Abstract
Objective
To examine whether first-trimester PAPP-A, free β-hCG, NLR, and PLR are associated with subsequent development of PE, and among women with PE, whether these biomarkers are associated with adverse perinatal outcomes.
Methods
In this retrospective case-control study, we analyzed 350 primigravid women, including 175 with PE (cases) and 175 with uncomplicated pregnancies (controls). All participants had singleton gestations and available first-trimester (11 + 0 to 13 + 6 weeks) serum measurements of PAPP-A, free β-hCG, and complete blood count. Univariable and multivariable logistic regression models assessed associations of biomarkers with PE and, separately among PE cases, with the composite adverse perinatal outcome. Receiver operating characteristic (ROC) curve analysis quantified discriminative performance.
Results
In multivariable analysis, first-trimester free β-hCG (OR = 1.358, 95% CI 1.206–1.530), PAPP-A (OR = 0.756, 95% CI 0.691–0.828), and NLR (OR = 1.655, 95% CI 1.214–2.256) were associated with PE. The combined model including these three markers showed improved discrimination for PE (AUC=0.793) compared with any marker alone. Among women with PE, higher β-hCG (OR = 1.489, 95% CI 1.242–1.784), elevated NLR (OR = 1.562, 95% CI 1.075–2.268), and lower PAPP-A (OR = 0.794, 95% CI 0.696–0.906) were associated with the adverse composite outcome in multivariable analysis. The multivariable model including these markers demonstrated an AUC of 0.804 for discrimination of the adverse composite outcome.
Conclusions
First-trimester levels of PAPP-A, free β-hCG, and NLR may be associated with the subsequent development of PE and with adverse perinatal outcomes among affected women. These findings suggest potential biological relevance but do not establish clinical utility and should be interpreted with caution given the observational study design.
Keywords
Introduction
Preeclampsia (PE) is a multisystem hypertensive disorder of pregnancy that affects roughly 2%–8% of pregnancies worldwide. 1 It remains a leading cause of maternal and perinatal morbidity and mortality globally, particularly in low-resource settings. 2 PE is especially associated with adverse outcomes such as preterm delivery, intrauterine growth restriction (IUGR), low birth weight, and perinatal death. 3 Early identification of women at risk is therefore critical to enable closer monitoring, timely intervention, and improved maternal and perinatal outcomes. 2
The pathogenesis of PE is multifactorial and not fully understood; abnormal placental development and chronic inflammation are considered key features. 4 In recent years, first-trimester screening markers have been investigated as predictors of PE. In particular, placental biomarkers used in routine aneuploidy screening—maternal serum free β-human chorionic gonadotropin (β-hCG) and pregnancy-associated plasma protein A (PAPP-A)—have been linked to placental function.5,6 Low first-trimester PAPP-A has been associated with an increased risk of subsequent PE; for instance, women with PAPP-A levels below the 10th percentile showed a more than threefold higher risk. 7 Similarly, elevated β-hCG levels have been associated with PE, with a recent meta-analysis reporting a twofold increase in PE risk among women with high hCG MoM values (≥2). 8 However, the predictive utility of individual biomarkers is limited, and combining multiple biomarkers—particularly those reflecting distinct biological pathways—may improve risk stratification.
Systemic inflammation is also implicated in PE. Chronic low-grade inflammation during pregnancy may be reflected in maternal blood cell profiles. 9 Ratios derived from complete blood counts, such as the neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR), are simple indices of systemic inflammation. These ratios have shown promise in chronic inflammatory conditions, 10 and several studies suggest alterations in NLR and PLR preceding the development of PE.11,12 For example, first-trimester NLR and PLR have been reported to be higher in women who subsequently develop PE. 12 However, results across studies have been inconsistent.13,14 More recent investigations continue to report heterogeneous associations between early inflammatory indices and PE risk, with some studies identifying NLR and composite indices as independent predictors, while others report weaker or population-specific associations.15–18 Because NLR and PLR are inexpensive and routinely available, they remain attractive candidate biomarkers for early PE risk assessment.
