Abstract
Objective
Evaluate a low-urapidil protocol (30 µg/kg bolus + 20 µg/kg/h infusion) on oxidative stress, inflammation, apoptosis, and matrix metalloproteinase−9 (MMP-9)-mediated matrix remodeling in a rat renal ischemia-reperfusion (I/R) injury model.
Methods
32 male Wistar rats were randomized into control (CR), sham (SH), I/R, and I/R + Urapidil (UR) groups. The CR group received no treatment, the SH group underwent laparotomy and intraperitoneal normal saline, the I/R group underwent laparotomy with 30-min of aortic clamping and 24 h of reperfusion, and the UR group received urapidil starting at 1 h before 30-min of aortic clamping (ischemia), followed by 24 h of reperfusion. Assessments included serum creatinine/urea, oxidative markers (total antioxidant status [TAS, total oxidant status [TOS]), inflammatory cytokines (tissue necrosis factor-alpha [TNF-α], interleukin [IL]-1β, IL-6, and MMP-9) via enzyme-linked immunosorbent assay, renal histopathology (hematoxylin and eosin staining), and immunohistochemistry (H-scores for endothelial nitric oxide synthase [eNOS], caspase-3, TNF-α, IL-1β, IL-6, and MMP-9).
Results
I/R significantly increased serum creatinine, urea, TNF-α, IL-1β, and TOS, and depleted TAS, indicating severe injury, oxidative stress, and inflammation. Extensive tubular necrosis, vacuolization, and edema were also observed. Immunohistochemistry revealed elevated levels of eNOS, caspase-3, cytokines, and MMP-9. Urapidil pretreatment effectively normalized serum creatinine/urea, restored TAS, reduced TOS and cytokines (TNF-α, IL-1β, IL-6) to baseline, and markedly alleviated histopathological damage. Urapidil significantly suppressed I/R-induced elevations in oxidative (eNOS), apoptotic (caspase-3), and inflammatory markers, and MMP-9 expression.
Conclusion
Low-dose urapidil confers significant protection against renal I/R injury by mitigating oxidative stress, inflammation, apoptosis, and MMP-9-mediated matrix degradation. The efficacy at microgram doses highlights its potential as a clinically relevant nephroprotective agent.
Keywords
Introduction
Ischemic reperfusion (I/R) injury is a serious clinical condition that occurs when blood flow to tissues is temporarily interrupted and then restored.1,2 This damage causes significant functional and cellular losses in various organs. Renal I/R injury is considered an important cause of acute kidney injury (AKI) and is frequently encountered during kidney transplantation, cardiovascular surgery, and shock.3,4 Recently, many complex mechanisms have been suggested to play a role in the pathophysiology of renal I/R injury, such as oxidative stress, inflammation, apoptosis, and ferroptosis.5,6 Among these, the increased production of reactive oxygen species (ROS) and insufficient antioxidant defense systems are considered critically important. 7
Urapidil (UP) blocks alpha-1 adrenergic receptors and activates serotonin (5-HT1A) receptors, helping to lower blood pressure by decreasing blood vessel resistance. The antioxidant, anti-inflammatory, and anti-apoptotic properties of UP have been previously investigated in various experimental I/R models, such as testicular torsion, ovarian torsion, intestinal ischemia, and cerebral ischemia.8,9 However, the number of studies examining the effects of UP in renal I/R injury is limited, and existing studies generally include intraperitoneal pre-ischemic dose applications at the milligram level. 10 Approaches such as the use of low-microgram doses, continuous intraperitoneal infusion, or application at earlier time points before ischemia have not yet been evaluated. In addition to the classically evaluated inflammatory (tissue necrosis factor-alpha [TNF-α], interleukin [IL]-1β), oxidative stress (total oxidant status [TOS], total antioxidant status [TAS]) and apoptosis (caspase-3) markers in renal I/R injury, the effects on new and different molecular mechanisms such as cellular stress responses (e.g., Nrf2, NF-κB), mitochondrial function markers, and ferroptosis have not been sufficiently investigated. UP has been shown to increase the level of nuclear factor-erythroid 2-related factor 2 (Nrf2), which controls the activity of enzymes involved in the antioxidant system and plays an important role in I/R damage.11,12 To address these gaps in the existing literature, in this study we aimed to comprehensively evaluate the effects of UP administered at a low microgram dose (30 µg/kg loading dose followed by 20 µg/kg/h continuous intraperitoneal infusion) on oxidative stress, inflammation, apoptosis, and matrix regulatory factors in the renal tissue of a renal I/R model. Through this, we aim to contribute to a better understanding of the renal protective mechanisms of UP and to evaluate its clinical potential by presenting a new dose and administration method.
Materials and methods
Study design
In this study, we used 4-month-old male Wistar albino rats (
Ethical approval
The Afyon Kocatepe University Experimental Animal Ethics Committee approved this study (approval number: 49533702/101; approval date: November 10, 2023). The experimental rats were placed in cages under standard laboratory conditions that included a 12 h light/dark cycle, at 22°C ± 2°C, and a humidity level of 55% ± 5%. The rats were provided ad libitum food and water.13,14 The study adhered to the ARRIVE reporting guidelines 15 and complied with the Guide for the Care and Use of Laboratory Animals, 8th Edition. 16 Every effort was made to minimize the number of animals used and reduce animal suffering.
