained with hematoxylin-eosin (H&E). Malondialdehyde (MDA) levels, total sulfhydryl (SH) levels, and histopathological parameters were evaluated in the tissue samples.
Results
MDA and total SH levels were significantly higher in the IR group than in the control group (p < 0.0001 and p = 0.001, respectively). MDA and total SH levels were significantly lower in the IR-HRS group than in the IR group (p < 0.0001 and p = 0.013, respectively). A histopathological examination revealed that neutrophil infiltration/aggregation, alveolar wall thickness, and total lung injury score were significantly higher in the IR group than in the control group (p < 0.0001, p = 0.001, and p < 0.0001, respectively). Similarly, alveolar wall thickness and total lung injury scores were significantly higher in the IR-HRS group than in the control group (p = 0.009 and p = 0.004, respectively). A statistically significant decrease was observed in neutrophil infiltration/aggregation and total lung injury scores in the IR-HRS group compared to those in the IR group (p = 0.023 and p = 0.022, respectively).
Conclusion
HRS at a dose of 20 mg/kg, administered intraperitoneally 30 min before ischemia in rats, reduced lipid peroxidation and oxidative stress, while also reducing IR damage in lung histopathology. We believe that HRS administered to rats prior to IR exerts a lung-protective effect.
Keywords
Introduction
Ischemia-reperfusion (IR) injury in skeletal muscles occurs when oxygenated blood is reintroduced into ischemic tissues. 1 Such injuries may occur as a result of various medical incidents, including severe limb trauma, vascular damage, limb replantation, thrombosis, or the use of a surgical tourniquet. Skeletal muscle IR injury may lead to muscle spasms and fibrotic changes, as well as multiple organ dysfunction syndrome affecting the lung, heart, and brain, which can result in high mortality rates after cardiovascular and orthopedic surgeries.2–5
Although lung injury induced by skeletal muscle IR is well documented, it remains inadequately elucidated in the current literature. Endothelial cell damage resulting from neutrophil activation is one of the primary pathological dysfunctions in lung injury. Skeletal muscle IR can release inflammatory cytokines, endogenous danger molecules, and oxygen-free radicals, and activate the complement system.6–9 In normal physiological states, the venous circulation passing through the pulmonary vasculature acts as a neutrophil pool. In the lungs, there is an increased potential for neutrophil–endothelial interactions compared to other vascular beds. Together with the large surface area of the endothelium, the pulmonary vasculature is a quantitatively significant site for neutrophil–endothelial interactions. All these responses lead to the accumulation of inflammatory infiltrates in the alveoli and interstitium.6–9 These factors are responsible for decreased gas exchange and increased work of breathing, exposing the lungs to acute respiratory distress syndrome (ARDS), 10 which may lead to mortality rates of up to 40%. 11 Thus, developing effective treatments for skeletal muscle IR injury is imperative to protect the lungs.
Molecular hydrogen (H2) acts as an antioxidant by selectively quenching detrimental reactive oxygen species.12,13 Unlike most antioxidants, H2 can penetrate and diffuse into cell organelles membranes due to its advantageous distribution characteristics, 14 and inhalation of H2 gas can reduce IR injury through its antioxidant effect.12,15–17 However, the inhalation of H2 gas is neither convenient nor safe for clinical use because it is combustible and explosive. In contrast, hydrogen-rich saline (HRS) has been demonstrated to have protective effects in multiple organs.12,15–21
Experimental animal studies have demonstrated that the administration of hydrogen gas and HRS effectively mitigates IR injury in skeletal muscle by preventing endoplasmic reticulum stress, reducing apoptosis, and modulating the JNK signaling pathway.22,23 Furthermore, the administration of HRS for acute lung injury following burns results in reduced levels of pulmonary inflammation mediators and myeloperoxidase. 24 HRS also enhances pulmonary function by mitigating oxidative stress and inflammatory responses. 24 Similarly, using a hydrogen-rich preservation solution during lung transplantation attenuates IR injury in rat lungs by exerting antioxidant and anti-inflammatory effects during cold ischemia. 25
The protective effects of HRS on skeletal muscle IR and the protective effects of oral and inhaled hydrogen and HRS on the lungs have been demonstrated in the literature.23,25–27 However, there is no evidence to suggest that HRS provides a protective effect on the lungs during skeletal muscle IR. HRS is easily produced, stored, and transported, so it may provide a safe, low-cost option for skeletal muscle IR injury and distant organ damage caused by skeletal muscle injury. Therefore, this study aimed to evaluate the efficacy of HRS for mitigating lung damage during skeletal muscle IR.