Despite these findings, the combined predictive value of first-trimester PAPP-A, β-hCG, and inflammatory indices (NLR, PLR) for PE remains unclear. Few studies have evaluated all four markers concurrently, and evidence regarding NLR and PLR in early pregnancy PE prediction is still evolving. Moreover, PE is clinically heterogeneous, and the extent to which early first-trimester markers relate to subsequent adverse perinatal outcomes remains incompletely defined. Investigating these associations may contribute to improved understanding of PE pathophysiology and risk stratification. Conceptually, placental dysfunction and systemic inflammation represent interrelated but distinct pathways in PE development. We therefore hypothesized that first-trimester biochemical screening markers reflecting placental function (PAPP-A and free β-hCG) and CBC-derived inflammatory indices (NLR and PLR) may provide complementary information when evaluated together.
Accordingly, in this case-control study, we aimed to evaluate whether first-trimester serum levels of PAPP-A and free β-hCG, and the hematologic indices NLR and PLR, are associated with the subsequent development of PE. As a secondary objective, we assessed whether these markers were associated with composite adverse perinatal outcomes among women with PE.
Methods
Study design and setting
This case-control study was conducted at Yongkang Maternal and Child Health Hospital between May 2023 and July 2025.
Participants
Cases were pregnant women with a confirmed diagnosis of PE according to the American College of Obstetricians and Gynecologists (ACOG) criteria 19 who presented consecutively to Yongkang Maternal and Child Health Hospital during the study period. All cases were screened for eligibility using predefined inclusion and exclusion criteria, and 175 cases were included in the final analysis. Controls were pregnant women with an uncomplicated pregnancy who presented consecutively to the same hospital during the same study period, attended routine antenatal care, and delivered at term. Controls were screened using the same predefined inclusion and exclusion criteria, and 175 controls were included in the final analysis. No individual matching was performed between cases and controls. The participant flow diagram is shown in Figure 1.

The participant flow diagram.
Inclusion and exclusion criteria
Inclusion criteria were age 18–40 years, primigravida status, singleton pregnancy, available first-trimester serum results for PAPP-A and free β-hCG, as well as a complete blood count (CBC), all measured at the same visit between 11 + 0 and 13 + 6 weeks of gestation.
Exclusion criteria included chronic or systemic disorders (e.g. preexisting hypertension, diabetes, renal or cardiac disease, autoimmune or hematologic disorders), active infection or inflammatory condition during first-trimester screening, malignancy, gestational diabetes mellitus (GDM), assisted reproduction, smoking, and pregnancies complicated by fetal structural anomalies or suspected or confirmed chromosomal abnormalities, including aneuploidy identified through first-trimester screening, invasive prenatal testing (including amniocentesis), or postnatal evaluation.
Data collection
For all participants, data were abstracted from hospital records, laboratory databases, and a medical history obtained at the time of recruitment. Variables included maternal age, pre-pregnancy BMI, gestational age at first-trimester screening, PAPP-A, free β-hCG, NLR, and PLR. First-trimester serum PAPP-A and free β-hCG concentrations were measured as part of routine aneuploidy screening at the hospital laboratory. Values were expressed as multiples of the median (MoM) as provided by the laboratory. MoM calculations were performed using the laboratory's standard automated screening software, based on gestational age-specific reference medians derived from the local screened population and according to routine clinical protocols. Adjustments routinely applied in first-trimester screening, including those for gestational age and selected maternal characteristics, were incorporated into the laboratory-generated MoM values. All assays were performed in a single certified laboratory using standardized procedures and quality control measures. NLR and PLR were calculated from CBC by dividing absolute neutrophil and platelet counts by the absolute lymphocyte count, respectively. All CBC samples were collected concurrently with first-trimester serum screening during scheduled routine antenatal visits between 11 + 0 and 13 + 6 weeks of gestation.
