Abstract
Objective
Bacterial infections (BIs) in decompensated cirrhosis are associated with high morbidity and mortality but remain diagnostically challenging due to limited conventional biomarker accuracy. We aim to evaluate the utility of serum Delta-like ligand 1 (DLL1) for BI detection in patients with decompensated cirrhosis.
Methods
In this dual-cohort prospective study, 320 hospitalized patients with decompensated cirrhosis were consecutively enrolled and stratified into derivation (n = 224) and independent validation (n = 96) cohorts. Serum DLL1 levels were quantified at admission with an enzyme-linked immunosorbent assay. Diagnostic performance was assessed through receiver operating characteristic (ROC) curve analysis and multivariable logistic regression.
Results
Compared with their noninfected counterparts, patients with decompensated cirrhosis and BI had elevated serum DLL1 levels (P < 0.001). DLL1 was an independent predictor of BIs (adjusted odds ratio (OR) = 5.495, 95% confidence interval (CI): 3.022–9.992) in multivariate analysis. DLL1 demonstrated robust diagnostic performance (area under the curve (AUC) = 0.863, 95% CI: 0.814–0.911), which was further improved when combined with C-reactive protein (CRP) in a dual-marker model (AUC = 0.918, P < 0.001) and validated in an independent cohort (AUC = 0.925). Decision curve analysis confirmed the clinical utility of this combination across threshold probabilities of 10% to 80%. Notably, DLL1 levels were weakly correlated with total bilirubin levels (Spearman's ρ=0.24, P < 0.001), suggesting limited confounding effects from hepatic inflammation.
Conclusion
Serum DLL1 was a clinically viable diagnostic biomarker for BIs in patients with decompensated cirrhosis and demonstrated weak confounding effects from hepatic dysfunction. The CRP–DLL1 combined model achieved superior diagnostic accuracy with cross-cohort validation robustness.
Introduction
The global burden of infections among patients with liver cirrhosis is increasing, particularly in those with decompensated cirrhosis. The elevated incidence of infections has a profound impact on patient outcomes. 1 Research indicates that, owing to compromised immune function, individuals with liver cirrhosis exhibit higher susceptibility to bacterial, fungal, and viral infections than those without liver cirrhosis. 2 This immunodeficient state increases the likelihood of multiorgan failure and is closely linked to elevated mortality rates.3,4 Patients with decompensated liver cirrhosis are at risk for bacterial infections (BIs), which are difficult to diagnose because of the subtle onset of BIs and low detection rate via cultures. C-reactive protein (CRP) is a commonly used biomarker, but its reliability is questionable because of inflammation due to cirrhosis itself. 5 Procalcitonin (PCT) has high diagnostic accuracy for severe infections, although its performance may be influenced by hepatic and renal function status. 6 Other biomarkers investigated in this field include interleukin-6, the neutrophil-to-lymphocyte ratio, presepsin, resistin, blood metabolites, and circulating microRNAs (miRNAs).7–11 However, the diagnostic accuracy of single biomarkers remains limited; therefore, novel biomarkers should continue to be explored and their clinical utility in practice should continue to be evaluated in future research.
Notch ligand delta-like 1 (DLL1), a transmembrane protein of the Delta/Jagged family, activates downstream signaling pathways upon binding to its receptor, and DLL1 plays critical roles in embryogenesis, angiogenesis, and hematopoiesis. 12 Additionally, the Notch signaling pathway regulated by DLL1 modulates immune cell differentiation and function, thereby influencing host defence against pathogen infections.13–16 Upon stimulation by bacteria or endotoxins, DLL1 is secreted by monocytes and triggers the Notch signaling pathway in neighboring cells; this cascade amplifies proinflammatory responses, damages vascular endothelial cells, and increases vascular permeability. 17 Hildebrand et al. demonstrated that DLL1 expression is upregulated in primary human monocytes exposed to various bacterial pathogens in vitro, with abundant soluble DLL1 detected in cell supernatants. 14 Notably, plasma DLL1 levels are significantly elevated in sepsis patients but remain unchanged in noninfectious inflammatory conditions (e.g. surgery or trauma), exhibiting diagnostic value superior to that of PCT and CRP. 18 Emerging evidence suggests that postliver transplantation DLL1 levels predict infection occurrence and correlate with clinical complexity. 19 While DLL1 shows promise in the diagnosis of infections and the assessment of infection prognosis, the role of DLL1 in decompensated cirrhosis remains unexplored. The aim of this study was to evaluate the utility of serum DLL1 for BI detection in patients with decompensated cirrhosis.
