Abstract
Acute kidney injury (AKI) is a common condition in intensive care units (ICUs) and is associated with high mortality rates, particularly when kidney replacement therapy (KRT) becomes necessary. The optimal timing for initiating KRT remains a subject of ongoing debate. Emerging tools and methodologies, such as machine learning and advanced sub-phenotyping, offer promising insights into refining AKI management. Moving beyond the traditional “early” versus “delayed” paradigm and the heavy reliance on serum creatinine measurements, there is an opportunity to develop treatment strategies tailored to the unique pathophysiological and medical context of each patient. Such individualized approaches could potentially improve outcomes and transform AKI management into ICUs. However, KRT is not without risks. Hemodynamic instability poses a significant challenge, complicating the management of critically ill patients. The selection of a KRT modality—whether intermittent hemodialysis, continuous renal replacement therapy, or peritoneal dialysis—introduces additional complexities. Each modality has distinct advantages and limitations, requiring a careful, patient-specific approach to ensure optimal care. This decision-making process is further influenced by the availability of specialized equipment and trained personnel, resources that may be limited in some settings. Notably, current evidence does not demonstrate a clear survival or recovery benefit from the early initiation of dialysis. This narrative review explores the ongoing debates surrounding KRT timing and methodology, highlighting the importance of adopting patient-centric, individualized strategies to navigate the evolving landscape of AKI management.
Introduction
Acute kidney injury (AKI), characterized by its significant heterogeneity, has profound implications for both short- and long-term patient outcomes. 1 Within the intensive care unit (ICU) environment, AKI manifests at an alarming prevalence, affecting 30–60% of admissions and mortality rates over 50% when necessitating kidney replacement therapy (KRT).2,3 While current clinical practice guidelines articulate the initiation of KRT in addressing severe AKI-related complications, such as hyperkalemia, metabolic acidosis, and fluid overload-induced pulmonary edema, there remains a lack of consensus on the optimal timing for KRT initiation when these severe complications are not present. This lacuna in the guidelines has instigated extensive deliberations and divergence of opinions among ICU practitioners.4–8 The confluence of this lack of directive clarity and apprehensions regarding the potential adverse events of KRT has culminated in a cautious approach to KRT initiation by many clinicians. 8 Expanding the introduction further, it is crucial to balance the benefits and risks associated with KRT. While KRT can be life-saving in acute settings, its adverse effects, such as hemodynamic instability and the challenge of choosing the appropriate modality, must be considered. Major clinical trials have provided insights but have also highlighted the complexities and nuances of KRT management in different patient populations.
In this narrative review, we aim to examine the current evidence on the ongoing controversies around KRT timing in critically ill patients with AKI, discuss why solely relying on the “early vs. delayed” paradigm may be insufficient, and consider how emerging biomarkers and machine learning (ML) approaches might inform more individualized care.
Methods
In accordance with the Scale for the Assessment of Narrative Review Articles (SANRA) 9 and to ensure methodological rigor, a structured literature search was conducted using PubMed, Scopus, and Web of Science to identify studies published from January 2018 through July 2024. We used the following search terms (including both free-text and, where applicable, MeSH terms): “acute kidney injury,” “AKI,” “kidney replacement therapy,” “renal replacement therapy,” “timing of dialysis,” “early initiation,” “late initiation,” “indications,” “sub-phenotyping,” “critical care,” and “ICU.” To ensure comprehensive coverage, we also screened reference lists from key research articles, reviews, and meta-analyses for additional sources. Only peer-reviewed English language-publications were included. Conference abstracts, commentaries without primary data, and papers focusing exclusively on pediatric populations were excluded.
An initial screening based on titles and abstracts was conducted by the first author, and it was followed by full-text review of potentially relevant articles by two authors. Studies were selected if they addressed the timing, modality choice, or individualized management of KRT in adult ICU patients with AKI, with particular emphasis on hemodynamic implications, outcome measures, and novel methodologies (e.g. ML-based approaches). Where multiple publications from the same research group overlapped, the most recent and comprehensive article was chosen.