Participants were followed until delivery. Adverse perinatal outcomes were assessed only among women with PE, as pregnancies with complications were excluded from the control group by design. A composite adverse perinatal outcome was defined a priori as the occurrence of at least one of the following events: preterm delivery (<37 completed weeks of gestation), low birth weight (<2500 g), intrauterine growth restriction (estimated fetal weight <10th percentile for gestational age), neonatal respiratory distress syndrome (NRDS diagnosed by neonatology staff based on clinical and radiographic criteria), low 5-min Apgar score (<7), 20 perinatal death (stillbirth or neonatal death within the first 7 days of life), placental abruption (clinically diagnosed), or neonatal sepsis (culture-proven or clinically diagnosed requiring antibiotic treatment). Each woman with PE was considered to have the composite outcome if one or more of these complications occurred.
Sample size
Sample size was determined according to events-per-variable (EPV) recommendations for logistic regression. With seven predictors in the planned multivariable model (age, BMI, gestational age, NLR, PLR, β-hCG, and PAPP-A) and considering conservative recommendations (≥20) to avoid model overfitting, an EPV of 25 was targeted. Accordingly, a sample size of 175 cases and 175 controls was estimated, which exceeds the commonly cited minimum threshold of ≥10 EPV. 21
Statistical analysis
Statistical analyses were performed using SPSS (version 25.0), R (version 4.3.1), and MedCalc (version 23.4.5). Continuous variables were summarized as mean ± standard deviation (SD) and median (interquartile range [IQR]) and were compared between groups using independent-samples t-tests. Categorical variables were presented as frequencies and percentages.
Univariable and multivariable logistic regression analyses were conducted to assess the association of study biomarkers with PE and, within the PE group, with the composite adverse perinatal outcome. All variables included in the multivariable models were selected a priori based on the study hypothesis and prior evidence. A forced-entry (Enter) approach was used to include all prespecified variables simultaneously, rather than data-driven stepwise selection. Known baseline confounders, including maternal age, pre-pregnancy BMI, and gestational age, were included in all multivariable models for adjustment. Multicollinearity among predictors was assessed using variance inflation factors (VIFs), with values <5 considered indicative of acceptable collinearity. Despite moderate correlation between NLR and PLR, both variables were retained in the models because they reflect related but distinct inflammatory pathways and did not demonstrate problematic multicollinearity.
For analyses of the composite adverse perinatal outcome with a limited number of events (n = 72), penalized logistic regression using Firth's correction was performed as a sensitivity analysis in R (package logistf) to reduce small-sample bias and assess the robustness of multivariable estimates. Odds ratios (OR) with 95% confidence intervals (CI) were reported. Receiver operating characteristic (ROC) curve analyses were performed using MedCalc software to evaluate the discriminative ability of individual biomarkers and logistic regression-based prediction models. Areas under the curve (AUC) with corresponding 95% CI were calculated and graphically presented with confidence interval bands. Optimal cut-off values were determined using the Youden index, with corresponding sensitivity and specificity reported. A two-sided P-value <0.05 was considered statistically significant.
Ethics and consent
This study was conducted in accordance with the Declaration of Helsinki of 1975, as revised in 2024, and was approved by the Ethics Committee of Yongkang Maternal and Child Health Hospital (approval code: 20230426–46; approval date: 26 April 2023). All participants voluntarily provided written informed consent before enrollment in the study. Confidentiality of patient information was maintained throughout the study, and all patient data were fully anonymized with no identifiable information collected or reported.
Reporting standards
This case-control study was reported in accordance with the STROBE guidelines, as recommended by the EQUATOR Network. 22
Results
This case-control study included 175 women with PE and 175 controls with uncomplicated pregnancies. Maternal characteristics, first-trimester laboratory parameters, and adverse perinatal outcomes are summarized in Table 1. The two groups were comparable with respect to maternal age, pre-pregnancy BMI, gestational age at first-trimester screening, neutrophil count, and platelet count. In contrast, women who later developed PE exhibited significantly higher first-trimester β-hCG, NLR, and PLR values, along with lower PAPP-A and lymphocyte counts.
Maternal characteristics, first-trimester laboratory parameters, and adverse perinatal outcomes of the study participants.

Data are presented as mean ± SD, median (IQR), and frequency (%).