Materials and methods
Study population
This prospective cohort study enrolled consecutive adults with decompensated cirrhosis admitted to Ganzhou People's Hospital (China) between June 2023 and June 2024, with written informed consent obtained from all participants following approval by the Clinical Research Ethics Subcommittee of Ganzhou Hospital-Nanfang Hospital, Southern Medical University (TY-ZKY2023-023-1), all the patients’ details have been de-identified. This study was conducted in accordance with the ethical principles of the Declaration of Helsinki (2024 revision). The reporting of this study conformed to the STARD guideline. 20 Participants were subsequently randomized through stratified sampling into derivation (70%) and validation (30%) cohorts to ensure balanced representation of BI and noninfection (NO BI) subgroups. The study population inclusion required confirmed decompensated cirrhosis diagnosis, whereas the exclusion criteria were severe extrahepatic organ dysfunction, immunosuppressant use, an organ transplantation history, hospitalization <48 hours, or unavailable biospecimens.
Sample size calculation
The sample size was prospectively estimated using a single-arm diagnostic accuracy study design. Assuming DLL1 sensitivity and specificity of 75% (derived from its established performance in sepsis populations 18 and conservative adjustment for cirrhosis-associated immune dysfunction and severity of infection), with 30% infection prevalence 4 and 5% attrition rate for biospecimen quality control, we calculated a minimum requirement of 73 infected patients and 73 noninfected patients to achieve 95% confidence intervals (CI) within ±10%. This translated to a total recruitment target of 256 participants.
Clinical definitions
The diagnostic criteria adhered to international guidelines: decompensated cirrhosis according to the European Association for the Study of the Liver, acute-on-chronic liver failure (ACLF) per the Asian Pacific Association for the Study of the Liver criteria, and acute kidney injury (AKI) based on the International Club of Ascites consensus.21–23
Spontaneous bacterial peritonitis was diagnosed by ascitic fluid neutrophil count criteria with secondary peritonitis exclusion; urinary tract infections required clinical symptoms with microbiological/pyuria confirmation; pneumonia involved radiographic evidence plus clinical/microbiological findings; spontaneous bacteremia mandated positive blood cultures without identifiable source; other infections followed Centers for Disease Control and Prevention (CDC) criteria. 24
Data collection and laboratory assays
Demographic, clinical, and laboratory data were systematically extracted from electronic medical records by trained clinicians. Blood samples were collected within 24 hours of admission, centrifuged at 3000×
Statistical analysis
Continuous variables are presented as the means ± standard deviations or medians (interquartile ranges) on the basis of distribution normality, as assessed via the Shapiro–Wilk test, whereas categorical variables are presented as frequencies (%). Between-group differences in baseline characteristics were evaluated using the Mann–Whitney U test or independent t test for continuous variables and the χ² test or Fisher's exact test for categorical variables. Univariate and multivariate binary logistic regression analyses were performed to identify predictors of BI, with variables achieving P < 0.10 in univariate analysis entered into a backwards elimination multivariate model (retention threshold: P < 0.05). Diagnostic performance metrics (area under the curve (AUC), sensitivity, and specificity) for the CRP, DLL1, and combined models were calculated using receiver operating characteristic (ROC) curve analysis (R package pROC), with optimal cut-offs determined by Youden's index and AUC differences tested via DeLong's method. Decision curve analysis (DCA) was used to quantify the clinical net benefit across threshold probabilities, and calibration curves and the Hosmer–Lemeshow test (P > 0.05 indicating good fit) were used to assess the agreement between the predicted and observed probabilities. Spearman's rank correlation (ρ) was used to analyze associations between DLL1 levels and clinical parameters, with P values adjusted for multiple comparisons via the Benjamini–Hochberg method. All analyses were performed in R 4.2.1 (pROC, rmda, rms, ggplot2 packages), and statistical significance was defined as a two-tailed P value <0.05.
Results
Study population and baseline characteristics
A total of 343 patients with decompensated cirrhosis were initially eligible. After applying the inclusion/exclusion criteria, 320 patients were enrolled, including 123 with BI and 197 without BI (NO BI). These patients were randomly divided into derivation (n = 224, BI = 86) and validation (n = 96, BI = 37) cohorts (Supplemental Figure 1).
