Abstract
Objective
To compare the effectiveness, safety, and cost outcomes of brand-name febuxostat (feburic) and generic febuxostat (feuri) in patients with hyperuricemia or gout in a real-world clinical setting.
Methods
We conducted a retrospective cohort study using electronic medical records from Taichung Veterans General Hospital between November 2021 and December 2023. Adult patients with hyperuricemia or gout who initiated febuxostat therapy and had no prior urate-lowering treatment in the preceding 3 months were included. Serum uric acid levels were assessed at baseline, 3 months, and the final follow-up. Propensity score matching (1:1) was applied to balance baseline demographic, clinical, laboratory, cardiovascular, and renal characteristics. Clinical outcomes, laboratory parameters, and medication costs were compared between groups.
Results
Both feuri and feburic were associated with significant reductions in serum uric acid levels after 3 months of treatment. At the 3-month assessment, the mean serum uric acid level was modestly lower in the feuri group (5.41 ± 2.06 mg/dL) than in the feburic group (5.75 ± 2.12 mg/dL;
Conclusions
This real-world study suggests that generic febuxostat is associated with safety outcomes comparable to those of the brand-name formulation and may be prescribed at lower doses and lower medication costs in routine clinical practice.
Introduction
Gout is biochemically characterized by the saturation of extracellular fluid with urate. Both the incidence 1 and prevalence 2 of gout increased from 2011 to 2018. Approximately 12 million patients are affected by gout. 3 The condition is primarily caused by elevated blood uric acid (UA) levels, with hyperuricemia being a necessary factor for the development of urate crystal deposition disease. Consequently, the treatment of gout focuses on UA-lowering agents, including uricosuric drugs and xanthine oxidase inhibitors (XOI) such as allopurinol and febuxostat. Uricosuric agents have limited effectiveness in patients with renal dysfunction. 4 For most patients requiring UA-lowering therapy, allopurinol is recommended as the first-line XOI. However, allopurinol dosing must be adjusted in patients with renal dysfunction. 5 Though rare, severe adverse reactions like Stevens-Johnson syndrome 6 may occur, with a higher but still low incidence in individuals with the human leukocyte antigen (HLA) B58:01 allele, chronic kidney disease (CKD), or those using thiazide/loop diuretics. Allopurinol should be avoided in HLA-B5801-positive individuals, especially among Korean, Thai, and Chinese populations, where this genetic marker is more prevalent.7,8 For patients with reduced kidney function, febuxostat, initiated at 40 mg daily, serves as an alternative XOI option. Febuxostat is more effective than allopurinol at reducing uric acid levels.9,10 However, the cost of treatment with brand-name febuxostat (Feburic®, Astellas Pharma Singapore Pte Ltd) is significantly higher compared to allopurinol, whether in its generic form or brand-name equivalents.
There are many advantages of generic medications compared to brand-name medications. Generic medications offer the same efficacy and safety, containing identical active ingredients, strength, dosage form, and route of administration as brand-name drugs. 11 They are also required to meet strict bioequivalence standards, 12 ensuring that the generic version releases its active ingredient into the bloodstream at nearly the same rate and in nearly the same amounts as the original drug. However, since they do not require the same investment in research, development, and marketing, generic drugs are typically 80–85% cheaper than their brand-name counterparts according to the Food and Drug Administration. 11 This lower cost increases accessibility for a wider population, particularly for individuals or healthcare systems that may struggle with the high cost of brand-name medications. Additionally, because multiple manufacturers can produce generic versions once the patent expires, they are more readily available, reducing the risk of shortages, which sometimes occur with single brand-name producers. For example, during the COVID-19 pandemic, many countries faced medication shortages, and the wider availability of generic medications helped address these challenges. 13
However, generic medications only undergo studies for bioequivalence—meaning the generic version is tested to ensure it releases its active ingredient into the bloodstream at a similar rate and quantity as the original drug. 12 These studies are typically conducted on a relatively small group (24–36) of healthy volunteers. In Taiwan's regulations, 14 both brand-name and generic drugs must have the same dosage form, strength, and efficacy, which is primarily based on bioequivalence testing. Their therapeutic equivalence may not always be exactly the same. It's also important to note that excipients (inactive ingredients) always differ between brand-name and generic products. 15 Many excipients have not been thoroughly tested for their impact on drug absorption in the intestine. 15 Further laboratory studies are needed to understand the potential differences in excipients between brand-name and generic products, particularly regarding their safety and effectiveness in real-world human studies.