GA at FTS: gestational age at first-trimester screening; NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-to-lymphocyte ratio; IUGR: intrauterine growth restriction; NRDS: neonatal respiratory distress syndrome.
*Composite adverse perinatal outcome was defined as the occurrence of one or more individual adverse outcomes, as described in the Methods section.
In univariable logistic regression analyses, higher first-trimester β-hCG, NLR, and PLR levels were positively associated with subsequent PE, whereas lower PAPP-A values were inversely associated with PE. In multivariable logistic regression, only β-hCG, PAPP-A, and NLR remained independently associated with PE (Table 2).
Logistic regression analyses of maternal and biochemical predictors for preeclampsia and composite adverse perinatal outcome.

OR: odds ratio; CI: confidence interval; GA at FTS: gestational age at first-trimester screening; NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-to-lymphocyte ratio.
β-hCG and PAPP-A were analyzed per 0.1 MoM increase in all logistic regression models.
Composite adverse perinatal outcome was defined as the occurrence of one or more individual adverse outcomes, as described in the Methods section.
ROC curve analysis was performed to evaluate the discriminative ability of the study biomarkers for predicting PE (Figure 2). The AUC for β-hCG was 0.658 (95% CI: 0.606–0.707, P < 0.001), for PAPP-A 0.696 (95% CI: 0.645–0.744, P < 0.001), for NLR 0.660 (95% CI: 0.608–0.710, P < 0.001), and for the combined logistic regression model including β-hCG, PAPP-A, and NLR 0.793 (95% CI: 0.747–0.834, P < 0.001). The combined model demonstrated the highest discriminative ability among the evaluated models. Optimal cut-off points were determined using the Youden index. The optimal cut-offs were ≥1.08 for β-hCG (sensitivity 56.0%, specificity 68.0%), ≤0.93 for PAPP-A (56.0% and 72.6%), ≥3.45 for NLR (52.6% and 77.1%), and ≥0.57 for the combined model (65.1% and 84.6%). Individually, biomarkers showed moderate discriminative performance, whereas their combination showed improved discrimination.
