Abstract
Objectives
Fluid management in critically ill obese patients is challenging due to the risk of fluid overload when using standard weight-based formulas. This study aims to evaluate the association between hydration status and mortality in elderly obese critically ill patients with acute kidney injury (AKI).
Methods
This retrospective cohort study included elderly obese critically ill patients with AKI admitted to a tertiary care ICU between January 2020 and December 2022. AKI was diagnosed according to the Kidney Disease: Improving Global Outcomes (KDIGO) criteria. Fluid overload was defined as body weight-adjusted fluid balance >10%. The primary outcome was 90-day all-cause mortality. Statistical analyses included Kaplan–Meier survival curves and multivariate Cox regression.
Results
A total of 539 elderly obese critically ill patients with AKI were included, of whom 244 (45.3%) developed fluid overload. During the 90-day follow-up, 209 patients (38.8%) died. Patients with fluid overload had significantly higher 90-day mortality compared to those without fluid overload (50.4% vs. 29.2%, P < 0.01). Kaplan–Meier analysis showed a higher mortality risk for fluid-overloaded patients (P < 0.01). Multivariate Cox regression indicated that fluid overload (HR = 2.04, 95% CI: 1.24–3.36, P < 0.01) and SOFA score (HR = 1.13, 95% CI: 1.04–1.22, P < 0.01) were independent predictors of increased mortality.
Conclusions
Fluid overload is significantly associated with increased 90-day mortality in elderly obese critically ill patients with AKI.
Introduction
Acute kidney injury (AKI) is a common and serious condition in critically ill patients, characterized by a sudden decline in kidney function that can lead to significant morbidity and mortality. 1 AKI affects approximately 13.3 million people globally each year, with an estimated mortality rate ranging from 20% to 50% in critically ill patients.2,3
The management of AKI is intrinsically associated with fluid status. On one hand, hypovolemia is a major cause of AKI, making prompt and adequate rehydration a cornerstone of treatment to restore renal perfusion. 4 On the other hand, the kidneys’ impaired ability to excrete excess fluid in AKI creates a high risk for fluid overload. 5 This paradoxical situation makes precise fluid management critically important yet challenging. 6 This challenge is particularly magnified in obese patients. Conventional fluid calculation formulas, which are often based on total body weight, are poorly suited for this population. 7 Adipose tissue has a lower metabolic rate and fluid content compared to lean body mass, leading these formulas to overestimate fluid requirements. 8 Consequently, obese critically ill patients are at a heightened risk of iatrogenic fluid overload, which can exacerbate conditions such as pulmonary edema, heart failure, and impaired wound healing, thereby increasing mortality. 9
The interplay between obesity, AKI, and fluid status creates a complex clinical scenario. Fluid overload may be a critical, modifiable risk factor that disproportionately affects outcomes in obese patients with AKI. However, the specific influence of hydration status on mortality in the high-risk subgroup of elderly obese critically ill patients with AKI has rarely been investigated. Therefore, the objective of this study was to evaluate the association between hydration status and mortality in this unique and vulnerable population.
Materials and methods
Patients
This retrospective cohort study was conducted at Pu’er People's Hospital, utilizing consecutive data from the first ICU admissions between January 2020 and December 2022. Patients were included if they: (a) aged≥65 years; (b) had obesity 10 defined as body mass index (BMI) ≥ 28 kg/m2; (c) were diagnosis of AKI based on kidney disease: Improving Global Outcomes (KDIGO) criteria 11 ; (d) needed rehydration fluid management. Patients with chronic kidney disease (CKD) stage 4 or higher (estimated glomerular filtration rate <30 mL/min/1.73 m2) 12 or active malignancy were excluded based on review of electronic medical records and laboratory results at admission. All patient details have been de-identified.
The study was approved by the Institutional Review Board of Pu’er People's Hospital (approval no. PEIEC2023124, approval date: 2023/03/22). The study was conducted in accordance with the principles of the Declaration of Helsinki of 1975 as revised in 2024. Informed consent was waived by the Institutional Review Board of Pu’er People's Hospital due to the retrospective nature of the study and the use of de-identified data. All data were stored securely, and confidentiality was maintained throughout the study. In addition, the reporting of this study conforms to STROBE guidelines. 13
Data collection
Patients’ baseline demographic, medical history, clinical and laboratory data and severity of illness were extracted from electronic medical records. The Acute Physiology and Chronic Health Evaluation III (APACHE III) score 14 was calculated based on the worst values within the first 24 hours of ICU admission. Sequential Organ Failure Assessment (SOFA) scores were calculated daily during the ICU stay, and the highest (worst) value was used to represent the severity of organ dysfunction. Organ failure was defined according to SOFA criteria (organ-specific score ≥2). 15 Sepsis was defined according to the Sepsis-3 criteria. 16 Ventilation status was defined as the need for invasive mechanical ventilation at any time during the ICU stay.