Materials and methods
This study was conducted at the Gazi University Animal Laboratory in Ankara, Turkey, using ethical procedures approved by the Gazi University Ethical Committee of Experimental Animals (G.Ü.ET-22-096). Adult male Wistar albino rats weighing 250–280 g, sourced from the Gazi University Experimental Animals Research Center, were used in the experiment. The animals were housed individually in a controlled environment with temperature and humidity set at 20–21°C and 45–55%. The rats received a 12-h light/dark cycle and had free access to food and distilled water.
Hydrogen-rich saline production
Hydrogen was dissolved in physiological saline for 6 h under high pressure (0.4 MPa) to a supersaturated level using our department's HRS-producing apparatus. Saturated hydrogen saline was stored in an aluminum bag with no dead volume at 4°C under atmospheric pressure. The HRS was sterilized with gamma radiation and freshly prepared to maintain a concentration of 0.6 mmol/L. The method described by Ohsawa et al. was used to determine the hydrogen content of saline using gas chromatography. 12
Experimental protocol
Eighteen rats were randomly assigned and equally (n = 6) divided into three groups: control, IR, and IR-HRS. All surgical procedures were performed under general anesthesia. An intramuscular injection of 50 mg/kg ketamine hydrochloride (500 mg/10 ml; Ketalar®vial; Parke-Davis; Pfizer, Inc.) + 10 mg/kg xylazine hydrochloride (Alfazyne® vial 2%; Ege Vet, Ltd) was administered to induce anesthesia.
Anesthesia was maintained with injections of 20 mg/kg ketamine and 5 mg/kg xylazine if a positive reaction to surgical stress or intermittent tail pinch was observed. After 30 min, the procedure was performed under a warming lamp, with the rats in the supine position.
In the control group, midline laparotomy was the sole surgical procedure performed, without any additional intervention. After 4 h of follow-up, the lung tissue of the rats was excised following sacrification under anesthesia.
In the IR group, an atraumatic microclamp was placed on the infrarenal aorta for 120 min, after which the clamp was withdrawn and reperfused for another 120 min. Subsequently, the rats were sacrificed, and the lung tissues were excised.
In the IR-HRS group, HRS (20 mg/kg) was administered intraperitoneally after the laparotomy. After 30 min of HRS administration, an atraumatic microclamp was placed on the infrarenal aorta for 120 min., and the clamp was then withdrawn and reperfused for another 120 min. The lung tissues of the rats were excised after they were sacrificed under anesthesia.
All rats were sacrificed by collecting blood (5–10 mL) from the abdominal aorta after the administration of (100 mg/kg) and xylazine (10 mg/kg). After the heartbeat and respiration ceased, the rats were monitored for 2 minutes to confirm their death.
Histopathological evaluation
After sacrification, the lungs were harvested and preserved in formalin solution. A histological examination was performed by a pathologist who was blinded to the experimental protocol. Tissues were stained with hematoxylin-eosin, and 10 lung tissue sections were examined. A 200- to 400-times magnified light microscope was used to observe the stained slides. In each specimen, neutrophil infiltration and alveolar thickness were assessed to determine the severity of the lung injury. The severity of the injury indicated by each parameter was categorized as falling into one of four levels: none, minimal (+), moderate (++), or severe (+++). The individual scores for each parameter were totaled and recorded as the total injury score. 28
Biochemical evaluation
Lipid peroxidation was measured using the Esterbauer method. The interaction between malondialdehyde (MDA) and thiobarbituric acid at a temperature of 90–95 °C resulted in the formation of a pink chromogen. After collection, the lung specimens were promptly cooled, and their absorbance was measured using spectrophotometry at a wavelength of 532 nm. The results were expressed in nmol/g. 29 A simple spectrophotometric method was used for the routine concomitant determination of sulfhydryl (SH) groups in the PB-SH, NP-SH, and T-SH fractions in various tissues. These spectrophotometric procedures are based on the method of Ellman,30,31 who reported that 5.5′-dithiobis- (2,-nitrobenzoic acid) is reduced by SH groups to form 1 mole of 2-nitro-5-mercaptobenzoic acid per mole of SH. The Sedlak method was employed to determine the total SH content, and the results were reported as µMol/L. 32
Statistical analysis
SPSS (version 26.0; IBM Corp.) was used for the statistical analysis. The data distribution was analyzed using a Shapiro–Wilk test. The data were evaluated using a Kruskal–Wallis test (histopathological parameters), Dunn's test, or one-way ANOVA (biochemical parameters), followed by a Bonferroni test. Statistical significance was set at p < 0.05. Data were shown as either median [min–max] or mean ± standard error, depending on the overall variable distribution.