Acute kidney injury in the intensive care unit
Prevalence and setting
AKI is characterized by a rapid decline in estimated glomerular filtration rate, signifying impaired kidney function. 10 AKI occurring in the ICU stands as an independent risk factor for mortality with rates ranging from 40 to 55% exceeding the mortality rates observed in serious conditions such as myocardial infarction, sepsis in the absence of AKI, and acute respiratory distress syndrome.10,11 The pathogenesis of AKI in ICU patients is multifactorial. Disturbances in glomerular blood flow can arise from hypovolemic and ischemic conditions exacerbated by factors such as periglomerular shunting and endothelial dysfunction. 12 Additionally, inflammatory responses triggered by damage-associated and pathogen-associated molecular patterns (DAMPs and PAMPs) lead to immune cell activation and subsequent harmful effects. Tubular disruptions through processes such as ferroptosis and mitochondrial dysfunction add to AKI's varied presentations. 13 The overall complexity is further compounded by organ crosstalk and nephrotoxic effects of some medications. 14
AKI in the context of sepsis is particularly significant, as sepsis is the most common cause of AKI in ICU settings and the focus of many clinical trials. The inflammatory and hemodynamic alterations induced by sepsis compound the risk of AKI, leading to worse outcomes. Studies have shown that septic AKI is associated with higher mortality and prolonged hospital stays compared to non-septic AKI. 2 Following AKI onset, its repercussions extend beyond immediate threats. Long-term AKI predisposes patients to recurrent episodes, increased chances of hospital readmission, cardiovascular complications, oncological issues, and hastened progression to chronic kidney disease. 11
Basic management
AKI management involves carefully considering volume status and hemodynamic management. Both volume depletion and fluid overload can harm kidney function, and striking the right balance is crucial. 15 Intravenous fluids can be beneficial in cases of true hypovolemia, but diagnosing this condition is challenging. Empiric fluid resuscitation based solely on serum creatinine (sCr) levels or oliguria can be misguided, especially in patients with fluid intolerance syndrome. Although fluid responsiveness tests can identify patients who are unlikely to benefit from fluid administration, the presence of fluid responsiveness does not necessarily mean that fluid therapy is indicated or safe. Individualized care, including monitoring blood pressure and cardiac function, is crucial for AKI. 16 Vasopressors are essential in managing hypotension when volume resuscitation alone is insufficient or in severe vasodilatory shock, including septic shock, where they help preserve renal perfusion only after intravascular volume is restored. Norepinephrine is first-line, but vasopressin may better protect kidneys by minimizing afferent arteriolar constriction while constricting efferent arterioles. Other vasopressors offer inconsistent kidney outcomes. Goal-directed therapy, combining fluid and vasopressor titration, can further prevent AKI. 17 Nephrotoxic management of AKI involves the prompt identification and discontinuation of all potentially nephrotoxic drugs and agents. Avoidance of nephrotoxic agents is a crucial element in preventing AKI, especially if associated with risk factors (pre-existing CKD, dehydration, other nephrotoxic medications). However, it is necessary to emphasize the importance of adapting dosages to renal function in order to avoid underdosing or overdosing which can lead to renal toxicity and the occurrence of AKI. When such drugs are essential for treatment, prophylactic volume expansion with crystalloids should be used to prevent AKI, with careful monitoring of the drug concentrations and kidney function. Contrast agents in the context of investigations or interventions should not be delayed for potential preventative measures. 18
While these management approaches are vital, an equally critical intervention in AKI is the initiation of KRT. The dilemma is not just about deciding to initiate KRT but more pertinently, when to initiate KRT. A plethora of research exists, yet a clear consensus has yet to be reached. The discrepancies in various studies underline the need to further explore this topic.
Evidence on KRT timing: What do recent research reveal?