Therefore, in this study, we will investigate the cost-effectiveness and safety of brand-name febuxostat (Feburic®, Astellas Pharma Singapore Pte Ltd) compared to generic febuxostat (Feuri®, Chunghwa Yuming Healthcare Co., Ltd) in a post-marketing surveillance study.
Materials and methods
Study design
We included outpatients aged 18 and older from Taichung Veterans General Hospital between November 1, 2021, and December 31, 2023. Initially, patients diagnosed with hyperuricemia or gout were enrolled. The index date was defined as the date when patients started taking UA-lowering agents, including feburic, feuri, allopurinol, or benzbromarone. Only UA-lowering drug-naive patients (for at least 3 months) without any medication changes were included. Drug-naive status was defined as not having taken any UA-lowering agents between November 1, 2021, and February 1, 2022. Only patients with a medication duration of at least 90 days were included. The index event corresponds to the initiation of UA-lowering agents, with the commencement date serving as the index date. It is essential that the diagnosis of hyperuricemia or gout precedes the start of UA-lowering agents. For both groups (feburic and feuri), the baseline data collection window extends from 1 year to one day before the index date, using the most recent available data. To mitigate reverse causality, outcome data collection began 1 month after the index event and continued until the end of the follow-up period. The study protocol was approved by the Institutional Review Board of Taichung Veterans General Hospital (Approval No. SE24291C). The requirement for informed consent was waived because this study used retrospective, de-identified patient data. All patient information was anonymized prior to analysis. The study was conducted in accordance with the Declaration of Helsinki (1975), as revised in 2024. This study was a retrospective, real-world cohort study conducted using electronic medical records from Taichung Veterans General Hospital. The reporting of this observational study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 16
Definition of population and baseline data collection
Hyperuricemia was defined using ICD-10-CM code: E79.0, and gout was defined using ICD-10-CM codes: M10, M109, M1A, M1A9XX0, and M1A9XX1. Demographic data, including age (years), gender, and body mass index (kg/m2), were collected. Laboratory data were also included, such as white blood cell count (WBC) (/cumm), hemoglobin (g/dL), platelet count, serum UA (mg/ dL), aspartate transaminase (AST) (U/L), alanine transaminase (ALT) (U/L), serum albumin (g/dL), low-density lipoprotein (LDL) (mg/dL), fasting glucose (mg/dL), glycated hemoglobin (HbA1c) (%), total cholesterol (TC) (mg/dL), and triglyceride (mg/dL). Baseline comorbidities, such as diabetes mellitus (DM) (ICD-10-CM codes E10 and E11), allergy (ICD-10-CM code T78.40XA), and hepatitis (ICD-10-CM codes B15.X-B19.X), were also included.
Baseline renal function data were collected, including blood urea nitrogen (BUN) (mg/dL), serum creatinine (SCr) (mg/dL), estimated glomerular filtration rate (eGFR) (mL/min/1.73 m2), urinary protein-to-creatinine ratio (UPCR (mg/g)), urinary albumin-to-creatinine ratio (UACR)(mg/g), acute kidney injury (AKI) (ICD-10-CM code N17.9), chronic kidney disease (CKD) (ICD-10-CM codes N18.1-N18.5, N18.9), and end-stage kidney disease (ESKD) (ICD-10-CM code N18.6). Baseline CV status was defined as follows: heart failure (ICD-10-CM codes I11.X, I13.X, I50.X), hypertension (ICD-10-CM codes I10 and I15) (including systolic (SBP) and diastolic blood pressure (DBP) (mmHg)), ischemic heart disease (ICD-10-CM codes I24.8, I24.9, I25.89, I25.9), myocardial infarction (ICD-10-CM codes I21.X, I22.X, I23.X, I24, I25.2), stroke (ICD-10-CM codes I60-I69), and angina pectoris (ICD-10-CM codes I20.X, I23.7, I25.X, K70.X-K71.X, K73.X, K75.X, Z20.5, Z22.5.X). Left ventricular ejection fraction (LVEF) (%) and NT-proBNP (pg/mL) were also included.
Medications associated with CV outcomes were also collected according to ATC codes, including renin–angiotensin–system inhibitors, angiotensin receptor-neprilysin inhibitors, beta blockers, sodium-glucose cotransporter-2 inhibitors, mineralocorticoid receptor antagonists, aspirin, warfarin, and direct oral anticoagulants (edoxaban, apixaban, rivaroxaban, and dabigatran).
