Abstract
X-ray computed tomography (CT) and magnetic resonance (MR) imaging are essential tools in modern medical diagnosis and treatment. However, traditional contrast agents are inadequate in the diagnosis of various health conditions. Consequently, the development of targeted nano-contrast agents has become a crucial area of focus in the development of medical image-enhancing contrast agents. To fully understand the current development of nano-contrast agents, this review provides an overview of the preparation methods and research advancements in CT nano-contrast agents, MR nano-contrast agents, and CT/MR multimodal nano-contrast agents described in previous publications. Due to the physicochemical properties of nanomaterials, such as self-assembly and surface modifiability, these specific nano-contrast agents can greatly improve the targeting of lesions through various preparation methods and clearly highlight the distinction between lesions and normal tissues in both CT and MR. As a result, they have the potential to be used in the early stages of disease to improve diagnostic capacity and level in medical imaging.
Introduction
With the development of medical technology, imaging has become increasingly important in the diagnosis and treatment of various diseases of the human body. The use of imaging techniques, such as X-ray, ultrasound (US), computed tomography (CT), magnetic resonance (MR), and nuclear medicine imaging, has increased, with CT and MR being the most commonly used. These techniques reliably aid in the definitive diagnosis, treatment, and follow-up of many diseases.1,2 For the differential diagnosis of various diseases, targeted image enhancement processing is necessary to improve the image resolution and diagnostic accuracy.
Contrast agents, the most crucial medical image-enhancement methods, are required to achieve targeted image-enhancement processing of the target area. Traditional contrast agents are mostly small molecules that have several disadvantages, such as rapid metabolism and poor targeting in human circulation and tumors, limiting their clinical efficacy. 3 Nano-formulations have garnered significant attention in basic research into disease diagnosis and treatment owing to their unique properties, including enhanced permeability and retention (EPR) in solid tumors and surface modifiability.4–47 Research into the development of more targeted nano-enhanced contrast agents aims to combine the potential advantages of nanomaterials to overcome the shortcomings of traditional small-molecule contrast agents, such as rapid metabolism and poor targeting. Improvement of the diagnostic ability and level of medical imaging has become an important research area in the preparation of medical imaging-enhanced contrast agents. This review aims to systematically describe recent advances in nanotechnology research into CT- and MR-targeted enhancement contrast agents to act as a reference for further research. Figure 1 shows the multiple nanomaterials used in CT and MR contrast agents, several of which are described in the following sections.
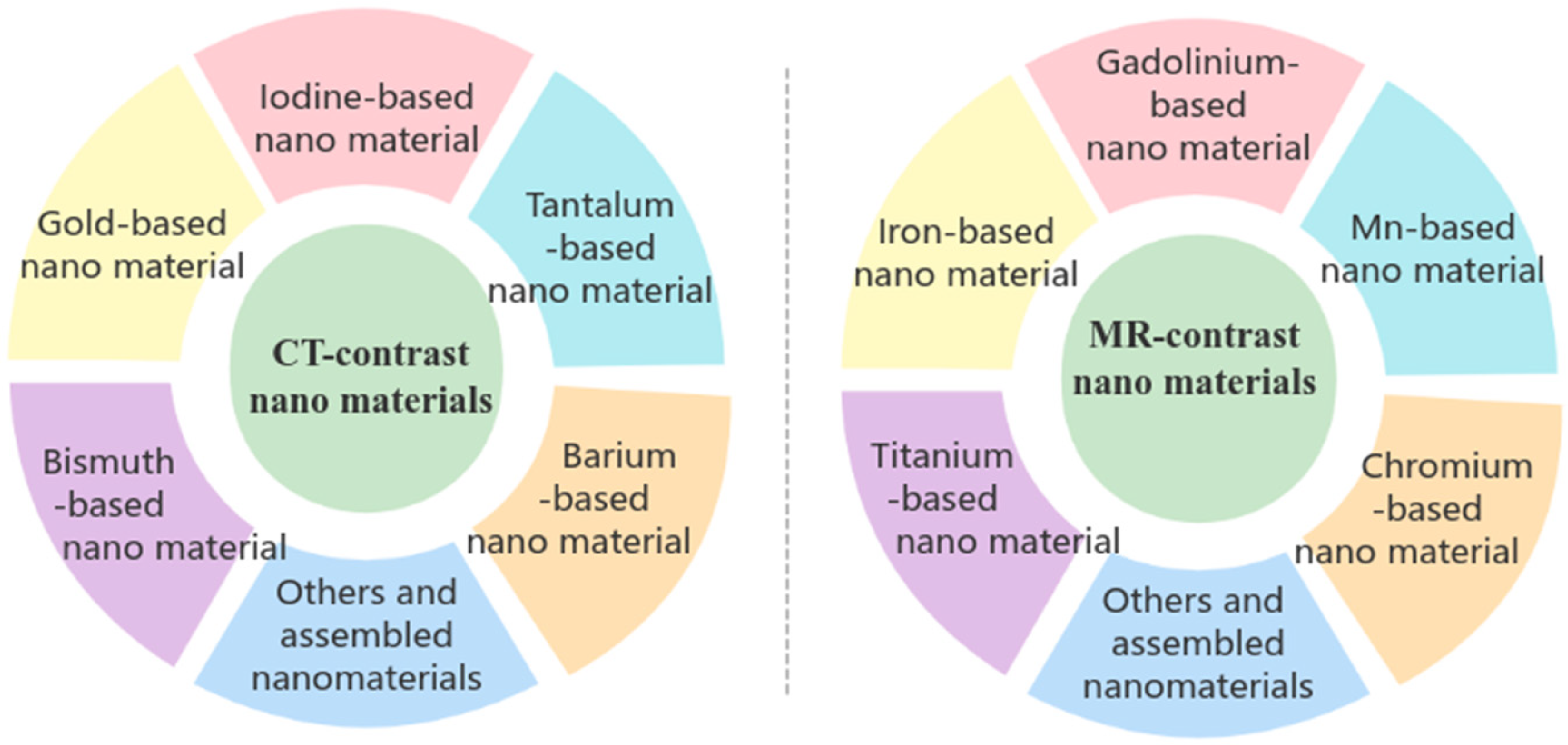
Multiple nanomaterials used in CT and MR contrast agents.