As shown in Table 1, the study population was predominantly male (75%), with a mean age of 57 years. Hepatitis B virus (HBV) infection was the leading etiology, followed by alcohol-related cirrhosis and HBV-alcohol dual etiology. Spontaneous bacterial peritonitis (derivation and validation, 42.8% and 29.7%, respectively) and pulmonary infections (derivation and validation, 36% and 54.1%, respectively) were the most common infection types. Ascites was present in 73.3% and 83.3% of the derivation and validation cohorts, respectively. ACLF was observed in 33.9% (derivation) and 29.2% (validation) of patients at baseline. The median Child–Pugh score was 9 in both cohorts. No significant differences in demographics, etiology, infection sites, liver function parameters (total bilirubin (TBIL), creatinine, and INR), inflammatory markers (CRP and PCT), or DLL1 levels (P > 0.05 for all) were detected between the cohorts.
Baseline characteristics of patients from the derivation and validation cohorts.

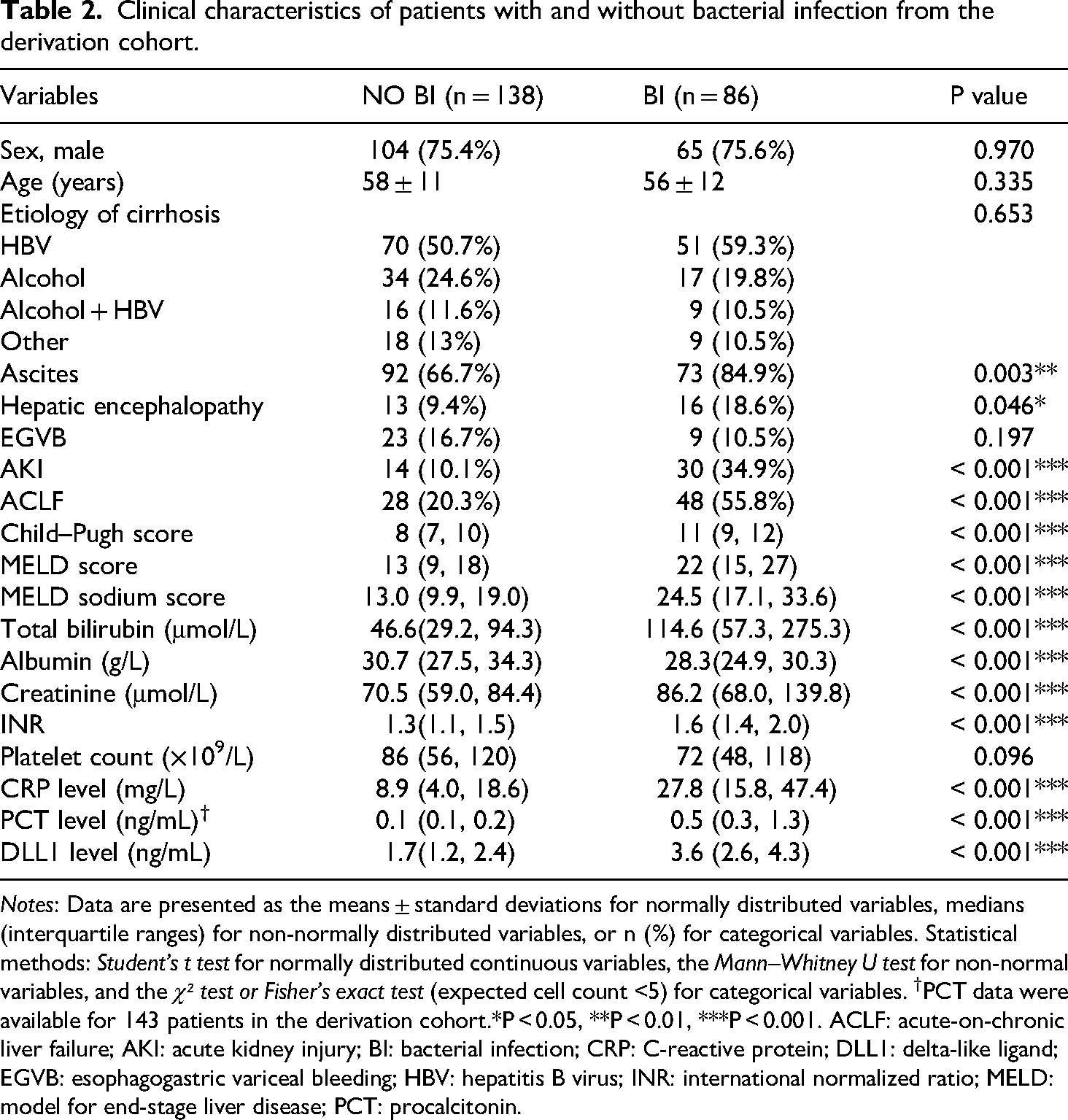
Patients with BI presented higher Child–Pugh scores and model for end-stage liver disease (MELD) sodium scores; creatinine, CRP, PCT, and DLL1 levels; and INRs and lower albumin levels (P < 0.05 for all) than patients with NO BI did in both cohorts (Table 2 and Supplemental Table 2). No significant differences were observed in age, sex, etiology, or platelet count (P > 0.05). Serum DLL1 levels were significantly elevated in patients with BI compared with NO BI controls in the derivation cohort (P < 0.001; Figure 1(a)), but no significant differences in the expression levels of DLL1 were observed among different infection sites (Figure 1(b)).