Pre-specified outcomes
Serum UA levels were collected at three time points: baseline (before medication), the first value after medication (at 3 months), and the last value during the follow-up period. The dosage of feburic or feuri was also recorded. The initial dose of feburic or feuri was universally 40 mg per day. The final stable dose was determined based on the last recorded dose during the study period.
Laboratory outcomes in pre-propensity score matching (PSM) cohort of hemogram (WBC, hemoglobin, and platelet), liver function (AST, ALT, and serum albumin), renal function (BUN, SCr, eGFR, UPCR, and UACR), heart function (LVEF and NT-proBNP), and metabolic parameters (TC, LDL, and triglyceride) were collected to comparsion before and after either febuic or feuri. Outcomes (both incidence and prevalence) between feburic and feuri groups wre compared in post-PSM groups, inclduign renal function (BUN, eGFR, ESKD, CKD, AKI, UPCR, and UACR), CV outcome (LVEF, NT-proBNP, ischemic heart disease, myocardiala infarction, stroke, hearrt failure, and angina pectoris), safelty toucome (allergy, Stevens-Johnson syndrome, hepatitis, diabes melltis and hypertension), hospital visit per year and usage of painkiller (according to ATC code of steroid, acetaminophen, opioid, and non-steroidal anti-inflammatory drugs) (as surrogat outcome of gout attack).
Statistical analyses
Normality was assessed using the Kolmogorov–Smirnov test (if two groups) and Kruskal–Wallis test (if three more groups). The
Baseline characteristics between feburic and feuri before and after propensity score matching.

PSM: propensity score matching
SMD: standard mean difference
Statistical analyses were performed using SPSS software version 22 (SPSS Inc., Chicago, Illinois, USA) and R software (version 4.3.1) with the MatchIt package. A two-tailed
Results
Patient selection algorithms
The detailed patient selection process is shown in Figure 1. Initially, we identified 39,818 patients diagnosed with gout or hyperuricemia in our outpatient department since November 1, 2021. We excluded patients who had never received treatment with any uric acid-lowering agents (
