Abstract
Background
Benzodiazepines (BZDs) are commonly prescribed as adjunctive drugs for patients with cardiovascular diseases (CVDs), particularly those who experience anxiety or insomnia. However, the relationship between the use of BZDs and incident risk of sudden cardiac arrest (SCA) has not been well investigated. In this study, we aimed to examine the association between the use of BZDs and the incident risk of SCA among patients with CVD.
Method
In this retrospective cohort study, a total of 74,715 eligible patients with new-onset CVD as a primary cause of hospitalization between July 2016 and August 2022 were included from the health information platform in Shenzhen, China. Among them, 61,761 BZD non-initiators were identified and matched to 12,954 BZD initiators by propensity score at a maximum ratio of 5:1. Propensity score-matched Cox proportional hazard models were used to estimate the hazard ratios (HRs) and 95% confidence intervals (CIs).
Results
Over a 12-month follow-up period, 29 (2.24 per 1000 person-years) and 137 (2.22 per 1000 person-years) SCA cases occurred among propensity score-matched BZD initiators and non-initiators, respectively. Patients who initiated BZD treatment were associated with a 101% increased risk of SCA incidence compared with patients without BZD treatment (adjusted HR: 2.01, 95% CI: 1.42, 2.83). Furthermore, compared with the non-use (0 defined daily dose, DDD), the adjusted HR was 1.43 (95% CI: 1.32, 1.56) for the BZD consumption of ≤1 DDD and 2.58 (95% CI: 2.37, 2.81) for the BZD consumption of >1 DDD (
Conclusion
This study provides evidence that BZD initiation may be associated with an increased incident risk of SCA in patients with CVD. Our finding highlights the importance of cautious prescribing BZDs in the health management of patients with CVD.
Keywords
Introduction
Sudden cardiac arrest (SCA) refers to sudden, unexpected, pulseless conditions of likely cardiac origin. 1 A SCA event can compromise the heart's ability to pump blood efficiently, leading to tissue or organ hypoxia. If the spontaneous circulation cannot be resuscitated immediately, the patient will suffer irreparable damage or death. There are at least 2 million cases of SCA reported worldwide each year, 2 which poses a huge disease and economic burden. According to the latest report on cardiac arrest and cardiopulmonary resuscitation in China, the incidence rate of SCA has been increasing over the past decade and the overall incidence rate of out-of-hospital SCA was 97.1 per 100,000 population in 2020. 3 Meanwhile, the survival rate was less than 1.2% among patients who experienced SCA outside of the hospital in China. 4 Considering the low survival likelihood of SCA patients, it is estimated that SCA accounts for over 544,000 deaths annually in China. 5 Moreover, the SCA is a severe outcome that contributes to approximately 50% of deaths among patients with cardiovascular diseases (CVDs). 6
CVD is an important public health issue in China, responsible for 4.58 million deaths in 2020. 7 Patients with CVD frequently experience symptoms such as anxiety, depression, and sleep disorders8,9 due to long-term treatment, complications, and medication side effects. Benzodiazepine (BZD) is one of the representatives of sedative-hypnotic medications and is commonly used as an adjunctive drug in the prescriptions for CVD patients, particularly for those with anxiety or insomnia. 10 Although BZDs can effectively alleviate insomnia and anxiety, some argue that they may not be suitable as first-line medications for anxiety intervention among CVD patients11,12 due to their potential for dependency and the association with respiratory depression,13,14 QT interval prolongation, increased incidence of myocardial ischemia, and worsened prognosis for patients with CVD.15–17 Moreover, the respiratory depression and QT interval prolongation caused by BZDs are also significant factors contributing to the onset of SCA.
Despite potential risk pathways connecting BZDs to SCA, there is currently limited evidence to establish a clear relationship between BZDs and the risk of SCA. To provide insights that reflect real-life experiences, we evaluated the risk of SCA incidence in patients with CVD associated with BZD use from a large retrospective cohort based on the medical registry database in Shenzhen, China.
