Abstract
Objective
This retrospective study aimed to investigate the correlation between neutrophil gelatinase–associated lipocalin (NGAL) levels and the clinical progression and severity of diabetes-related acute kidney injury (AKI). The quantitative determination of NGAL in plasma on the Beckman Coulter AU480 analyzer was measured using the Bioporto NGAL TestTM, a particle-enhanced turbidimetric immunoassay with hospitalized patients at an East Central Georgia Medical Center.
Methods
The clinical determination of plasma NGAL included a retrospective cohort study where 45 adult patients were selectively recruited. The selective criteria were patients with and without diabetes mellitus (DM) at risk for developing AKI admitted to the Medical Center between January and November 2023. All patients included in the study had pNGAL levels measured upon admission and up to 96 h post-admission. Receiver operating characteristics and likelihood ratio methods were used to determine optimal sensitivity, specificity, and cutoff value of pNGAL in AKI patients associated with and without DM.
Results
The intra-assay and interassay imprecision percent relative standard deviation was between 2.7% and 4.2%. pNGAL levels were higher for patients with AKI compared to non-AKI patients, regardless of DM status. The optimal cutoff value for pNGAL to predict AKI for patients with DM was 293 ng/mL, with a sensitivity of 80% and specificity of 87%. In a multivariate logistic regression model, pNGAL levels at 48 h post-admission were determined to be associated with diabetes-related AKI patients.
Conclusion
Plasma NGAL levels at 48 h are associated with patients with diabetes-related AKI. The specific cutoff values for AKI for early diagnosis and risk stratification and its association with comorbidities must be determined to improve patient outcomes.
Introduction
Neutrophil gelatinase–associated lipocalin (NGAL) is a protein of the lipocalin family and has variable degrees of gene expression in human tissues, such as the prostate, uterus, kidney, lung, stomach, salivary glands, trachea, and colon. 1 Neutrophil gelatinase–associated lipocalin is highly expressed in pathological states, including acute kidney injury (AKI) because it is synthesized by the thick ascending limb of Henle's loop and the collecting duct in the kidneys. 2
Neutrophil gelatinase–associated lipocalin was first proposed to be an early biomarker of AKI by Mishra et al. and was found to be upregulated as early as 2 h in urine following ischemia. Neutrophil gelatinase–associated lipocalin can be quantified in plasma, serum, and urine. It has been shown that NGAL levels can still increase or remain elevated during and after kidney injury, which makes it beneficial for clinicians to determine treatment protocols.
The AU480 Chemistry Analyzer (Beckman Coulter, Pasadena, CA) is a reliable, cost-effective, and easy-to-use diagnostic chemistry analyzer that uses the principle of photoelectric colorimetry to measure a specific chemical composition in a sample. The AU480 chemistry analyzer has a 400 photometric test per hour capability that requires a small amount of sample volume. This analyzer has several strengths in regard to biomarker validation as it is fully automated, reliable, and has a short turnaround time for results. In addition, it reduces sample volume, offers sample tracking, patient statistics, easy maintenance procedures, a refrigerated carousel, and supports multiple sample types such as serum, plasma, and urine. These are all critical characteristics needed for an ideal biomarker testing platform. 3
Remarkably, several studies have reported that NGAL may be used as an efficient biomarker for early diagnosis and predicting the severity and progression of AKI. 4 However, the usefulness of its clinical relevance in diabetic neuropathy and immune-mediated glomerulonephritis is still unclear and lacking. In addition, it is noteworthy to point out that many studies used urine instead of plasma or serum as a specimen. The novelty of this study is the use of plasma. 5
Therefore, the present study aimed to validate the analytical and clinical performance of the Bioporto NGAL Test on the Beckman Coulter AU480 chemistry analyzer for measuring NGAL in human EDTA plasma and to assess pNGAL levels with the clinical progression and severity of diabetes-related AKI. People with diabetes are more likely to develop AKI compared to the general population. This is because diabetes can lead to chronic complications like diabetic nephropathy, which can affect kidney function. Recognizing this association between diabetes and AKI is important in real world and clinical settings, as it has significant implications for patient care.
Methods
This retrospective study selectively recruited adult patients over 18 years old from the emergency room at a Medical Center in East Central Georgia. There were three groups: a control group and two AKI experimental groups with and without type 1 or type 2 diabetes mellitus (DM).
The control group (DM (+) AKI (−)) consisted of adults over 18 years old hospitalized at the Medical Center with DM and without AKI. The first AKI group (DM (+) AKI (+)) included subjects confirmed and diagnosed with diabetes and AKI according to KDIGO criteria and clinical assessment. The second group (DM (−) AKI (+)) included subjects confirmed and diagnosed with AKI but not diabetes. Exclusion criteria include individuals with any of the following conditions: (a) patients with serum creatinine above 6 mg/dL; (b) any malignancy or cancer, including pancreatic cancer and multiple myeloma; (c) inflammatory variations in leukocyte count; (d) renal transplant or end-stage renal disease; and (e) undergoing treatment with steroids or immunosuppressant to minimize potential confounding factors. Ethical approval was obtained from the Institutional Review Board (IRB) at Augusta University Medical Center. The study was categorized as exempt (category 4) since it involved the use of secondary data from the patient's medical records (IRB # 1840264) and as well as the use of discarded blood samples for pNGAL measurements. A HIPAA Waiver of Authorization was granted to access the subjects’ medical records and extract past and present health information for research purposes. All patients have been deidentified in accordance with the HIPPA waiver. This study was conducted following the Helsinki Declaration of 1975, which was revised in 2013.