Nano-contrast agents in CT
A CT scanner is a tomographic imaging device for the human body that uses X-ray imaging. It provides high contrast for tissues and structures with large density differences but poor contrast for soft tissues because of the small differences in X-ray absorption coefficients. Contrast agents are often administered orally or via injection during CT scans to enhance the imaging effects and strengthen the identification of soft tissue abnormalities, which, by increasing the contrast between tumor or lesion tissues and normal tissues, results in high-contrast CT images that can significantly improve disease diagnosis.
Currently, the X-ray contrast agents commonly employed in clinical applications and research include high-atomic-number elements, such as IO, Au, barium, bismuth, and tantalum. Among them, IO-containing small molecules have garnered great interest and have been used in clinical settings. However, small molecules containing IO are quickly excreted by the kidneys, resulting in a brief contrast duration and poor targeting. Additionally, large doses of these molecules can cause serious adverse reactions and allergies in some patients. 4 Ideal CT contrast agents have five characteristics: high structural stability, good biocompatibility, high water solubility, strong targeting, and controlled retention, and slow metabolism. High structural stability, biocompatibility, and water solubility are essential for effective drug preparation. Strong targeting, controllable retention, and slow metabolism allow the contrast agent to remain for the anticipated duration at the target site, thereby significantly improving diagnostic sensitivity and efficiency. Processing high-atomic-number materials to the nanoscale, characterizing and modifying these materials for different anatomical sites and diseases, and preparing nano-targeted reagents to achieve these five properties are now the primary focus of research into nano-CT contrast agents.8–27
Iodine nanometer Ct contrast agents
Nanoscale iodinated contrast agents, such as micelles, polymers, and liposomes, have been increasingly developed based on their EPR in solid tumors in nano-formulations and surface modifiability to meet clinical needs for CT diagnostic contrast agents.8–12
Iodine nanoparticles (NPs) offer numerous research opportunities, one of which is the development of iodine nanoliposome contrast agents. Iohexol and iodixanol are formed by polyethylene glycol (PEG)-type liposome encapsulation. When liposomes approximately 100 nm in size are generated, they can notably increase the rate of blood circulation in mice. Additionally, they can reduce the phagocytosis rate of liposomes by macrophages, making them highly effective for the imaging of cardiac and vascular structures. 13 Iodine nanoliposome contrast agents are eliminated from the body via a pathway that favors the reticuloendothelial system over glomerular filtration, thereby reducing nephrotoxicity. In addition to the physical encapsulation approach described above, the introduction of iodine atoms into amphiphilic molecules, which is necessary for the formation of liposome-like lipid bilayer membranes, is another potential method of fabrication. In liposome-type CT contrast agents featuring a phospholipid bilayer structure comprising amphiphilic monomers, iodine molecules are integrated via covalent bonds, and a cavity in the interior of liposome enables the loading of additional drugs. Figure 2 illustrates the structure and imaging of this nanoliposome. 14

(A) Nanoliposomes that co-encapsulate CT contrast agents and photosensitizers. (B) Transmission electron microscopy images of TPPS4 in NL (LT) and NL that co-encapsulate iodixanol and TPPS4 (LIT). (C) Three-dimensional volume-rendered images of nude mice with HeLa tumor xenografts injected with free iodixanol or NL-co-encapsulated iodixanol and TPPS4 (LIT) into the tail vein. The location of the tumor is indicated by arrows. Reproduced with open access and meets Creative Commons protocols from Ref. 14 ©Theranostics, 2019.