Method
Sources of data
Shenzhen is one of China's most economically developed cities with a population of over 17 million residents. We designed a retrospective cohort study by collecting the data from the Electronic Health Records of the Shenzhen Health Information Platform,18,19 a comprehensive medical and health-related database. It covers various healthcare facilities, including hospitals, community health service centers, and public health institutions. Using this platform, we could collect participants’ (1) medical records, including outpatient and inpatient medical records, disease diagnosis and corresponding codes of International Classification of Diseases 10 (ICD-10), and prescription records; (2) death registration records; (3) resident registration. These data could be linked to each other by using encrypted, unique ID numbers.
Study population
All patients with new-onset CVD as a primary cause of hospitalization between 1 July 2016 and 31 August 2022 were studied. CVD was defined as ischemic heart disease (ICD-10: I20-I25), heart failure (I50), stroke (I61-I64, I69.1-I69.4), and related subcategories (
Exposure definition
Referencing the

Study diagram in the present study. CVD: cardiovascular disease; BZDs: benzodiazepines.
Outcome definition
The outcomes of this study were incident SCA (ICD-10: I46, I46.0, I46.1, I46.9, R96, R96.0, and R96.1; Supplementary Table S1) subsequent to the CVD diagnosis and initiation of BZD treatment. We tracked the SCA events in a medical registry-based setting, and identified the SCA events from the inpatient/outpatient medical records and death registration records by ICD-10 codes or terminologies in Chinese words with the regular expression method (Supplementary Figure S2). Patients were followed up in an intention-to-treat approach 25 from the incident date until a diagnosis of SCA, death, the end of the observation period (6/12 months), or 3 August 2022, whichever came first. We considered 6-month and 12-month observations as the principal analysis periods (Figure 1).
Covariates
Patients’ baseline characteristics at or up to six months before the incident date were analyzed, including demographic information (age, sex, ethnic groups, marriage status, education levels, occupation, and payment), medicine usages (antiplatelet agents, anticoagulant agents, antihypertensive agents, anti-arrhythmia agents, hypoglycemic agents, statins, antidepressants, and opioids; Supplementary Table S2); comorbidities (hypertension, diabetes, arrhythmia, chronic respiratory diseases (CRD), hyperlipidemia, depression, Parkinson, asthma, alcohol withdrawal, epilepsy, insomnia, and anxiety; Supplementary Table S1), and healthcare utilization (the days with prescriptions and the numbers of outpatient or hospitalization before the incident date). We calculated the Charlson comorbidity index 26 (Supplementary Table S3) to reflect comorbidity condition for each participant, which had been validated for the Chinese population and utilized in previous studies.27,28
Statistical analysis
We cross-tabulated patients’ baseline characteristics between those who started or did not start BZD treatment and calculated the events of outcomes and incidence rates. To address potential biases caused by lack of randomization, we matched the BZD non-initiators to BZD initiators by using the propensity scores at a maximum ratio of 5:1. The propensity score matching (PSM) took into account age (±3 years), sex, ethnic groups, marriage status, education levels, occupation, payment, and prevalent comorbidities like ischemic heart disease, heart failure, stroke, arrhythmia, hypertension, diabetes, CRD, hyperlipemia, depression, Parkinson, asthma, alcohol withdrawal, epilepsy, insomnia, and anxiety. The BZD non-initiators were selected without replacement using the nearest neighbor matching algorithm with propensity scores calculated by logistic regressions, and a caliper was set to 0.10. Eventually, 12,954 BZD initiators and 61,761 non-initiators were paired and included in the present study. We calculated the standardized mean difference to assess the balance between the two groups before or after PSM. If the standardized mean difference was less than 0.10, we could consider the two groups to be comparable. 29
