Abstract
Objective
It is to evaluate the efficacy and safety of regional citrate anticoagulation (RCA) in patients with hepatic insufficiency who develop acute kidney injury (AKI) and require continuous renal replacement therapy (CRRT).
Methods
This single-center retrospective cohort study included intensive care unit (ICU) patients with hepatic insufficiency–associated AKI who were treated with CRRT between May 2024 and May 2025. After excluding incomplete records and performing 1:1 propensity score matching, 66 patients were analyzed: 39 received RCA and 27 received systemic heparin or no anticoagulation (non-RCA). Clinical characteristics, Acute Physiology and Chronic Health Evaluation II (APACHE II) score, Child–Pugh class, filter lifespan, coagulation indices, metabolic parameters, and adverse events were also collected. The primary endpoints were filter lifespan, bleeding, and citrate accumulation. The secondary endpoints included filter lifespan, bleeding, citrate accumulation, treatment-related metabolic, coagulation changes and 28-day mortality. Multivariate logistic regression and receiver operating characteristic analyses were performed.
Results
Baseline illness severity was higher in the RCA group (APACHE II 30.46 ± 7.1 vs. 16.07 ± 5.3, p < .0001; median Child–Pugh score 8 vs. 7, p = .002). RCA showed a nonsignificant trend toward longer median filter lifespan compared with non-RCA (40.0 vs. 36.0 h, p = .219), with the largest numerical difference in Child–Pugh class C. Lactate clearance after CRRT was greater in the RCA group (Δlactate 2.96 ± 7.56 vs. 0.10 ± 2.40 mmol/L, p = .037), while bleeding rates and coagulation changes were comparable. Citrate accumulation occurred in 12.5% of cases, and Child–Pugh class B was independently protective. Age and APACHE II scores independently predicted 28-day mortality. Although the unadjusted mortality was higher in the RCA group, the difference was not significant after adjustment. A combined model including age, APACHE II score, and change in activated partial thromboplastin time showed a good predictive performance (area under the curve = 0.778).
Conclusion
RCA is safe and feasible in patients with hepatic insufficiency undergoing CRRT.
Keywords
Introduction
Acute kidney injury (AKI) frequently complicates liver failure, and the bidirectional pathophysiological interaction between the two organs can lead to a downward spiral, culminating in hepatorenal syndrome or multiorgan dysfunction.1–3 Continuous renal replacement therapy (CRRT) is crucial for correct fluid overload, remove circulating toxins, and stabilizing the internal milieu. 4 However, the effectiveness of CRRT is closely tied to the choice of anticoagulation strategy, which must preserve the filter patency while minimizing iatrogenic harm. This issue is particularly pertinent in the pediatric intensive care unit, where simultaneous management of AKI and liver failure remains a clinical challenge. Our study evaluated the safety and efficacy of regional citrate anticoagulation (RCA) in this complex setting and provided clinically valuable insights into its use in critically ill patients.
Systemic unfractionated heparin remains the traditional anticoagulant for CRRT, 5 however, its safety profile deteriorates markedly in patients with hepatic insufficiency complicated by AKI. 6 Impaired hepatic synthesis of clotting factors, altered platelet function, and reduced renal clearance of heparin metabolites increase the risk of hemorrhage and drug accumulation, rendering systemic heparin either contraindicated or usable with extreme caution in this population.
RCA therapy is an attractive alternative. By chelating ionized calcium within the extracorporeal circuit, citrate locally suppresses the coagulation cascade within the extracorporeal circuit. 5 Because its systemic effect is minimal, quickly reversible with calcium infusion, and associated with a lower bleeding risk, RCA is particularly well suited for patients who are predisposed to hemorrhage. 7 RCA is generally used with caution in patients with liver cell failure given the risks of citrate accumulation and metabolic disturbances. However, our retrospective analysis suggests cautious optimism, as RCA appears to extend filter life without significantly exacerbating the metabolic parameters. While citrate accumulation remains a theoretical concern, particularly in the context of impaired liver function and AKI, our findings indicate that RCA can be safely used when monitored closely. The technique offers potential benefits, including a longer filter lifespan and careful biochemical surveillance to mitigate risks such as metabolic acidosis or hypocalcemia, particularly in patients with advanced liver dysfunction.8,9 Consequently, the balance between circuit longevity and metabolic safety during RCA in patients with dual-organ failure remains unclear.
Preliminary reports, largely limited to static biochemical snapshots and small cohorts, suggest that RCA can be employed safely in selected patients with hepatic impairment when dosing is adjusted and biochemical surveillance is rigorous. 10 However, dynamic assessments, stratified risk analyses, and robust predictors of citrate-related complications are lacking in the literature.
In the present retrospective cohort study, we aim to carefully evaluate the safety and efficacy of RCA in patients with hepatic insufficiency and concurrent AKI undergoing CRRT, given the cautious optimism regarding its ability to increase the filter life without exacerbating metabolic parameters. We analyzed the filter lifespan, metabolic and electrolyte perturbations, bleeding events, and 28-day mortality. Propensity score matching (PSM) and multivariate regression were applied to identify the independent predictors of complications, with the ultimate goal of informing individualized anticoagulation strategies in this high-risk population.
Materials and methods
Study design and participants
This single-center retrospective cohort study examined the efficacy and safety of RCA in adult patients with concomitant liver dysfunction and AKI who received CRRT between May 2024 and May 2025 at Longhua District Central Hospital, Shenzhen City. Clinical data for all eligible cases (n = 80) were retrieved from the hospital electronic medical record system. The reporting of this study conforms to STROBE guidelines. 11
Inclusion criteria
Age ≥18 years Liver dysfunction confirmed by Child–Pugh or Model for End-Stage Liver Disease score above prespecified thresholds AKI diagnosed according to KDIGO criteria CRRT duration ≥24 h Availability of complete clinical and laboratory data for outcome evaluation
Exclusion criteria
Active bleeding or documented bleeding diathesis (e.g. gastrointestinal or intracranial hemorrhage) Use of concurrent anticoagulation regimens (e.g. heparin combined with citrate) Uncontrolled arrhythmias or severe electrolyte imbalances Premature discontinuation of CRRT for nonmedical reasons (e.g. voluntary discharge) Missing critical data preventing primary outcome assessment
Patients were selectively classified into the citrate-based regional anticoagulation (RCA) and non-RCA (systemic unfractionated heparin or no anticoagulation) groups. The selection of RCA was based on specific clinical criteria, including the patient's bleeding risk, liver function, and coagulopathy severity. RCA was preferred in patients who were at a high risk of bleeding due to severe hepatic dysfunction or those who had contraindications to systemic heparin, such as a history of hemorrhagic complications or significant coagulopathy. To minimize baseline imbalances, 1:1 nearest-neighbor PSM was performed using sex, age, Acute Physiology and Chronic Health Evaluation II (APACHE II) score, and Child–Pugh class as matching covariates. The inclusion of these clinical criteria for RCA selection adds clarity to the treatment strategy and strengthens the rationale for their use in this cohort.
The study protocol was conducted in accordance with the declaration of Helsinki of 1975 as revised in 2024 and was approved by the Medical Ethics Committee of Longhua District Central Hospital, Shenzhen City in 12 March 2024 (No. 2024-016-01), and the requirement for written informed consent was waived owing to the retrospective observational design and use of deidentified data.