A large number of recent systematic reviews and meta-analyses, summarized in Table 1, revealed a predominant result on the impact of early KRT initiation. A significant majority of these studies, hailing from diverse global locations and encompassing varying numbers of randomized controlled trials (RCTs), converge on the notion that early KRT initiation does not confer a notable survival advantage in critically ill patients with AKI.18–34 This consensus remains robust, even when studies dissect the results in varying populations, such as those with septic AKI or those in surgical ICUs.20,23 However, nuances emerge, most prominently from the Cochrane reviews by Fayad et al., where an initial 2018 study suggested the potential benefits of early KRT initiation, 19 but only for a subsequent 2022 study to echo the prevailing sentiment. 30 Examination of the previously mentioned meta-analyses necessitated a comprehensive review of the primary RCTs from 2016 to 2020, providing an in-depth perspective on the dynamics of early KRT initiation in patients with AKI (Table 2). Published in esteemed academic journals, these seminal studies, while united in their focus, manifest marked heterogeneity in their definitions of “early” initiation, reflecting the complexities and nuances of the issue at hand.35–40 For instance, “early” in AKIKI STUDY is delineated as a mere 6-h post-diagnosis of Kidney Disease: Improving Global Outcomes (KDIGO) stage 3 AKI, 35 while the STARRT AKI study perceives early as within 12 h following a KDIGO stage 2 or 3 diagnosis. 38 Notably, the ELAIN study, which sets its early initiation threshold at 8 h for stage 2 AKI, stands apart by indicating a benefit associated with the early initiation strategy 36 ; a finding that contrasts with the majority of other studies, such as the FST, which defines “early” within 6 h of any KDIGO stage of AKI without a response to the Furosemide Stress Test. 39 This variability in defining “early” illuminates the absence of a standardized, universally accepted criterion, a challenge that significantly impacts clinical decision-making and research outcomes synthesis. In the 2021, AKIKI 2 study conducted across 39 French ICUs, comparing “delayed” versus “more-delayed” KRT initiation strategies revealed that postponing KRT for an extended duration was associated with an increased risk of 60-day mortality. 41 Beyond these definitions, the interventions present their own tapestry of diversity (Table 2), with modalities mostly left to the physician's discretion, ranging from intermittent hemodialysis and Continuous Veno-venous Hemodiafiltration to Continuous Veno-venous hemofiltration and Sustained Low-Efficiency Dialysis.35–41 This landscape of findings underscores the multifaceted nature of the impact of KRT initiation and accentuates the need for nuanced interpretations tailored to specific patient profiles.
Recent metanalysis on the timing of initiating kidney replacement therapy in AKI.
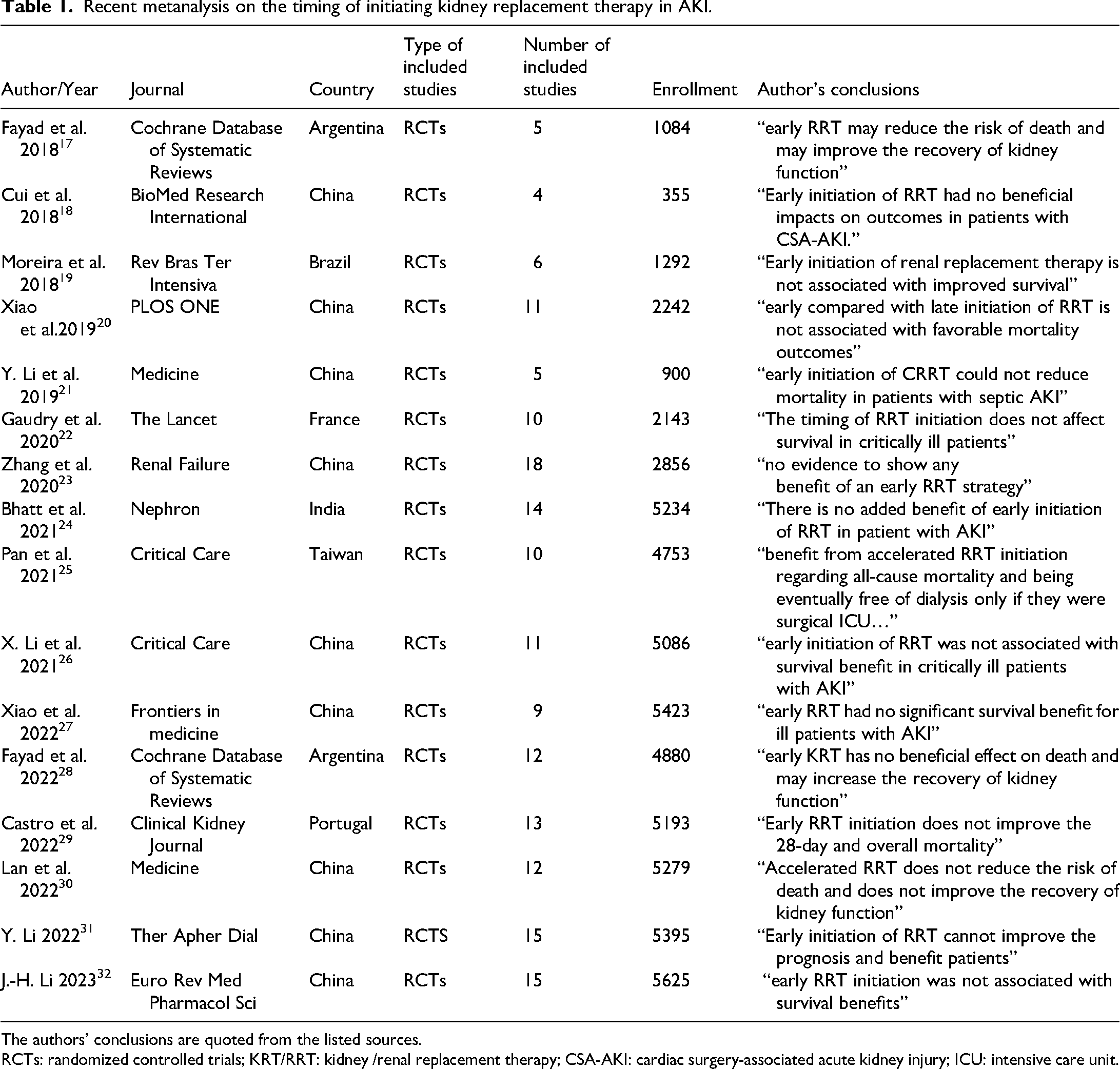
The authors’ conclusions are quoted from the listed sources.