Propensity score-matched Kapan-Meier method and log-rank test were used to fit and compare the survival curves of initiators and non-initiators groups. We employed the propensity score-matched Cox regression models (i.e. stratified Cox models) to evaluate the association between BZD usage and the incident risk of SCA. The hazard ratios (HRs) and 95% confidence intervals (CIs) were estimated by three separate sets of models. Crude model was fit without any adjustments. Model 1 was adjusted for age, sex, ethnic groups, marriage status, education levels, occupation, and payment; Based on Model 1, the prevalent comorbidities (ischemic heart disease, heart failure, stroke, arrhythmia, hypertension, diabetes, CRD, hyperlipemia, depression, Parkinson, asthma, alcohol withdrawal, epilepsy, insomnia, and anxiety) and the healthcare utilization indicators (the days with prescriptions and the numbers of outpatient or hospitalization before the incident date) were further adjusted in Model 2. The World Health Organization established the defined daily dose (DDD) system (see the ATC/DDD Index, http://www.whocc.no/atc_ddd_index/) as a standardized metric for drug utilization studies, enabling horizontal comparisons of drug consumption. In our study, we assessed the utilization of various BZDs within 14 days after the incident date, using DDD as the measurement.30,31 As a result, we considered three categories of drug consumption: 0 DDD, ≤1 DDD, and >1 DDD. We chose the 1 DDD as the nodal point because low-dosage use of BZDs was more common in this study. Additionally, we performed tests for linear trend by modeling the ordered categories of drug consumption as a one degree-of-freedom linear term.
We performed a set of subgroup analyses stratified by age (≤65/>65 years old), sex (men/women), drug groups defined by acting time (short/long-acting agents; Supplementary Table S2), ischemic heart disease (yes/no), heart failure (yes/no), and stroke (yes/no) within 12 months of follow-up period. And the likelihood-ratio test was used to assess the interactions between treatment effects and subgroups. We selected this period because the effect of BZD usage tended to be more stable in our study or statistically significant in a previous study. 24 Several sensitivity analyses were conducted: (1) further adjusting medicine usages based on Model 2; (2) extending the observation periods to 24 months of follow-up; (3) excluding patients who experienced SCA within 14 days after the incident date to avoid potential reverse causality; (4) considering the possibility of “Drugs that are prescribed are not necessarily dispensed, and drugs that are dispensed are not necessarily ingested,” 32 we excluded patients who started BZD treatment outside of the hospitalization period, because only the prescriptions during hospitalization were known to be strictly adhered to; (5) revising the matching factors to avoid potential over-matching, we matched the BZD non-initiators to initiators at a maximum ratio of 5:1, and the factors encompassed age (±3 years), sex, and prevalent comorbidities closely related to BZD treatment including alcohol withdrawal, epilepsy, insomnia, and anxiety; (6) matching the healthcare utilization indicators further to ensure a more rigorous equality in healthcare utilization between the two groups; (7) excluding patients who experienced SCA resulting from accidents (Supplementary Table S1). Given that SCA occurring within a 14-day period after the incident date was more likely to be caused by complications related to CVDs or other etiologies, we compared the number and cumulative incidence rate of SCA within this period between BZD initiators and non-initiators using chi-square and log-rank tests, respectively.
All the analyses were performed using R software (version 4.1.2, R Foundation for Statistical Computing, Vienna, Austria; https://cran.r-project.org/). Statistical tests were performed using a two-sided approach, and a
Results
Patient characteristics
A total of 158,585 eligible subjects were identified in this study, including 13,023 BZD initiators and 145,562 BZD non-initiators. Before PSM, BZD initiators and BZD non-initiators were mainly men, accounting for 54.3% and 61.0%, respectively, and their average ages were 55.3 (standard deviation: 15.1) and 52.7 (15.3) years old, respectively. Compared to non-initiators, BZD initiators tended to have a higher proportion of Han ethnicity, no partner, higher education, technical professionals, and basic medical insurance. Moreover, BZD initiators had higher burden of comorbidities, especially in conditions such as alcohol withdrawal, insomnia, and anxiety. And they were also more likely to use other medications (Supplementary Table S4). Furthermore, BZD initiators were more likely to have a higher number of days with prescriptions and a greater frequency of outpatient visits or hospitalizations before the incident date (Table 1). After PSM, the two groups were comparable (most standardized mean difference <0.10). And a total of 12,954 BZD initiators and 61,761 non-initiators were included in the primary analysis, with a mean age of 55.3 (15.1) and 54.9 (15.1) years old, respectively (Table 1).