CRRT prescription and anticoagulation
CRRT was administered using Prismaflex® machines (Baxter) with AN69 high-flux filters (1.9 m2). Blood flow was set at 100–150 mL/min, and the effluent dose was 25–30 mL/kg/h, typically in the CVVHDF mode. In the RCA group, 4% trisodium citrate was infused prefilter to achieve postfilter ionized calcium 0.25–0.35 mmol/L, with systemic ionized calcium maintained at 1.0–1.2 mmol/L via continuous calcium chloride infusion. In the non-RCA group, unfractionated heparin was administered as a bolus (30–50 U/kg) plus continuous infusion (5–10 U/kg/h) or omitted in patients at a high bleeding risk. The filter was replaced when the transmembrane pressure exceeded 250 mm Hg or clotting was observed.
The detailed prescription parameters for the RCA and non-RCA groups are summarized in Supplemental Tables S1 and S2.
Data collection and variable definitions
All data were extracted from hospital electronic medical records and CRRT machine logs including admission notes, laboratory results, nursing documentation, CRRT operational parameters, and medication records. Data collection was conducted independently by two trained researchers and discrepancies were resolved by a third investigator to ensure accuracy and completeness. The data acquisition process was methodologically strong with a rigorous approach to ensure reliability. To further enhance the clarity of the methodology, the RCA management protocol used in this study is outlined in Supplemental Table S1. This protocol provides a detailed description of the RCA dosing and monitoring strategy, including prefilter citrate infusion, calcium maintenance, and management of citrate-related complications, ensuring consistency in treatment administration across the cohort.
Baseline characteristics
The collected baseline data comprised demographic data (sex and age), bleeding status at enrollment, and severity of illness assessed using the APACHE and Chronic Health Evaluation II scores. Liver function was stratified according to the Child–Pugh classification. The filter lifespan, defined as the interval from CRRT initiation to an unplanned circuit exchange for clotting, was recorded in hours.
Laboratory metrics and longitudinal monitoring
Laboratory parameters were assessed at baseline and serially monitored during CRRT to track organ functions and metabolic trends. The liver function indices included alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin (TBIL), and prothrombin time (PT). Renal function was assessed based on serum creatinine (Scr), blood urea nitrogen (BUN), and electrolyte (sodium and potassium) levels. Acid–base status was monitored using arterial pH, bicarbonate (HCO₃⁻), and lactate levels. Calcium metabolism was evaluated based on the total-to-ionized calcium ratio (tCa/iCa). Citrate accumulation was defined as a total-to-ionized calcium ratio (tCa/iCa) ≥ 2.5. Safety monitoring included citrate accumulation (tCa/iCa ratio ≥2.5) as the primary safety endpoint. In addition, trends in acid–base status (pH and HCO₃⁻), electrolytes (ionized calcium, sodium, and magnesium), and lactate levels were evaluated as metabolic and biochemical safety indicators.
Anticoagulation-related adverse events
Anticoagulation-associated complications included hemorrhagic events (gastrointestinal bleeding, catheter site oozing, subcutaneous ecchymosis, and major bleeding requiring transfusion). In addition, biochemical and metabolic indicators, including ionized calcium, sodium, and magnesium levels, and electrolyte disturbances were systematically monitored as safety outcomes. For analytic purposes, metabolic alkalosis was defined as arterial pH > 7.5 or HCO₃⁻>30 mmol/L; hypocalcemia as ionized calcium<0.9 mmol/L; hypernatremia as serum sodium>145 mmol/L; hypomagnesemia as serum magnesium<0.7 mmol/L; and hyperlactatemia as lactate>2.0 mmol/L. Circuit-related outcomes included the rate and causes of unplanned filter replacement and any therapy interruption.
Outcome measures
The primary endpoints were filter lifespan and bleeding or citrate accumulation, whereas the secondary endpoints included per-treatment metabolic/coagulation shifts and 28-day mortality.
Statistical analysis
All statistical analyses were performed using the R software (version 4.5.1) and SPSS (version 26.0). Continuous variables were assessed for normality and are presented as mean ± standard deviation or median with interquartile range. Normally distributed data were compared using two-sample t-tests, whereas non-Gaussian data were compared using Mann–Whitney U tests. Categorical variables were summarized as counts (percentages) and evaluated using χ2 or Fisher's exact test, as appropriate. Statistical significance was set at p < .05.
Propensity score matching
To minimize baseline confounding, PSM was conducted using a 1:1 nearest-neighbor approach with a caliper width of 0.2 standard deviations, incorporating age, sex, APACHE II score, Child–Pugh class, baseline Scr, and albumin levels. After matching, 66 pairs of patients were obtained with a satisfactory covariate balance (Figure 1), and all subsequent analyses were performed within the matched cohort.

Study flow diagram. Eighty intensive-care admissions with hepatic insufficiency-associated acute kidney injury (AKI) who underwent continuous renal replacement therapy (CRRT) between May 2024 and May 2025 were screened. Five patients were excluded for not meeting prespecified eligibility criteria. Propensity-score matching (1:1 nearest neighbor) was then applied to the remaining 75 cases, yielding 66 well-balanced participants for analysis: 39 managed with regional citrate anticoagulation (RCA) and 27 with systemic heparin or no anticoagulation (non-RCA).
Primary outcome analysis
The primary outcome, filter lifespan, was compared between groups using the Mann–Whitney U test because of the nonnormal distribution. The incidences of RCA-related metabolic complications, bleeding events, and 28-day mortality were analyzed using chi-square or Fisher's exact tests, depending on the expected frequencies.
Regression modeling
Univariate and multivariate logistic regression analyses were conducted to identify independent risk factors for RCA-associated metabolic complications. Variables with p < .10 in univariate analysis were entered into the multivariate model. Adjusted effects are reported as odds ratios (OR) with 95% confidence intervals (CI). Statistical significance was set at p < .05.
Diagnostic performance assessment
Receiver operating characteristic (ROC) curve analysis was performed to evaluate the predictive performance of candidate variables for citrate-related metabolic disturbances. The area under the curve (AUC) was calculated and the optimal cutoff points were derived using the Youden index.
Sensitivity analyses
Robustness was explored through (i) stratified analyses by Child–Pugh class (A, B, C) to examine effect consistency across liver disease severity and (ii) exclusion of statistical outliers and influential observations to confirm model stability.
Results
Baseline characteristics
Table 1 summarizes the baseline characteristics of 66 patients with hepatic insufficiency and AKI, including 39 in the RCA group and 27 in the non-RCA group. Sex distribution (76.9% vs. 70.4% male, p = .566) and age (median 57.0 vs. 48.0 years, p = .736) were comparable between the groups. In contrast, illness severity was markedly higher in the RCA cohort, as reflected by significantly higher APACHE II (median 30.0 vs. 16.0, p < .0001) and Child–Pugh scores (median 8.0 vs. 7.0, p = 0.024). Following PSM, the difference in severity between the RCA and non-RCA groups remained consistent, suggesting that RCA was selected for patients with more severe hepatic and renal dysfunction, which may have impacted clinical outcomes. However, the application of PSM helps mitigate potential confounders, allowing a clearer comparison of the effects of RCA on filter lifespan and metabolic outcomes, regardless of baseline severity differences between groups. Baseline coagulation indices also differed significantly, with the RCA group exhibiting longer activated partial thromboplastin time (APTT) and PT, and lower platelet counts (PLT) (all p < .05), consistent with more advanced hepatic coagulopathy. Secondary bleeding occurred more often in the RCA group (20.5% vs. 3.7%), but the difference was not statistically significant (p = .061). Moreover, metabolic derangement was greater in the RCA arm, with significantly higher serum lactate levels, lower bicarbonate (HCO₃⁻) levels, and more acidotic arterial pH (all p < .05). These findings suggest that anticoagulant selection was likely influenced by clinical judgment of the initial severity of liver dysfunction and metabolic status, reflecting the retrospective, nonrandomized nature of the study.