Research into the use of emulsified nano IO-containing non-water-soluble contrast agents is a crucial area in the study of iodine nano-contrast agents. Surfactants allow the encapsulation of non-water-soluble colloidal dispersions to form nanosuspensions, a technique that has been successfully used for imaging the cardiovascular system of rabbits and lung tissues in mice.15,16 In the preparation of non-water-soluble contrast agents containing iodine, there is the possibility of iodine leakage or loss during NP synthesis using various methods. To reduce this risk, iodine can be incorporated into macromolecular particles via direct polymerization. In one study, the physical form of P(MAOETIB-GMA) copolymers synthesized with an average particle size ranging from 30 to 50 nm via emulsion polymerization to serve as CT contrast agents against macrophages remained stable. 17 A range of preparation methods has enabled us to design and produce distinctive nano-type CT contrast agents containing iodine that meet particular specifications.
Gold nano-CT contrast agents
Gold (Au) nanomaterials have been extensively studied and used in the biomedical field owing to their chemical inertness, oxidation resistance, and excellent biocompatibility. 18 Au has a higher X-ray absorption capacity and provides a contrast effect per unit mass approximately 2.7 times better than IO. To improve the stability and solubility of Au nano-contrast agents, researchers have utilized different surface modification techniques for Au NPs and analyzed their contrast effects. Commonly used stabilizers are amine and carboxylate compounds, sulfur compounds, and specific polymers. In a study by Kim et al., 19 use of mono-poly(ethylene glycol)-encapsulated Au NPs resulted in clear CT images of subcutaneous tumors in mice. When Kattumuri et al. 20 utilized Au NPs encapsulated in natural gum Arabic (GA-AuNPs), they observed their high biocompatibility and stability, as indicated by the stability of the Au content remaining stable within 72 h of injection.
Research into modifying stabilizers as well as surface molecules is an essential area of research into Au nanomaterials. Preparation efficiency is also important in nano-agent research. When Li et al. 21 adapted the surface of Au NPs with two thiol-binding molecules while considering the acidic microenvironment of tumor tissue, they found that this modification enabled the Au NPs to remain stable at the normal blood and tissue pH while leading to aggregation of the NPs outside of acidic cells, achieving pH-controlled-induced aggregation of the Au NPs. When Gomes et al. 22 used UV irradiation to accelerate the reaction for the synthesis of Au NP–Pluronic nanocomposites, they found that the use of UV irradiation at 254 nm resulted in a 10-fold increase in the preparation rate of the nanocomposites. They also observed that the dispersion sizes were smaller than those obtained using the control method under ambient light. In vitro experiments have subsequently confirmed the potential of Au as a contrast agent in enhanced X-ray CT imaging for clinical use. Figure 3 shows the in vitro imaging results.

The CT images in (A) show the contrast agents AuNP-PLU:2.0UV and optiray 3201, both with a concentration of 1.23 mg/mL of gold and iodine; AuNP-PLU:2.0, with a concentration of 1.09 mg/mL of Au; and Optiray 3202, with a concentration of 1.09 mg/mL of iodine. The measurements were obtained for beam energies of 80, 100, 120, and 140 KV. (B) Variations in CT values with beam energy for commercial IO-containing contrast agent Optiray 320 and photochemically produced Au-PLU:2.0UV. Reproduced with open access and meets Creative Commons protocols from Ref. 22. ©Polymers (Basel), 2023.
Au is a strong contrast element that is used for X-ray absorption. Current research into Au nano-contrast agents focuses on compounding or modifying stabilizers and various types of small molecules to improve the stability and biocompatibility of the agents or to enable targeted and multimodal imaging functions. However, as Au NPs may accumulate in vivo, ensuring biosafety remains a critical challenge for the clinical utilization of AU nano-contrast agents.
Other metallic nano-Ct contrast agents
In addition to Au NPs, tantalum, tungsten, bismuth, and other metals have received increasing attention as CT contrast agents. Hyeon et al. 23 modified silane derivatives on the surface of tantalum oxide NPs and attached fluorescent molecules and PEG to provide dual-mode CT and fluorescence imaging. Similarly, Zha et al. 24 utilized liquid-phase dispersive polymerization to produce polypyrrole NPs containing tantalum oxide. These NPs can aggregate in the blood flow-rich region of tumors, providing a good CT contrast effect and absorbing near-infrared light for photoacoustic imaging of deep tissues. The ability of tungsten oxide NPs to attenuate X-rays is significantly greater than that of IO NPs. When Jakhmola et al. 25 modified tungsten oxide NPs with biodegradable caprolactones to stabilize them under physiological conditions, they achieved significant CT-enhanced imaging results with low concentrations of tungsten oxide NPs. Bismuth (Bi) has several attractive properties, such as a relatively low cost, high atomic number (83), and high X-ray attenuation coefficient, making it a candidate material for CT contrast agents. In previous studies, Rabin et al. 26 used ethylene- and alkanone-coated bismuth(III) sulfide (Bi2S3) for vascular CT imaging in mice. When Perera et al. 27 used KBi(H:O)2(Fe(CN)6) as a substitute for Bi2S3, they observed enhanced stability and reduced cytotoxicity by means of ethylene and alkanone surface modification.