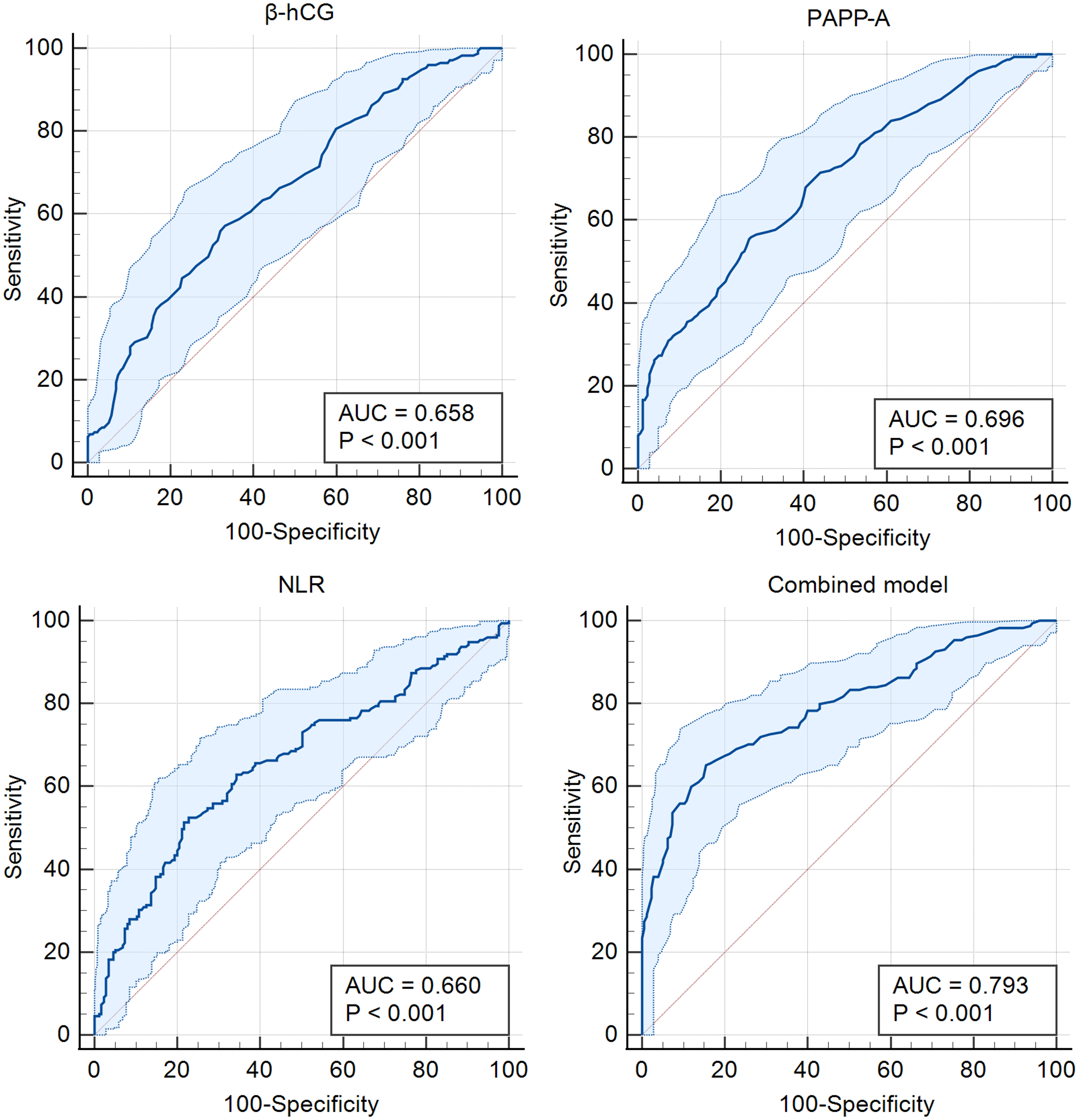
Receiver operating characteristic (ROC) curves of first-trimester biomarkers for prediction of preeclampsia.
Among women with PE, 72 (41.1%) experienced at least one adverse perinatal outcome. The distribution of individual adverse outcomes is presented in Table 1. Logistic regression analyses were conducted to examine the associations between first-trimester biomarkers and the composite adverse perinatal outcome. In univariable models, higher β-hCG, elevated NLR and PLR, and lower PAPP-A values were significantly associated with increased risk of the adverse outcome. After multivariable adjustment, β-hCG, NLR, and PAPP-A remained independent predictors (Table 2). To assess model robustness, penalized logistic regression with Firth correction was performed. Results were consistent with standard logistic regression, with β-hCG, PAPP-A, and NLR remaining significantly associated with the composite adverse outcome, while PLR and maternal covariates did not reach statistical significance. These findings support the stability of the observed associations despite the limited number of adverse events.
The discriminative performance of biomarkers for predicting the composite adverse outcome within the PE group was further evaluated using ROC analysis (Figure 3). The AUC was 0.690 (95% CI, 0.616–0.758; P < 0.001) for β-hCG, 0.648 (95% CI, 0.572–0.718; P < 0.001) for PAPP-A, and 0.694 (95% CI, 0.620–0.761; P < 0.001) for NLR. When combined in a multivariable model, discriminative performance improved, yielding an AUC of 0.804 (95% CI, 0.738–0.860; P < 0.001).