Baseline characteristics of patients grouped by predicted anticoagulation strategy.
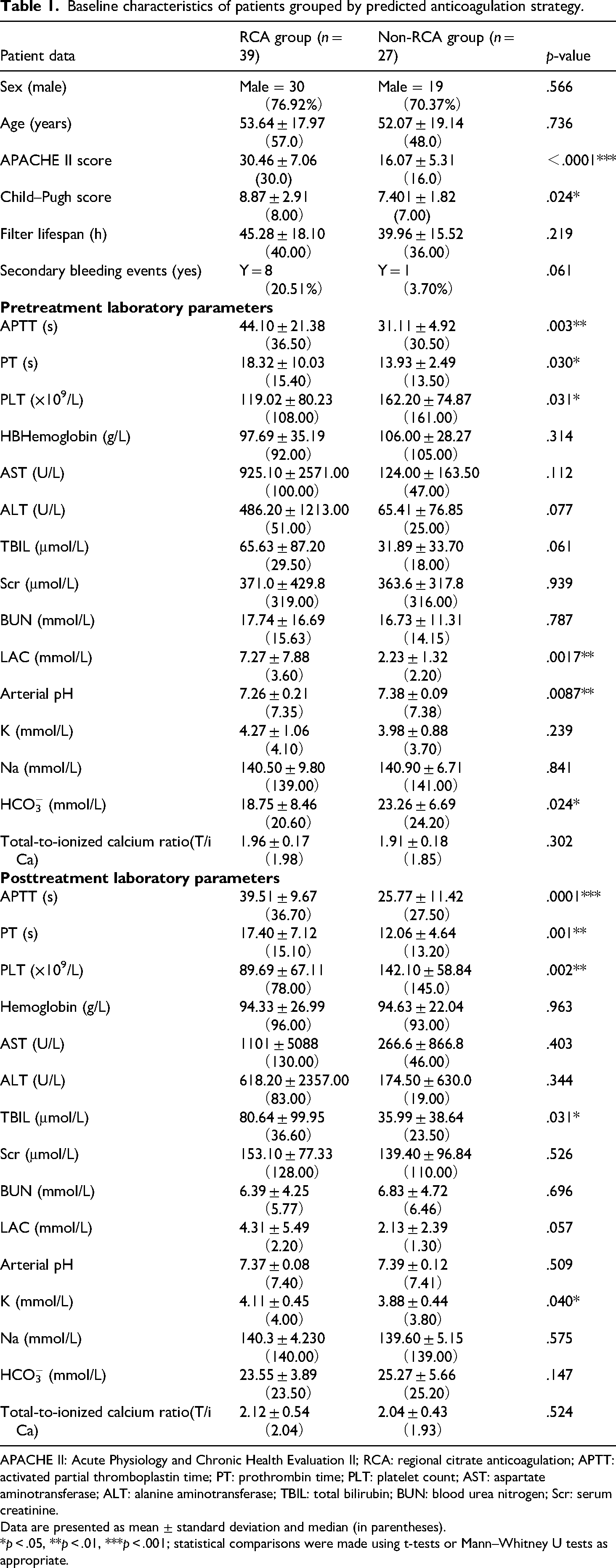
APACHE II: Acute Physiology and Chronic Health Evaluation II; RCA: regional citrate anticoagulation; APTT: activated partial thromboplastin time; PT: prothrombin time; PLT: platelet count; AST: aspartate aminotransferase; ALT: alanine aminotransferase; TBIL: total bilirubin; BUN: blood urea nitrogen; Scr: serum creatinine.
Data are presented as mean ± standard deviation and median (in parentheses).
*p < .05, **p < .01, ***p < .001; statistical comparisons were made using t-tests or Mann–Whitney U tests as appropriate.
Circuit lifespan assessment
The median filter survival time was numerically longer with RCA anticoagulation than with non-RCA anticoagulation (40.0 vs. 36.0 h), although the difference was not statistically significant (p = .219). While this trend suggests the potential benefits of RCA in preserving filter patency, it is important to consider methodological limitations, particularly the imbalance in coagulation status between the two groups. The PSM model effectively balanced key variables such as age, sex, APACHE II score, and Child–Pugh class, but it did not account for baseline differences in coagulation status, which directly affects filter lifespan. This limitation may introduce confounding factors, potentially influencing the assessment of RCA effectiveness in prolonging filter survival. Therefore, while RCA shows promise in extending the filter lifespan, further investigation that includes coagulation parameters as matching covariates is needed to confirm these findings (Figure 2A). Nonetheless, the direction of this change points to a possible advantage of the RCA in preserving extracorporeal circuit patency.
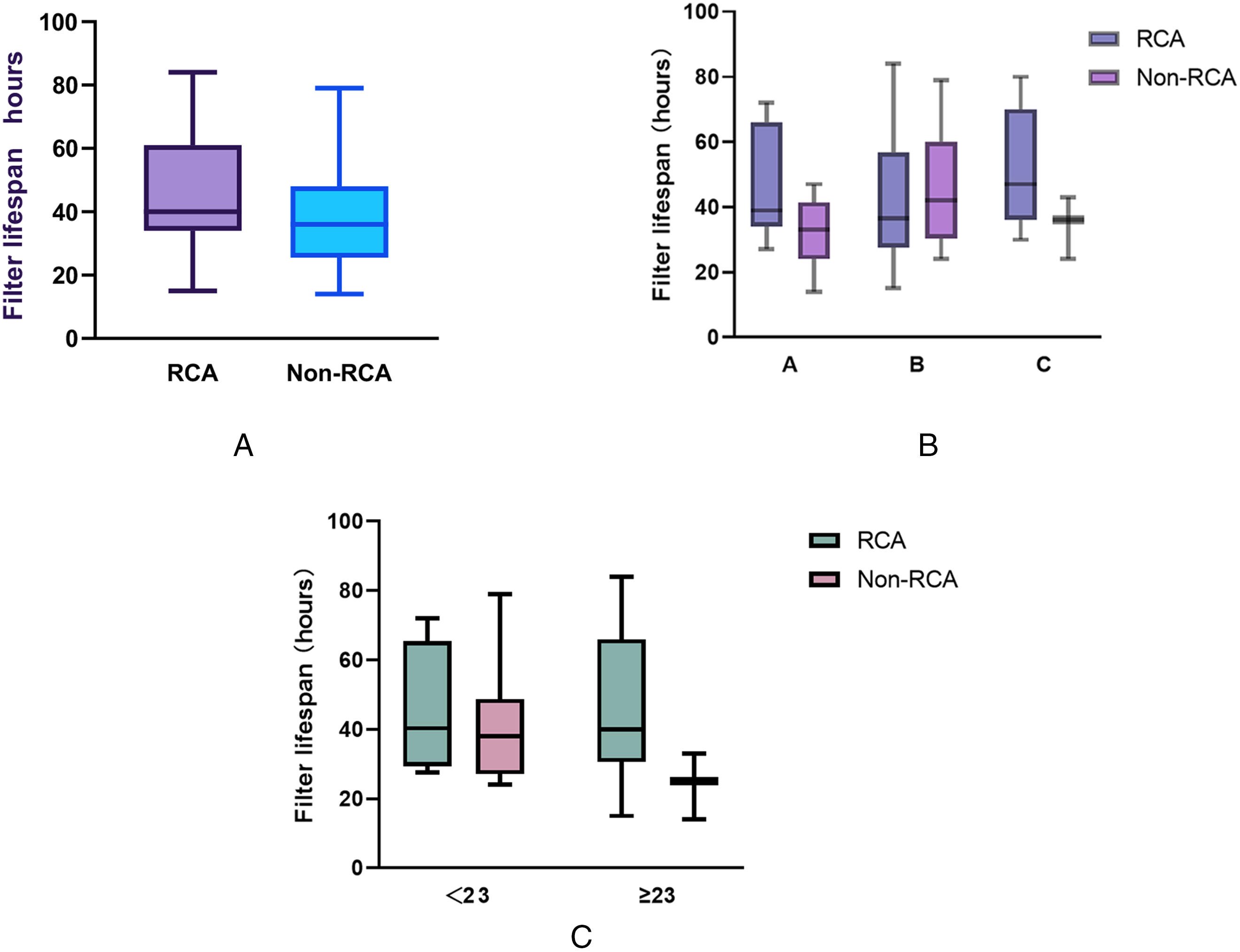
Comparison of filter lifespan by anticoagulation strategy and clinical subgroups. (A) Box-and-whisker plots for the regional citrate anticoagulation (RCA) and non-RCA cohorts. Median circuit life was longer with RCA (40.0 h, IQR 24.0–52.5) than with systemic heparin or no anticoagulation (36.0 h, 18.0–45.0), but the difference did not reach statistical significance (p = .219). (B) Subgroup analysis by Child–Pugh class. In the RCA cohort, median filter life rose as hepatic impairment worsened (Class A 35.0 h; B 38.0 h; C 47.0 h), whereas the non-RCA cohort showed shorter survival in Class C (32.0 h). The anticoagulation by Child–Pugh interaction was not significant (p = .124), yet RCA displayed a clear trend toward longer circuit life in Class C cirrhosis. (C) Stratification by illness severity using the cohort median APACHE II score (< 23 vs. ≥ 23). RCA yielded numerically longer predicted mean filter lifespans in both strata (45.65 h vs. 32.77 h for non-RCA), but the interaction between anticoagulation strategy and APACHE II category was not significant (p = .194).