To determine the performance characteristics of Bioporto Diagnostics NGAL Test on the automated AU480 chemistry analyzer (Beckman Coulter, Pasadena, CA), the levels of pNGAL were measured using a particle-enhanced turbidimetric immunoassay according to the manufacturer's standard protocol. This particle-enhanced turbidimetric immunoassay required approximately 3 μL of the sample, 50-μL antibody, and 150 μL of reaction buffer. The reaction started by adding the polystyrene microparticles coated with mouse monoclonal antibodies against human NGAL. After NGAL was detected in the sample, immune particles aggregate, and the level of aggregation was measured by the amount of light scattering at an absorption of 700 nm. To determine the pNGAL concentration in the sample, a calibration curve was generated using NGAL calibrators. Their corresponding absorption values were obtained during the analytical phase, and the concentration was determined where the interpolation occurred. The assay was completed in 10 min. 6 The reporting of this study conforms to the STROBE guidelines. 7
Accuracy
The accuracy of the test was measured using nine determinations over three concentration levels (Low, Normal, and High). Then, the percent of error was determined based on equation (1) 8 (Please refer to the Supplementary File for the equation).
Precision
Guidelines from the Clinical and Laboratory Standards Institute EP15-A2 document were adopted to determine repeatability and reproducibility. Using Bioporto quality control reagents, the reproducibility was determined by taking measurements three times a day for five consecutive days using the two-level dedicated quality control materials (low and high). The repeatability was measured by analyzing the low and high control levels in 10 replicates within each run. 9 Repeatability and reproducibility were reported using the standard deviation (SD), percent of relative SD (RSD; coefficient of variation), and confidence interval.
Limit of detection
To determine the limit of detection (LoD), the limit of blank (LoB) was first calculated. The limit of the blank was estimated by measuring replicates of saline (n = 20), and then the mean and SD were calculated according to equation (2) (Please refer to the Supplementary File for the equation).
After the LoB was calculated, the dilution of the lowest NGAL calibrator level (50 ng/mL) provided by the manufacturer was measured in duplicates (n = 20), and then the mean and SD values were calculated. 10 The LoD was calculated using equation (3) (Please refer to the Supplementary File for the equation).
Limits of quantitation
To determine the limit of quantitation (LoQ), also known as functional sensitivity, nine serial dilutions in saline (i.e. from 1:2 to 1:512) of a plasma sample with an NGAL concentration of 1441 ng/mL were performed. Each dilution was measured in duplicates, and the imprecision for each dilution was calculated. A model was then created to extrapolate the NGAL value associated with a coefficient of variation 11 of 20%; see equation (4) 12 (Please refer to the Supplementary File for the equation).
Specificity
To verify that the assay only detected NGAL, interference studies were performed to determine the analytical specificity. The interference screening occurred by testing two plasma samples from subjects diagnosed with leukocytosis with and without AKI to see the correlation between NGAL levels and leukocytosis. 13
Linearity
The linearity study was performed using the Bioporto NGAL calibration kit and quality controls with 10 different concentrations. Each concentration was measured in triplicates, and the measurement range was determined using linear regression analysis and Pearson's correlation coefficient. The clinical reportable range was determined from the linearity study by using the lowest and highest analyte concentration that produces a valid value. 13
Sample carryover
To evaluate if there is any sample carryover between samples, the Bioporto NGAL low and high controls were measured in the following order: three low controls (208 ng/mL), one high control (474 ng/mL), and one low control (208 ng/mL). The percent carryover (k) was calculated according to equation (5) (Please refer to the Supplementary File for the equation). Positions 1 through 3 and 5 were low controls, and position 4 matched the high control. A carryover < 1 was denoted as acceptable.
Clinical determination
Measurements of NGAL were performed on human plasma from adults with confirmed DM and/or AKI diagnosis according to KDIGO criteria. 4 Three groups were included, subjects with (DM (+) AKI (+), n = 15), subjects with DM without AKI (DM (+) AKI (−), n = 15), and subjects without DM but with AKI (DM (−) AKI (+), n = 15), to assess NGAL function in AKI disease progression and severity of DM-related AKI during hospitalization at various time points (admission (day 0), 24 h (day 1), 48 h (day 2), 72 h (day 3), and 96 h (day 4), and if available, before discharge). Patients were not followed up after discharge. The patient's EMR was used to extract all other relevant clinical data, such as demographic characteristics, lab values (except pNGAL), medical history, and risk factors.
Blood samples were collected into EDTA tubes using standard procedure protocols and centrifuged at 3000 rpm at 4°C for 15 min to obtain the plasma. Afterward, a minimum of 100 µL of the plasma was dispensed into plastic 16 mm × 60 mm sterile transport tubes (Market Lab, Caledonia, MI) and stored at −80°C. All samples were analyzed within six months in two batches using the same lots of reagents from the manufacturer.
Sample size calculation
The primary endpoint of this study was to investigate the usefulness of pNGAL as an early diagnostic of AKI in adults with DM with NGAL values upregulating earlier than serum creatinine. Therefore, rejection of Ho occurs when the specificity of the pNGAL in predicting AKI in DM patients is higher than the specificity of serum creatinine.
The Buderer formula, using the Excel Software (equation (6), please refer to the Supplementary File for the equation), was used to estimate the sample size needed to determine the diagnostic accuracy of NGAL to predict AKI in DM patients. 14 where n = required sample size,SP = anticipated specificity,α = size of the critical region (1 − α is the confidence level),z1−α/2 = standard normal deviate corresponding to the specified size of the critical region (α), andL = absolute precision desired on either side (half-width of the confidence interval) of specificity
This study used the following parameters: a prevalence of 40%, 15 a precision of 10%, and a specificity of 0.96 from a pilot study 16 to calculate the minimum sample size. Using an alpha of 5% and a confidence interval of 95%, the Buderer formula estimated the minimum sample size for the study population to be 25. However, in this study, 45 subjects (15 per group) were enrolled to account for specimens that did not meet the inclusion criteria or were excluded for not having all relevant data.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics for Macintosh Version 28.0 (IBM Corp, Armonk, NY). Statistical significance was set at α = 0.05. All continuous data related to patients and control subjects were stated as means, SD, or median values if data had normal distribution according to the Shapiro–Wilk test for normality and interquartile range (IQR) if the data had a skewed distribution. All categorical variables were expressed as proportions. The Chi-square test was used to evaluate the association between categorical variables. Continuous variables were compared using One Way ANOVA for parametric data and the Kruskal–Wallis One-Way ANOVA test for nonparametric data. For any variables found statistically significant, the Dunn's Multiple Comparison Test, which is a post hoc nonparametric test, was performed to determine which groups were statistically significant. A multivariate binary regression model was used to assess the association between pNGAL levels and the risk for AKI and to control for potential confounders. Initially, a univariate regression was performed, and variables with a p-value < 0.20 were then included in the multivariate binary logistic regression model (equation (7), please refer to the Supplementary File for the equation).