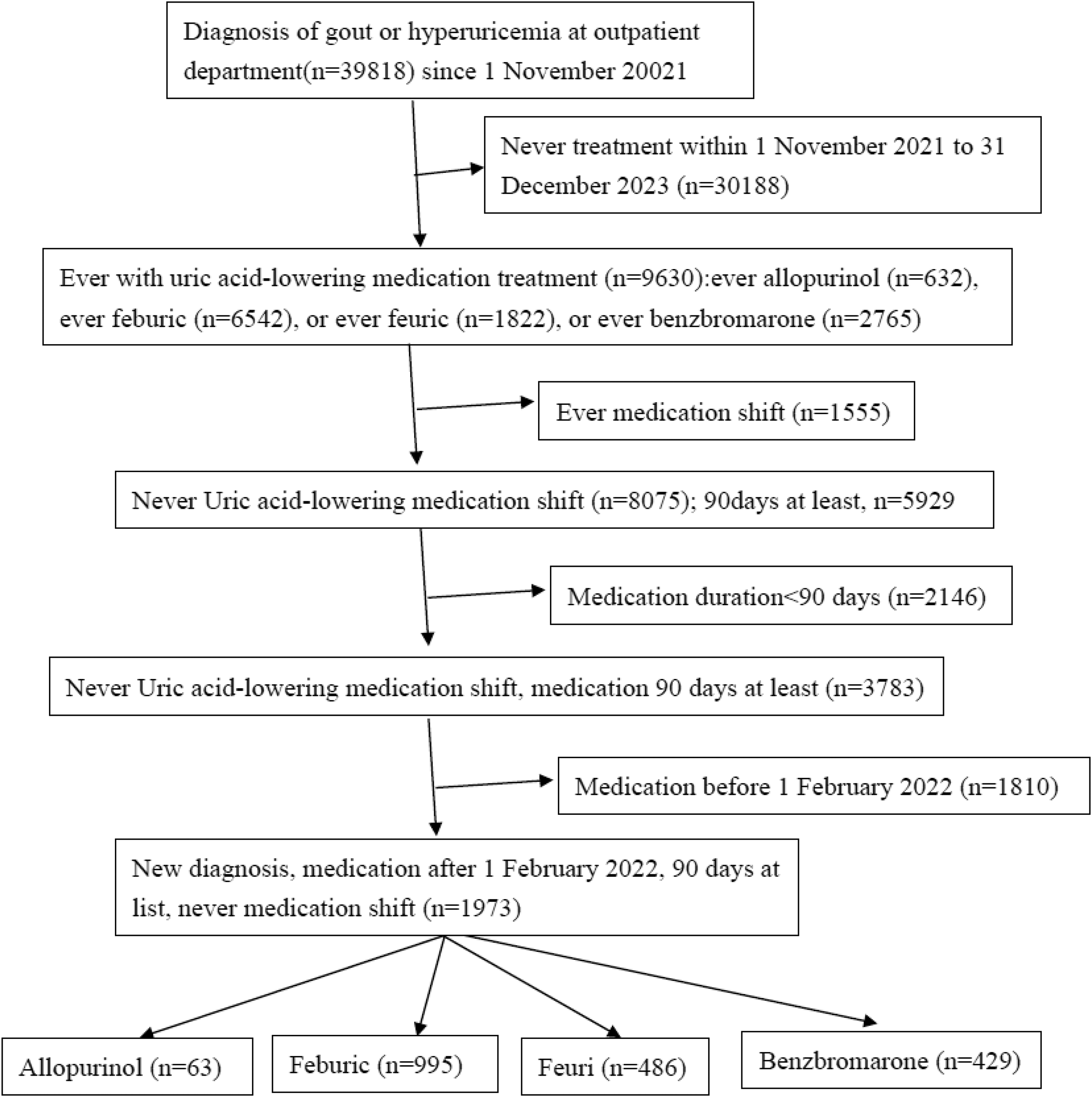
Algorithm for participant recruitment.
Baseline characteristics of this cohort before and after propensity score matching
Table 1 compares the baseline characteristics of the two patient groups, feburic (
In this cohort (after PSM), the patients are relatively young, with an average age of 63, and predominantly male (67%). The metabolic conditions are generally mild, including mild hypertension (mean SBP of 141 mmHg), mild hyperuricemia (serum UA levels of 8.1–8.2 mg/dL), LDL of 104 mg/dL, triglycerides of 150 mg/dL, fasting glucose levels of 117–119 mg/dL, HbA1c of 5.59–5.90%, and 38% prevalence of DM. Regarding CV conditions, 30% of patients had heart failure, 4% had ischemic heart disease, 1.9% had a history of myocardial infarction, 10–11% had a history of stroke, 50% had reduced LVEF, and NT-proBNP levels ranged from 322 to 3653 pg/mL Renal function was notably impaired, with SCr levels of 2.64–2.80 mg/dL, an eGFR of 41 mL/min/1.73 m2, 68% prevalence of CKD, and a high prevalence of proteinuria (UPCR of 1840–2011 mg/g and UACR of 1054–1143 mg/g). In terms of medication, 43% of patients were taking renin-angiotensin system inhibitors. In summary, this cohort demonstrates moderate renal dysfunction (eGFR of 41 mL/min/1.73 m2), mild metabolic conditions, and mild to moderate CV conditions. Most patients in this cohort were primarily from the nephrology department: 49.9% in either the feburic or feuri group, 49.9% in the feburic group, and 68.5% in the feuri (Supplementary Figure 2).
Outcomes of serum uric acid in feburic (brand) and feuri (generic, CYH) groups
The UA levels at baseline (before medication), at the first follow-up (after 3 months of a universal dose of 40 mg per day), and the final follow-up for both the feburic and feuir groups (before PSM) are shown in Figure 2. Table 2 presents a comparison between the two groups, while Table 3 shows a comparison of UA levels at different time points within each group (feburic and feuir).