When stratified by Child–Pugh classification, the median circuit lifespan increased progressively within the RCA arm with worsening liver function: 35.0 h in Class A, 38.0 in Class B, and 47.0 in Class C. In contrast, the corresponding non-RCA cohort displayed 33.0, 42.0, and 32.0 h, respectively (Figure 2B). A two-way ANOVA showed no significant interaction between anticoagulation strategy and Child–Pugh class (p = .124); however, the least-squares mean difference favored RCA by 9.38 h (p = .076). Although the non-RCA Class C subgroup was small (n = 3), RCA appeared to confer a clinically meaningful benefit in patients with severe hepatic dysfunction (p = .139), warranting further prospective confirmation.
Dichotomization of the cohort at a median APACHE II score of 23 revealed no statistically significant main effect of the anticoagulation strategy on circuit longevity (p = .078) (Figure 2C). Nevertheless, the estimated mean filter lifespan remained appreciably longer with RCA therapy (45.65 h) than with non-RCA therapy (32.77 h). The APACHE II risk grouping itself exerted no significant influence on filter duration (p = .261), although patients in the lower-severity group demonstrated numerically longer circuit survival than their higher-severity counterparts (43.27 vs. 35.15 h). Importantly, the interaction term between anticoagulation strategy and APACHE II category was not nonsignificant (p = .194), indicating that the favorable trend associated with RCA is consistent across the spectrum of global illness severity.
Collectively, these data suggest a consistent, though not statistically definitive, trend toward a longer circuit lifespan with RCA, particularly in patients with advanced hepatic dysfunction. However, this observation should be cautiously interpreted. Although PSM balanced the key indicators of disease severity (age, sex, APACHE II score, and Child–Pugh class), baseline coagulation tendency, which may directly influence filter survival, was not explicitly included in the matching model. Residual confounding related to coagulation status may therefore have influenced circuit lifespan, and the apparent protective trend associated with RCA should be considered hypothesis generating rather than confirmatory. Larger prospective studies are required to confirm these observations and define the subgroups most likely to benefit.
Impact of CRRT on metabolic, coagulation, and biochemical safety indicators
To delineate the physiological effects of the two anticoagulation strategies, we compared pre- and posttreatment changes (Δ) in the key laboratory indices (Table 2). Overall, the median Δ values were similar between the groups (p > .05) for most variables, indicating a comparable systemic safety profile. Nonetheless, RCA displayed directionally favorable trends, including a smaller decline in hemoglobin levels (Δ 6.00 [−34.0, 31.0] vs. 13.0 [−15.0, 28.0] g/L, p = .393) and a greater decrease in TBIL (Δ −7.80 [−37.3, 14.3] vs. −5.60 [−22.8, 31.5] μmol/L, p = .482). Notably, the RCA group exhibited a significantly greater reduction in lactate levels compared to the non-RCA group (Δ 2.96 ± 7.56 vs. 0.10 ± 2.40 mmol/L, p = .037), indicating potentially improved tissue perfusion and metabolic control.
Comparison of changes (Δ) in metabolic and coagulation parameters before and after CRRT between the RCA and non-RCA groups.

CRRT: continuous renal replacement therapy; RCA: regional citrate anticoagulation; APTT: activated partial thromboplastin time; PT: prothrombin time; PLT: platelet count; AST: aspartate aminotransferase; ALT: alanine aminotransferase; TBIL: total bilirubin; BUN: blood urea nitrogen; Scr: serum creatinine; HB: hemoglobin.
Data are presented as mean ± standard deviation or median (interquartile range), as appropriate. Δ represents the difference between post- and pretreatment values.
p-values refer to between-group comparisons of Δ values using the t-test or Mann–Whitney U test depending on distribution. *p < .05 indicates statistical significance.
In the subgroup analysis stratified by Child–Pugh class, the effects of CRRT on coagulation and metabolic parameters varied with the hepatic functional reserve (Figure 3). The median APTT shortened in Class A (−2.2 s) and B (−3.6 s) but increased slightly in Class C (+2.0 s), although the difference was not statistically significant (p = .573). PT remained essentially unchanged across classes (p = .439), indicating a minimal coagulation impact. Notably, lactate clearance was significantly pronounced in Child–Pugh Class C patients (p = .029), which is consistent with their higher baseline lactate burden. Arterial pH remained stable in both groups, with no significant intergroup differences observed (p = .815). These findings underscore the modulatory role of hepatic function in the physiological response to CRRT and highlight the need for intensified monitoring in patients with advanced cirrhosis (Child–Pugh Class C) owing to their greater vulnerability to dynamic changes in coagulation and metabolic burden.