The intravenous input and output data were obtained from the first ICU admission until the ICU discharge. To quantify cumulative fluid balance in relation to body weight, the “body weight-adjusted fluid balance” was calculated as (total fluid intake - total fluid lost)/baseline body weight. Fluid overload was defined as “body weight-adjusted fluid balance” > 10%, consistent with previous studies.17,18 Fluid administration in the ICU generally followed a standardized protocol based on body weight, hemodynamic status, urine output, and comorbidities; however, physician discretion was applied for individualized adjustments based on the patient's acute condition and laboratory results. Consequently, some patients experienced fluid overload >10%, particularly elderly obese patients with complex critical illness, reflecting real-world ICU practice.
The primary outcome was the 90-day all-cause mortality, which was collected from medical records and/or telephone interview.
Statistical analysis
Statistical power was determined on the primary outcome of 90-day mortality. It was estimated that 244 patients in each group would provide >90% power with an alpha of 5% to detect a 10% difference in 90-day mortality between patients with and without fluid overload.
All variables had <5% missing values; missing data were handled using multiple imputation to minimize potential bias. Differences in time-to-event outcomes were assessed using log-rank tests and graphically presented with Kaplan–Meier curves. Univariate analyses were performed to evaluate associations between each variable and 90-day mortality. Variables with a P value <0.1 in univariate analysis were entered into the multivariate Cox regression model. Multicollinearity among variables was assessed using variance inflation factors (VIF), with VIF >5 indicating potential collinearity. The proportional hazards assumption for the Cox model was tested using Schoenfeld residuals, and all variables satisfied the assumption.
Subgroup analyses were further conducted to evaluate the consistency of the association between fluid overload and mortality across age groups (65–74 years and ≥75 years) and BMI categories (28–32.4 kg/m2 [Grade 1 obesity] and ≥32.5 kg/m2 [Grade 2 obesity]) according to the suggestions of the WHO for Chinese population. 19 Sensitivity analyses were performed by applying alternative thresholds for defining fluid overload (≥5% and ≥15% of body weight-adjusted cumulative fluid balance) to assess the robustness of the primary findings.
All tests were 2-sided and a P value <0.05 was considered statistically significant. All statistical analyses were performed with the SPSS statistical software program package (SPSS version 22.0 for Windows, Armonk, NY: IBM Corp.).
Results
A total of 692 elderly obese critically ill patients with AKI were identified between January 2020 and December 2022. After excluding 153 patients with CKD stage 4 or higher or active malignancy, 539 patients were finally included in the analysis. Among them, 244 (45.3%) experienced fluid overload, while 295 (54.7%) did not (Figure 1). Patients’ demographics, medical history, severity of illness scores, and clinical and laboratory data stratified by the presence or absence of fluid overload are included in Table 1. Compared to those without fluid overload, patients with fluid overload had significantly higher APACHE III and SOFA scores, a greater number of failed organ systems, and higher proportions of sepsis and ventilator requirement.

Patient flowchart.
Patients’ characteristics based on fluid overload status.

APACHE, Acute Physiology and Chronic Health Evaluation; BMI, body mass index; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; FiO2, inspired oxygen fraction; SOFA, Sequential Organ Failure Assessment.
During the 90-day follow-up, 209 deaths (38.8%) occurred in the overall population. Mortality was significantly higher in patients with fluid overload compared to those without (50.4% vs. 29.2%, P < 0.01). As shown in Figure 2, the Kaplan–Meier survival curve demonstrated a significantly higher cumulative risk of mortality among patients with fluid overload compared to those without (log-rank P < 0.01) (P < 0.01).