Other metallic materials, such as barium, holmium, gadolinium, rhenium, and ytterbium, have also been extensively studied as CT imaging agents at the nanoscale level. However, most of these metals do not occur naturally in the human body, and the mechanisms of their degradation, metabolism, and excretion are not well understood in current research. Therefore, a thorough investigation of the biocompatibility of these nano-contrast agents is necessary.
Nano-contrast agents in Mr
Magnetic resonance imaging (MRI) is an imaging method that uses atomic nuclei to resonate in a powerful magnetic field to generate a signal that is reconstructed into an image. MRI has become a vital tool for contemporary medical diagnosis because of its excellent soft-tissue resolving capacity and multiparameter characteristics. During MRI examinations, exogenous substances, such as contrast agents, may be necessary to enhance the image quality. After injection into the veins, MRI contrast agents circulate throughout the body. These agents can be categorized into three types based on their magnetic sensitivity—paramagnetic chelates, superparamagnetic particles, and ferromagnetic particles—that have different effects on T1 and T2 imaging sequences.
Paramagnetic chelates, such as gadopentetates, can decrease T1 relaxation time, resulting in a high T1 signal. In contrast, superparamagnetic and ferromagnetic particles can shorten the T2 relaxation time, thereby producing a low T2 signal. The most frequently used contrast agent for MRI in clinical settings is gadodiamide, which has been favored by clinics and imaging departments since its introduction in 1988. However, some healthy individuals report experiencing head discomfort after using gadolinium (Gd)-based contrast agents. 28 Moreover, gadodiamide preparation is complex and lacks targeting, and free Gd3+ is associated with an elevated risk of developing nephrogenic systemic fibrosis (NSF). 29 Owing to these concerns, as well as the unique physicochemical properties of nanomaterials, such as EPR and surface modifiability, the development of functional nano-MRI contrast agents has become a crucial focus of clinical and foundational research.
Gadolinium-based paramagnetic nano-MRI contrast agents
In MRI, gadolinium elements use their strong paramagnetism (dipole-dipole interaction) to alter the relaxation of surrounding protons, significantly shortening T1 and T2 relaxation times and thus increasing the contrast between tissues, with the most obvious change being in the T1-weighted imaging signal. Although Gd chelates are currently the most widely used T1 contrast agents for MRI, conventional Gd chelates have inadequate relaxivity, which hinders their efficacy in clinical diagnostics. The creation of nanosized Gd-based T1 contrast agents, such as gadolinium chelates loaded into nanocarriers, offers a practical approach to improving the T1 contrast capability of Gd chelates. Xu et al. 30 utilized a reverse microemulsion technique for the coating of iron oxide (IO) with IO@SiO2 followed by the loading of Gd chelates onto the surface of silica-coated IO NPs, ultimately leading to the creation of Gd-based silica-coated magnetite (IO@SiO2-DTPA-Gd) NPs. They found that the resulting contrast agent demonstrated high T1 contrast ability, indicating that these nanostructures may create new avenues for the production of high-performance T1 contrast agents. Figure 4 illustrates the structure and in vivo imaging effects of their experiment.

(1) synthesis of IO@SiO2-DTPA-Gd nanoparticles (NPs). (1A) Schematic cartoon illustrates the synthesis of IO@SiO2-DTPA-Gd NPs. Transmission electron microscopy images of (1B) iron oxide core, (1C) IO@SiO2, and (1D) IO@SiO2-DTPA-Gd NPs. (2) In vivo MRI. (2A) T1-weighted MR images of the tumor were captured at various intervals (15, 30, 45, 60, and 120 min) post-intravenous introduction of IO@SiO2-DTPA-Gd NPs. (2B) Signal-to-noise ratio changes in the images were observed during these different time points. Reproduced with permission from Ref. 30. ©International Journal of Nanomedicine, 2019. Originally published by and used with permission from Dove Medical Press Ltd.