RCTs: randomized controlled trials; KRT/RRT: kidney /renal replacement therapy; CSA-AKI: cardiac surgery-associated acute kidney injury; ICU: intensive care unit.
Comparison of main recent trials in timing of initiating kidney replacement therapy in the ICU.

AKI: acute kidney injury; KDIGO: Kidney Disease Improving Global Outcome; IHD: intermittent hemodialysis; CRRT: continuous renal replacement therapy; CVVHDF: continuous Veno-venous hemodiafiltration; SLED: slow low-efficiency dialysis; ICU: intensive care unit.
Dissecting the problematic of AKI
The Kidney Disease: Improving Global Outcomes definition
The KDIGO group established a standard AKI definition, emphasizing the changes in sCr levels over a week or the onset of oliguria. This consensus has played a key role in shaping research on AKI. 7 While sCr remains a conventional marker for kidney function, its use as a sole determinant for initiating KRT in critically ill patients is fraught with limitations. The inherent variability in creatinine production in critically ill individuals coupled with fluctuating fluid balances makes its interpretation challenging. However, the advent of “KDIGO AKI stages” based on sCr and urine output fails to capture the nuanced etiological, anatomical, and cellular responses of kidney injuries. 42 Such categorizations, particularly in critically ill patients, risk oversimplifying a multifaceted condition, which could lead to suboptimal clinical decisions. In the context of KRT for critically ill patients, there is an urgent need to move beyond the sCr-centric paradigm.43,44
A dichotomous approach: Early vs delayed initiation of dialysis
AKI is not a singular, homogenous entity, but rather a loose collection of syndromes, each with unique pathophysiological underpinnings. Recognizing this heterogeneity is crucial when formulating therapeutic strategies and interpreting clinical research. 12 The prevailing dichotomic classification of KRT initiation into “early” versus “delayed” categories, while offering some semblance of clinical simplicity, becomes particularly limited in the context of AKI's diverse spectrum. In a sense, such binary classification seeks to fit a multifaceted clinical phenomenon into a one-size-fits-all framework, which can be problematic. 45 The lack of universally consistent definitions for “early” and “delayed” across studies amplifies these challenges. For instance, while AKIKI and STARRT-AKI studies employ differing timelines for “early” initiation, their definitions become even more convoluted when considered in meta-analyses.35,38 Collating data from studies with divergent definitions dilutes the precision of the findings, potentially hindering scientific progress and yielding less actionable insights (Figure 1). Approaching a therapy as sensitive as KRT with dichotomous reasoning for a heterogeneous clinical syndrome, such as AKI, can lead to oversimplification. This may culminate in premature interventions for some patients and undue delays in essential treatment for others. The variability inherent in AKI mandates an approach that goes beyond dichotomies, emphasizing the need for individualized, patient-centric strategies.