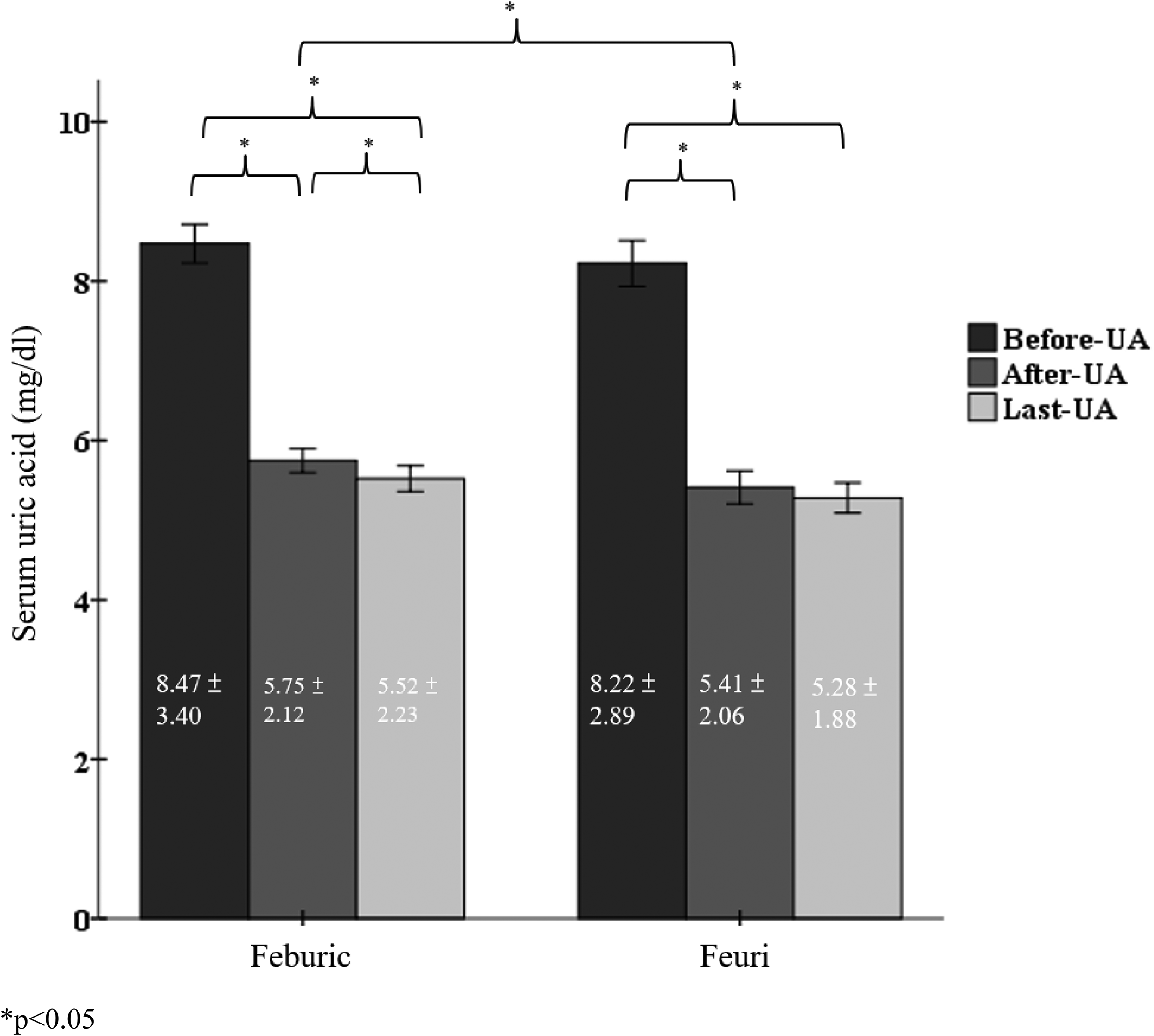
Serum uric acid before and after each feburic and feuri.
Comparisons of serum uric acid between feburic and feuri groups during different timings (before propensity score matching).

*The last uric acid is the serum uric acid value while end of follow-up: 341 ± 131 days for feburic group and 330 ± 133 days for feuri group.
Comparisons of serum uric acid during different timings in either feburic or feuri group (before propensity score matching).

n/a: not available
In Figure 2 and Table 3, feburic significantly lowered serum UA levels after 3 months of treatment (from 8.47 ± 3.40 mg/dL to 5.75 ± 2.12 mg/dL,
As seen in Figure 2 and Table 2, during the first follow-up (3 months), feuri reduced UA levels more effectively than feburic (5.41 ± 2.06 mg/dL vs. 5.75 ± 2.12 mg/dL,
In addition to comparisons of mean serum UA levels, we evaluated the proportion of patients achieving the commonly recommended target serum UA level (<6 mg/dL) (Supplementary Table 1). At the 3-month follow-up, a numerically higher proportion of patients in the feuri group achieved the target UA level compared with the feburic group. However, after PSM, the difference in target attainment between groups was not statistically significant. Similar findings were observed at the final follow-up, with comparable proportions of patients achieving target UA levels in both groups. These results indicate that, despite modest differences in mean serum UA values, the likelihood of achieving clinically relevant UA targets was broadly similar between generic and brand-name febuxostat in real-world practice.
Does comparison between feburic (brand) and feuri (generic, CYH) groups
The dose and price information for feburic (brand) and feuri (generic, CYH) are presented in Supplement Table 2. Both groups started with a uniform initial dose of 40 mg per day. However, the mean final dose was lower in the feuri group compared to the feburic group (41.58 mg vs. 45.96 mg per day,
Other laboratory outcomes before and after treatment either feburic (brand) and feuri (generic, CYH) (before PSM)
Other laboratory outcomes before and after treatment with either feburic or feuri are presented in Table 4. In both groups, liver enzymes (AST and ALT) did not show significant changes (all
Comparisons of all laboratory outcomes before and after either feburic or feuri treatment (before propensity score matching).