Receiver operator characteristics (ROC) curve analysis was performed to identify the optimal cutoff value of pNGAL levels, sensitivity, specificity, and positive and negative predictive value for the prediction of AKI in patients with DM. The area under the curve (AUC) was calculated as a measure of the accuracy of the test. The highest likelihood ratios (LRs) were used to determine the appropriate cutoff value. Picking a cutoff that is based on the LR is appropriate for this study because it produces a balance between sensitivity and specificity; so that neither a true nor a false positive is amplified nor sacrificed. To determine if pNGAL can predict AKI before 48 h, the discriminatory power of pNGAL and serum creatinine were determined at admission, 24 h, and 48 h using the ROC curve, LRs, and AUC.
Results
Linearity and imprecision studies
On the AU480 system, the NGAL Test assay showed good precision at all two control concentrations with an intra-assay (repeatability) and interassay (reproducibility) % RSD < 5%. Supplementary Table 1 summarizes the mean, SD, and %RSD results. The obtained analytical measurements range for AU480 is 50–3000 ng/mL, verified by the manufacturer 17 (Supplementary Figure 1), and the dilution studies extended the clinical reportable range to 20–3000 ng/mL.
Accuracy
The analytical accuracy of NGAL Test™ on Beckman Coulter AU480 was calculated based on the percent error of the system as indicated by the manufacturer. The accuracy of the three determinants of low, normal, and high was found to be similar to the manufacturers’ claim with a percent error of 3%, 0%, and 1% for the low (208 ng/mL), normal (474 ng/mL), and high (1399 ng/mL) determinants, respectively.
Carryover
No significant sample carryover (k) was observed during the analytical validation procedures. The calculated k was 0.14%, which is below the acceptable 1% carryover determined by the manufacturer. Supplementary Table 2 summarizes the carryover results.
Limit of blank, LoD, and limit of quantitation
The LoB and LoD of the NGAL TEST 18 were found to be 2.2 ng/mL and 15.4 ng/mL, respectively. The approximation of the limit of quantitation is shown in Supplementary Figure 2. A 20% CV of the assay corresponded to an NGAL concentration of 9.9 ng/mL.
Specificity
Neutrophil gelatinase–associated lipocalin is known to be present in neutrophils, and patients diagnosed with leukocytosis can have elevated NGAL, giving false-positive testing results. For this reason, the present study performed a specificity test using patients with leukocytosis samples to evaluate the effect of leukocytes on pNGAL levels in patients with and without AKI. Results showed that samples without AKI diagnosis have an elevated pNGAL level, indicating leukocytosis in plasma causes false-positive NGAL results (Supplementary Table 3).
Clinical determination of subject population
A total of 45 adults were recruited during the study period (Figure 1), out of which 24 were African Americans. The median age of the study population was 55.2 years old. Among the 45 patients, 30 (66.7%) had DM, and out of these 30 patients, 16 (53.3%) were African American. Among all the patients with AKI (n = 30), more than half (n = 17, 56.6%) were African Americans. Table 1 presents key baseline characteristics according to the presence of baseline DM and the occurrence of AKI.
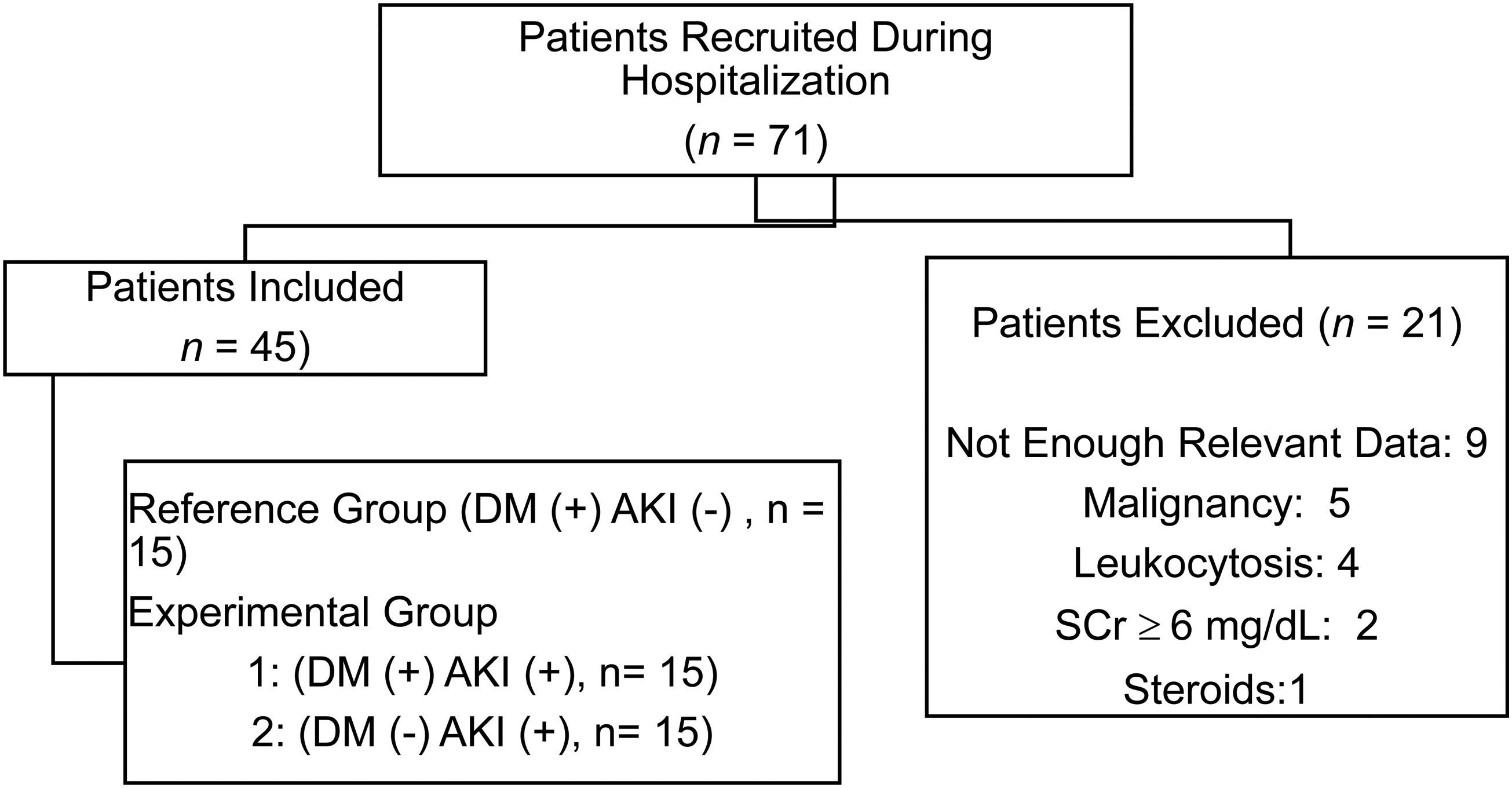
Study flowchart.
Baseline characteristics of hospitalized patients based on presence or absence of DM and AKI.

Note: Data are expressed as mean ± SD, median (25–75% interquartile range) for nonparametric variables, or number (percentage).