Baseline characteristics before and after propensity score matching.
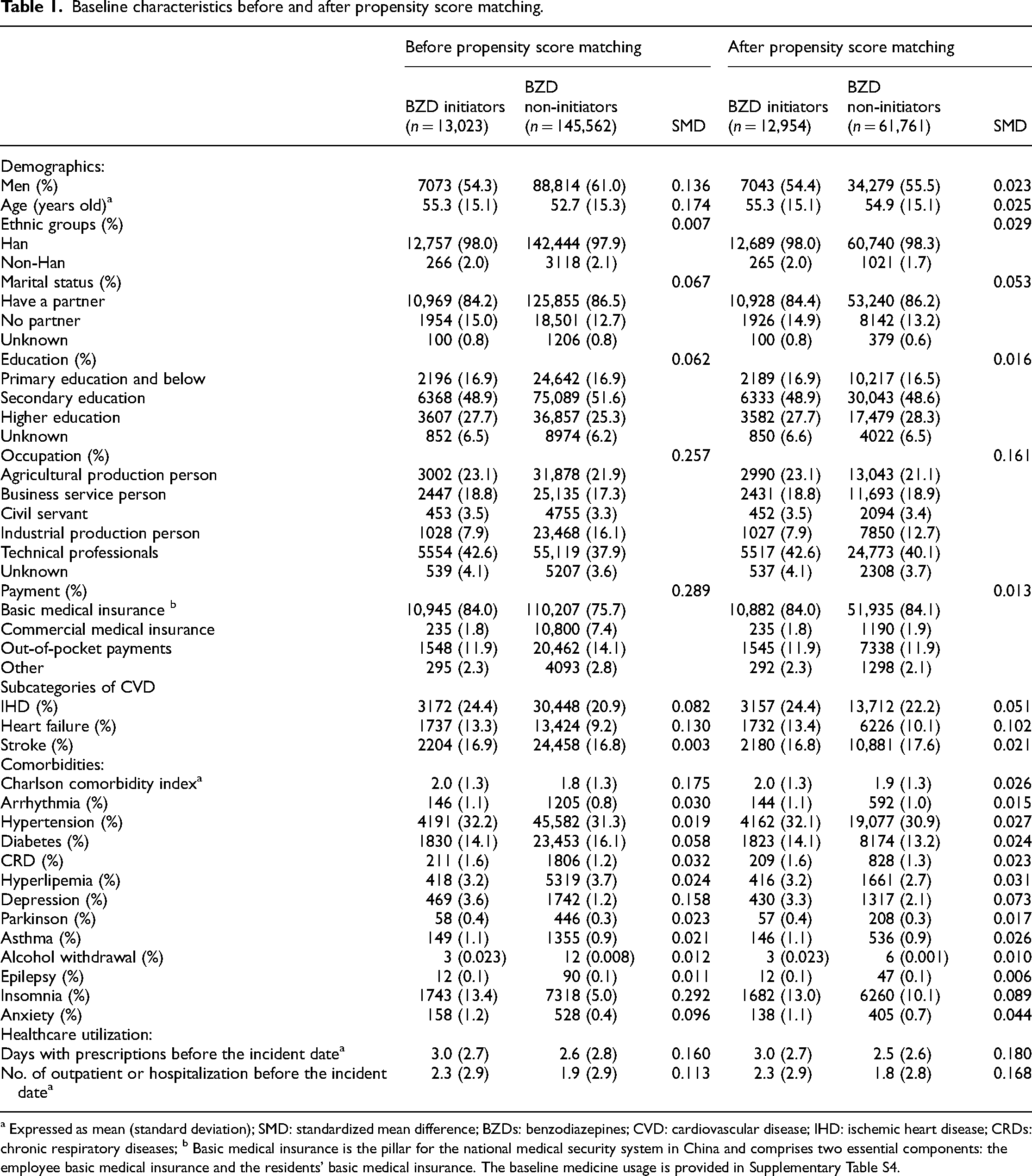
a Expressed as mean (standard deviation); SMD: standardized mean difference; BZDs: benzodiazepines; CVD: cardiovascular disease; IHD: ischemic heart disease; CRDs: chronic respiratory diseases; b Basic medical insurance is the pillar for the national medical security system in China and comprises two essential components: the employee basic medical insurance and the residents’ basic medical insurance. The baseline medicine usage is provided in Supplementary Table S4.