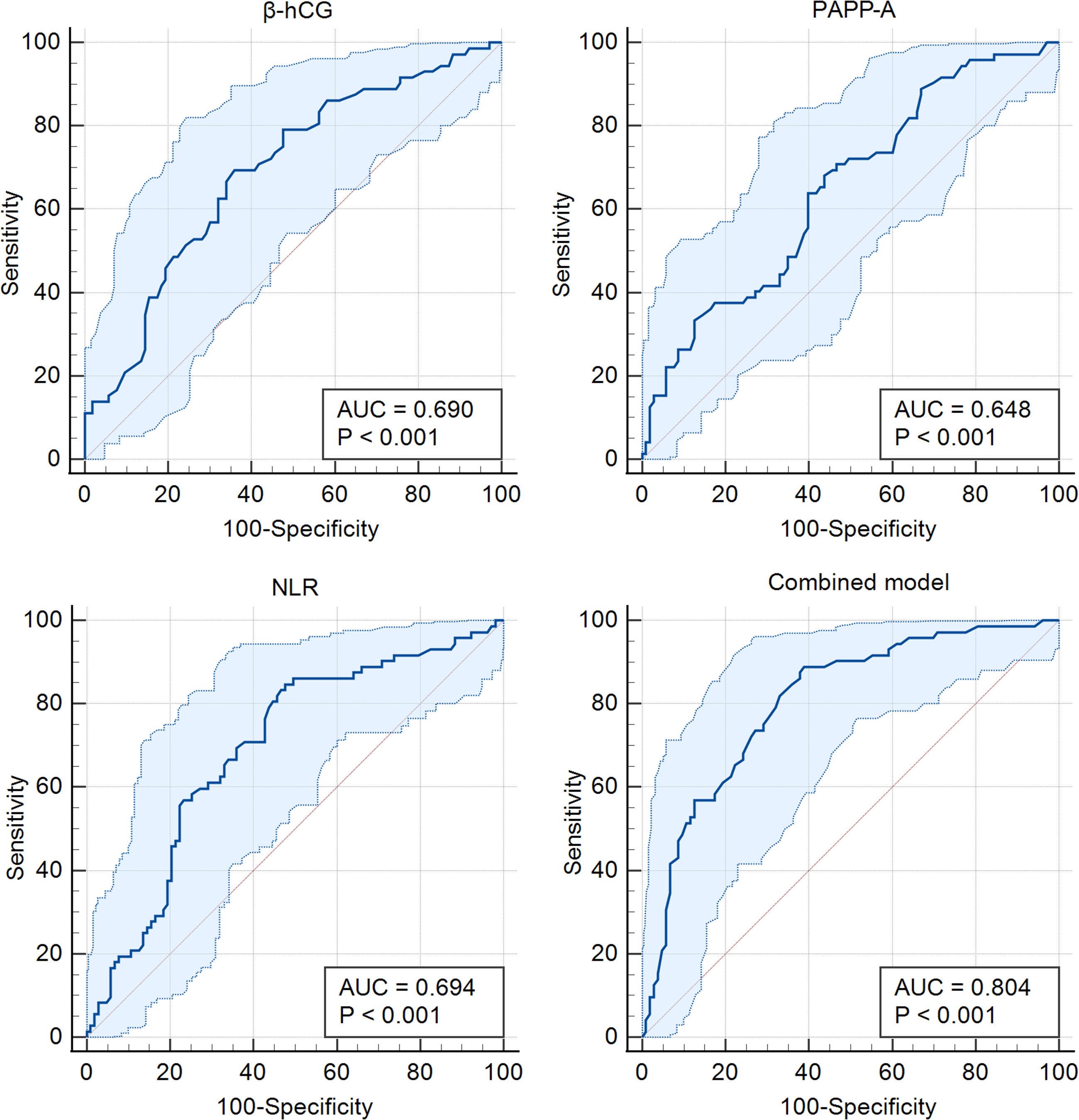
Receiver operating characteristic (ROC) curves of first-trimester biomarkers for prediction of composite adverse perinatal outcome among women with preeclampsia.
Discussion
Our study identified significant associations between early-pregnancy biomarkers and the later development of PE, as well as with composite adverse perinatal outcomes among women who developed PE. Notably, lower first-trimester PAPP-A and higher free β-hCG and NLR were independently associated with subsequent PE, aligning with current understanding of placental and inflammatory involvement in PE.