“Early” definitions in major recent clinical trials (based on Table 2).
Can biomarkers help in initiating KRT?
sCr has been a conventional marker for kidney function, yet its use as the sole determinant for initiating KRT in critically ill patients is fraught with limitations. The variability in creatinine production in critically ill individuals, coupled with fluctuating fluid balances, makes its interpretation challenging. As such, relying exclusively on sCr can lead to suboptimal clinical decisions.
46
Therefore, several urine and plasma biomarkers have been explored in the setting of AKI, and a three-tiered biomarker framework has emerged, encompassing functional, stress, and damage biomarkers.
However, their accuracy in anticipating the requirement for KRT at the bedside remains under investigation. For instance, a previous systematic review and meta-analysis included 41 studies evaluating 13 different biomarkers showed promising results in predicting the need for KRT but the strength of evidence could not recommend their routine use in clinical decision-making regarding KRT initiation. 48 A further meta-analysis from 2020 proposed threshold values for both urinary and plasma NGAL that correspond to an increased risk of severe AKI or AKI requiring dialysis. These cutoffs could aid clinical research and practice by highlighting patients at greater renal risk. 49 Moreover, exosomes are small messengers that help cells communicate and play a role in both damage and healing in AKI. Researchers are using multi-omics (studying genes, proteins, and metabolites together) to better understand AKI, detect it earlier, and find new treatments. There is also growing interest in using exosomes to deliver medicine directly to the kidneys, which could improve recovery and reduce complications. 50
The furosemide stress test has potential, especially if integrated with others tests and clinical features in critically ill patients.51–53 The Doppler-based resistive index has not been successful in differentiating transitory AKI from persistent AKI.54,55 Finally, biomarkers may prove valuable when used in conjunction with other diagnostic methods and clinical features. 46
Emerging perspectives: Phenotyping and machine learning
The management and understanding of AKI are witnessing significant advancements propelled by the integration of clinical insights, novel sub-phenotyping methods, and innovative ML techniques. Sitbon et al. highlighted the significance of clinical judgment in predicting KRT needs in patients with AKI. Their study underscored that model incorporating physician judgment, especially in conjunction with metrics such as SOFA score, consistently outperformed those that did not. 56 Additionally, Pike et al. highlighted that conventional clinical models that consider parameters such as age and mean arterial pressure are valuable for predicting KRT outcomes. 57 In a more recent study, the authors applied unsupervised consensus clustering, a ML technique, to discern distinct sub-phenotypes among patients with dialysis-requiring sepsis-associated AKI, showing differing clinical outcomes. Intriguingly, one identified subgroup; with a higher severity of acute illness; displayed a heightened risk of mortality and a diminished chance of being free from KRT compared to others, even though they were younger and had fewer comorbidities. 58 This ML enabled sub-phenotyping resonated in other studies, further drawing attention to its potential to elucidate AKI's intricacies.59,60 While the value of prognostic enrichment in AKI management is recognized, the overarching goal of sub-phenotyping diverse disorders leans towards predictive enrichment or tailoring specific treatments to individuals; however, these sub-phenotyping tools have not made it to clinical practice because of challenges such as wide-scale validation, timely availability of biomarkers, and model complexities 45
Beyond the realm of sub-phenotyping, recent advances in artificial intelligence by ML have shown promise in providing accurate, data-driven predictions across a range of clinical domains, including sepsis management, ventilator weaning, and acute organ failure.61–64 By integrating numerous clinical variables (such as laboratory parameters, physiological measures, and patient demographics), ML models can potentially detect subtle patterns that might be overlooked during routine clinical assessment. In the context of AKI, emerging evidence suggests ML-based predictive tools could offer earlier and more precise estimation of which patients are likely to require KRT, thus enhancing resource allocation and patient care. 65 A proof-of-concept study demonstrated the feasibility of using a recurrent neural network to predict, up to 48 h in advance, the need for KRT, admission to intensive care, or death in patients with AKI. Trained on over 21,000 AKI episodes (25 million data points), the model exceeded 99% in both specificity and negative predictive value. Its positive predictive value for dialysis reached 62%. 66 Collectively, these studies highlight the evolving trajectory of research and management of AKI. The combination of clinical insights, advanced sub-phenotyping, and ML promises the future of more tailored and precise interventions for AKI.