Short-acting BZDs were prescribed more frequently than long-acting ones, accounting for 94.6% of all BZD prescriptions. Among these, Alprazolam (40.9%), Midazolam (37.2%), and Estazolam (15.2%) emerged as the three most commonly prescribed medications (Supplementary Figure S3).
Association between BZD initiation with incident risk of SCA
After PSM, the number of SCA events was 24 (3.76 per 1000 person-years) and 93 (3.02 per 1000 person-years) within a 6-month follow-up, and 29 (2.24 per 1000 person-years) and 137 (2.22 per 1000 person-years) within a 12-month follow-up among BZD initiators and non-initiators groups, respectively (Supplementary Table S5). The results from different models consistently showed adverse associations between BZD initiation and increased incident risks of SCA. In the full model (i.e. Model 2), the BZD initiators were linked to a 199% increased incident risk of SCA within a 6-month follow-up (adjusted HR: 2.99, 95% CI: 2.03, 4.41), compared with BZD non-initiators. When extending the follow-up period to 12 months, the association remained stable, with an adjusted HR of 2.01 (95% CI: 1.42, 2.83; Table 2). Moreover, propensity score-matched Kaplan-Meier curves comparing the survival probability between two groups were consistent with the primary findings (Figure 2). We further estimated the potential impact of the consumption of BZDs on the risk of SCA incidence among all study participants. Compared to the no BZD consumption (0 DDD), the HR of incident risk of SCA was 2.64 (95% CI: 2.31, 3.02) for the BZD consumption of ≤1 DDD and 2.90 (95% CI: 2.52, 3.33) for the BZD consumption of >1 DDD, with a

Propensity score matched Kaplan–Meier curves of survival probability since start of follow-up, and the number of patients at risk. BZDs: benzodiazepines.

Association between the duration of benzodiazepines (BZDs) use with the incident risk of sudden cardiac arrest after propensity score matching. HR: hazard ratio; 95% CI: 95% confidence interval; Adjusted: adjusted for age, sex, ethnic groups, marital status, education levels, occupation, payment, arrhythmia, hypertension, diabetes, chronic respiratory diseases, hyperlipemia, depression, Parkinson, asthma, alcohol withdrawal, epilepsy, insomnia, anxiety, days with prescriptions, and numbers of outpatient or hospitalization; **
Risk of incident sudden cardiac arrest associated with BZD initiators versus BZD non-initiators within 6/12 months of follow-up period after PSM.