These findings are consistent with prior evidence demonstrating reduced first-trimester PAPP-A and elevated free β-hCG levels in pregnancies complicated by PE.23,24 The observed association between NLR and PE risk further supports the contribution of inflammatory processes. 25 Recent studies support an association between early inflammatory indices and PE, while highlighting substantial heterogeneity in observed effects across populations and study designs.15–18 In contrast, PLR showed only a univariable association and did not remain independently predictive, consistent with recent meta-analytic evidence. 26 The combined biomarker model demonstrated improved discriminative ability compared with individual markers, which is consistent with the multifactorial nature of PE. However, the observed AUC values indicate moderate rather than strong discrimination and should not be interpreted as evidence of clinical screening performance. Limitations related to study design, lack of external validation, and absence of comparison with established screening algorithms preclude conclusions regarding clinical applicability.
In this context, the potential role of second-trimester uterine artery (UtA) Doppler as a complementary assessment in pregnancies with abnormal first-trimester serum markers merits consideration. Recent evidence suggests that in women with low PAPP-A and/or abnormal free β-hCG values, the addition of second-trimester UtA Doppler pulsatility index may further refine risk assessment for PE and adverse perinatal outcomes. 27 Although UtA Doppler was not available in the present study, our findings are consistent with a stepwise approach in which early biochemical markers identify higher-risk pregnancies that may benefit from targeted Doppler surveillance rather than serving as standalone screening tools. Previous studies also indicate that integrating biochemical markers with uterine Doppler assessment may improve risk prediction.28,29 Thus, our results support the exploration of multimarker strategies, while emphasizing the need for prospective validation and integration with established screening models.
Among women with PE, those who experienced the adverse composite perinatal outcome showed more pronounced first-trimester biomarker abnormalities. Specifically, elevated β-hCG and NLR and reduced PAPP-A were significantly associated with higher odds of the composite adverse outcome, and these associations remained independent in multivariable analyses. These findings are in line with previous reports. For instance, Papamichail et al. reported that women with very low first-trimester PAPP-A (≤0.4 MoM) had nearly threefold higher odds of a composite adverse outcome, including PE, pregnancy-induced hypertension, and IUGR. 30 This finding suggests that reduced first-trimester PAPP-A levels, as a marker of early placental dysfunction, may be associated with increased susceptibility to both PE and its downstream perinatal sequelae. While literature on first-trimester free β-hCG and neonatal outcomes remains limited, our findings suggest that elevated β-hCG MoM may mark more severe placental pathology among women who subsequently develop PE. Similarly, elevated first-trimester NLR among PE cases may reflect a heightened inflammatory milieu associated with worse perinatal outcomes, consistent with evidence that NLR is higher in severe versus mild PE. 25
Inflammatory markers derived from routine complete blood count, including NLR, have also been evaluated in other obstetric complications such as GDM. Although recent data indicate that second-trimester NLR values may be higher in women with GDM, their standalone predictive value appears limited and inconsistent. 31 These findings suggest that CBC-derived inflammatory markers may reflect a nonspecific inflammatory state common to multiple pregnancy complications rather than disease-specific mechanisms. Accordingly, our results support a contributory—but not exclusive—role for NLR in PE-related risk stratification.
Overall, these findings suggest that among women who develop PE, early biomarkers of placental insufficiency and systemic inflammation are associated with an increased risk of adverse perinatal outcomes. However, these associations should not be interpreted as evidence of screening performance or early diagnostic accuracy. Rather, the use of a composite adverse perinatal outcome reflects overall morbidity among PE cases and supports the interpretation of these biomarkers as informative for future research aimed at refining early risk stratification and clinical surveillance.
Limitations
This study has several limitations that should be considered when interpreting the results. First, as a retrospective hospital-based case-control study, causal inferences cannot be drawn, and the potential for selection bias cannot be fully excluded. However, cases and controls were recruited from the same source population and time period using identical eligibility criteria, which helps mitigate differential selection. In addition, key confounders were addressed through study design and multivariable adjustment. Nevertheless, residual confounding from unmeasured factors, such as socioeconomic or environmental variables, may still have influenced the observed associations.