Perspectives: From dilemma to direction
Sean Bagshaw et al. in 2009 proposed an algorithm for starting KRT in adult critically ill patients, giving a starting point to a more patient-centric approach. In this strategy, the authors integrate two important aspects that may influence the decision of initiating kidney support, especially “dynamic factors” like the severity of the illness by a rapid progression of kidney and non-kidney organ dysfunction. In addition, there is the need for co-interventions that could increase kidney striving, such as surgery, large fluid volumes, mechanical ventilation, and corticosteroids, etc. 67 In 2012, Ostermann and colleagues proposed another algorithm to guide decision-making regarding AKI-KRT that emphasizes the importance of regular evaluation of patients’ features and trends. 68 The latter was readapted by adding the notion of kidney baseline function and reserve and also highlighting the need for consideration of all patients’ trajectories rather than relying only on laboratory threshold values. 69
The concept of a balance between kidney capacity and metabolic demands has been suggested to be taken into consideration when deciding to initiate KRT. However, the exact method used to assess this balance at the bedside is still unclear.7,43,70 Another major point of focus is the implications of patient and family in such decisions by delivering clear simple language concerning the risks and potential benefits of such therapy and overall prognosis. 7 A more recent proposition by Bagshaw et al. summarized the prevailing approach of the “watch and wait strategy” by incorporating all patient's features. 71 These algorithms have the merit of proposing a patient-centered approach, taking into consideration key factors (Figure 2), such as the severity of causal disease, kidney reserve, clinical and biological trends, overall prognosis, and patient and family preferences rather than the urine output/ sCr-centric paradigm.67–71 Nevertheless, the vast heterogeneity of AKI syndrome still stands against precision medicine approaches. The challenge that remains before the scientific community is to identify relevant clinical phenotypes based on “evidence-based medicine” and thus develop distinct management algorithms.72,73