Anesthesia
All rats were premedicated by intramuscular administration of 13 mg/kg Xylazine HCl ((Xylazinbio, %2, 50 ml Vial, Bioveta, Bioveta, a.s., Komenskeho Czech Republic). General anesthesia with Ketamine HCl ((Ketasol, %10, 10 ml vial; İnterhas Medical and Chemical Products Industry and Inc. Ankara-Turkey) was administered intramuscularly at a dose of 87 mg/kg intramuscularly. 17
Surgical procedure
After dissection of the abdominal cavity, the aorta was located and exposed from its origin to the distal bifurcation of the left renal artery, where it was proximally clamped. Similar to other ischemia-inducing methods, I/R injury was induced by infrarenal clamping of the abdominal aorta (Aesculap, Tuttlingen, Germany), which has been shown to cause I/R injury18–21 In a previous experimental study, 22 a dose of 20 µg/(kg⋅h) UP, the maximum dose that does not impact mean blood flow in the kidneys, arterial blood pressure, and heart rate, was administered intraperitoneally. The pulse of the femoral artery was not detected throughout the 30-min ischemia phase; however, it returned on reperfusion. 17
Groups and the I/R model
Group 1 ( Group 2 ( Group 3 ( Group 4 (
Euthanasia
After a 24-hour reperfusion period all rats were deeply anesthetized via intraperitoneally Thiopental Sodium 150 mg/kg (Pental, 0,5 g fl. İ.E. Ulagay Drug Manifacture Türk A.Ş. Istanbul-TURKEY) and subsequently euthanized by overdose.
Source of animals
All rats were certified Wistar Albino rats obtained from the licensed breeding colony of the Afyon Kocatepe university Experimental Animals Application and Research Center. All procedures were conducted in the same facility.
Histopathological examination
Re-laparotomy was performed under anesthesia, as in the I/R procedure, and a left nephrectomy was performed. The renal capsule was excised. Half of each kidney was preserved in 10% formalin for histopathological examination. Hematoxylin and eosin (HE) staining and scoring were performed to determine the morphological effects of UP on I/R injury. The expressions of endothelial nitric oxide synthase (eNOS) for oxidative stress; caspase-3 for apoptosis; and IL-1β, IL-6, TNF-α, and matrix metalloproteinase-9 (MMP-9) for inflammation were examined using immunohistochemistry (IHC) and given a H-score. The remaining half of the kidneys were transported under a cold chain and stored at –85°C for biochemical measurements in renal tissues using enzyme-linked immunosorbent assay (ELISA) kits with rat enzyme-linked immunosorbent for determining the levels of IL-6, IL-1β, TNF-α, MMP-9, TAS, and TOS for ELISA.
HE staining
Specimens were taken from all kidneys and fixed in a 10% buffered neutral formaldehyde solution in the pathology laboratory. Sections were blocked in paraffin after routine tissue tracking. Then, 4- to 6-µm sections were cut from the blocks. Sections were stained with HE for histopathological evaluation. Ten randomly selected fields from all HE-stained sections were examined at 20 × magnification. The extent of lesions observed in the samples, including congestion, vacuolar degeneration, interstitial edema, proteinaceous material accumulation in the lumen, tubular separation, and necrosis, was scored on a scale ranging from 0 to 4. Scoring was based on the following criteria: 0 was normal; 1was mild lesions affecting 25% of the sample; 2 was lesions affecting 25–50% of the sample; 3 was lesions affecting 50–75% of the sample; and 4 was lesions affecting more than 75% of the sample. The samples were evaluated for glomerular changes, tubular dilatation, cellular vacuolization, presence of proteinaceous casts, necrosis, interstitial inflammation, and congestion.23–27
Immunohistochemical staining
Indirect IHC was performed on formalin-fixed samples after deparaffinization and antigen retrieval. The sections were then incubated in Tris-buffered saline Tween-20 (TBST, pH 7.4) for 15 min and subsequently washed with TBST. Furthermore, 1% hydrogen peroxide (Lab Vision; Thermo Scientific) was administered for 30 min to avoid non-specific binding. Following the completion of the blocking procedure, the samples were treated with primary antibodies, including eNOS (Boster Bio., catalog number: A01604-2, diluted at a ratio of 1:250), caspase 3 (Thermo Scıentıfıc, catalog number: RB-1197-P0, diluted at a ratio of 1:100), TNF-α (Proteintech, catalog number:60291-1-Ig, diluted at a ratio of 1:300), IL-1β (Bioss, catalog number:bs-6319R, diluted at a ratio of 1:150), IL-6 (ST John’s, catalog number:STJ1110424, diluted at a ratio of 1:100), and MMP-9 (Boster Bio, catalog number: PA2140-2, diluted at a ratio of 1:100). The samples were then subjected to overnight incubation with the primary antibody at 4°C. The following day, cells were incubated with a biotin-conjugated secondary antibody (Lab Vision; Thermo Scientific, CA, USA) for 20 min at room temperature. Subsequently, sections were incubated with streptavidin-peroxidase for 20 min (Lab Vision, Thermo Scientific, CA, USA). Then, the sections underwent washing using TBST. The final step involved the development of a color reaction by incubating the sections with a chromogen substrate containing diaminobenzidine (DAB) for approximately 5–10 min at room temperature. Mayer’s hematoxylin was used for counterstaining. The H-score method was used to examine the semi-quantitative results obtained from immunohistochemical staining. Staining intensity was evaluated as weak (1), medium (2), and strong (3). For each intensity, three different microscopic fields were used to count cells. The following formula was used to determine the relevant score: H-score = ∑Pi (staining intensity + 1). Where the rate of labeled cells is presented as Pi, ranging from 0% to 100% for intensity values.28,29
Biochemical examination
For biochemical examination, 2 mL blood samples were collected from rats in all groups by intracardiac puncture before they were sacrificed. Blood urea, calcium (Ca+2), creatinine, chloride (Cl-), magnesium (Mg+2), phosphorus (P-3), potassium (K+), sodium (Na+), and total protein (TP) levels in the serum and plasma samples were analyzed using an autoanalyzer. The levels of IL-1 (BT LAB, Rat ELISA Kits, Bioassay Technology Lab, Zhejiang, China, Cat No.: E0119Ra), IL-6 (BT LAB, Bioassay Technology Lab, Zhejiang, China, Cat No.: SEA079Ra), TNF-α (BT LAB, Rat ELISA Kits, Bioassay Technology Lab, Zhejiang, China, Cat No.: SEA133 Ra), TAS (Andygene, Rat ELISA Kits, Bioassay Technology Laboratory, Shanghai, China, Cat No.: AD3283Ra), TOS (BT LAB; Rat ELISA Kits, Bioassay Technology Lab, Zhejiang, China, Cat No.: E1512Ra,), NO (BT LAB; Cat No.: E0703Ra, Bioassay Technology Lab, Zhejiang, China), eNOS (Sunlong; Rat ELISA Kits, Sunlong Technology Co., LTD, China, Cat No.: SL1159Ra), and MMP-9 (YL BioNT; YL Biont, Shanghai YL Technology Co, Ltd, China Cat No.: YLA0585Ra) were analyzed using the ELISA kit.