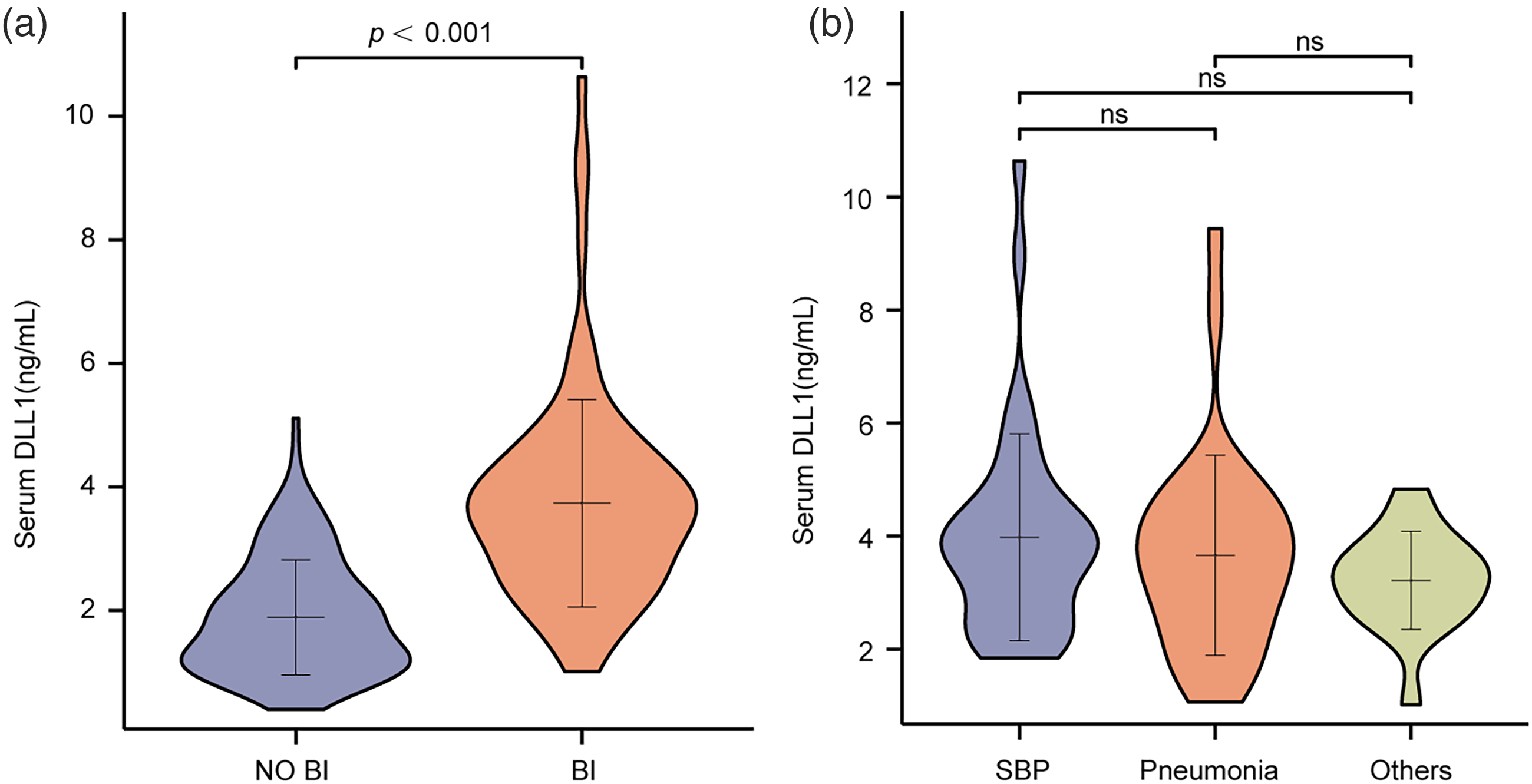
Differential expression of DLL1 according to infection status and infection site in decompensated cirrhosis patients (derivation cohort). (a) Comparison of serum DLL1 levels between patients with BIs and noninfected controls in the derivation cohort. Compared with non-BI controls, patients with BI presented significantly elevated DLL1 levels (P < 0.001, Mann–Whitney U test). (b) Analysis of DLL1 expression stratified by infection type, including SBP, pneumonia, and others. No statistically significant differences were observed across infection types (Kruskal–Wallis test, P = 0.214). BI: bacterial infection; DLL1: Delta-like ligand 1; SBP: spontaneous bacterial peritonitis.
Clinical characteristics of patients with and without bacterial infection from the derivation cohort.
Independent predictors of BIs
Univariate analysis of the derivation cohort (Supplemental Table 1) revealed that the Child–Pugh score (odds ratio (OR) = 1.725, 95% CI: 1.459–2.039), MELD sodium score (1.161, 1.112–1.213), albumin level (0.884, 0.833–0.939), CRP level (1.070, 1.046–1.094), and DLL1 level (3.836, 2.685–5.480) were significant predictors of BI (P < 0.001). Ascites, AKI, ACLF, TBIL level, creatinine level, and INR were also associated with BI but lost significance