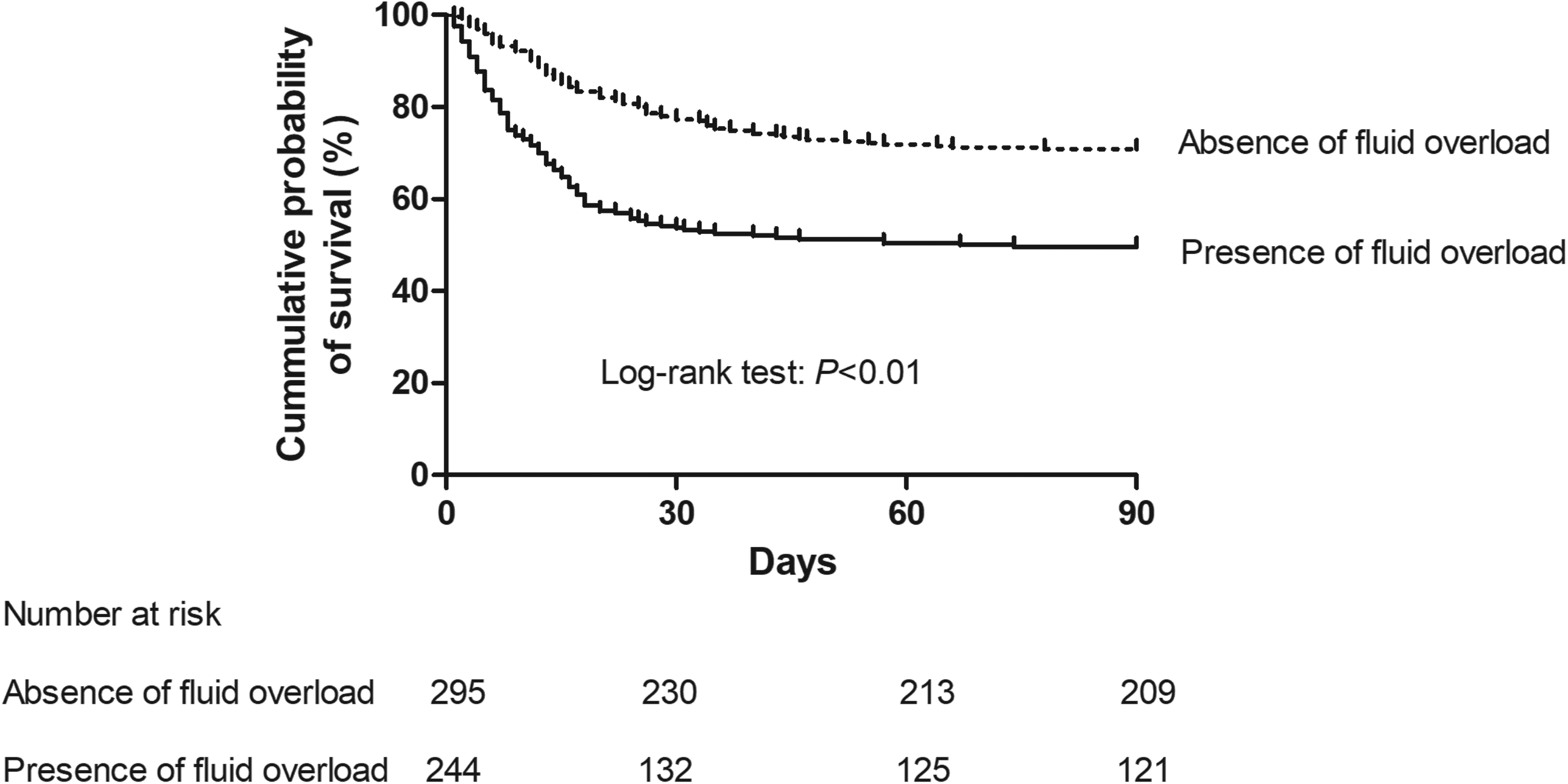
Kaplan–Meier survival analysis for 90-day mortality stratified by fluid overload status. The solid line represents patients with fluid overload, and the dashed line represents those without. The difference between the curves was assessed using the log-rank test (P < 0.01). The number of patients at risk for each group at pre-specified time points (0, 30, 60, and 90 days) is presented in the table beneath the figure. The declining numbers in the “fluid overload” group correspond to its higher mortality rate, confirming the association between fluid overload and reduced survival over time.