Statistical analysis
Biochemical analyses: Groups were compared based on each parameter using the Kruskal–Wallis
Histopathological analyses: Images were scored and averages were calculated. Data are presented as means ± standard deviations (
Results
Comparison of biochemical measurements between groups.

*:
Comparison of inflammatory markers between groups.

*:
A significant decrease in the TAS level was observed in the I/R group compared with the CR group (
The glomerular and tubular structures of the HE-stained samples were compared with those of the CR, less-damaged SH, and I/R groups. Damage, including Bowman space expansion in the glomerulus, degenerative and necrotic changes in tubular epithelial cells, inflammatory cell infiltration in the interstitial area, and vacuolization in the glomerular capillary bundle, increased significantly in the I/R group. No pathological deformities were observed in the kidneys, tubules, or glomeruli in the CR group. The SH group had a small amount of damage. Pathological distortions of the epithelial structure of the glomeruli and tubules were observed in the I/R group. Intensive degenerative structures and fluid accumulation associated with dilatation and edema were observed in the lumen, particularly in the epithelium of the proximal tubules surrounding the glomeruli. The lumen of the tubules was found to be extremely dilated owing to sloughing and deformation of the epithelium. The Bowman’s capsule and visceral cells were edematous, and the filtration space was dilated. The UR group had less damage than the I/R group (Figure 1). Pathological details were observed in all groups with HE staining (Figure 2). Statistical evaluations were performed by scoring the general morphological lesions after HE staining. The CR and SH groups had significantly higher numbers of lesion ( Transverse section of kidney cortex (figure above) and medulla with hematoxylin and eosin (HE) staining. Severe tubular damage, necrosis, tubular dilatation, vacuolization, loss of brush