Despite these promising results, the build-up of contrast agents in body tissues at the nanoscale level can lead to negative reactions linked to the liberation of free Gd ions from nanoscale systems, including nephrogenic systemic fibrosis and gadolinium deposition in brain tissues. To address this issue, Guo et al. 31 proposed a new formulation strategy for Gd-based macromolecular MRI contrast agents (Gd-mCAs) that significantly reduces Gd3+ deposition in the human body while maintaining adequate imaging contrast. Although MRI contrast agents with particle sizes of 3–10 nm can be selectively absorbed by lymphatic vessels and filtered in the kidneys for final excretion, no Gd-complex-based MRI contrast agents are available in this range for selective imaging of the lymphatic system. Yano et al. 32 succeeded in achieving selective imaging of the lymphatic system by incorporating Gd-divinyltriamine penta (DTPA), a common contrast agent, with carboxylated nanodiamond (CND) particles to fabricate a nanoscale MRI contrast agent for the targeted imaging of the lymphatic system. Imaging experiments showed that the CND contrast agent with dadoterate meglumine (DOTAREM) exhibited enhanced MRI visibility in water and serum compared with the CND contrast agent with DTPA. These results suggest that the DOTAREM-loaded CND contrast agent is a promising nano-agent for the development of selective MR of the lymphatic system.
Magnetic and superparamagnetic nano MRI contrast agents
Magnetic NPs are an important group of nanomaterials that have been extensively studied as contrast agents for MRI and drug-delivery vehicles. Magnetic and superparamagnetic materials utilized in MRI enhance the differentiation between unhealthy and healthy tissues by shortening the proton spin–spin transverse relaxation time (T2) and the effective transverse relaxation time (T2*). In an investigation of the use of magnetic cobalt ferrite spinel (MCFS) NPs to develop a multifunctional drug-delivery system with MRI capabilities for cancer detection and treatment, Mikhaylov et al. 33 found that the MCFS NPs significantly improved the MRI contrast and enabled high-resolution T1- and T2-weighted MRI signal detection. Kang et al. 34 synthesized magnetic nanocrystals via thermal decomposition in organic solvents to obtain a highly magnetic methoxylated PEG-polylactic acid as an amphiphilic polymer. Constructing magnetic nanoclusters using PEG-polymerized magnetic nano-assemblies, they found that the reagent assemblies were stably dispersed in the aqueous phase with uniform size, exhibited sufficient MR contrast, extended retention, and had high biocompatibility, all of which improved the diagnostic efficiency and visibility of the disease site. When Thangudu et al. 35 synthesized ultrafine iron carbonate NPs (FeCO3 NPs) and evaluated their MRI contrast agent potential, they observed that FeCO3 NPs significantly enhanced the T2-weighted MRI contrast in a concentration-dependent manner, with an r2 value of 730. 9 ± 4.8 mM−1 S−1 at 9.4 T. These findings confirmed that FeCO3 NPs have potential for clinical applications in the early detection of lung tumors owing to their excellent biocompatibility and T2-weighted contrast ability. Figure 5 illustrates the in vivo imaging results.

In vivo MR contrast properties of FeCO3 nanoparticles (NPs). (A) In vivo T2-weighted MR images of lung tumors (n = 3) pre and post 1-, 3-, and 5-h intravenous injection of phosphate-buffered saline (PBS) and FeCO3 (10 mg Fe ion/kg) NPs. MRI scans were performed on a 9.4 T MRI scanner, circle/pseudo color indicates the tumor region. (B) Kinetic plot of in vivo contrast ability of PBS and FeCO3 NPs. (C) Schematic representation of in vivo MR imaging of lung tumors using FeCO3 NPs on a 9.4-T MRI scanner. Reproduced with open access and meets Creative Commons protocols from Ref. 35. ©J Nanobiotechnology, 2022.
Aiming to achieve a therapeutic diagnosis of cartilage damage using a multifunctional nanoprobe consisting of chitosan-modified Fe3O4 NPs self-assembled with the small-molecule growth factor kartogenin (KGN), Hong et al. 36 found that the use of the nanoprobe enhanced T2-weighted images when MRI was performed to detect cartilage damage. Nevertheless, the aggregation and accumulation of bare Fe3O4 magnetic NPs in vivo have significant disadvantages owing to their lack of biocompatibility. Improving the biocompatibility of contrast agents requires surface modifications. Danafar et al. 37 synthesized magnetic Fe3O4-Au hybridized NPs by merging the advantages of Fe3O4 and Au NPs in a simple manner by encapsulating NPs with bovine serum albumin (BSA) to enhance their biocompatibility. Modality experiments revealed a noteworthy decrease in T2 signal intensity on MR images, supporting the enhancement of nanoplatforms for accumulation and aggregation in tumors, a leading trend in the advancement of precision nanomedicine diagnostics.
Superparamagnetic substances can decrease T2 relaxation time and boost a low T2 signal to a certain degree. Luo et al. 38 created a nanoplatform consisting of IO@MnO2@DOX (IMD) hybrid NPs composed of superparamagnetic IO (SPION), manganese dioxide (MnO2), and doxorubicin (DOX). They found that its use allows efficient T2-weighted MRI and switchable photoacoustic (PA) imaging. SPION NPs (SPIONs) are typically enclosed in drug-carrying NPs or microparticles for synchronous MRI and treatment of diseased tissues. While conventionally encapsulated SPIONs typically have a limited ability to enhance T2-weighted relaxation, Lee et al. 39 revealed that SPIONs immobilized on 200 nm diameter lactic-co-glycolic acid (PLGA) NPs using Pickering emulsification had an 18-fold higher relaxation rate than encapsulated SPIONs and a 1.5-fold higher relaxation rate than free SPIONs. In contrast, SPIONs immobilized on PLGA particles with a diameter of 10 µm displayed a minor enhancement in MR relaxivity. This discovery is compelling and will have a significant impact on current advancements in the synthesis and assembly of advanced MR contrast agents.