In univariate Cox regression analysis (Table 2), several variables were associated with 90-day mortality, including fluid overload, age, heart rate, FiO2, APACHE III score, SOFA score, number of organ failures, need for ventilator, and sepsis/septic shock (all P < 0.10). Variables with a P < 0.10 in univariate analysis were entered into the multivariate Cox regression model. After adjustment, only fluid overload (HR = 2.04, 95% CI: 1.24–3.36, P < 0.01) and SOFA score (HR = 1.13, 95% CI: 1.04–1.22, P < 0.01) remained independent predictors of mortality. Subgroup analyses revealed that the association between fluid overload and mortality remained consistent across age and BMI strata (Table 3). In patients aged 65 to 74 years, fluid overload was associated with a 2.46-fold increased risk of death (95% CI: 1.64–3.68; P < 0.01), while in those aged ≥75 years, the risk increased to 5.89-fold (95% CI: 2.87–12.09; P < 0.01). Similarly, among patients with a BMI of 28 to 32.4 kg/m2 and ≥32.5 kg/m2, fluid overload was associated with a 4.05-fold (95% CI: 2.41–6.83; P < 0.01) and 2.84-fold (95% CI: 1.64–4.94; P < 0.01) higher mortality risk, respectively.
Univariate and multivariate cox regression for exploring the association of fluid overload with all-cause 90-day mortality.

APACHE, Acute Physiology and Chronic Health Evaluation; BMI, body mass index; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; FiO2, inspired oxygen fraction; SOFA, Sequential Organ Failure Assessment.
Subgroup analysis of the association between fluid overload and 90-day all-cause mortality across age and body mass index (BMI) strata using multivariate cox regression.

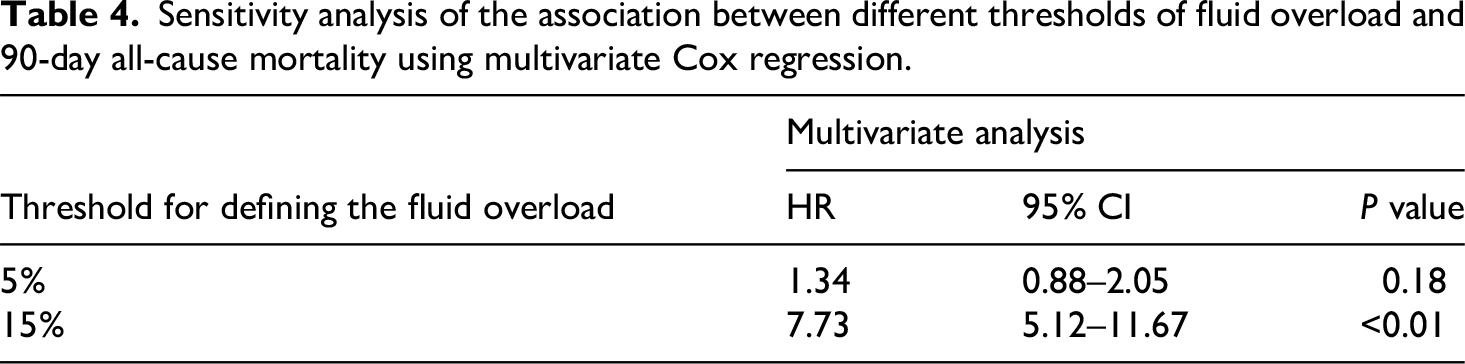
In sensitivity analyses using different definitions of fluid overload, the results were directionally consistent (Table 4). When the cutoff was set at 5%, the association with 90-day mortality was weaker and not statistically significant (HR = 1.34, 95% CI: 0.88–2.05; P = 0.18). At a 15% cutoff, the association strengthened and remained significant (HR = 7.73, 95% CI: 5.12–11.67; P < 0.01).
Sensitivity analysis of the association between different thresholds of fluid overload and 90-day all-cause mortality using multivariate Cox regression.
Discussion
This study aimed to investigate the association between hydration status and mortality in the specific high-risk subgroup of elderly obese critically ill patients with AKI—a population that has been underrepresented in prior research on fluid overload. While the detrimental effects of fluid overload in general AKI populations are well established, its impact within this unique demographic remains poorly characterized. Our findings confirm that fluid overload is not only prevalent but also strongly and independently associated with increased 90-day mortality in this vulnerable group, thereby addressing a critical gap in the literature.