Results
Biochemical results
The ANOVA indicated significant interactions between all groups in terms of MDA and total SH levels (p < 0.0001 and p = 0.003, respectively). As indicated in Table 1, both MDA and total SH levels were significantly higher in the IR group than in the control group (p < 0.0001, p < 0.0001, respectively). However, the IR-HRS group exhibited significantly lower levels of MDA and total SH compared to the IR group (p < 0.0001, p = 0.013, respectively, Table 1).
Biochemical analyses of the lung tissue [mean ± SE].
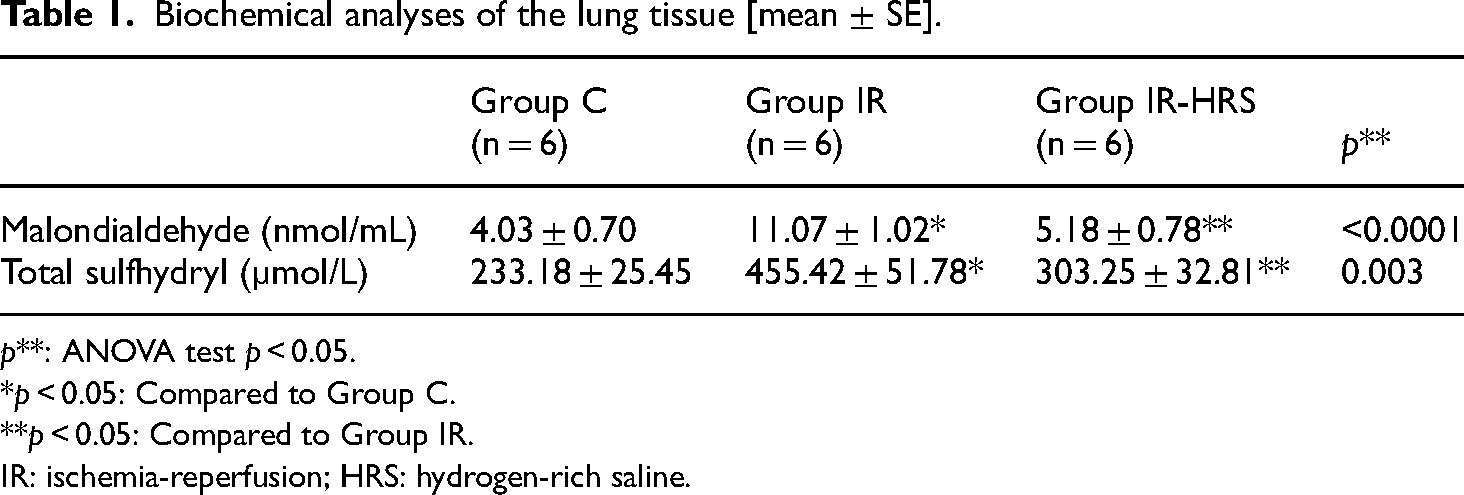
p**: ANOVA test p < 0.05.
*p < 0.05: Compared to Group C.
**p < 0.05: Compared to Group IR.
IR: ischemia-reperfusion; HRS: hydrogen-rich saline.
Histopathological results
The results of pathological staining analysis using light microscopy revealed significant differences between the groups in terms of neutrophil infiltration/aggregation, alveolar wall thickness, and total score (p = 0.005, p = 0.007, and p < 0.0001, respectively, as shown in Table 2, Figures 1–3). Specifically, the IR group demonstrated significantly higher levels of neutrophil infiltration/aggregation, alveolar wall thickness, and total scores than the control group (p < 0.0001, all). Additionally, the IR-HRS group demonstrated significantly higher levels of alveolar wall thickness and total scores than the control group (p = 0.009 and p = 0.004, respectively, Table 2, Figures 1–3). Rats in the IR-HRS group showed a substantial reduction in the degree of neutrophil infiltration and total score (p = 0.023 and p = 0.022, respectively, Table 2, Figures 1–3).