border, and hyaline cast formation in the tubular lumens (blue arrows) are observed for ischemia-reperfusion (I/R). HE staining of kidney samples from each group. In contrast to the CR and slightly damaged SH samples showing normal morphology, the damage is more pronounced in the I/R samples and less in the UP samples. HE, hematoxylin and eosin; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil. General morphological disorders in hematoxylin and eosin (HE) staining were scored and statistically evaluated. SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil.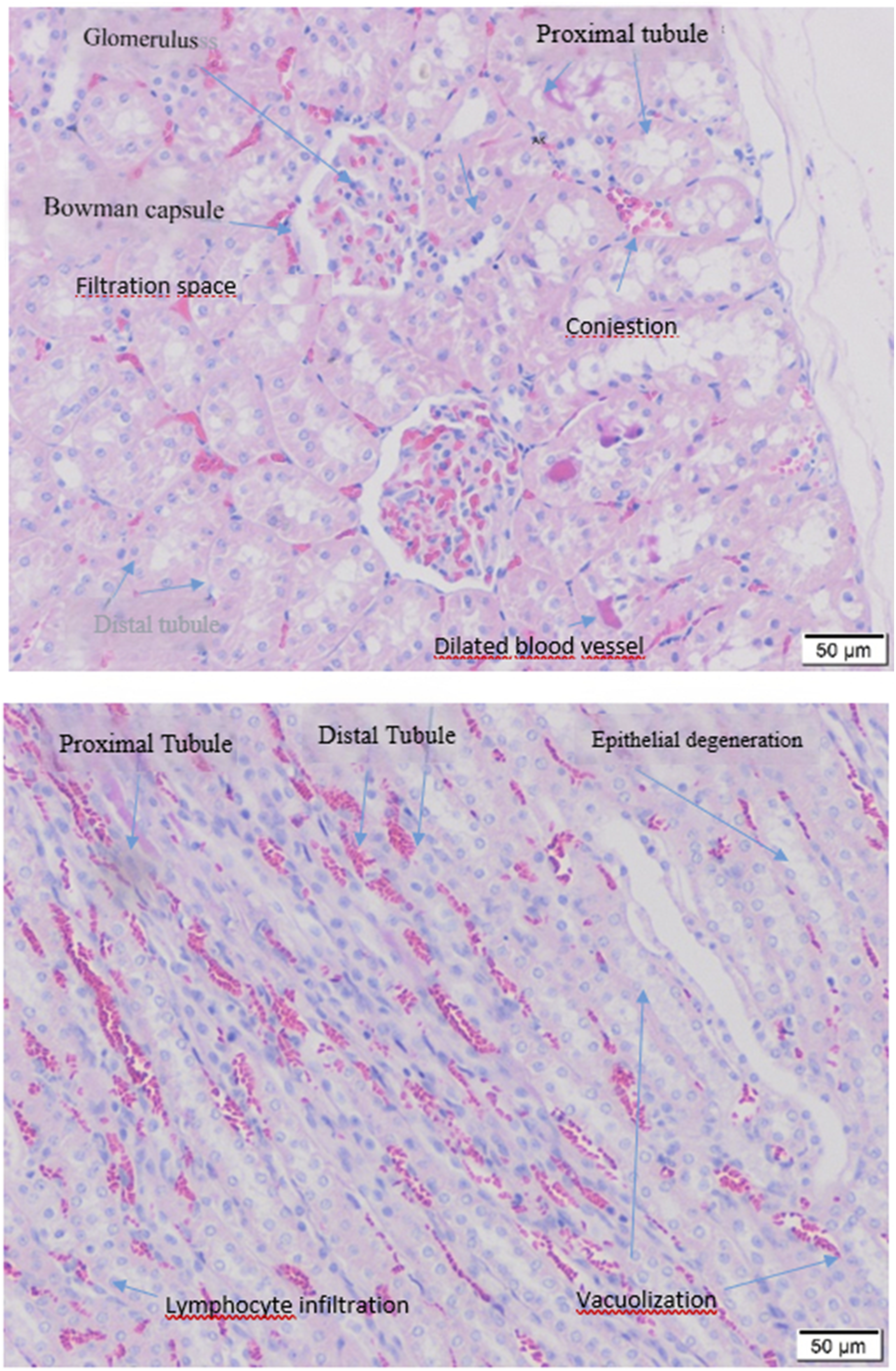


As an oxidative stress marker, eNOS immunoreactivity increased in the CR and SH groups after I/R and decreased after UP administration (Figure 3). Statistical evaluation was conducted by scoring eNOS immunoreactivity using IHC. Immunoreactivity was significantly ( eNOS IHC of kidney samples from each group. When eNOS IHC was compared with the CR and SH groups, increased oxidative stress is seen in the I/R group which is reduced in the UP samples. eNOS, endothelial nitric oxide synthase; IHC, immunohistochemistry; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil. Statistical evaluations were made with H-scoring in IHC eNOS markings. IHC, immunohistochemistry; eNOS, endothelial nitric oxide synthase; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil.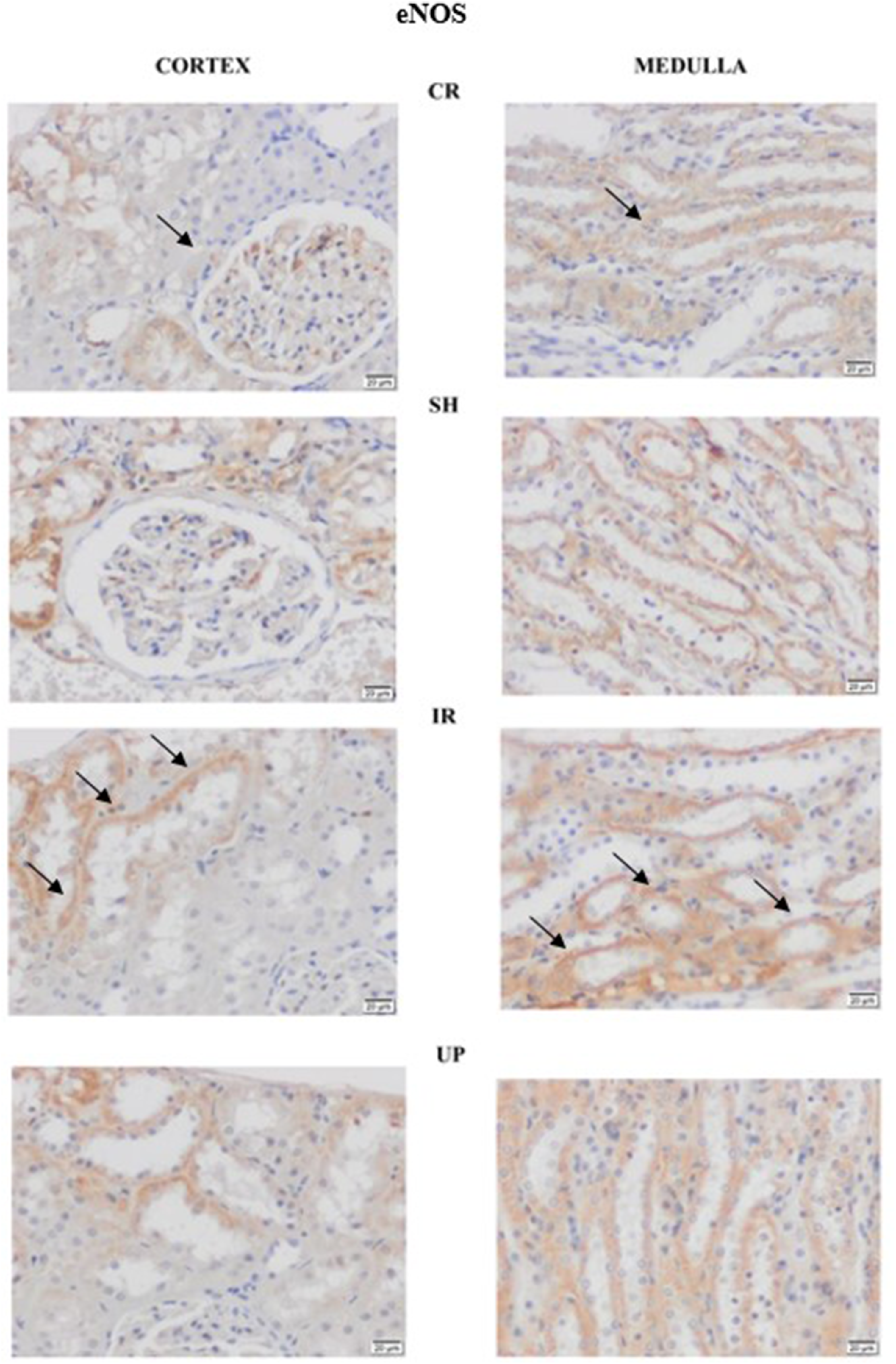

Caspase 3, a marker of apoptosis, had increased basal immunoreactivity in the CR and SH samples after I/R, which decreased after UP administration (Figure 4). Caspase 3 immunoreactivity was scored, revealing that immunoreactivity significantly ( Comparison of Caspase 3 IHC between the kidney samples from each group. Details of Caspase 3 labeling were observed with IHC. IHC, immunohistochemistry; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil. Statistical evaluations were made with h-scoring in IHC Caspase 3 labeling. IHC, immunohistochemistry; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil.