Our findings align with existing literature that highlights the detrimental effects of fluid overload in critically ill patients. Several studies have documented that fluid overload can increase the risk of mortality and adverse outcomes, especially in patients with AKI. The Program to Improve Care in Acute Renal Disease (PICARD) program showed that mortality at 30 days (37% vs. 25%, P = 0.02), 60 days (46% vs. 32%, P = 0.006), and at hospital discharge (48% vs. 35%, P = 0.01) was significantly higher in patients with fluid overload. 20 Similarly, the RENAL Replacement Therapy Study indicated that a positive mean daily fluid balance during study treatment was independently associated with an increased risk of death at 90 days (P = 0.0017). 21 Our study extends this evidence by focusing specifically on elderly obese patients, who represent a unique and growing subgroup of the critically ill population often underrepresented in prior research. The sensitivity analysis further demonstrated that the association between fluid overload and mortality was dose-dependent, becoming more pronounced at higher thresholds, which reinforces the clinical relevance of our findings.
Although our study did not directly compare this cohort with the general ICU population, elderly obese patients likely had a higher prevalence of chronic comorbidities, which may further increase their vulnerability to fluid overload. Previous data support this notion. For instance, a study from a tertiary ICU in Turkey reported that compared with adult patients of normal weight, elderly obese patients had significantly higher incidences of hypertension (54.2% vs. 27.5%), diabetes (36.7% vs. 15.0%), chronic obstructive pulmonary disease (14.2% vs. 8.7%), and chronic renal failure (11.1% vs. 8.2%) (all P < 0.05). These findings suggest that the elevated comorbidity burden in elderly obese individuals could partly explain their increased susceptibility to the adverse effects of fluid overload. In addition, they are physiologically more susceptible to the adverse effects of fluid overload due to reduced renal reserve, impaired cardiac function, and altered fluid distribution. Epidemiological data suggest that AKI occurs in approximately 20% to 50% of critically ill patients, and obesity has been identified as an independent risk factor for AKI through mechanisms such as chronic inflammation, renal hyperfiltration, and elevated intra-abdominal pressure. 22 Therefore, elderly obese patients likely represent a subgroup at higher baseline risk for both AKI and fluid accumulation, which may explain the relatively high prevalence of fluid overload (45%) observed in our study. Several factors may further contribute to this finding, including limitations of body weight-based fluid calculations, age-related organ dysfunction, and clinical variability in fluid administration under critical conditions.
The mechanisms behind the increased mortality observed in fluid-overloaded patients are multifaceted. Obese patients have a higher baseline risk for conditions such as sleep apnea, hypertension, and cardiovascular diseases, which can be exacerbated by fluid overload. 23 The reduced respiratory reserve due to excess adipose tissue further complicates the clinical scenario. 24 Moreover, the altered pharmacokinetics in obese patients can affect drug distribution and efficacy, potentially leading to suboptimal treatment responses. 25 Fluid overload can also lead to increased intra-abdominal pressure, impairing renal perfusion and exacerbating AKI. 26
Despite the significant findings, this study has several limitations. Being a retrospective cohort study, it is susceptible to selection bias and residual confounding, as some potential confounders, such as diuretic use, timing of dialysis, and vasopressor support, were not available in the dataset and could not be included in the analysis. The single-center design may limit the generalizability of the results to other settings with different patient populations and treatment protocols. Additionally, the definition of fluid overload based on body weight-adjusted fluid balance, while commonly used, may not fully capture the complexity of fluid dynamics in critically ill patients.
Conclusion
In conclusion, our study demonstrates a significant association between fluid overload and increased mortality in elderly obese critically ill patients with AKI. These findings highlight the urgent need for tailored fluid management strategies to improve clinical outcomes in this high-risk population. Efforts to optimize fluid therapy prevent fluid overload, and monitor patient response closely are essential components of care for obese patients with AKI in the ICU setting.
Footnotes
Acknowledgments
The authors thank all the authors who participated in this study.
Ethical considerations
The study was approved by the Institutional Review Board (IRB) of Pu’er People's Hospital (approval no. 2023124, approval date: 2023/03/22). The study was conducted in accordance with the principles of the Declaration of Helsinki of 1975 as revised in 2024.
Consent to participate
Since all data were fully anonymized before we accessed them, informed consent was waived by the IRB of Pu’er People's Hospital. All data were stored securely, and confidentiality was maintained throughout the study.
Consent for publication
Not applicable.
Author contributions
Junhua Yang conceived the study idea and designed the study; Jiesheng Yang collected the data, performed the data analysis, and drafted the manuscript; All authors reviewed the manuscript and agreed to submit it.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