Normal lung tissue in group C, HE × 100. Abbreviations: A—alveolus; TB—terminal bronchiole; ias—interalveolar septum; ↓↓—thickening of interalveolar septum; inf—inflammation.

Severe neutrophilic infiltration and increased alveolar wall thickness in the IR group, HE × 100. Abbreviations: A—alveolus; da—ductus alveolaris; conj—congestion; neutrophil infiltration indicated by white arrow; inf—inflammation; ↓—pulmonary vascular thickening. IR: ischemia-reperfusion.

Mild neutrophilic infiltration and increased alveolar wall thickness in the IR-HRS group, HE × 100. Abbreviations: A—alveolus; tb—terminal bronchiole; SA—saccus alveolaris; DA—ductus alveolaris; conj—congestion; neutrophil infiltration indicated by white arrow. IR-HRS: ischemia-reperfusion hydrogen-rich saline.
Histopathological analyses of the lung tissue [median (min–max)].

p**: Kruskal–Wallis test p < 0.05.
*p < 0.05: Compared to Group C.
**p < 0.05: Compared to Group IR.
IR: ischemia-reperfusion; HRS: hydrogen-rich saline.
Discussion
In this study, we established an animal model of skeletal muscle IR injury and demonstrated that intraperitoneal administration of HRS protected the lungs against skeletal muscle IR injury. HRS treatment ameliorated histopathological changes and alleviated MDA and SH levels, leading to an overall improvement in the lungs.
Skeletal muscle IR, a prevalent pathophysiological phenomenon encountered in surgical practice, can occur due to severe crush injuries, peripheral arterial clamping during orthopedic surgery or trauma, embolectomy, or abdominal aortic aneurysm surgery. Unfortunately, the deleterious effects of limb IR are not limited to muscle tissues; it can also damage distant organs, including the lungs. 3 The lungs are particularly prone to IR injury, which can lead to the development of ARDS, a severe condition with high morbidity and mortality rates.11,33
Hydrogen, an element commonly found in nature, was previously thought to be an inert gas with no biological activity. However, unlike the most well-known strong reduction agents, hydrogen has specific features that allow it to target organelles. It can rapidly diffuse across the membranes of subcellular organelles and has a rapid onset of activity. 34 Therefore, it is a promising option for preventing IR-induced injuries. In a study that has attracted the interest of researchers worldwide, Ohsawa et al. demonstrated the effects of inhaled hydrogen on brain IR damage. 12 However, given its limited accessibility and flammability, the application of inhaled hydrogen in clinical practice is limited.
In contrast, hydrogen-enriched water is safer, more portable, and more feasible than the inhalation of hydrogen gas. Hydrogen-enriched water has the same antioxidant, anti-inflammatory, and antiapoptotic effects as those of hydrogen inhalation. 35 Indeed, studies have emphasized that HRS has preventive and therapeutic effects in many pathologies, including aplastic anemia, 36 subarachnoid hemorrhages, 37 intestinal damage in hemorrhagic shock, 38 heart dysfunction, 20 cardiopulmonary bypass-induced cerebral damage, 39 renal damage after IR, 40 Alzheimer, 41 burn damage, 42 erythrocyte deformability after limb ischemia, 19 and postoperative cognitive function. 21 Others have demonstrated that HRS may potentially alleviate primary lung IR injury. 43 However, it is important to acknowledge that remote lung injury resulting from skeletal muscle IR is more frequently observed in clinical practice, and studies exploring the efficacy of HRS in this context are limited. 44 Accordingly, this study aimed to investigate the potential effects of HRS on lung injury caused by skeletal muscle IR injury.
Increased production of oxygen radicals has been shown to play a key role in the pathogenesis of remote lung injury in animal models.45,46 Lung injury frequently occurs during skeletal muscle ischemia due to free radicals released by reperfusing tissues. 46 Lipid peroxidation, triggered by oxygen-free radicals, is an important cause of lung injury. MDA is the principal and most studied product of lipid peroxidation.43,44 Here, we observed that lung injury resulting from muscle IR was associated with increased MDA levels in the lungs. Our finding consistent with previous studies showing that HRS inhibits lipid peroxidation. 47 SH groups, commonly considered the most important and most frequently encountered antioxidants, act as powerful antioxidants that play a vital role in interrupting the oxidation process. Evaluating protein-based SH groups is essential for measuring oxidative stress and determining the effectiveness of antioxidant therapies. 48 In this study, we showed that lung injury after muscle IR was correlated with increased SH levels in the lungs. Treatment with HRS considerably lowered SH levels, indicating a potential protective effect against lung injury.