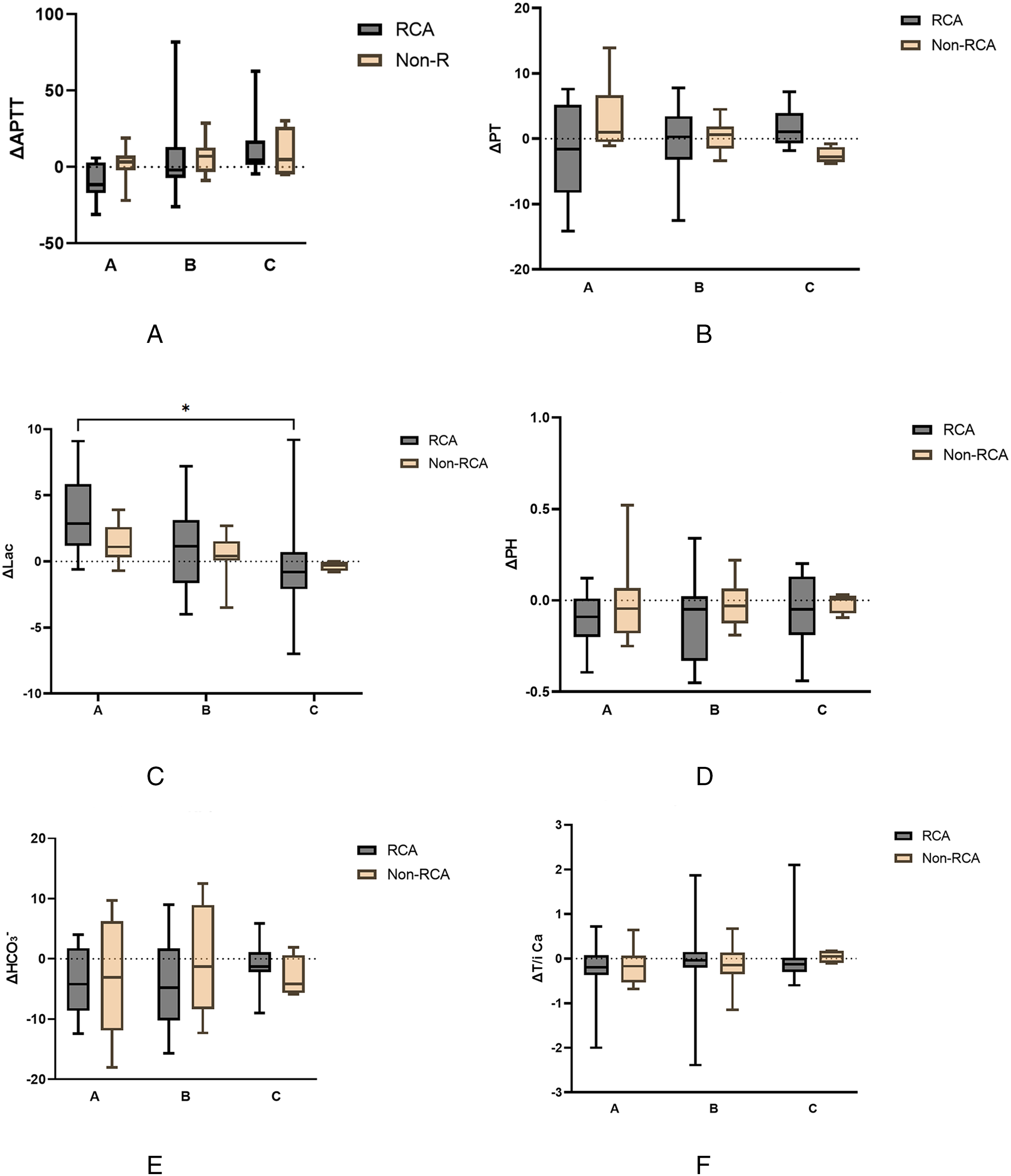
Post-CRRT changes in coagulation and metabolic indices, stratified by Child–Pugh class. (A) Activated partial thromboplastin time (ΔAPTT) across Child–Pugh classes (A)–(C) in RCA versus non-RCA cohorts. No significant differences were detected, although Class C patients exhibited greater variability. (B) Prothrombin time (ΔPT) by Child–Pugh class. Both anticoagulation strategies produced minimal PT shifts, with no intergroup or interclass significance. (C) ΔLactate (ΔLac) stratified by Child–Pugh class. RCA achieved a significantly greater lactate reduction in Class C (p < .05), with analogous trends in Classes A and B that did not reach significance. (D) ΔpH values across liver-function strata. Arterial pH remained within normal bounds in both groups, showing no significant intergroup or interclass differences. (E) ΔHCO₃⁻ (serum bicarbonate) by Child–Pugh class. Changes were small and comparable between RCA and non-RCA in all classes. (F) Total-to-ionized calcium ratio (ΔtCa/iCa) across Child–Pugh categories. Both cohorts demonstrated variable shifts without statistically significant differences.
Analysis of factors associated with citrate accumulation
To identify predictors of citrate accumulation, we first performed univariate logistic regression on 20 candidate variables encompassing age, APACHE II score, Child–Pugh score, Child–Pugh classification, filter lifespan, secondary bleeding events, and pre- to posttreatment changes (Δ) in laboratory parameters (ΔAPTT, ΔPT, ΔPLT, ΔHB, ΔAST, ΔALT, ΔTBIL, ΔScr, ΔBUN, ΔLactate, ΔpH, ΔK, ΔNa, and ΔHCO₃⁻) (Table 3). None of the variables reached statistical significance (all p > .05), although three parameters approached the threshold: Child–Pugh score showed a negative association with citrate accumulation (β = −1.203, OR = 0.299, 95% CI [0.054–1.653], p = .166). ΔLactate exhibited a borderline inverse relationship (β = −0.170, OR = 0.843, 95% CI [0.703–1.011], p = .066). ΔAPTT also showed a weak trend (β = −0.030, OR = 0.970, 95% CI [0.930–1.011], p = .156).
Univariate logistic regression analysis of factors associated with citrate accumulation.
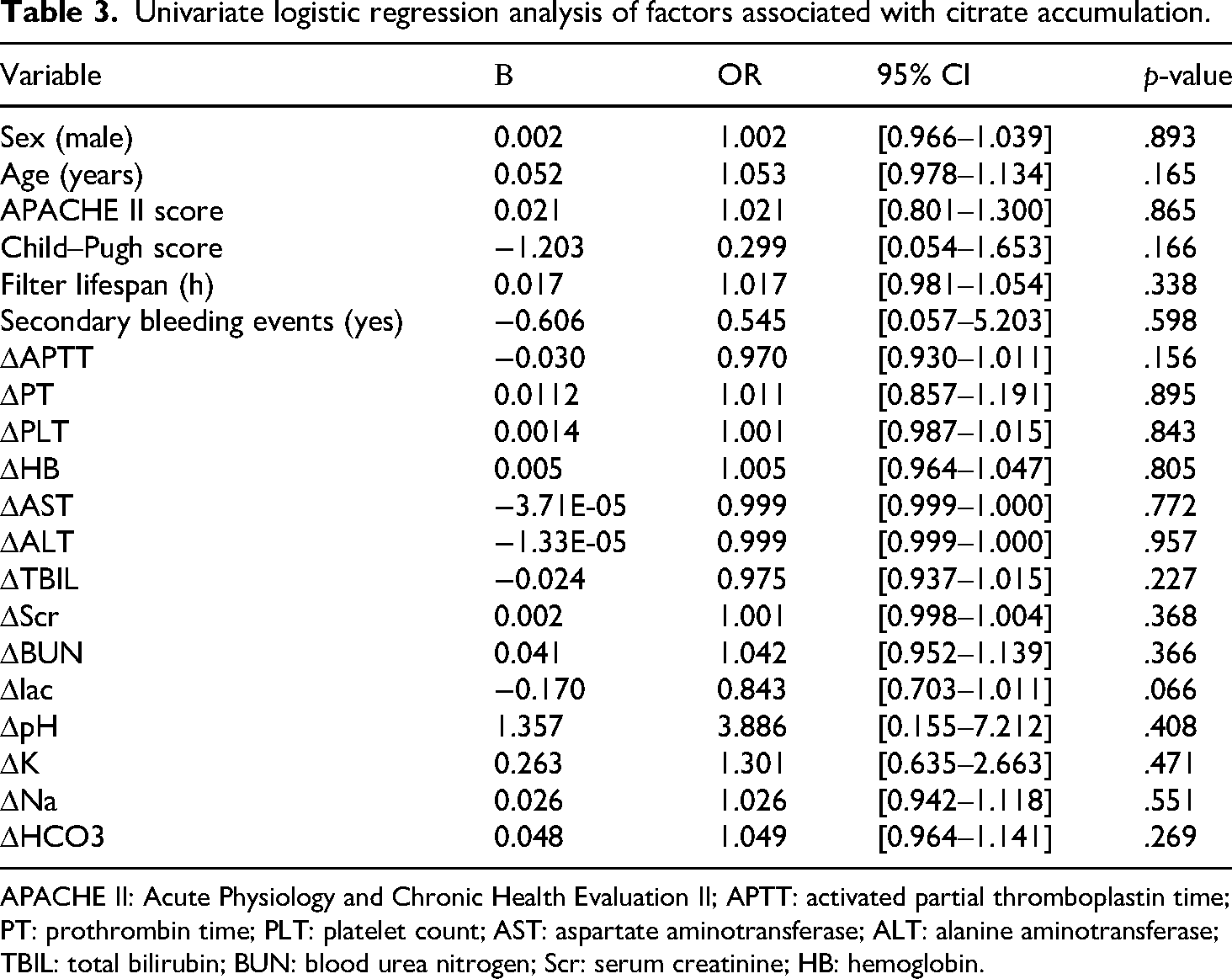
APACHE II: Acute Physiology and Chronic Health Evaluation II; APTT: activated partial thromboplastin time; PT: prothrombin time; PLT: platelet count; AST: aspartate aminotransferase; ALT: alanine aminotransferase; TBIL: total bilirubin; BUN: blood urea nitrogen; Scr: serum creatinine; HB: hemoglobin.