BMI: body mass index; CAD: coronary artery disease; CHF: congestive heart failure; CKD: chronic kidney disease; HLD: hyperlipidemia; HTN: hypertension; LoS: length of stay. Baseline SCr values are lab values from previous hospital visit before recruitment of this study.
*p < 0.05 (The p-value denotes a comparison between three groups no-DM with AKI (DM (−) AKI (+)), DM without AKI (DM (+) AKI (−)) and DM with AKI (DM (+) AKI (+)) using Chi-Square and Kruskal–Wallis One-Way ANOVA Test).
Associations between AKI with and without DM and clinical laboratory characteristics
A Chi-square test of independence was performed to examine the relation between the presence or absence of DM with AKI and the diagnosis of other comorbidities such as congestive heart failure (CHF) or hypertension (HTN). The relation between the presence or absence of DM and AKI with HTN was statistically significant, χ2 (2, N = 45) = 9.5, p = 0.009 (Table 1). Supplementary Tables 4 and 5 present key laboratory findings at admission (0 h) and 48 h of hospitalization to the presence of baseline DM and AKI incidence, respectively.
A Kruskal–Wallis test was conducted to determine whether there is an effect on diagnosis groups based on DM and AKI and laboratory values. A total of 15 subjects were included in each group, and all laboratory values were measured at admission until 96 h or discharge. The results indicate there was statistically significant difference in laboratory values: BUN (H (2) = 20.7, p ≤ 0.001), CO2 (H (2) = 9.3, p = 0.009), eGFR (H (2) = 28.5, p ≤ 0.001), Glucose (H (2) = 17.5, p ≤ 0.001), pNGAL (H (2) = 12.7, p = 0.002, and SCr (H (2) = 25.9, p ≤ 0.001 between at least two groups at admission, where H denotes the test statistic used in Kruskal–Wallis test for hypothesis testing (Supplementary Table 4).
A Dunn's multiple comparison test was performed to determine which groups were statistically significant for these laboratory tests. The data are presented in Supplementary Table 6. On day 2, all the laboratory values listed above were also found to be statistically significant except for CO2. However, Cl was found statistically significant at this time point: BUN (H (2) = 16.2, p ≤ 0.001), Cl (H (2) = 8.6, p = 0.014), eGFR (H (2) = 22.9, p ≤ 0.001), Glucose (H (2) = 11.3, p ≤ 0.004), pNGAL (H (2) 16.4, p ≤ 0.001, and SCr (H (2) = 20.4, p ≤ 0.001 (see Supplementary Tables 6 and 7 for Dunn's Multiple Comparisons post hoc).
Association between pNGAL, SCr, and eGFR in hospitalization of AKI patients with and without DM
Figure 2 demonstrates serial measurements of pNGAL, SCr, and eGFR mean levels stratified by the presence or absence of baseline DM and the development of AKI. Patients who develop AKI superimposed on DM demonstrated higher mean plasma NGAL levels and serum creatinine. Regarding biomarkers levels at admission (Table 2), the median (IQR) for plasma concentration of NGAL was significantly higher in subjects that had AKI superimposed on DM compared to those who had DM, but AKI was absent (360.9 ng/mL [294.9–747.7] vs. 190.2 ng/mL [86.6–284.1], p = 0.003), respectively. pNGAL levels in the group with AKI superimposed on DM were also higher than in the group with AKI, but no DM, but the differences were not statistically significant (see Supplementary Table 6).

Serial measurement of pNGAL, SCr, and eGFR at 0 h, 24 h, 48 h, 72 h, and 96 h stratified by DM and AKI.
Serial measurements presentation of pNGAL, SCr, and eGFR stratified by DM and AKI.

Note: Data are expressed as median (25–75% interquartile range).
AKI: acute kidney injury; DM: diabetes mellitus; eGFR: estimated glomerular filtration rate; pNGAL: plasma neutrophil gelatinase–associated lipocalin; SCr: serum creatinine.