All outcomes compariosn between feburic (brand) and feuri (generic, CYH) (after PSM)
All outcome comparisons between the feburic and feuri groups are shown in Supplementary Table 3 (before PSM) and Table 5 (after PSM). Although the incidence of heart failure was significantly higher in the feburic group (2.7%) compared to the feuri group (0.4%), the prevalence of heart failure was similar between the two groups (30%). Other CV outcomes were also comparable. The similarity in cardiovascular outcomes between feburic and feuri is further illustrated in Figure 3, showing Kaplan-Meier survival curves (all log-rank
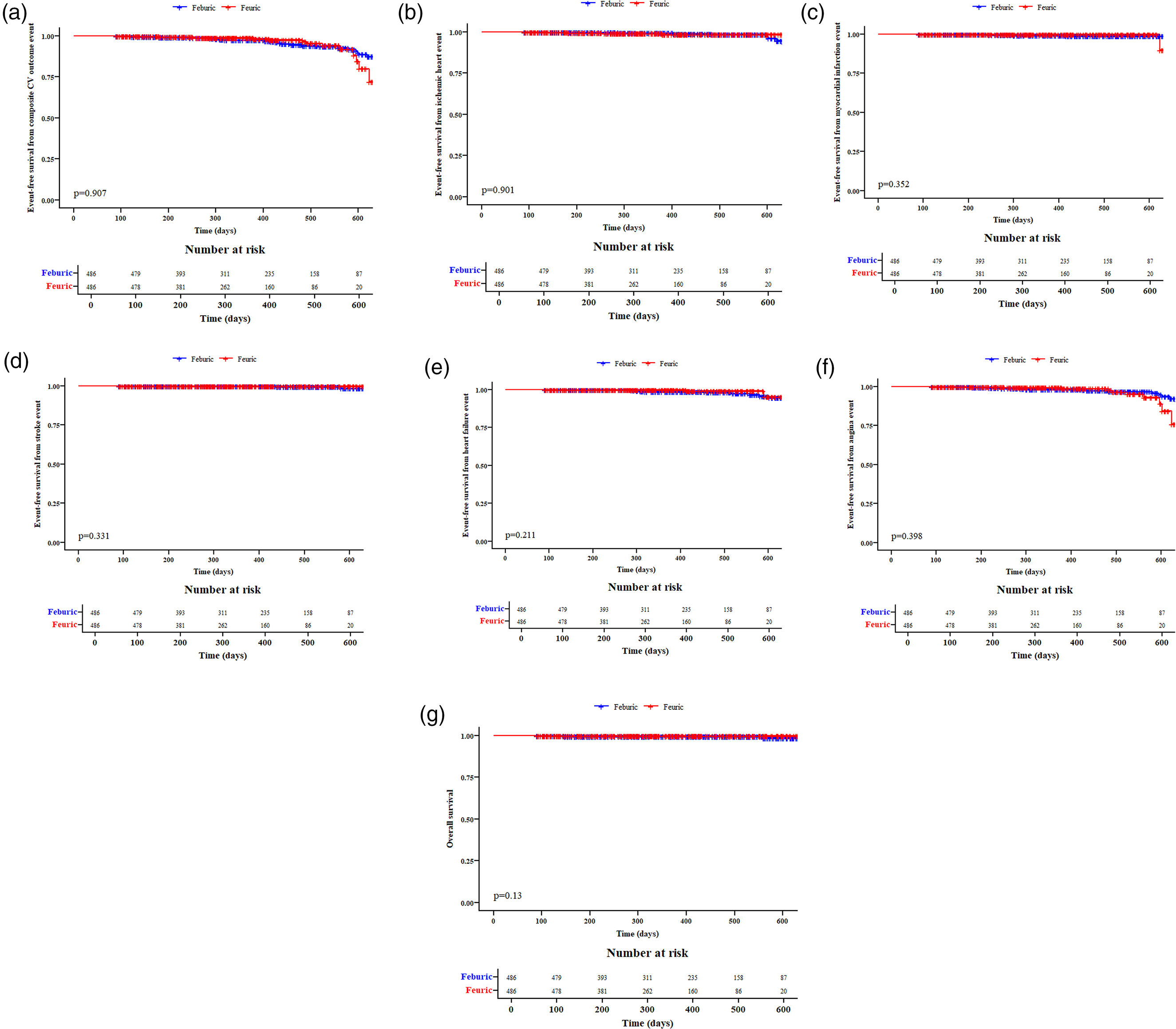
Kaplan–Meier survival curves between feburic and feuri (after Propensity Score Matching) for CV composite outcome, ischemic heart disease, myocardial infarction, stroke, heart failure, angina pectoris, and all-cause mortality.