Manganese-based nano MRI contrast agents
Hollow nanoplatforms based on manganese (Mn), a metallic element paramagnetic in nature, are currently receiving significant attention in the field of MR-enhanced imaging contrast agents and diagnostic-therapeutic integrated platforms. 38 These platforms provide the advantages of both the hollow structures and the intrinsic diagnostic and therapeutic properties of Mn2+. Empty spaces in particular can contain various small-molecule medications. Upon decomposition within the tumor microenvironment, liberated Mn2+ functions as both a contrast agent for MRI and sensitizer for chemodynamic therapy (CDT). Several hollow Mn-based nanoplatforms can be used for concurrent cancer therapy and MRI, including hollow manganese and its oxides (MnxOy), hollow matrix-loaded MnxOy, hollow Mn-doped NPs, hollow Mn complex NPs, hollow Mn cobalt (Co)-based NPs, and hollow Mn–Fe-based NPs. 40 Nevertheless, interference effects and lack of selectivity present challenges in the development of dual-mode contrast agents for MRI. To address these issues and surpass the limitations of single-mode imaging contrast, Xie et al. 41 successfully synthesized and used MgMnAl layered bis-hydroxide@iron oxide NPs (MgMnAl-LDH@IO NPs) as a dual-mode enhancement contrast agent for MRI. Their NP innovation addresses the obstacles encountered during the creation of contrast agents for dual-mode MRI. In vivo dual-mode MRI showed that MgMnAl-LDH@IONPs displayed significant T1- and T2-weighted MR in mice with mammary tumors. Its facile synthesis process, excellent biocompatibility, sensitive stimulation response, and concurrent enhancement of T1/T2 MRI signals in vitro and in vivo indicate that MgMnAl-LDH@IONPs may be a promising option for clinical MR imaging applications. Aiming to detect breast cancer cells using in vitro and in vivo using T1-weighted MRI, Xie et al. 42 synthesized NPs composed of gadolinium-manganese-molybdenum polymetallic oxide (Gd-Mn-Mo POM NPs) and coated them with chitosan-imidazole to create POM@CSIm NPs. Their experiments showed that the nanoclusters adhered to the cancer cells and selectively accumulated in the tumor regions, indicating their potential as MR nano-agents for early cancer detection.
CT/MR multimodal nano-contrast agent research
In clinical diagnosis, objective evaluation often requires the use of two or more imaging methods. Therefore, researchers have specifically selected nanomaterials targeting different modalities for image-enhancing contrast and prepared composite nano-agents through self-assembly and surface modification to develop multifunctional nano-contrast agents. These agents exhibit simultaneous multimodal image-enhancing contrast to meet the needs of patients who require multiple modality imaging examinations simultaneously. Enhanced contrast imaging is facilitated by a multifunctional nano-contrast agent that can simultaneously accommodate various imaging modalities with a single drug application. This approach offers significant advantages in terms of convenience and economic feasibility. One example is the fusion of magnetic and Au elements, which yields MRI/CT bimodal imaging, or the merging of graphene oxide and Au nanomaterials in the form of polylactic acid microcapsules, resulting in CT/US bimodal imaging.43,44 Wang et al. 45 synthesized ultrasmall Ag/Gd2O3 (Ag/Gd@BSA) heterogeneous NPs using BSA as a template and a biomineralization strategy 44 to achieve high aqueous dispersity. Using hybrid Ag/Gd@BSA-Gd2O3 NPs for multimodal enhancement imaging-guided photothermal therapy (PTT) with MR, CT, and photoacoustic (PA) imaging, they found that this approach effectively provided bimodal contrast enhancement for CT and MR.
Peng et al. 46 created Au nanoflowers (NFs) containing macrophages (MAs) and ultra-small IO NPs (USIO NPs) for dual-mode enhanced imaging of tumors using CT and MR techniques. They were able to produce Fe3O4/Au dendrimer-stabilized Au nanoflowers (DSNFs) with an average size of approximately 90 nm that exhibited high r1 relaxivity (1.22 mM−1 s−1) as well as notable colloidal stability and cytocompatibility. Because EPR is critical for accurate cancer diagnosis, Xu et al. 47 synthesized a multifunctional Arg-Gly-Asp (RGD)-Gd@Au-dendrimer-based (DNG)-PS (R-G@ADP) nanogel, a stable colloid with an average diameter of 122 nm, and found that it exhibited a high X-ray attenuation coefficient and excellent r1 relaxivity (9.13 mM−1 s−1). Furthermore, it displayed desired protein resistance and cytocompatibility via amphiphilic ion modification. Because of its specificity in targeting via RGD and superior infiltration into tumors compared with single dendritic-embedded Au NPs, multifunctional R-G@ADP can effectively target pancreatic tumor models and improve CT/MR dual-modality imaging in vivo. Figure 6 illustrates the structure and in vivo imaging effects of multifunctional R-G@ADP.