*p < 0.05. (The p-value denotes a comparison between three groups no-DM with AKI (DM (−) AKI (+)), DM without AKI (DM (+) AKI (−)) and DM with AKI (DM (+) AKI (+)) using Kruskal–Wallis One-Way ANOVA Test.)
The median serum creatinine concentration at admission was higher between subjects that had DM with AKI vs. DM without AKI (2.31 mg/dL [1.62–3.03] vs. 0.83 mg/dL [0.68–1.06], p = 0.001), respectively. The median SCr concentrations were slightly lower by one-fold between subjects that had AKI with DM vs. AKI without DM (2.31 mg/dL [1.62–3.03] vs. 2.48 mg/dL [1.62–3.14], p = 0.961) but not statistically significant (see Supplementary Table 6).
eGFR median values for subjects with DM but without AKI were significantly higher (90 mL/min/1.73 m2 [79–90]) compared to subjects with AKI and DM (28 mL/min/1.73 m2 [18–41] and those with AKI no DM (25 mL/min/1.73 m2 [16–52], p ≤ 0.001), respectively (see Supplementary Table 6). This trend between biomarkers pNGAL and SCr was seen among the other time points (see Supplementary Tables 7 and 8).
Optimal pNGAL threshold for early detection of AKI in DM
Receiver operator characteristic curve analysis showed a better discriminatory capacity for SCr in terms of early diagnosis of AKI in DM at 0 h, 24 h, and 48 h, with the AUC being 0.98 for SCr and 0.85 for pNGAL at admission (Figure 3A). At 24 h, the AUC for SCr and pNGAL was 0.97 and 0.81, respectively (Figure 3B). At 48 h, the AUC for SCr was 0.93 and 0.88 for pNGAL (Figure 3C). At all three time points, SCr and pNGAL significantly differed with p < 0.05. The ROC curves for the 72 h and 96 h time points can be found in Supplementary Figures 3 and 4, respectively.

ROC curve analysis for early prediction in diabetes-associated AKI.
Weighing the AUC for AKI prediction for groups with and without DM and timing of measurements, admission was selected as the optimal point for the pNGAL threshold in determining DM-related AKI prediction. The highest LR method was used to calculate the cutoff value of the positive diagnosis of AKI related to DM at admission. The calculated cutoff value for pNGAL using this method was 293 ng/mL with a sensitivity of 80%, specificity of 87%, and AUC of 0.85. The study had a 92.3% power to discern between true positive result (true positive rate) and the false positive result of 13% (false positive rate) (Supplementary Table 9). The positive and negative predictive values were 92.3% and 68.4%, respectively, and a prevalence of 66.7% was determined.
Comparing renal indices and clinical characteristics in predicting AKI development in DM
To further explore the association of pNGAL levels with the possibility of developing AKI in diabetic patients, we performed univariate and multivariate logistic regression analysis. As can be seen, the univariate analysis indicated that pNGAL levels at 48 h (OR = 1.004, 95%; CI = 0.999–1.010; p = 0.150) and age (OR = 1.036, 95%; CI = 0.988–1.086; p = 0.144) were associated with a higher probability of developing AKI. Other variables such as sex (OR = 0.250, 95%; CI = 0.055–1.0141; p = 0.074) and diuretics (OR = 0.167, 95%; CI = 0.033–0.853; p = 0.032) were associated with lower probabilities of developing AKI (Table 3).
Odds ratio and 95% confidence interval for the possible prognostic factors of AKI development.