Variables with clinical plausibility or low p-values in univariate analysis, including age, APACHE II score, filter lifespan, secondary bleeding events, Child–Pugh classification, and representative Δ laboratory indices, were subsequently entered into a multivariable logistic model after confirming the absence of multicollinearity (Table 4). The final model exhibited acceptable goodness of fit, with variance inflation factor values within acceptable ranges. Among the included variables, Child–Pugh Class B emerged as an independent protective factor against citrate accumulation (OR = 0.054, 95% CI [0.003–0.921], p = .044). A similar nonsignificant trend was observed for Class C (p = .361), reflecting the wide confidence intervals stemming from the limited sample. Other variables, including age (p = .968), APACHE II score (p = .466), and various Δ biochemical parameters, failed to predict citrate accumulation independently (Table 4).
Multivariate logistic regression analysis of factors associated with citrate accumulation.

APACHE II: Acute Physiology and Chronic Health Evaluation II; APTT: activated partial thromboplastin time; PT: prothrombin time; PLT: platelet count; AST: aspartate aminotransferase; ALT: alanine aminotransferase; TBIL: total bilirubin; Scr: serum creatinine; HB: hemoglobin.
*p < .05 indicates statistical significance.
Taken together, these results suggest that moderate hepatic impairment (Child–Pugh B) may confer relative protection against citrate overload during CRRT, whereas other routinely measured clinical or laboratory parameters offer limited predictive value. Larger, prospective cohorts are needed to validate these observations and refine risk-stratification tools.
Risk factors for short-term mortality and predictive model construction
The 28-day mortality rate was significantly higher in the RCA group (37.1%) than in the non-RCA group (12.0%) according to Fisher's exact test, confirming the association (p = .040) (Figure 4A). Univariate logistic regression analysis, with 28-day mortality as the dependent variable, and patient baseline characteristics along with changes in biochemical parameters as covariates, identified APACHE II score as a significant risk factor for 28-day mortality (OR = 1.073, 95% CI [1.004–1.146], p = .038). Two additional variables, ΔAPTT (OR = 1.071, 95% CI [0.997–1.150], p = .060) and ΔHemoglobin (ΔHb) (OR = 1.026, 95% CI [0.990–1.064], p = .160), also showed potential associations, although these trends were not statistically significant (Table 5).
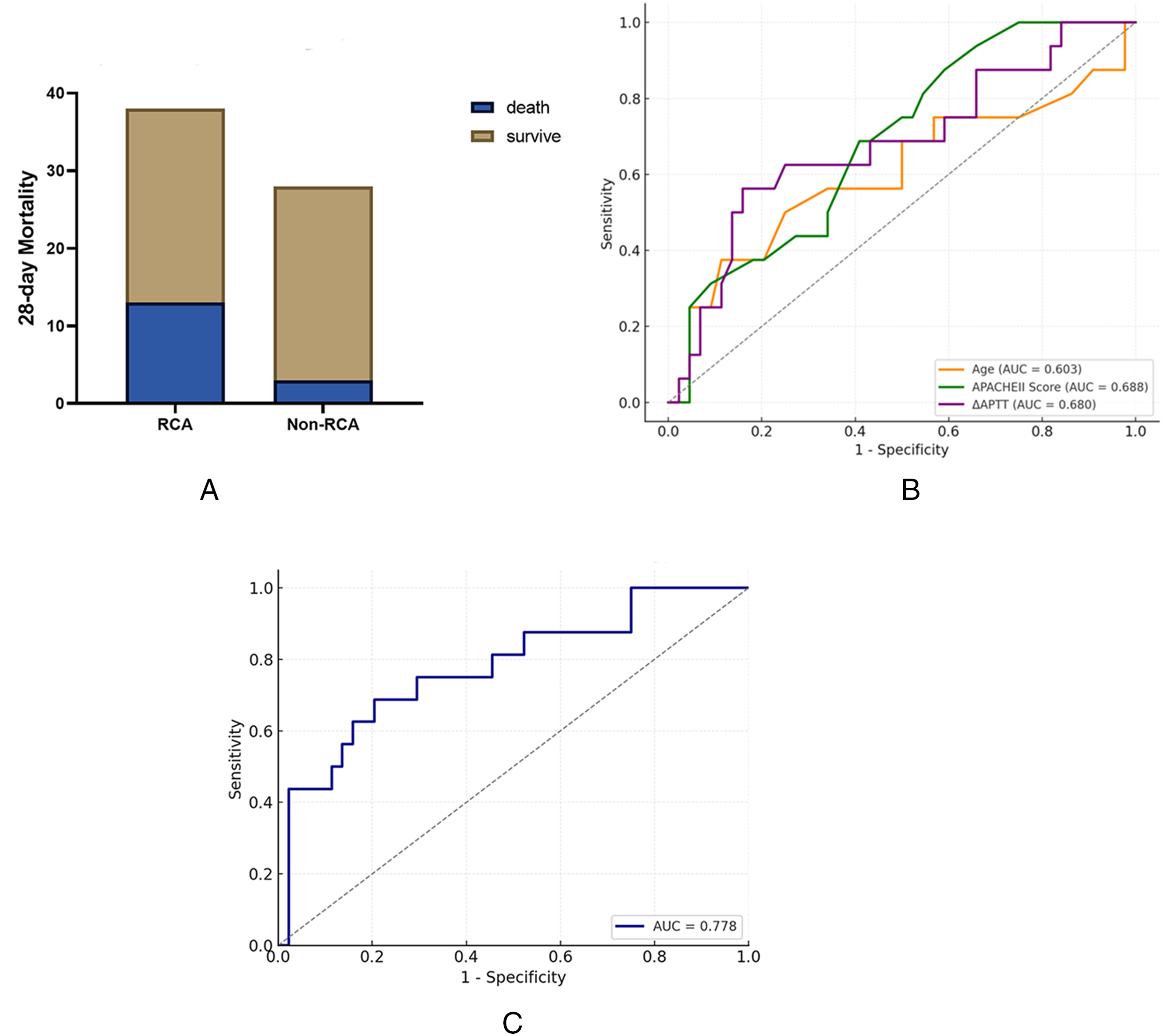
Twenty-eight-day mortality and predictive model performance. (A) Bar graph showing 28-day mortality in the RCA versus non-RCA cohorts. The RCA group experienced higher mortality (37.1%) compared with non-RCA (12.0%), a difference that reached statistical significance (Fisher's exact p = .040). (B) Receiver-operating-characteristic (ROC) curves for individual predictors of 28-day mortality: age (AUC = 0.603), APACHE II score (AUC = 0.688), and ΔAPTT (AUC = 0.680). APACHE II demonstrated the strongest single-variable discrimination. (C) ROC curve for the multivariable logistic regression model incorporating age, APACHE II score, and ΔAPTT. The combined model yielded an AUC of 0.778, indicating good overall predictive accuracy for 28-day mortality.