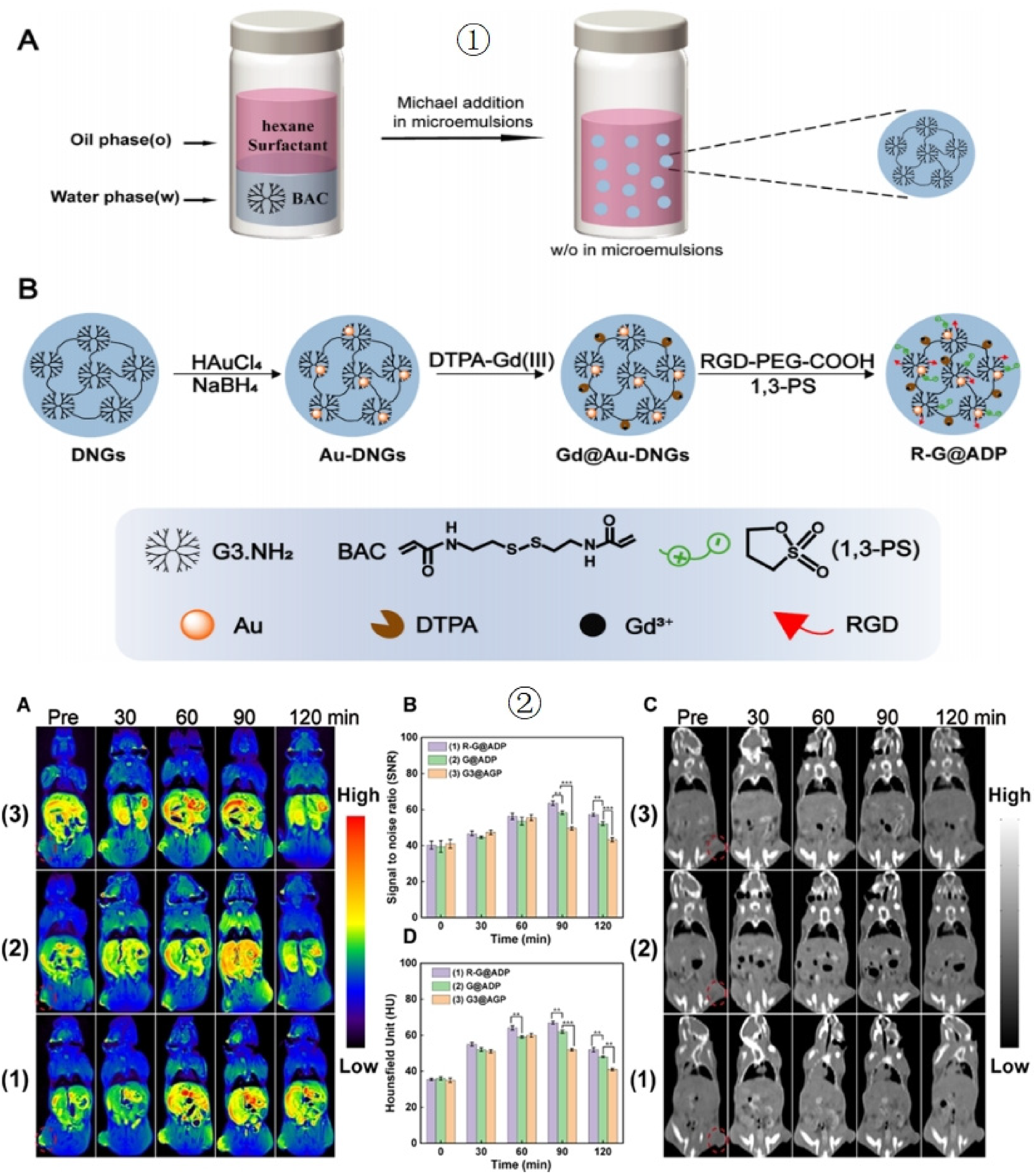
Schematic illustration of the synthesis of dendrimer-based nanogels (1A) and R-G@ADP (1B). Representative T1-weighted MR images (2A), T1 MR signal-to-noise ratios (2B), CT images (2C), and CT values (2D) of Panc-2 tumors at various time intervals post-intravenous injection of targeted R-G@ADP (1), non-targeted G@ ADP (2), and G3 @AGP (3) (Au = 0.1 M, Gd = 0.01 M). Red circles highlight tumor areas. Reproduced with permission from Ref. 47. ©Biomacromolecules, 2023.