Histopathologically, lung injury is characterized by alveolar wall thickening, interstitial edema, and activation, adhesion, and release of toxic chemicals by polymorphonuclear neutrophils. 49 A substantial amount of toxin present in the bloodstream directly affects the lungs, resulting in edema of endothelial cells, thickening of alveolar walls, diminished oxygen exchange, infiltration of inflammatory cells, and a significant increase in the levels of oxygen-free radicals and lipid peroxidation products within the lung tissue. Furthermore, such toxins decrease the antioxidant capacity of lungs. 50 Numerous histopathological studies have indicated that HRS decreases tissue inflammation following IR injury.12,15–21 Histopathological examination of the lung tissue samples in the present study revealed a statistically significant difference in neutrophil infiltration or aggregation and alveolar wall thickening. Specifically, the extent of neutrophil infiltration or aggregation was significantly higher in the IR group than in the control and IR-HRS groups.
The study has some limitations. Firstly, no power analysis was conducted to determine the appropriate sample size. Each experimental group consisted of six animals, in adherence to the guidelines outlined by the ethics committee. The inclusion of six animals per group represents the maximum permissible number sanctioned by the ethics committee under the purview of the 3Rs rule (replacement, reduction, and refinement). Secondly, due to constraints in sample volume, only total SH and MDA levels were measured, precluding the assessment of other biochemical parameters such as glutathione, catalase, and superoxide dismutase. Obtaining the necessary large quantities of samples for additional oxidative stress assays was not feasible within the scope of this study.
Conclusion
In conclusion, the outcomes of this study provide considerable insight into the possible therapeutic effects of HRS in minimizing the pulmonary damage caused by skeletal muscle IR. Because of its efficacy, ease, and low cost, HRS can be used for the routine treatment of organ dysfunction, including lung injury. The underlying mechanisms of its protective effects may involve the prevention of neutrophil aggregation and oxidative damage in injured lungs.
Footnotes
Authors’ contributions
MA, AK, and GLO were responsible for designing the study and analyzing and interpreting the data. AÖ and HD performed the study in the laboratory in accordance with the methodology and approved. MA was responsible for the acquisition, analysis, and interpretation of the data. MA and AÖ confirmed the authenticity of the raw data. SE and HD provided scientific and technical assistance for the experiments and critically revised the article for important intellectual content. HB and ŞCS collected samples and were responsible for the execution of the project. ŞCS was responsible for cellular and molecular experiments. All authors have read and approved the final version of the manuscript.
Availability of data and materials
The analyzed data for this study are fully documented and included in this published article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Ethical approval for this study was obtained from the Gazi University Experimental Animal Ethics Committee (Ankara, Turkey; approval no. G.Ü.ET-22-096).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable.
Author biographies
Abdullah Özer is Medical Doctor of Cardiovascular Surgery. His research area is phlebologie and ischemia reperfusion
Selin Erel is medical doctor of Anesthesiology and Reanimation. Her research area is ischemia reperfusion, nanoparticles and anesthesia applications.
Ayşegül Küçük is a Professor Physiology. Her research area: ischemia-reperfusion injury, neuroscience, nanoparticles and Alzheimer's
Hüseyin Demirtaş is Medical Doctor of Cardiovascular Surgery. His research area is phlebologie and ischemia reperfusion
Şaban Cem Sezen is a specialist in Histology and Embryology. His research area: histology, histopathology, ischemia-reperfusion injury
Hakan Boyunağa is a Professor of Medical Biochemistry. His research areas are free radicals and antioxidants, oxidative stress, toxicology, purification and kinetic properties of enzymes (protein), flavonoids and other basic and clinical research related to medical biochemistry.
Gürsel Levent Oktar is Professor of Cardiovascular Surgery. His research area is phlebologie and ischemia reperfusion
Mustafa Arslan is Professor of Anesthesiology and Reanimation. His research area is ischemia reperfusion, nanoparticles, Alzheimer's and anesthesia applications.