Reference to BZD non-initiators, PSM: propensity score matched; HR: hazard ratio; 95% CI: 95% confidence interval; Model1: adjusted for age, sex, ethnic groups, marital status, education levels, occupation, and payment; Model 2: Model 1+ adjusted for arrhythmia, hypertension, diabetes, chronic respiratory diseases, hyperlipemia, depression, Parkinson, asthma, alcohol withdrawal, epilepsy, insomnia, anxiety, days with prescriptions, and numbers of outpatient or hospitalization; *
Supplementary analysis
During a 12-month follow-up period, the association of BZD use with the incident risk of SCA reached the statistically significant level in some subgroups, with this association being more pronounced in women patients compared to men (

Stratified analysis of the incident risk of sudden cardiac arrest associated with BZD initiators versus BZD non-initiators after propensity score matched during 12-month follow-up. BZDs: benzodiazepines; IHD: ischemic heart disease; HR: hazard ratio; adjusted, adjusted for age, sex, ethnic groups, marital status, education levels, occupation, payment, arrhythmia, hypertension, diabetes, chronic respiratory diseases, hyperlipemia, depression, Parkinson, asthma, alcohol withdrawal, epilepsy, insomnia, anxiety, days with prescriptions, and numbers of outpatient or hospitalization; the hazard ratio could not be estimated in the subgroup of long-acting by using the propensity score matched Cox regression model, due to the limited sample sizes. In the long-acting and short-acting subgroups, BZD initiators were compared with their matched BZD non-initiators. The event numbers (total numbers) for matched BZD non-initiators in the long-acting and short-acting subgroups were 14 (3279) and 123 (58,482), respectively. *
Discussion
In the retrospective cohort study of over 74,000 CVD patients from a large medical registry database in Shenzhen, China, we presented novel evidence regarding the negative impact of the BZD initiation on the risk of SCA incidence. Specifically, patients who initiated BZDs were associated with a 199% increased risk of SCA incidence within the 6-month follow-up and a 101% increased risk within the 12-month follow-up. Furthermore, as compared with non-use (0 DDD), the risks of SCA incidence seemed to increase gradually with BZD consumption from ≤1 DDD to >1 DDD within the 12-month follow-up (
BZDs are widely prescribed psychotropic drugs in developed countries such as the USA and Canada.33,34 Previous studies have extensively explored the increased risk of SCA incidence associated with certain psychotropic medications, such as antidepressants and antipsychotics. 35 Serge Sicouri et al. 36 extensively reviewed the potential mechanisms underlying SCA associated with antidepressants and antipsychotic drugs. This review indicated that drug-induced long QT syndrome, torsade de pointes, and the Brugada syndrome phenotype may contribute to the high risk. However, given the variations in pharmacological mechanisms and target receptors, direct comparisons of our findings with those of other studies on antidepressants and antipsychotic drugs were not feasible, despite these medications belonging to the same category of psychotropic drugs as BZDs. Therefore, to our knowledge, this study provided a novel comprehensive report on the detrimental effect of initiating BZDs on the risk of SCA incidence in patients with CVD.
The association between BZDs and adverse cardiovascular events and overall mortality had been widely reported. In a study comprising a total of Dutch 4837 post-myocardial infarction patients, Shengxin et al.
37
found that BZDRs (benzodiazepine and drugs related to benzodiazepine) use was related to a higher risk of CVD mortality (adjusted HR: 1.43, 95% CI: 1.14, 1.81) or all-cause mortality (adjusted HR: 1.31, 95% CI: 1.14, 1.52), and these relationships were dose-dependent (
The mechanisms underlying the effect of BZDs on the risk of SCA in patients with CVD are not well established. However, it is hypothesized that BZDs may increase the risk of SCA incidence through two main pathophysiological pathways: respiratory depression
41
and arrhythmia.
42
BZDs exert their effects by binding to γ-amino-butyric acid (GABA) type A receptors in the central nervous system, which play a key role in the negative regulation of rhythmic respiration.
42
In addition, as a neurotransmitter, BZDs may also have a direct inhibitory effect on respiration. Evidence from a randomized-controlled, double-blind trial revealed that Flurazepam, a type of BZDs, was associated with significant increases in the frequency and the total duration of apnea episodes.
13
Other studies corroborated these findings by demonstrating a meaningful dose–response relationship between BZD use and the central apnea index (coefficient = 0.247,
Our study had several strengths. Insomnia and anxiety were the primary indications for BZD treatment, making them essential factors that could not be ignored in observational studies on BZDs. In contrast to prior studies,37–39 using the health information platform, we were able to obtain diagnoses of insomnia or anxiety from inpatient and outpatient settings, which to some extent reflected the presence of insomnia or anxiety in the present study population. Then, we employed PSM to match the BZD non-initiators with initiators with similar baseline characteristics, including insomnia and anxiety. After PSM, the distribution of insomnia and anxiety tended to be balanced between these two groups, with standardized mean differences of 0.089 and 0.044, respectively. This approach was consistent with the study conducted by Elisabetta et al., 24 where similar factors and matching techniques were employed. Furthermore, other indications for BZD prescription, including alcohol withdrawal and epilepsy, were also taken into consideration and addressed in this study. Finally, in China, BZDs were classified as Schedule II psychotropic drugs and were subject to rigorous regulation and supervision. This stringent oversight helped to reduce the possibility of patients obtaining BZDs through unregulated channels, such as self-purchasing from pharmacies. As a result, the utilization of BZDs as reflected in prescription records tended to provide a more accurate representation of the actual usage scenario.