Comparisons of all outcomes between feburic and feuri (after propensity score matching).

Renal and liver function showed no significant differences between the feburic and feuri groups (all
Discussion
This study represents a real-world comparison of the cost, effectiveness, and safety outcomes between generic febuxostat (Feuri, CYH) and brand-name febuxostat (Feburic). Overall, the two groups demonstrated comparable cardiovascular, renal, and hepatic outcomes, and no allergic reactions were observed in either group. A lower use of acetaminophen was noted among feuri users compared with feburic users. In addition, both formulations were associated with significant reductions in serum UA levels; feuri was prescribed at a lower final daily dose and was associated with lower overall medication costs. However, the absolute differences in serum UA levels were modest. Importantly, when treatment effectiveness was evaluated using target serum uric acid attainment rather than mean differences alone, the proportions of patients achieving recommended uric acid goals were similar between groups, suggesting broadly comparable clinical effectiveness in routine practice. Taken together, these findings suggest that generic febuxostat may offer effectiveness and safety outcomes comparable to those of the brand-name formulation in routine clinical practice. The generalizability of these findings may be limited, as the study population predominantly consisted of patients with impaired renal function, reflecting local reimbursement policies in Taiwan, where febuxostat is primarily reimbursed for patients with an estimated glomerular filtration rate below 45 mL/min/1.73 m2. 17
While feuri demonstrated a statistically lower mean serum uric acid level at the 3-month follow-up compared with feburic, the magnitude of this difference was modest (approximately 0.3 mg/dL). Such a difference may not be translated into clinically meaningful benefits for individual patients. To provide a more clinically relevant perspective, we additionally evaluated target uric acid attainment, which may better reflect real-world therapeutic goals than mean differences alone. Overall, our findings suggest comparable urate-lowering effectiveness between the two formulations rather than clear superiority of either agent.
An analysis 18 of National Health Insurance claims data from 2009 to 2015 showed that spending on febuxostat gradually increased from 11.61% in 2013 to 49.19% in 2015, while spending on allopurinol decreased from 29.67% to 15.12%, and on other UA-lowering agents from 58.72% to 35.69%. The number of patients treated with febuxostat also steadily increased over time. The study recommended evaluating the cost-effectiveness of febuxostat use in clinical settings. Another study, using electronic medical records from Chang-Gung Memorial Hospital for adult patients between 2010 and 2015, also showed an increase in costs following the introduction of febuxostat. 19 Balancing safety concerns (fewer severe allergic reactions with febuxostat compared to allopurinol), and the higher cost of febuxostat, prescribing feuri over feburic may be considered in clinical practice. Based on our study, the annual costs for feuri and feburic were 79.07 and 99.31 US dollars, respectively, resulting in a yearly difference of 20.25 US dollars per patient. Currently, there are approximately 600,000 gout patients in Taiwan, with nearly 50% receiving treatment with febuxostat. If feuri were used instead of feburic, it could result in an annual savings of 6 million US dollars in Taiwan.