TNF-α, a marker of inflammation, had increased basal immunoreactivity in the CR and SH samples following I/R, which decreased after UP administration (Figure 5). TNF-α immunoreactivity was scored, revealing that the immunoreactivity increased significantly ( Comparison of TNF-α IHC between the kidney samples from each group. Details of TNF-α labeling were observed with IHC. TNF-α, tissue necrosis factor-alpha; IHC, immunohistochemistry; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil. Statistical evaluations were made with h-scoring in IHC TNF-α labeling. IHC, immunohistochemistry; TNF-α, tissue necrosis factor-alpha; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil.

IL-1β, an inflammatory marker, had increased basal immunoreactivity in the CR and SH samples following I/R, which decreased after UP administration (Figure 6). IL-1β immunoreactivity was scored, and statistical evaluations were made. Immunoreactivity increased significantly ( Comparison of IL-1β IHC between the kidney samples from each group. Details of IL-1β labeling with IHC were observed at this figure. IL-1β, interleukin-1 beta; IHC, immunohistochemistry; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil. Statistical evaluations were made with H-scoring in IHC IL-1β markings. IHC, immunohistochemistry; IL-1β, interleukin-1 beta; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil.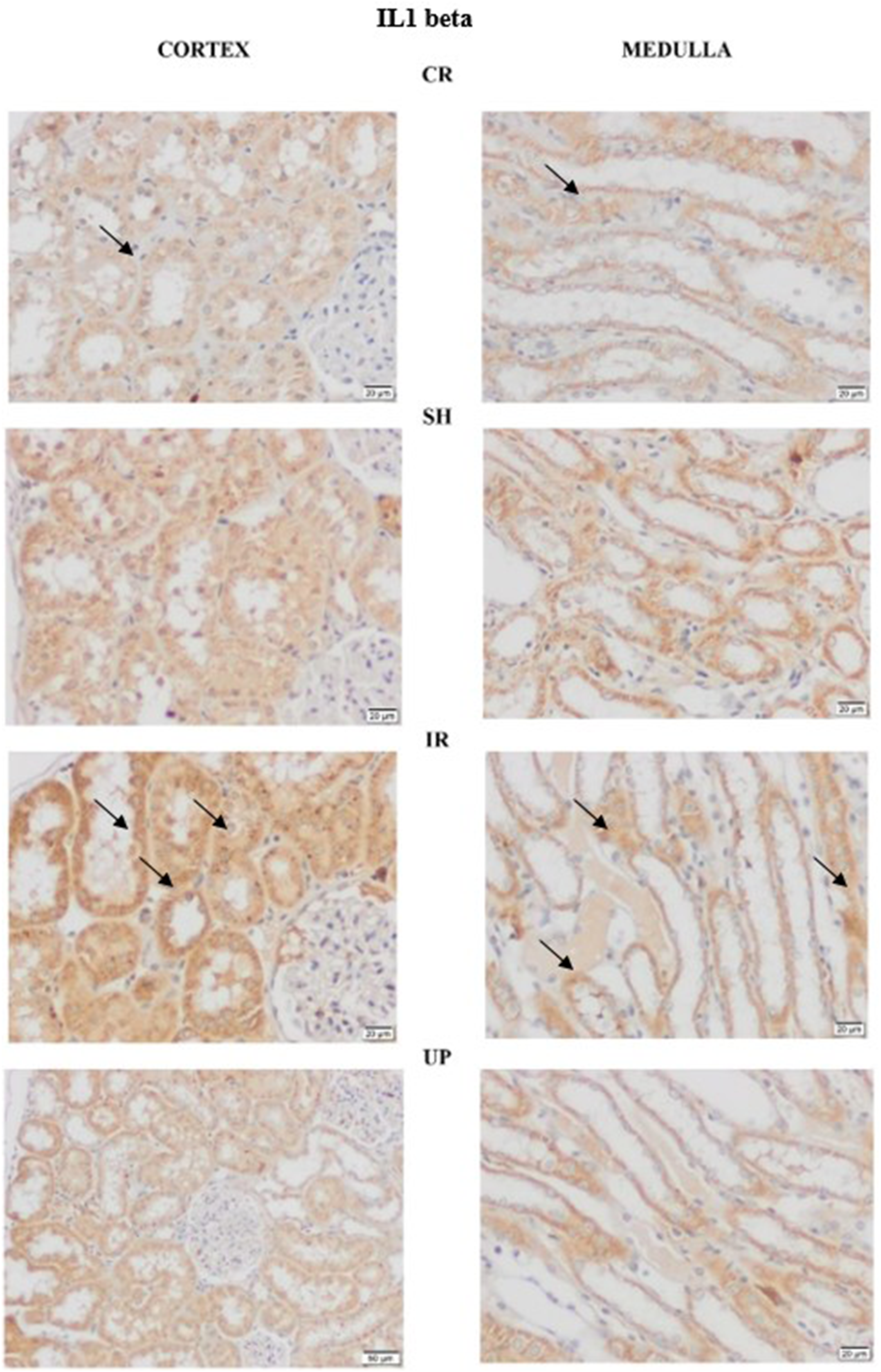