in the multivariate analysis. Multivariate regression analysis (Table 3) revealed four independent predictors of BI in the derivation cohort: the Child–Pugh score (adjusted OR (aOR) = 1.714, 1.110–2.645; P = 0.015), MELD sodium score (1.121, 1.007–1.249; P = 0.037), CRP level (1.068, 1.032–1.105; P < 0.001), and DLL1 level (5.495, 3.022–9.992; P < 0.001). In the validation cohort (Supplemental Table 3), the Child–Pugh score (aOR = 1.987, 1.014–3.892; P = 0.045), CRP level (1.045, 1.014–1.077; P = 0.004), and DLL1 level (4.698, 2.332–9.467; P < 0.001) remained significant in the multivariate analysis.
Independent predictors of bacterial infections in decompensated cirrhosis patients from the derivation cohort.

Diagnostic performance analysis
In the derivation cohort (Figure 2(a)), the combined model (CRP + DLL1) achieved superior discriminative performance (AUC = 0.918, 95% CI: 0.882–0.953) compared with the CRP model (AUC = 0.787, 0.726–0.849) and DLL1 model (0.863, 0.814–0.911) alone (DeLong test: CRP vs. combined P < 0.001; DLL1 vs. combined P = 0.002). At the optimal cut-offs (Table 4), the DLL1 model demonstrated greater sensitivity (0.826 vs. 0.774) and specificity (0.754 vs. 0.710) than did the CRP model, with the combined model further increasing the specificity to 0.906.