CT/MR dual-mode nano-contrast agents possessing specific targeting abilities are predominantly crafted through intricate characterization and assembly processes involving high-atomic-number metallic or magnetic materials. However, utilizing multi-material assembly and modification characterization methods not only intensifies the level of preparation intricacy but also increases its cost. Therefore, future research and development should aim to create CT/MR dual-mode targeted nano-contrast agents that are simple, cost-effective, and efficient while maintaining imaging performance.
Although MRI is commonly used for imaging prostate cancer (PCa), more advanced detection techniques are necessary for precise localization of metastatic PCa. The limitations of single-modality imaging pose a significant challenge for healthcare providers because the current ability to use distinct methods to detect PCa and its metastases in patients is inadequate. To overcome this limitation, Wang et al. 48 developed a targeted therapeutic diagnostic platform for PCa multimodality imaging-guided photothermal therapy utilizing an Au/Mn nanodot-luteinizing hormone-releasing hormone (AMNDs-LHRH) nanosystem. This system can precisely target both gonadotropin-releasing hormone receptor (GnRH-R)-positive PCa and metastasis for precise preoperative CT/MR diagnosis while enabling fluorescence (FL) visualization to guide surgical intervention, showing its promise in clinical cancer detection and surgical guidance.
Discussion
Although CT and MRI are essential to medical diagnosis and treatment, conventional contrast agents cannot meet diverse demands for disease diagnosis and treatment, requiring the development of novel contrast agents. With advancements in nanotechnology, researchers have discovered the numerous benefits of nano-contrast agents that can address some of the difficulties associated with traditional contrast agents. To enhance agent solubility, stability, and biocompatibility, strict control of the size of nano-contrast agents, adjustment of nanomaterial environments, and surface modification of NPs are crucial. An important direction for the development of nano-targeted contrast agents is the modification of nanomaterial combinations to achieve multimodal CT and MR contrasts. By doing so, disease diagnosis efficiency can be improved while reducing patient pain and treatment costs. However, the design of nano-contrast agents should consider the relative contents of various elements to avoid concentration variations among different imaging systems. Another area of development involves combining nanomaterials with ligands specific to different tumors, thereby improving tumor targeting of nano-targeted contrast agents and significantly enhancing their diagnostic efficacy in tumors. By implementing this strategy, tumors can be detected and diagnosed at an earlier stage, which can increase the cure rate. The development of diagnostic and therapeutic integration platforms is a current focus in nano-reagent research. By assembling and modifying nanomaterials with different properties, imaging enhancement, drug-carrying therapy, radiotherapy, and photothermal therapy can be achieved. The development of these platforms expands the possibilities of nano-reagent research.49–52 Future reviews will provide a detailed description of these promising diagnostic and therapeutic integrated nano-reagent platforms.
Conclusion
The development of nano-contrast agents for medical imaging has led to several developments that show promising potential for applications. However, most nano-contrast agents are still in the research stage, and challenges related to biotoxicity and pharmacokinetic evaluation remain to be addressed. Moreover, prioritizing the preparation methods for nano-contrast agents to reduce expenses, increase production efficiency, and achieve mass production is essential. Nevertheless, as nano-contrast agents continue to advance, they will likely have a significant impact on the diagnosis and treatment of diseases as their limitations are resolved.
Footnotes
Acknowledgments
Author contributions
Writing–original draft preparation, Jianjun Lai; writing–review and editing, Zhizeng Luo and Zhibing Wu; funding acquisition, Liting Chen and Zhizeng Luo. All the authors have read and agreed to the published version of the manuscript.
Data availability statement
Data availability is not applicable to this article, as no new data were created or analyzed in this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Zhejiang Medicine and Health Science and Technology Program under Grant 2021KY434, the Zhejiang Provincial Natural Science Foundation Key Project under Grant LZ23F030005, and the National Natural Science Foundation of China under Grant 62171171.
Author biographies
Jianjun Lai, holds a PhD in Control Science and Engineering, is engaged in the study of tumor radiotherapy physics, radiographic physics technologies, and the application of nanotechnology in cancer radiotherapy. His work focuses on enhancing the precision and efficacy of radiotherapeutic methods in oncology.
Professor Zhizeng Luo, an academic PhD supervisor, specializes in pattern recognition technologies within the life and health sciences domain. His research contributes to the development of innovative analytical tools for biomedical applications.
Liting Chen, an attending physician in oncology, is involved in the research of new precision diagnostic and therapeutic technologies for cancer, aiming to integrate these advancements into clinical practice for improved patient care.
Zhibing Wu, a chief physician in radiation oncology, focuses on precision diagnosis and treatment in oncology, particularly exploring multimodal physical therapy approaches for cancer treatment.