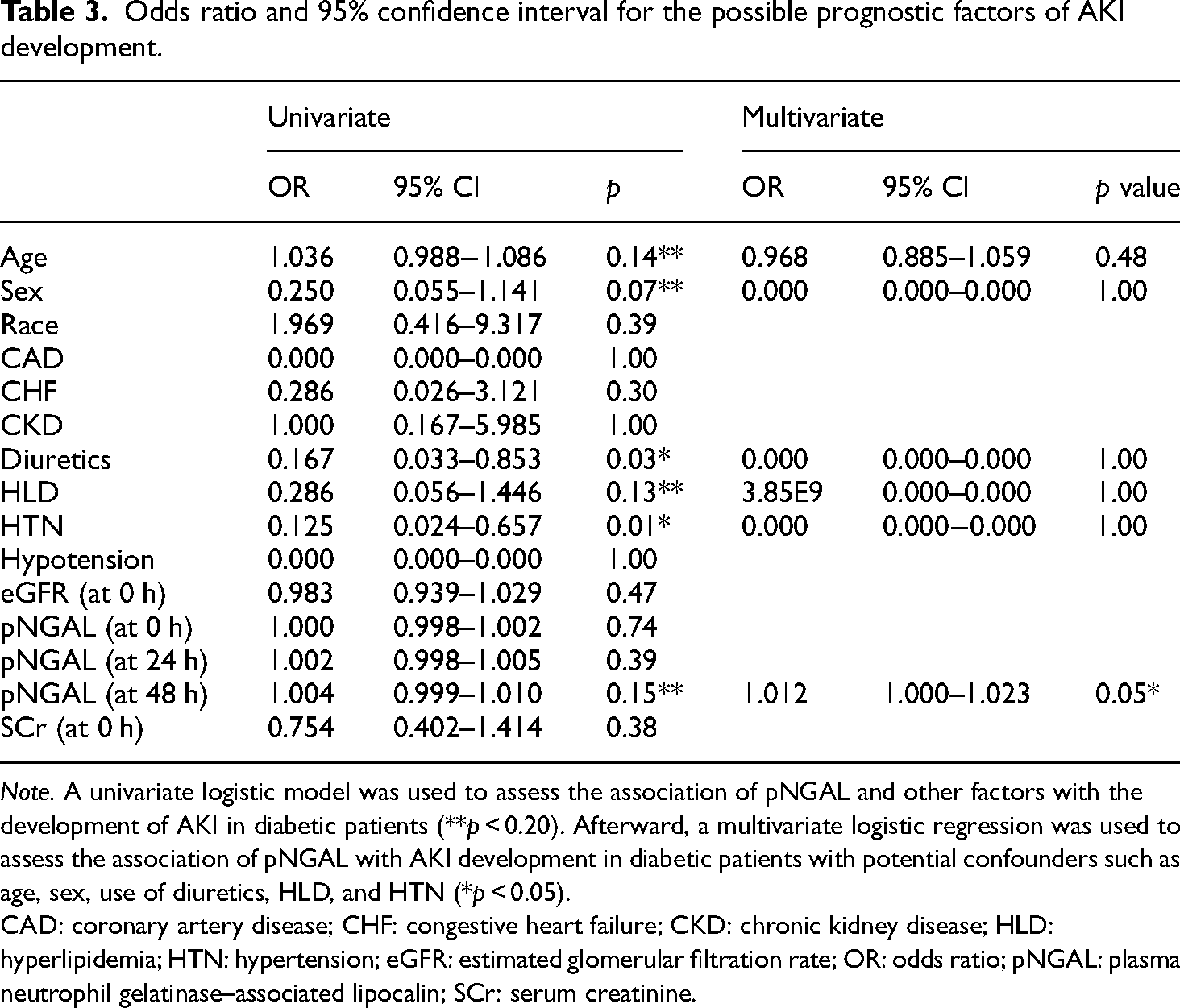
Note. A univariate logistic model was used to assess the association of pNGAL and other factors with the development of AKI in diabetic patients (**p < 0.20). Afterward, a multivariate logistic regression was used to assess the association of pNGAL with AKI development in diabetic patients with potential confounders such as age, sex, use of diuretics, HLD, and HTN (*p < 0.05).
CAD: coronary artery disease; CHF: congestive heart failure; CKD: chronic kidney disease; HLD: hyperlipidemia; HTN: hypertension; eGFR: estimated glomerular filtration rate; OR: odds ratio; pNGAL: plasma neutrophil gelatinase–associated lipocalin; SCr: serum creatinine.
The multivariate analysis showed that pNGAL levels at 48 h are associated with AKI development (OR = 1.012, 95%; CI = 1.000–1.023; p = 0.051) (equation (8), please refer to the Supplementary File for the equation). Additionally, the univariate analysis indicated that age was associated with a higher probability of developing AKI; however, in the multivariate analysis, it was not associated with AKI development, probably due to the effect of other cofounders.
Discussion
Findings from this study showed pNGAL is a valuable biomarker for DM-related AKI with an optimal cutoff value of 293 ng/mL. At 48 h, pNGAL levels are associated with diabetes-related AKI with a specificity of 93%.
To provide valuable information, a biomarker should indicate normal and abnormal pathogenic processes or responses to exposure or interventions in the clinical setting so that an appropriate diagnosis can be made. 19 An analytical validation is applicable to establish experimental evidence that the performance characteristics of a biomarker are suitable for the test to produce reproducible, reliable, highly sensitive, and specific results. 20 Therefore, the study's first aim was to establish the performance characteristics of the BioPorto NGAL Test on the Beckman Coulter AU480 analyzer.
The measurement of biomarkers for structural injury for the prediction, diagnosis, and prognosis of AKI has been identified and studied extensively in clinical trials to determine how best to integrate them into clinical practice. 21 Neutrophil gelatinase–associated lipocalin has been found to be a promising biomarker of AKI. 22 Neutrophil gelatinase–associated lipocalin is expressed at low levels in several human tissues, including the kidneys, and is known to increase rapidly after acute renal injury. 23 Using NGAL to test for patients with suspected AKI is linked with a lower cost in comparison to other diagnostic methods. 24 Until recently, the accessibility of commercial methods for measuring NGAL in serum, plasma, or urine was inadequate, mainly consisting of ELISA methods. Now fully automated immunoassay (BioPorto NGAL Test and Gentian NGAL particle-enhanced turbidimetric immunoassay [PETIA]) for use on conventional clinical chemistry analyzers6,12,25 and the immunochemistry Abbott ARCHITECT platform 26 has been introduced to the market for research used. In this study, an analytical validation was performed using the BioPorto NGAL Test using plasma on the Beckman AU480, a commonly used clinical chemistry analyzer for diagnostics. In contrast, other reported studies mainly have used urine or serum.