Univariate logistic regression analysis of factors associated with 28-day mortality.

APACHE II: Acute Physiology and Chronic Health Evaluation II; APTT: activated partial thromboplastin time; PT: prothrombin time; PLT: platelet count; AST: aspartate aminotransferase; ALT: alanine aminotransferase; TBIL: total bilirubin; BUN: blood urea nitrogen; Scr: serum creatinine; HB: hemoglobin.
*p < .05 indicates statistical significance.
To improve model robustness, variables with a p-value < .3 in univariate screening were subsequently entered into multivariate logistic regression after confirming the absence of multicollinearity. Age (OR = 1.056, 95% CI [1.005–1.111], p = .030) and APACHE II score (OR = 1.119, 95% CI [1.009–1.242], p = .035) emerged as independent predictors of 28-day mortality. Although ΔAPTT did not achieve statistical significance in the multivariate model (OR = 1.026, 95% CI [0.993–1.062], p = .108), it demonstrated a potential trend toward increased mortality risk (Table 6).
Multivariate logistic regression analysis of independent predictors of 28-day mortality.

APACHE II: Acute Physiology and Chronic Health Evaluation II; APTT: activated partial thromboplastin time; AST: aspartate aminotransferase; ALT: alanine aminotransferase; Scr: serum creatinine; HB: hemoglobin.
*p < .05 indicates statistical significance.
ROC curves were constructed for each significant predictor. The AUC was 0.603 for age, 0.688 for the APACHE II score, and 0.680 for ΔAPTT (Figure 4B). A composite model incorporating all three variables achieved an AUC of 0.778 (Figure 4C), indicating a good overall discrimination for predicting 28-day mortality.
Collectively, these findings highlight age and global illness severity (APACHE II) as robust determinants of short-term outcomes while suggesting a potential contribution of coagulation dynamics (ΔAPTT) to mortality risk. Larger prospective studies are needed to validate and refine this predictive paradigm.
Discussion
A principal observation of our retrospective analysis is the directionally longer filter lifespan achieved with RCA compared with noncitrate strategies; a difference that did not attain statistical significance in this modest cohort remained most apparent among individuals with advanced cirrhosis (Child–Pugh C). This trend is consistent with the existing CRRT literature. The multicenter, randomized RICH trial demonstrated an 11-h extension of the median circuit life with RCA relative to systemic heparin, despite enrolling a broad spectrum of critically ill patients. 12 A separate single-center study reported a nearly two-fold improvement in filter lifespan (49 h vs. 28 h, p = .004) in favor of RCA. 13 Meta-analyses have also confirmed that RCA significantly reduces the risk of premature circuit failure (hazard ratio ∼0.5 vs. regional heparin) and filter clotting (relative risk ∼0.70). 14
Our own Child–Pugh C subgroup highlights the practical relevance of these data: RCA preserved circuit patency despite profound coagulopathy, whereas noncitrate regimens showed markedly shorter survival. The likely mechanistic explanation involves the synergy of two factors: an intrinsically hypocoagulable systemic environment caused by hepatic synthetic failure, and the potent local anticoagulant effect of citrate through ionized-calcium chelation inside the extracorporeal circuit. Importantly, similar observations have been made for larger hepatology-focused cohorts. In the multicenter Liver Citrate Anticoagulation Threshold (L-CAT) observational study, 96% of CRRT circuits in patients with severe liver impairment remained functional at 72 h, and citrate accumulation was detected in only three of 133 cases, 15 underscoring both feasibility and biochemical safety when meticulous monitoring is applied even in the context of liver failure. 15
Taken together, our findings, along with existing literature, support that the superior circuit performance of RCA is not merely a technical victory; it translates into more reliable solute clearance, fewer treatment interruptions, and a lower burden of circuit exchanges, which are particularly valuable for hemodynamically unstable intensive-care patients in whom even a brief downtime can compromise metabolic control. Consistent filter longevity also minimizes nursing workload and preserves scarce CRRT resources, factors that acquire added significance during surges in critical care demand.
Beyond circuit mechanics, our investigation points to metabolic and coagulation stability as additional benefits of RCA. Across all Child–Pugh classes, we observed favorable trends in ΔAPTT and ΔPT, indicating that citrate did not exacerbate and may modestly ameliorate coagulation derangements. Notably, patients in Child–Pugh class C experienced the largest absolute decrease in serum lactate, suggesting improved lactate clearance and partial correction of metabolic acidosis. This finding aligns with prior work showing that citrate metabolism yields bicarbonate without introducing an exogenous lactate load, thereby supporting acid–base equilibrium. A recent systematic review focused on liver-failure populations receiving RCA and corroborated these metabolic advantages, documenting significant lactate reductions and stable coagulation parameters under vigilant biochemical surveillance. 16 Collectively, these data reinforce the notion that RCA not only optimizes circuit performance but also confers systemic physiological benefits in high-risk settings of concurrent hepatic and renal dysfunction.
Importantly, RCA did not exacerbate the systemic coagulopathy in our cohort. Because citrate chelates ionized calcium only within the extracorporeal circuit and no systemic heparin was administered, global clotting times remained stable. Indeed, APTT fell modestly in many Child–Pugh A and B patients, a change that is more plausible, explained by convalescence from critical illness rather than the pro-coagulant effect of citrate itself.12,17–19 The slight increase in APTT observed in Child–Pugh C participants is better attributed to the intrinsic severity of their hepatic synthetic failure than to the anticoagulation strategy. These observations reinforce earlier reports showing that RCA confines its action to the circuit and, when combined with supportive care, may even allow the gradual correction of systemic coagulation defects. The slight increase in APTT observed in Child–Pugh C participants is better attributed to the intrinsic severity of their hepatic synthetic failure than to the anticoagulation strategy. These observations reinforce earlier reports showing that RCA confines its action to the circuit and, when combined with supportive care, may even allow the gradual correction of systemic coagulation defects.