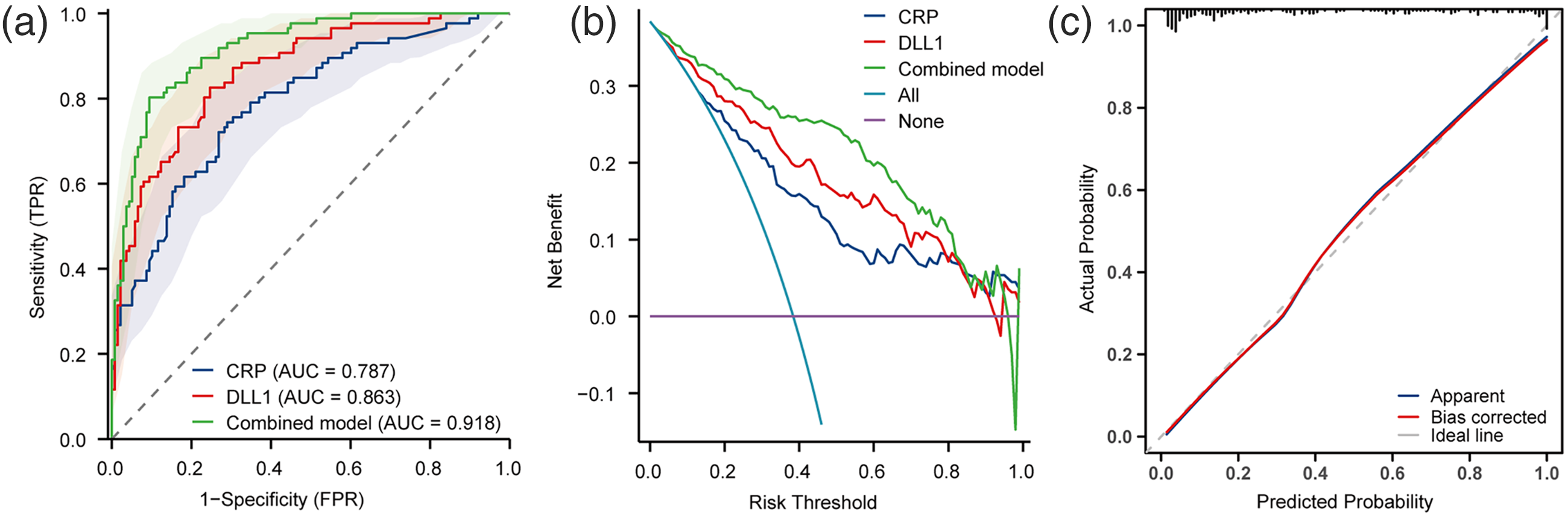
Diagnostic performance of serum biomarkers for BI (derivation cohort). (a) ROC curves for the CRP, DLL1, and combined models for the diagnosis of BI in patients with decompensated cirrhosis. DeLong's test revealed the following: CRP versus DLL1 (P = 0.062), CRP versus the combined model (P < 0.00), and DLL1 versus the combined model (P = 0.002). (b) DCA revealed that the combined model provided a net benefit superior to that of the CRP or DLL1 models alone across threshold probabilities of 10%-80%. (c) Calibration curve: the observed versus predicted probabilities aligned closely with the ideal line (Hosmer–Lemeshow, P = 0.73), indicating good model fit. The integrated analysis highlights the additive diagnostic value of combining CRP and DLL1 levels, with improved discrimination (AUC), clinical utility (DCA), and calibration accuracy. AUC: area under the curve; CRP: C-reactive protein; DCA: decision curve analysis; DLL1: delta-like ligand 1; ROC: receiver operating characteristic.
Diagnostic performance of individual and combined biomarkers for bacterial infections in the derivation cohort.

The validation cohort results (Supplemental Figure 2A and Table 4) confirmed the robust performance: the AUCs for the CRP, DLL1, and combined models were 0.836 (0.754–0.919), 0.881 (0.815–0.947), and 0.925 (0.877–0.974), respectively (DeLong test: CRP vs. combined P = 0.016; DLL1 vs. combined P = 0.048).
DCA (Figure 2(b)) revealed that the combined model provided greater net benefit than the CRP or DLL1 model alone across threshold probabilities of 10% to 80% in the derivation cohort. In the validation cohort (Supplemental Figure 2B), superiority persisted at the 10% to 30% and 40% to 100% thresholds. Calibration curve analysis (Figure 2(c), Supplemental Figure 2C) revealed excellent model fit (Hosmer–Lemeshow P = 0.73 and 0.35 for the derivation and validation cohorts, respectively).
In the subgroup with PCT data, CRP (AUC = 0.796), PCT (AUC = 0.819), and DLL1 (AUC = 0.901) showed comparable diagnostic efficacy(Figure 3(a)).The CRP–DLL1 dual-marker model demonstrates superior diagnostic accuracy compared to CRP–PCT combination and individual biomarkers (CRP, DLL1, and PCT) in the PCT-included subgroup (all DeLong's P < 0.05, Figure 3(b)). As shown in Supplemental Table 5, the PCT-included cohort demonstrated significantly higher age, hepatic encephalopathy frequency, ACLF prevalence, liver severity scores (Child–Pugh, MELD, MELD sodium), total bilirubin, and CRP levels, along with lower albumin and INR values compared to the NO PCT-included cohort (all P < 0.05), indicating substantial baseline disparities between groups.