IL-6, an inflammatory marker, had increased basal immunoreactivity in the CR and SH samples following I/R, which decreased after UP administration (Figure 7). IL6 immunoreactivity was scored using IHC, and statistical evaluations were made, revealing that the immunoreactivity in the I/R group increased significantly ( Comparison of IL-6 IHC between the kidney samples from each group. IL-6, interleukin-6; IHC, immunohistochemistry; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil. Statistical evaluations were made with H-scoring in IHC IL6 markings. IHC, immunohistochemistry; IL-6, interleukin-6; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil.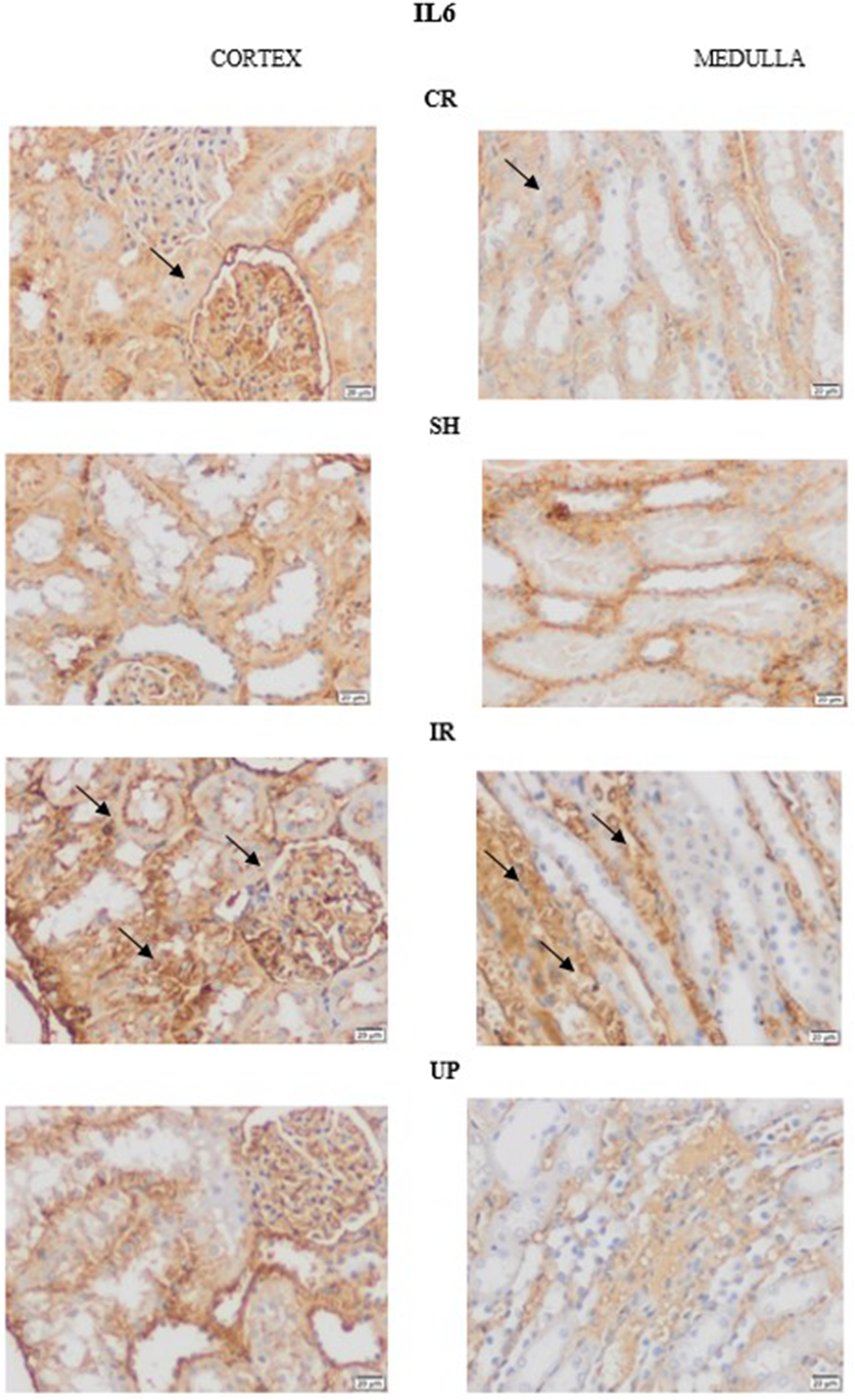

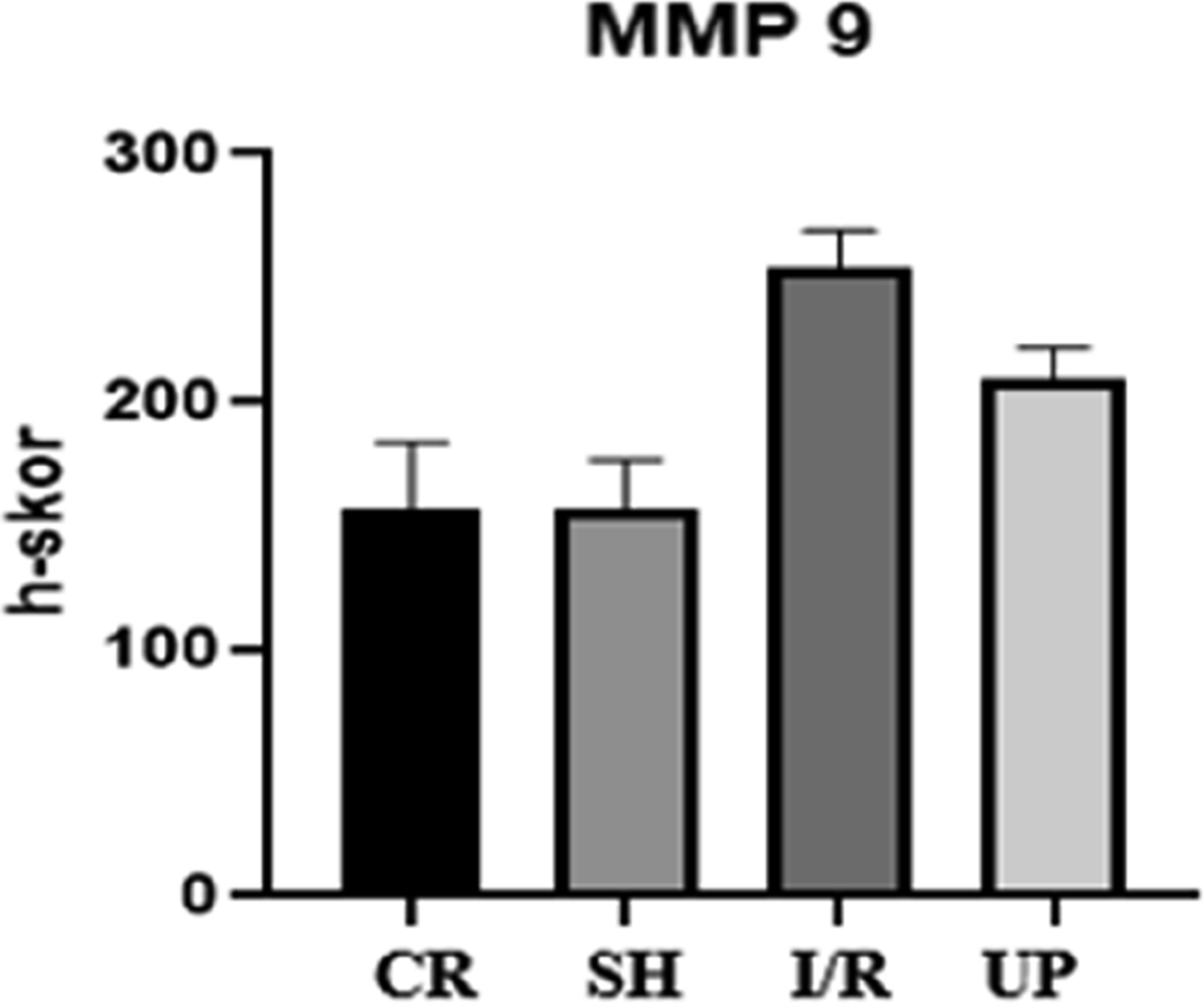
MMP-9 had increased basal immunoreactivity in the CR and SH samples following I/R, which decreased after UP administration (Figure 8). Statistical evaluations were conducted by scoring MMP-9 immunoreactivity using IHC. The immunoreactivity of the I/R group was significantly higher ( Comparison of MMP-9 IHC between the kidney samples from each group. Details of MMP-9 marking were observed with IHC. MMP-9, matrix metalloproteinase-9; IHC, immunohistochemistry; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil. Statistical evaluations were made with H-scoring in IHC MMP-9 markings. IHC, immunohistochemistry; MMP-9, matrix metalloproteinase-9; SH, sham group; CR, control group; I/R, ischemia-reperfusion injury; UP, urapidil.

Discussion
AKI is one of the leading causes of chronic kidney disease (CKD), accounting for 50–75% of deaths following severe kidney insults or organ transplantation.
33