Several limitations remain in the present study. Firstly, although this health information database allows us to track nearly recorded cases of SCA, it is challenging to trace unrecorded out-of-hospital SCA events. Consequently, the actual incidence rate of SCA in this context may be underestimated. Secondly, it is essential to acknowledge the potential for misclassification of SCA due to variations in diagnostic capabilities among different healthcare facilities. Thirdly, although our results were relatively robust by conducting PSM and a series of sensitivity and additional analyses, the residual confounding and potential immortal time bias were still possible. Fourthly, additional lifestyle data such as smoking and alcohol consumption, were not considered in the present study since they were not collected in the database we used currently. Finally, the findings were based solely on the population in Shenzhen, China, it should be a caution when extrapolated to other regions or countries, especially outside of China.
Conclusion
In the present propensity-score matched, retrospective cohort study, we observed that the use of BZDs may be associated with an increased incident risk of SCA in patients with CVD. This finding may emphasize the importance of cautious prescribing BZDs in the health management of patients with CVD.
Supplemental Material
sj-docx-1-sci-10.1177_00368504241295325 - Supplemental material for Benzodiazepine use and incident risk of sudden cardiac arrest in patients with cardiovascular diseases
Supplemental material, sj-docx-1-sci-10.1177_00368504241295325 for Benzodiazepine use and incident risk of sudden cardiac arrest in patients with cardiovascular diseases by Chunbao Mo, Shuang Wang, Xia Li, Furong Li, Cheng Jin, Bo Bai, Haolong Pei, Jing Zheng and Fengchao Liang in Science Progress
Supplemental Material
sj-docx-3-sci-10.1177_00368504241295325 - Supplemental material for Benzodiazepine use and incident risk of sudden cardiac arrest in patients with cardiovascular diseases
Supplemental material, sj-docx-3-sci-10.1177_00368504241295325 for Benzodiazepine use and incident risk of sudden cardiac arrest in patients with cardiovascular diseases by Chunbao Mo, Shuang Wang, Xia Li, Furong Li, Cheng Jin, Bo Bai, Haolong Pei, Jing Zheng and Fengchao Liang in Science Progress
Footnotes
Acknowledgments
We express our sincere gratitude to Prof. Dongfeng Gu from Southern University of Science and Technology for his invaluable support and guidance throughout this research endeavor. Additionally, we extend our appreciation to Zhiyuan Cheng, Jian Liao, and Keye Wu from the same institution for their valuable contributions in providing insightful advice during the writing process and meticulously reviewing the manuscript.
Author contributions
Fengchao Liang and Jing Zheng directed the study. Chunbao Mo analyzed the data and wrote the manuscript. Fengchao Liang, Jing Zheng, Shuang Wang, Xia Li, Furong Li, Cheng Jin, Bo Bai, and Haolong Pei contributed to the discussion and reviewed/edited the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the R&D Project of Pazhou Lab (Huangpu; Grant No. 2023K0610), the National Natural Science Foundation of China (Grant Nos. 82030102, 12126602, 42107465), the Shenzhen Medical Academy of Research and Translation (Grant No. C2302001), and the Ministry of Science and Technology of China (Grant No. 2022YFC3702703)..
Ethical approval
This study complied with the Helsinki Declaration of 1975 and was approved by the ethical review committee of the Southern University of Science and Technology (No. 20210067) on May 10, 2021, in Shenzhen, China. And all patient details were de-identified.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