Diagnostic performance of the CRP, PCT, DLL1, and combined models in the subgroup with PCT data. PCT data were available for 143 patients in the derivation cohort and 59 patients in the validation cohort. (a) CRP (AUC = 0.796), PCT (AUC = 0.819) and DLL1 (AUC = 0.901) showed comparable diagnostic efficacy. (b) The CRP–DLL1 dual-marker model demonstrates superior diagnostic accuracy compared to CRP–PCT combination and individual biomarkers (CRP, DLL1, PCT) in the PCT-included subgroup (all DeLong's P < 0.05). AUC: area under the curve; CRP: C-reactive protein; DLL1: delta-like ligand 1; PCT: procalcitonin.
Correlations between DLL1 levels and clinical parameters
The DLL1 level was not correlated with age (Spearman's |ρ| < 0.10) but was weakly associated with the Child–Pugh score, TBIL level, albumin level, and platelet count (|ρ| < 0.30; P < 0.001 for all; Supplemental Table 6, Figure 4). Moderate correlations of the DLL1 level with the MELD score (ρ=0.318), MELD sodium score (0.368), creatinine level (0.309), INR (0.319), CRP level (0.335), and PCT level (0.394) were observed (P < 0.001 for all).

Correlations between DLL1 levels and the levels of key clinical parameters (derivation and validation cohorts). (a) Spearman's correlation coefficient: total bilirubin level (R = 0.229, P < 0.001); (b) serum creatinine level (ρ=0.314, P = 0.002); and (c) CRP level (ρ=0.335, P < 0.001). CRP: C-reactive protein; DLL1: Delta-like ligand 1.
Discussion
A recent global epidemiological study on cirrhosis-associated infections revealed that the prevalence of infections at admission among hospitalized patients was 31.9%, with higher rates observed in lower-middle-income countries (LMICs) and lower rates of positive bacterial cultures. 25 Infections were associated with a fourfold increase in mortality risk for patients with cirrhosis, and the risk increases in parallel with the number of failing organs.26,27 Therefore, it is imperative to explore diagnostic biomarkers for BIs in patients with cirrhosis. Multiple studies have validated the diagnostic efficacy of DLL1 in sepsis.18,28 In this prospective cohort study with an independent validation cohort, the role of DLL1 in the diagnosis of BIs in patients with decompensated cirrhosis was evaluated. The main findings were as follows: (a) serum DLL1 levels were significantly elevated in the BI group compared with the NO BI group, with no significant differences observed across different infection sites; (b) the DLL1 level emerged as an independent predictor of BI; (c) the DLL1 level demonstrated robust diagnostic performance for BI, and the combined model (CRP + DLL1) achieved superior discriminative ability, DCA confirmed its clinical utility, and those results maintained robust in the independent validation cohort; and (d) the DLL1 level exhibited only weak correlations with the TBIL level, suggesting weak confounding effects from hepatic inflammation or dysfunction.