Renal I/R injury, which involves the transient interruption of renal blood flow followed by reperfusion, is a major precipitant of AKI and worsens patient outcomes.3,4 Despite various pharmacological interventions, AKI due to I/R has a poor prognosis, underscoring the urgent need for new treatment options. UP is an antihypertensive drug that acts as a peripheral alpha-1 adrenergic receptor blocker and has a central 5-HT1A agonist effects.
8
In this study, we investigated the anti-apoptotic, antioxidant, anti-inflammatory, and matrix-regulatory effects of UP using an experimental renal I/R model. We found that UP treatment significantly ameliorated I/R-induced oxidative stress, inflammation, and apoptosis in kidneys. Improvements in the TAS/TOS balance and histopathological protection observed in UP-treated kidneys support the notion that UP can mitigate I/R injury. Our results suggest that UP may serve as a potential therapeutic adjuvant for renal I/R injury in the kidney and possibly in other organs. Importantly, these protective effects were achieved with a
Numerous previous studies have investigated the pathogenesis of renal I/R injury and potential therapeutic interventions, highlighting elevated serum creatinine and urea as primary markers of renal dysfunction following I/R.1,2,4,6 Consistent with these findings, our study demonstrated significantly increased serum creatinine and urea levels in the I/R group. Notably, UP treatment substantially reduced these elevations, suggesting an improvement in renal function. This result aligns well with the limited available literature on UP’s renal protective effects, such as the findings of Erdoğan et al., 10 who also reported significant improvement in renal function following UP administration.