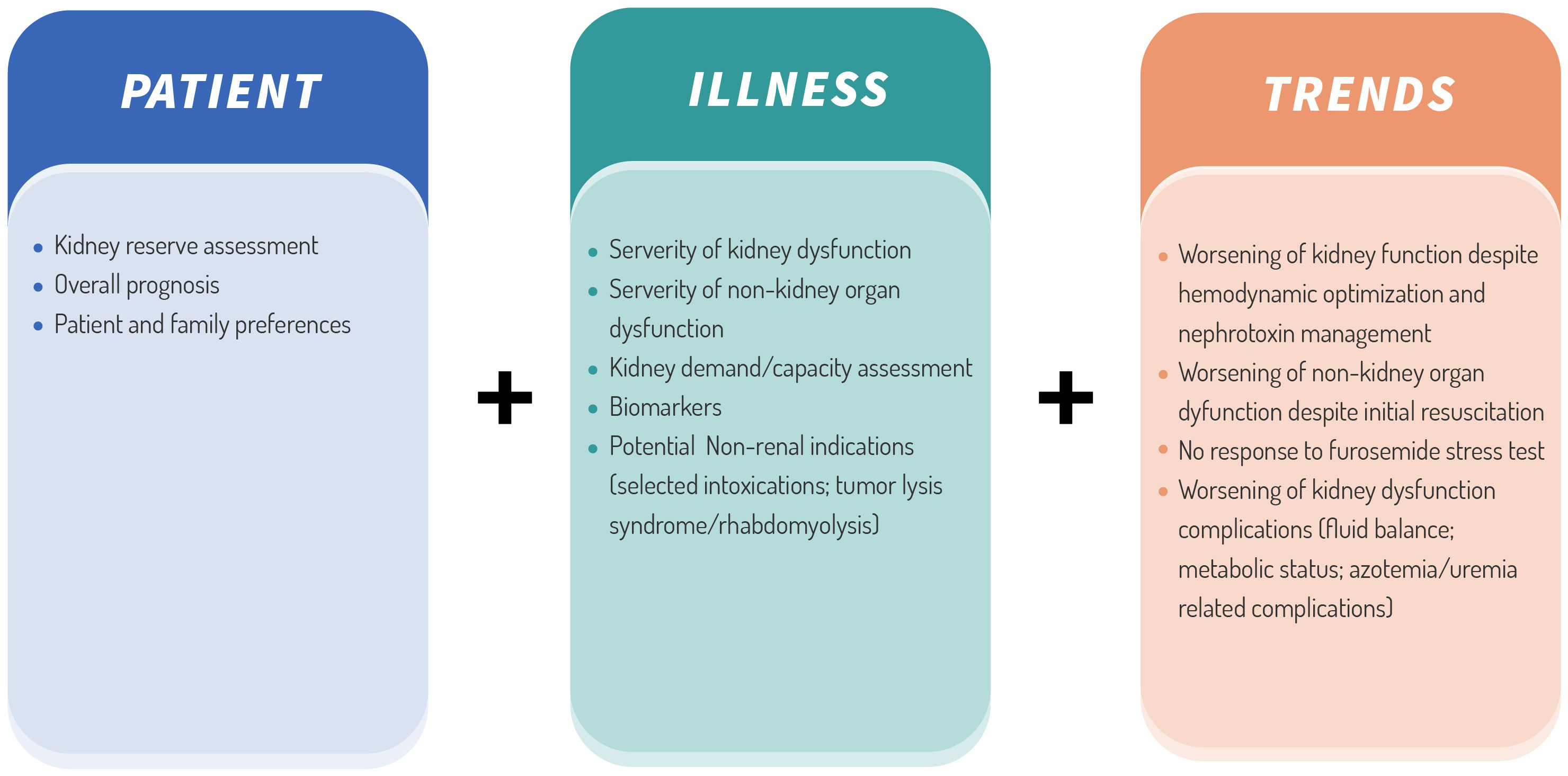
Assessment metrics for KRT initiations in AKI in the absence of life-threatening complications. AKI: acute kidney injury; KRT: kidney replacement therapy.
As a narrative review, our search and selection processes were not entirely systematic, increasing the possibility of selection bias. We did not perform a formal risk-of-bias assessment or quantitative synthesis of the available studies, which limits our ability to draw firm conclusions regarding the strength of the evidence. Moreover, we focused primarily on frequently cited and recent research; thus, relevant but less prominent studies may be underrepresented. These factors should be considered when interpreting our findings.
Conclusion
AKI in the ICU underscores the critical need to refine KRT initiation strategies. Current data question the presumed benefits of an “early” approach, advocating instead for individualized, evidence-based interventions that account for the complexity and heterogeneity of AKI. While many pivotal trials show no definitive advantage of early over delayed KRT, their methodological inconsistencies make broad generalizations risky. Even if consensus emerges regarding the optimal timing of KRT, additional questions on the modality (continuous vs. intermittent) and the appropriate timing to discontinue dialysis remain unresolved. Continued advancements in biomarkers, ML and sub-phenotyping promise new opportunities for personalized diagnosis and treatment. Ultimately, a paradigm shift is needed; one that moves beyond rigid dichotomies and leverages innovative technologies and clinical expertise to deliver more precise, patient-centered AKI care.
Footnotes
Ethical considerations
Not applicable, because this article does not contain any studies on human or animal subjects.
Author contributions
SE contributed to concept and design, literature search, article writing and drafting. AD and RA involved in literature search and review. YM and SM contributed to critical revision. All authors approved the final version of the article submitted for publication.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