By localizing anticoagulation, RCA circumvents the bleeding liability inherent to systemic heparin, a benefit that assumes particular importance in cirrhotic patients with a precarious hemostatic equilibrium. The parallel gains in metabolic control and coagulation stability represent a dual advantage, addressing two of the cardinal pathophysiological challenges, acidosis and coagulopathy, which complicate CRRT in patients with liver failure. Citrate accumulation remains the chief theoretical hazard of RCA in hepatic dysfunction, arising when impaired metabolism permits citrate to enter the systemic circulation, elevates the total-to-ionized calcium ratio, and provokes metabolic acidosis or hypocalcemia. Our analysis identified the Child–Pugh classification as the dominant determinant of this complication: Class C status conferred a markedly higher risk, whereas Class B appeared relatively protective. This finding is physiologically intuitive, as patients in Class B usually retain appreciable hepatocellular and extrahepatic metabolic capacity, whereas those in Class C do not, underscoring the critical role of hepatic function reserve in determining the safety of RCA.
Consistent with our results, current guidelines list severe acute hepatic failure or decompensated cirrhosis as relative contraindications to RCA.20,21 Nonetheless, accumulating evidence indicates that with vigilant biochemical surveillance, advanced liver disease does not invariably preclude safe use. In a multicenter L-CAT observational study, only 2% of patients with severe hepatic failure met the laboratory criteria for citrate accumulation, and metabolic outcomes were comparable to those in individuals with preserved liver function. 15 Similarly, a systematic review concluded that RCA “appears safe” when early markers of accumulation are tracked closely. 14 Our data refined this perspective by demonstrating that moderate hepatic impairment (Child–Pugh B) is generally compatible with RCA, whereas Class C disease demands individualized protocols and intensified monitoring. The predictive nomogram developed from our cohort, which integrates liver function metrics with systemic indicators, achieved good discrimination (AUC ∼ 0.8) for identifying patients at a risk of citrate accumulation. Such tools may facilitate tailored anticoagulation choices, ensuring that the metabolic and hemostatic benefits of RCA are realized without compromising safety.
We propose that the RCA should not be reflexively withheld in patients with moderate hepatic impairment. When ionized calcium, acid–base balance, and anion gap are checked at preset intervals, most Child–Pugh B patients can safely undergo RCA-CRRT. In contrast, patients with Child–Pugh C disease warrant a more conservative approach, starting with a reduced citrate load, tightening biochemical surveillance, and transitioning early to an alternative anticoagulant if any biochemical signature of accumulation appears. Protocols that begin with a reduced citrate dose have been shown to safely sustain circuit patency in patients with hepatic impairment. In particular, the L-CAT–guided approach demonstrated that lower citrate exposure can maintain effective anticoagulation while minimizing metabolic toxicity in liver-impaired cohorts.15,20,22
Evaluation of patient-centered outcomes revealed no difference in 28-day survival between citrate and noncitrate strategies, echoing randomized trials and meta-analyses that likewise failed to demonstrate a mortality advantage for RCA.13,23,24 For example, the RICH trial demonstrated a clear extension of circuit life with citrate, yet reported equivalent 90-day mortality when compared with systemic heparin, underscoring that outcomes in the intensive care setting are chiefly dictated by the severity of underlying disease, septic shock, multiorgan failure, or, in our cohort, advanced cirrhosis rather than by the anticoagulant used during CRRT.
Logistic regression analysis confirmed that age and APACHE II score were independent determinants of 28-day mortality, while ΔAPTT showed a non-significant trend toward association with mortality. Each variable was positively associated with risk; older patients, those presenting with greater baseline physiological derangement, and those who exhibited worsening coagulation profiles during CRRT experienced the poorest outcomes. The three-factor model displayed solid discriminatory power (AUC, 0.78), indicating that patient-specific characteristics, rather than the choice of anticoagulation strategy, primarily drive short-term survival. The APACHE II score is a long-standing predictor of intensive care unit mortality, integrating acute physiological stress and chronic comorbidities, while advanced age is well-recognized as a marker of diminished physiological reserve. Importantly, the inclusion of ΔAPTT underscores the potential prognostic value of dynamic coagulation monitoring during CRRT, aligning with emerging evidence that temporal trends in hemostatic indices carry greater predictive weight than single baseline measurements.25,26 A persistently elevated or rising APTT during CRRT likely signals unresolved coagulopathy, whether from ongoing disseminated intravascular coagulation or inadequate restoration of hepatic synthetic function, and therefore reflects more severe systemic derangement. 26
This observation is in accordance with previous work in critically ill and liver failure cohorts, where sustained abnormalities in coagulation tests (e.g. rising APTT or INR) have been associated with progression toward multiorgan failure and higher mortality, 27 including among patients receiving renal replacement therapy.28,29 Likewise, contemporary CRRT studies demonstrate that failure to metabolize citrate, evidenced by increasing systemic citrate concentrations, escalating APTT, or rising lactate, portends poor outcomes, underscoring the prognostic value of dynamic hemostatic and metabolic monitoring.15,29,30
Survival outcomes in patients receiving RCA-CRRT appear to be driven more by underlying illness severity than by anticoagulation strategies. A three-variable model (age, APACHE II score, ΔAPTT) showed good discrimination for 28-day mortality, helping identify patients at greatest risk who may benefit from escalated interventions and timely goal-of-care discussions. Several limitations warrant caution: the sample size was small (n = 66), limiting power; baseline imbalances existed, with higher APACHE II and Child–Pugh scores in the RCA group, raising residual confounding despite propensity matching; and the single-center retrospective design restricts generalizability. Thus, the findings should be viewed as hypothesis-generating rather than definitive, underscoring the need for larger multicenter studies.
Conclusions
In this single-center, propensity-matched retrospective study, RCA during CRRT in patients with hepatic dysfunction and AKI was feasible and generally safe. RCA showed a non-significant trend toward longer circuit survival and stable biochemical control, while mortality appeared to be driven more by illness severity than by anticoagulation strategies. Patient-specific citrate titration and close monitoring are essential, particularly in patients with advanced cirrhosis. Given the small sample size, baseline imbalances, and single-center design, these findings should be considered hypothesis generating. Larger multicenter studies are required to validate the safety, clarify the effects on outcomes, and refine patient-tailored RCA protocols.
Supplemental Material
sj-docx-1-sci-10.1177_00368504261433391 - Supplemental material for Regional citrate anticoagulation prolongs filter lifespan and improves metabolic control in CRRT for liver dysfunction-associated AKI: A retrospective cohort study
Supplemental material, sj-docx-1-sci-10.1177_00368504261433391 for Regional citrate anticoagulation prolongs filter lifespan and improves metabolic control in CRRT for liver dysfunction-associated AKI: A retrospective cohort study by Wenpeng Jin, Pengyu Chen, Xinyu Wang, Yunbo Du and Zhengxiong Li in Science Progress
Footnotes
Acknowledgements
Not applicable.
Ethics approval and consent to participate
This study was reviewed and approved by the Medical Ethics Committee of Longhua District Central Hospital, Shenzhen City in 12 March 2024 (No. 2024-016-01). All patient information in the database was anonymized, and therefore, informed consent was not required.
Ethical guidelines
The study protocol was conducted in accordance with the declaration of Helsinki.
Consent for publication
Not applicable.
Clinical trial number
Not applicable.
Author contributions
All the authors contributed to the conception and design of the study. Wenpeng Jin, Pengyu Chen, Xinyu Wang, and Yunbo Du performed the material preparation, data collection, and analysis. Wenpeng Jin prepared the first draft of this manuscript. Li reviewed and edited the manuscript. All authors contributed to the improvement of the manuscript. All authors have read and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Projects of Medical and Health Institutions of Longhua District, Shenzhen (Grant No. 2024061).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
All the data generated during this study are presented in the manuscript. The raw data files are available from the corresponding author upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