Renal I/R injury is strongly associated with oxidative stress caused by excessive ROS production and/or inadequate antioxidant defenses. 7 Excessive ROS generation triggers lipid peroxidation, DNA damage, and mitochondrial dysfunction, leading to cell injury and death. It also initiates an inflammatory cascade: post-ischemic tissue releases large amounts of cytokines such as TNF-α, IL-6, and IL-1β, which recruit inflammatory cells and amplify tissue injury. IL-1β, IL-6, and TNF-α are well-known mediators of renal damage in I/R injury.34–36 In our study, we observed that IL-1β and TNF-α levels increased with I/R and were significantly reduced by UP administration, both in tissue homogenates and at the tissue level (IHC). Although IL-6 levels did not show a significant change in the ELISA measurements, their immunoreactivity was elevated in I/R kidneys and decreased with UP, suggesting that localized cytokine production in the kidney was curbed by UP. These results clearly demonstrate the anti-inflammatory properties of UP in the context of renal I/R, as UP blunted the upsurge of key inflammatory cytokines (TNF-α, IL-1β, and IL-6) that drive AKI progression. This is consistent with previous reports on UP’s anti-inflammatory effects in other I/R models.8,9
Additionally, our findings highlight the impact of UP on
Another important aspect of our study was the
The antioxidant effects of UP observed in our study (increase in TAS and reduction in oxidative damage markers) align with previous findings that UP activates antioxidant defenses. Improved TAS in UP-treated kidneys indicated enhanced total antioxidant capacity, which is beneficial for counteracting I/R-induced ROS surges. Additionally, UP’s reduction of eNOS immunostaining, an oxidative stress marker, 39 in I/R kidneys suggests a normalization of endothelial function. Since eNOS can be upregulated by shear stress and oxidant stimuli during reperfusion, its reduction by UP may reflect decreased oxidative stress in endothelial cells or a stabilization of endothelial nitric oxide production. This effect contributed to better perfusion and less vascular injury in the UP-treated group, thereby complementing the direct cellular protective effects.
Furthermore, UP significantly reduced caspase-3, an indicator of inflammation and apoptosis, 40 and its activation in renal tissue, demonstrating an anti-apoptotic effect. Apoptosis is a key mechanism of cell loss in I/R injury, and limiting apoptosis helps preserve nephronal integrity. By reducing caspase-3, UP helps maintain cell survival in the post-ischemic kidney, which likely contributed to the improved renal function and histology observed.
Overall, our study highlights two novel and clinically relevant findings. First, we demonstrated that UP is effective at very low microgram doses, conferring significant protection against renal I/R injury. Second, we established that UP modulates MMP-9 expression, thereby influencing ECM remodeling during I/R injury. These findings not only extend the pharmacological profile of UP, but also open new avenues for its potential clinical applications. The efficacy observed at low doses suggests that the protective mechanism of UP may involve high-affinity receptor targets or highly sensitive signaling pathways that require minimal drug concentrations for activation. Additionally, the observed modulation of MMP-9 underscores a protective mechanism aimed at preserving tissue architecture, potentially reducing the risk of fibrosis and subsequent CKD following acute I/R injury.
The results of this study are particularly promising from a translational perspective. UP is an established antihypertensive medication with a well-characterized human safety profile, 10 facilitating its potential repurposing for renal protection in clinical settings, such as kidney transplantation, vascular surgery, or AKI management. Importantly, demonstrating renal protection at significantly lower doses than those typically used for hypertension control highlights an essential therapeutic advantage: effective renal protection can potentially be achieved without substantial systemic blood pressure alterations. Consequently, UP may serve as a viable adjunctive treatment for patients at risk of renal I/R injury, offering protective benefits while minimizing cardiovascular risk. Furthermore, through its inhibitory effects on MMP-9, UP may attenuate post-ischemic inflammatory responses and fibrosis, thereby improving long-term renal outcomes. Although additional clinical studies are necessary to refine dosing strategies and optimal timing of administration, our study provides robust proof-of-concept evidence for the nephroprotective effects of UP and introduces a novel concept of matrix protection via MMP-9 modulation. These mechanistic insights could significantly enhance the therapeutic design of combined approaches aimed at preventing and treating AKI.
Conclusion
In conclusion, we found that a low microgram dose of UP (30 µg/kg with infusion) significantly protects against renal I/R injury by reducing oxidative stress, apoptosis, inflammation, and downregulating MMP-9 expression. Our study uniquely demonstrated the efficacy of UP at minimal doses and its novel role in modulating ECM remodeling through MMP-9 inhibition. These findings suggest that UP could safely be repurposed in clinical scenarios such as kidney transplantation or vascular surgery, providing organ protection without substantial hemodynamic alterations. In addition, its matrix-protective effects may improve long-term renal outcomes by reducing fibrosis. Future clinical trials are warranted to further evaluate the nephroprotective effects of UP.
Footnotes
Ethical considerations
The Afyon Kocatepe University Experimental Animal Ethics Committee approved the study (Aproval No: 49533702/101, Dated 11.10.2023).
Author contributions
H.S, Z.K.S and A.B contributed to writing–original draft, methodology, and conceptualization. H.A.K and F.G contributed to writing–review & editing. B.D, Y.K, B.G.E and Z.D contributed to software and resources
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets and supporting the findings of this study are available from the corresponding author, Hazen Sarıtaş, upon reasonable request.