Previous studies comparing brand-name and generic febuxostat are scarce. Existing evidence generally suggests therapeutic equivalence between formulations that meet regulatory bioequivalence standards. Our results are consistent with this literature, demonstrating similar safety profiles and clinical outcomes in a real-world setting. Currently, there are over 10 generic versions of febuxostat available worldwide. However, only one study has compared generic febuxostat (Fengdingning, Ruiyang, and Youlitong) with the brand-name version, and it showed comparable effectiveness and safety. 20 Even with the same bioequivalence, therapeutic equivalence between generic and brand-name medications is not always identical. 21 There are several possible reasons why generic medications may sometimes appear more effective than brand-name medications, including manufacturing processes, formulation differences, and individual patient variability. First, while the active ingredients in generic and brand-name medications are the same, the excipients (inactive ingredients such as fillers, binders, and lubricants) differ between feburic and feuri (Supplementary Table 4). These differences might affect how the drug is released and absorbed in the gastrointestinal tract. Some patients might respond better to the excipients in the generic medication, leading to a perception of greater effectiveness. Second, bioavailability (the rate and extent of absorption) should be similar, usually within a ± 20% range. However, small variations within this range may occur, and in some cases, the generic medication might exhibit slightly higher bioavailability in certain patients, resulting in a stronger effect. Third, different manufacturers use distinct manufacturing processes and quality control measures, which might lead to minor variations in the final product. Although these differences are typically not clinically significant, there may be rare instances where a particular batch of generic medication performs better than the brand-name counterpart. Febuxostat was approved for medical use in the European Union in 2008 22 and in the United States in 2009. 23 However, feuri was produced by Chunghwa Yuming Healthcare in Taiwan on September 5, 2016. The manufacturing process incorporated more advanced techniques, potentially resulting in improved therapeutic effectiveness. Finally, the storage and transportation conditions for medications can affect their stability and potency. If a generic medication is better preserved during these processes, it may perform better in some cases. Since the generic medication (e.g., feuri) only has bioequivalence data, it is difficult to predict which factors are associated with better therapeutic effectiveness in feuri compared to feburic. Any observed differences between formulations should be interpreted as descriptive findings from real-world prescribing patterns rather than evidence of pharmacological or manufacturing superiority. Without direct pharmacokinetic, bioequivalence, or mechanistic data, no conclusions can be drawn regarding the underlying mechanisms of these observations.
There are some limitations to this study. First, we were unable to directly evaluate gout attacks based on our medical records and could only use a surrogate outcome, namely painkiller usage. Second, the findings of this study cannot be generalized to the general population, particularly those with normal renal function. This is due to the fact that Taiwan's National Health Insurance regulations for febuxostat are primarily based on an eGFR of less than 45 mL/min/1.73 m2. Therefore, the generalizability of this study is limited to patients with moderate CKD, with a mean estimated glomerular filtration rate of approximately 41 mL/min/1.73 m2. Third, we did not assess data regarding medical regimen change. Fourth, physicians may tend to prescribe brand-name medications to sicker patients, which could introduce selection bias. However, in our study, we did not observe worse baseline conditions, even prior to PSM. Despite comprehensive adjustment using PSM, residual confounding inherent to observational studies, including confounding by indication, cannot be completely excluded. Lastly, we did not investigate the differences between febuxostat and allopurinol. Despite these limitations, our study has strengths, particularly in demonstrating that the generic medication (feuri) showed similar effectiveness and outcomes in real-world evidence, all while being more cost-effective.
Conclusion
In this real-world retrospective cohort study, generic febuxostat was associated with effectiveness and safety outcomes comparable to those of brand-name febuxostat, while being prescribed at lower doses and lower medication costs. These findings suggest that generic febuxostat may represent a reasonable and cost-conscious alternative in routine clinical practice, particularly in populations with impaired renal function, although residual confounding inherent to observational studies cannot be fully excluded.
Supplemental Material
sj-docx-1-sci-10.1177_00368504261433091 - Supplemental material for A real-world retrospective cohort study comparing brand-name febuxostat (feburic) and generic febuxostat (feuri)
Supplemental material, sj-docx-1-sci-10.1177_00368504261433091 for A real-world retrospective cohort study comparing brand-name febuxostat (feburic) and generic febuxostat (feuri) by Shang-Feng Tsai, Ming-Ju Wu and Cheng-Hsu Chen in Science Progress
Footnotes
Human ethics and consent to participate declarations
The study protocol received approval from the Institutional Review Board of Taichung Veterans General Hospital (Approval number: SE24291C). Consent for participation was waived due to the use of de-identified patient data. The study protocol adhered to the Declaration of Helsinki (1975, as revised in 2024).
Acknowledgments
With thanks to the Division of Clinical Information, Center for Department of Digital Medicine of Taichung Veterans General Hospital. This study is based in part on data from the Taichung Veterans General Hospital Research Database and managed by Clinical Informatics Research & Development Center of Taichung Veterans General Hospital (Registered number F2024306). The interpretation and conclusions contained herein do not represent those of Taichung Veterans General Hospital.
Authors’ contribution
Conceptualization, S.F.T., W.M.J. and C.H.C.; methodology, T.S.F; formal analysis, S.F.T. resources, S.F.T., W.M.J. and C.H.C..; data curation, S.F.T; writing—original draft preparation, S.F.T., W.M.J. and C.H.C, project administration, S.F.T., W.M.J. and C.H.C. funding acquisition, W.M.J. and C.H.C.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chunghwa Yuming Healthcare Co., Taiwan,
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