In this study, EDTA plasma was selected over serum because studies have shown that serum and plasma NGAL levels are not equivalent. Serum values are higher than plasma. 27 Serum samples will likely consist of proteins produced and secreted during coagulation, whereas in plasma, the coagulation process is completely inactivated. 28 In addition, the excess of NGAL levels in serum could result from the in vitro release of NGAL from neutrophil granulocytes. Diminution of calcium by EDTA in the plasma can impede the calcium-dependent release of secondary granule contents, inactivating the excess release of NGAL into EDTA plasma even if neutrophils are activated. 27 In heparin plasma preparation, calcium levels are not decreased, and there is no inhibition of the calcium-dependent release, which explains the difference in NGAL concentrations from heparin plasma and EDTA plasma because heparin has the ability to provoke leukocyte and platelet aggregation.28,29 In Hansen et al. study, they found that NGAL levels in lithium heparin tubes were higher than EDTA and recommended that EDTA tubes should be used for blood samples to measure NGAL concentrations. 25 The International Council for Standardization in Hematology also recommends EDTA over other anticoagulants. 30
The BioPorto NGAL Test immunoassay has several technical and analytical advantages, including a low sample volume, a fast turnaround time, and low imprecision compared to other commercially available NGAL methods. The Beckman Coulter AU480 results are readily available within 10 min compared to 2 h with manual ELISA and 35 min for the Abbott ARCHITECT system. In addition, compared to ELISA method, there is no need for manual pretreatment steps with samples on the AU480. 26
In this study, we found that the BioPorto NGAL Test performed adequately in terms of imprecision, LoQ, and linearity, which is consistent with previous studies12,25,31,32 and the manufacturer's claimed performance. 18 The BioPorto NGAL assay demonstrated acceptable imprecision when using the manufacturer-supplied QC on the AU480 and was relatively comparable to the high plasma pooled patient material measured using The BioPorto NGAL assay on the ARCHITECT C16000 analyzer (Abbott Diagnostics, Chicago IL, USA). The interassay on the ARCHITECT C16000 analyzer displayed a %RSD of 2.9%, 32 slightly lower by 0.8-fold than our 3.7% on the Beckman Coulter AU480. However, the intra-assay consisted of the ARCHITECT C16000 and the Beckman Coulter AU480 analyzer, as %RSD was 2.5% 32 and 2.6%, respectively. The total imprecision was calculated as 3.4% on the Beckman Coulter AU480, which was marginally higher than that of the BioPorto NGAL Test (2.2%) on the Roche Cobas c501 chemistry analyzer. However, it wascomparable to that of the Abbott ARCHITECT NGAL assay (2.9–3.7%) and Gentian NGAL PETIA assay (3.7%) on the Roche Cobas c501 analyzer. 12
The LoQ (9.9 ng/mL) determined for the BioPorto assays in this study is below that the manufacturer (<25 ng/mL) 18 and Whitehead et al. (27.5 ng/mL) 32 claim on Abbott ARCHITECT c16000; but higher than Makris et al., (8.4 ng/mL) 31 for the assay run on an Abbott ARCHITECT c8000 analyzer. Overall, the BioPorto Assay reported higher concentrations on the Beckman AU480 than the Abbott methods. It is likely that discordance between these two analyzers is due to differences in assay design and calibration. For instance, the Abbott ARCHITECT c8000 Chemistry Analyzer does not have the capability of monitoring the reagent pressure and uses chemiluminescent microparticle immunoassay for the quantitative determination of NGAL, while the Beckman Coulter AU480 uses a particle-enhanced turbidimetric immunoassay to measure NGAL concentrations.6,26 Both instruments used human recombinant NGAL for the calibrators; however, there is no certified NGAL reference material. Due to this, there is a relative difference in the specificity and affinity of the antibodies against the different NGAL molecular forms and which epitopes the assay will target. 32
The linearity was excellent over a range of NGAL concentrations between 50 and 3000 ng/mL. Compared with the manufacturer's claim, the current reference immunoassay BioPorto NGAL Test on Beckman Coulter AU480 is accurate with a percent error of less than 4%. A specificity test was performed to assess whether the BioPorto NGAL test was detecting for NGAL only and not neutrophils present due to leukocytosis. The Medical Center where this study was conducted characterizes leukocytosis as having a WBC > 24.9 thous/mm3. False positive NGAL results in plasma with leukocytosis were previously reported in other studies with high neutrophil counts.33–35 Results show consistency in the limitations of NGAL as a biomarker in accurately assessing kidney injury in patients with leukocytosis. Therefore, patients with leukocytosis were excluded from this study to eliminate any confounding factors in the following two aims since NGAL is present in neutrophils. The results of analytical validation in this study show that BioPorto NGAL has acceptable performance characteristics on the AU480 Chemistry analyzer and is a viable option for assessing plasma NGAL in clinical settings.
To the best of our knowledge, this is the first study to assess plasma NGAL as an early biomarker of AKI in DM patients. The study's main finding was that the pNGAL level is associated with DM-related AKI at 48 h. In addition, pNGAL predicted AKI at 48 h with a relatively good specificity of 93% and AUC of 0.88 in DM patients compared to SCr with a lower specificity of 87% and AUC of 0.98, respectively. Additionally, pNGAL was associated with the severity and clinical prognosis of DM-related AKI as measurement levels showed a greater decrease over the time course compared to SCr. Thus, pNGAL can provide helpful opportunities to improve the therapeutic implications of AKI by conveying severity, diagnostics, and prognostic information at early time points compared to SCr.
The etiology of AKI in DM patients is complex and poorly understood. 36 It is known that the risk of AKI increases with type 2 DM combined with CHF and systemic HTN. 37 Previous studies have shown that HTN is linked to a 1.94-fold higher risk for AKI. 38 Hypertension occurred in 80% of patients in our cohort study that was admitted with DM caused as the primary diagnosis.
In the present cohort, 66.7% of patients showed signs of AKI at admission. When we classified the patients into three groups using AKI and preexisting DM, we demonstrated statistically significant elevated pNGAL levels in patients with DM (+) AKI (+) compared to DM (+) AKI (−) as well as DM (−) AKI (+) compared to DM (+) AKI (−). While pNGAL levels were associated with AKI in patients with DM, a cutoff value of pNGAL was identified for diagnosing DM-related AKI of 293 ng/mL with a sensitivity of 80% and specificity of 87%. This cutoff value was slightly higher by 1.2-fold compared to the 252 ng/mL cutoff found by Fodor et al. in the study of pNGAL in critically ill patients. The study by Fodor et al. had a sensitivity of 87% and a specificity of 83%, which was different from our studies, where we had a higher specificity and lower sensitivity. 39 Another study by Banai et al. determined the optimal cutoff to predict AKI for patients with CKD was 133 ng/mL, with a sensitivity of 73% and specificity of 75%. 40 Thus, pNGAL levels should be interpreted based on medical history. The data from the present study agrees with those of Fodor et al. and Banai et al., who also reported that NGAL levels were elevated in AKI patients compared to non-AKI patients.39,40
In the current cohort of patients, AKI demonstrated higher NGAL levels with the highest mean level among DM (+) AKI (+) patients. The elevated pNGAL levels may reflect the severity of tubular injury combined with the effects of a hyperglycemic crisis. Previous studies have shown that diabetes increases the risk of AKI for several reasons, such as medications, interventions, and cardiovascular diseases. 41
As the overall treatment of AKI in patients is scarce, the primary focus of management should lie on prevention. 40 In addition, early detection of kidney injury in critically ill or high-risk patients is significant for patient management and improved clinical outcomes. 42 Several recent studies suggested that NGAL has the potential to be an ideal biomarker of early kidney injury prediction.39,40,43–45 Early diagnosis and initiation of early therapeutic implications could help limit the need for transplants and reduce mortality. 40
Our study has notable limitations. This is a single-center study with a moderate sample size. The number of patients without DM (DM (−) AKI (+), n = 15) was insufficient to determine the reliability and generalizability of pNGAL within patients without DM and to make an overall comparison to the groups with DM (DM (+) AKI (+), n = 15, and DM (+) AKI (−), n = 15). This limited our capability of performing specific statistical testing. In addition, 21 patients were excluded from the study to avoid any potential confounding effect and selection bias. These patients presented the following clinical characteristics: malignancy, leukocytosis, not enough relevant medical data, or the presence of SCr level over 6 mg/dL. Also, this study was not designed to assess the mechanism of diabetes-related AKI, so a comprehensive evaluation of risk factors for AKI was not performed. As this is a retrospective inquiry, the study can only investigate associations, not causality. Medical history data were also extracted from the patient's EMR, and selection bias could be introduced as structured interviews were not conducted. Different treatment plans during admission were also a limitation of this study, as the researchers did not perform any intervention with treatment.