Sample size and event distribution represent important limitations. Although the overall cohort (n = 350) provided an adequate number of events per variable for the primary analysis of PE, statistical power was limited for the secondary analysis among women with PE, as only 72 cases experienced the composite adverse perinatal outcome. As a result, outcomes were analyzed in aggregate, precluding assessment of whether specific complications were differentially associated with the biomarkers of interest. The use of a composite adverse perinatal outcome, while common in obstetric research to address low event rates and improve statistical efficiency, may complicate interpretation when component outcomes differ in frequency, severity, and underlying pathophysiology. 32 Accordingly, unequal associations between predictors and individual components cannot be excluded, and findings should be interpreted with appropriate caution.33,34 For the secondary outcome, the limited number of events was below recommended thresholds for multivariable modeling, which may reduce the precision of effect estimates. Although sensitivity analyses using Firth penalized logistic regression supported the robustness of the main findings, this limitation should be acknowledged.
Stratified analyses according to early- versus late-onset PE were not performed due to limited event numbers and incomplete data on PE onset timing. Given the potential etiologic differences between these phenotypes, future adequately powered prospective studies should assess whether first-trimester biomarker associations vary by timing of disease onset. Additional limitations include the assessment of biomarkers at a single time point in early pregnancy, precluding evaluation of temporal trends, and the single-center nature of the study, which may limit generalizability. External validation in diverse cohorts is therefore warranted. Finally, multiple biomarkers and outcomes were evaluated without formal adjustment for multiple comparisons. Although analyses were hypothesis-driven and restricted to a predefined set of variables, the possibility of chance findings cannot be excluded and should be considered when interpreting statistically significant associations.
Conclusion
Our findings suggest that first-trimester levels of PAPP-A, free β-hCG, and NLR may be associated with the subsequent development of PE. Among women who developed PE, similar early biomarker abnormalities may also be associated with adverse perinatal outcomes. These observations indicate potential biological relevance but do not establish clinical utility. Given the observational design of this study, the results should be interpreted with caution. Further large-scale prospective studies are needed to confirm these associations and to evaluate their potential role in clinical risk assessment.
Supplemental Material
sj-docx-1-sci-10.1177_00368504261430053 - Supplemental material for First-trimester β-hCG, PAPP-A, and NLR in relation to preeclampsia and perinatal outcomes: A case-control study
Supplemental material, sj-docx-1-sci-10.1177_00368504261430053 for First-trimester β-hCG, PAPP-A, and NLR in relation to preeclampsia and perinatal outcomes: A case-control study by Xiongying Li, Jingxia Ying, Bingbin Xu, Xiaofeng Cui and Qiaoli Tong in Science Progress
Supplemental Material
sj-docx-2-sci-10.1177_00368504261430053 - Supplemental material for First-trimester β-hCG, PAPP-A, and NLR in relation to preeclampsia and perinatal outcomes: A case-control study
Supplemental material, sj-docx-2-sci-10.1177_00368504261430053 for First-trimester β-hCG, PAPP-A, and NLR in relation to preeclampsia and perinatal outcomes: A case-control study by Xiongying Li, Jingxia Ying, Bingbin Xu, Xiaofeng Cui and Qiaoli Tong in Science Progress
Footnotes
Acknowledgements
Not applicable.
Ethical considerations
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Yongkang Maternal and Child Health Hospital (approval code: 20230426-46; approval date: 26 April 2023).
Consent to participate
All participants voluntarily provided written informed consent before enrollment in the study.
Consent for publication
Not applicable.
Author contributions
[Conceptualization]: X.L., Q.T.; [Methodology]: X.L., J.Y., B.X., X.C., Q.T.; [Validation]: X.L., Q.T.; [Formal analysis]: J.Y., B.X., X.C.; [Investigation]: J.Y., B.X., X.C.; [Resources]: X.L., Q.T.; [Data Curation]: J.Y., B.X., X.C.; [Writing - Original Draft]: X.L., J.Y., B.X., X.C., Q.T.; [Writing - Review & Editing]: X.L., Q.T.; [Visualization]: J.Y., B.X., X.C.; [Supervision]: X.L., Q.T.; All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