In addition, the diagnosis of AKI was based on increased SCr concentrations according to the KDIGO classifications and not kidney biopsies. Modifications in SCr concentration might not detect small changes in glomerular filtration rate, and we could have only captured patients with more severe injury and underestimated renal injury. This can explain the fact that the data reported here showed SCr has a better discriminatory capacity in predicting AKI early.
It has been recommended by the 10th Consensus Conference of the Acute Dialysis Quality Initiative that a combination of functional markers such as SCr and damage markers like NGAL should be used to classify patients with AKI. 46 One limiting factor of NGAL is that it is not kidney specific. Thus, it can be a confounding predictive ability of pNGAL for AKI,39,42,47 and levels can be increased in other pathological conditions. The addition of urinary NGAL measurements would have strengthened our conclusions. Additional evaluation must be performed to understand the role of pNGAL for detecting structural AKI independently from SCr.
Conclusion
In conclusion, this study aimed to highlight the impact pNGAL has on early diagnostic accuracy in diabetes-related AKI. We found that pNGAL has acceptable performance characteristics on the Beckman Coulter AU480 chemistry analyzer and can be used in the clinical setting to produce results with a fast turnaround time. The diagnostic accuracy of pNGAL in predicting DM-related AKI was examined using ROC analysis and LR. ROC analysis showed that SCr was the better discriminatory factor for early diagnosis of DM-related AKI. In addition, pNGAL levels at 48 h are associated with DM-related AKI. The optimal cutoff for pNGAL in DM-related AKI is 293 ng/mL with a sensitivity and specificity of 80% and 87%, respectively. With an optimal cutoff of 293 ng/mL, pNGAL has a 92.3% capability to determine true positive DM-related AKI cases. However, these findings must be verified in a larger prospective study. Finally, we identified that the cutoff of pNGAL levels varies based on medical history. Identifying this cutoff value might be helpful in providing clinicians insight into therapeutic implications to reduce the worsening of AKI associated with DM.
Supplemental Material
sj-docx-2-sci-10.1177_00368504241288776 - Supplemental material for Validation of plasma neutrophil gelatinase–associated lipocalin as a biomarker for diabetes-related acute kidney injury
Supplemental material, sj-docx-2-sci-10.1177_00368504241288776 for Validation of plasma neutrophil gelatinase–associated lipocalin as a biomarker for diabetes-related acute kidney injury by Kendra B. Bufkin, Zubair A. Karim and Jeane Silva in Science Progress
Supplemental Material
sj-docx-4-sci-10.1177_00368504241288776 - Supplemental material for Validation of plasma neutrophil gelatinase–associated lipocalin as a biomarker for diabetes-related acute kidney injury
Supplemental material, sj-docx-4-sci-10.1177_00368504241288776 for Validation of plasma neutrophil gelatinase–associated lipocalin as a biomarker for diabetes-related acute kidney injury by Kendra B. Bufkin, Zubair A. Karim and Jeane Silva in Science Progress
Footnotes
Acknowledgments
The authors would like to thank Brett Rice for helping with sample collections and Vincent Robinson, for allowing the use of his laboratory and instrumentation.
Authors’ contribution
KB contributed to conceptualization, formal analysis, investigation, methodology, validation, visualization, writing original draft, and reviewing and editing. ZK was involved in the conceptualization and writing - reviewing & editing. JS was involved in conceptualization, funding acquisition, supervision, and reviewing & editing. All authors read and approved the final manuscripts and are responsible for the integrity of the data.
Data availability statement
The data presented in this study are available on request from the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical Approval was obtained from the Institutional Review Board (IRB) at Augusta University Medical Center, and the research study was categorized as exempt (category 4) since secondary data from the patient's medical records were used (IRB # 1840264) and discarded blood samples were used for pNGAL measurements. A HIPAA Waiver of Authorization was granted to access the subjects’ medical records and extract past and present health information for research purposes. This waiver eliminates the need for written and verbal informed consent from patients to publish their medical case details. Note that the waiver is granted when “the risk to the confidentiality of the participant's Protected Health Information (PHI) is minimal, and the research cannot be conducted without access to it.”
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Augusta University College of Allied Health Science Deans Office and School of Public Health.
Informed consent
Consent was not obtained from the patients for publication as a HIPAA Waiver of Authorization was granted to access the subjects’ medical records and extract past and present health information for research purposes. This waiver eliminates the need for written and verbal informed consent from patients to publish their medical case details. However, as this is an IRB-approved retrospective study, all patient information was deidentified.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