Patients with decompensated cirrhosis and concurrent BI at hospital admission frequently present with cirrhosis-related complications (ascites, hepatic encephalopathy, and AKI), higher composite liver function scores (Child–Pugh and MELD sodium scores), and a higher prevalence of ACLF; the most common infection types in this study were spontaneous bacterial peritonitis and pulmonary infections, and these findings align with findings from previous epidemiological studies.24,29 Among the independent predictors of infection, only the Child–Pugh score, MELD sodium score, CRP level, and DLL1 level retained statistical significance (PCT was excluded from the model owing to substantial missing data). These findings underscore the need for heightened vigilance against BIs in patients with advanced liver cirrhosis, necessitating prompt investigation and timely antibiotic treatment if BI is confirmed. In contrast, other individual markers (TBIL, albumin, and creatinine levels and the INR) showed no significant associations after adjustment, which aligns with the findings of previous research. 8
CRP, an inflammatory marker, is widely used in clinical practice for diagnosing and monitoring infections. However, CRP levels increase in various inflammatory conditions, including viral infections and noninfectious inflammatory responses. 30 In patients with cirrhosis, sustained inflammatory responses due to impaired liver function further compromise its reliability as an infection marker. 31 PCT is an effective biomarker for diagnosing severe BIs. Studies have revealed a moderate positive correlation between TBIL and PCT levels, necessitating diagnostic cut-off value adjustments based on TBIL levels. 32 In patients with AKI, their elevated PCT levels may compromise the predictive accuracy of PCT for infections, necessitating higher diagnostic thresholds. 33 Furthermore, PCT has limited diagnostic specificity in critically ill patients with compromised immunity. 34 This study also revealed that patients with decompensated cirrhosis and BI had a significantly greater baseline prevalence of AKI, along with elevated TBIL and serum creatinine levels, than NO BI patients did. This study revealed that CRP was an independent predictor of BIs (aOR = 1.068, 95% CI: 1.032–1.105; P < 0.001). At a cut-off value of 16.05 mg/L, the CRP level demonstrated a sensitivity of 77% for diagnosing BI, albeit with suboptimal specificity (71%). In the subgroup analysis of patients with PCT data available, this cohort exhibited significantly higher disease severity, as evidenced by elevated Child–Pugh and MELD sodium scores. While CRP, PCT, and DLL1 demonstrated comparable diagnostic efficacy as individual biomarkers, the CRP-DLL1 dual-marker model showed superior diagnostic accuracy compared to both the CRP–PCT combination and single-marker approaches. This finding underscores the robustness of the CRP–DLL1 model in decompensated cirrhosis patients with advanced disease severity.
Hildebrand et al. first reported that plasma DLL1 levels were significantly elevated during the early phase of sepsis, whereas surgical and trauma patients presented no such elevation. 18 Hölle et al. subsequently confirmed these findings, demonstrating that septic patients exhibited substantially higher DLL1 levels than did those with nonseptic infections or noninfectious systemic inflammation; importantly, DLL1 demonstrated superior diagnostic performance over conventional biomarkers, including CRP, PCT, and leukocyte count, for sepsis identification. 35 Further evidence from Schneck et al. revealed that septic shock patients had markedly elevated DLL1 levels relative to postoperative cohorts (abdominal surgery and coronary artery bypass grafting), establishing its predictive value for both sepsis progression and AKI. 36 Notably, a secondary analysis of the Sodium Selenite and Procalcitonin Guided Antimicrobial Therapy in Severe Sepsis (SISPCT) trial provided critical insights: DLL1 levels were significantly higher in patients with septic shock than in uncomplicated sepsis patients; these elevations were correlated with infection foci (abdominal infections, primary bacteremia, and surgical site infections) and pathogen type (gram-negative infections); and DLL1 was independently associated with mortality. 37 This study is the first to establish DLL1 as a novel diagnostic biomarker for BI in patients with decompensated cirrhosis. While DLL1 demonstrated numerically superior sensitivity (82.6% vs. 77.4%) and specificity (75.4% vs. 71.0%) compared with CRP, this difference did not reach statistical significance (DeLong test, P = 0.062), potentially due to sample size limitations. Notably, the combined CRP-DLL1 model achieved significantly improved diagnostic performance in both the derivation (AUC = 0.918) and validation cohorts (AUC = 0.925). However, dynamic variations in sensitivity and specificity between cohorts may be attributable to baseline imbalances (e.g. pathogen distribution or infection severity) and threshold discrepancies, highlighting the need for context-specific dynamic threshold adjustment strategies.
Mechanistically, the weak correlation of DLL1 with liver function indices (e.g. TBIL, Spearman's ρ=0.24) suggests reduced interference from hepatic inflammatory “noise,” a critical advantage over CRP and PCT in patients with end-stage liver disease. Despite moderate correlations with renal dysfunction (creatinine, ρ=0.31) and conventional inflammatory markers (CRP and PCT), DLL1 maintained independent predictive value for BI regardless of the MELD sodium score or Child–Pugh score, supporting its potential as a “liver-agnostic” biomarker reflective of distinct pathophysiological pathways. The synergistic diagnostic performance improvement achieved by combining DLL1 and CRP underscores their complementary roles in infection detection. As a pivotal ligand in the Notch signaling pathway, DLL1 plays critical roles in immune regulation by modulating the differentiation and functional polarization of immune cells, thereby strengthening host defence mechanisms against pathogens. 38 Mechanistic studies revealed that DLL1-mediated Notch signaling specifically promotes the differentiation of Ly6Chi monocytes into Ly6Clo-p patrolling monocytes, a process markedly amplified under inflammatory conditions. 39 In the context of sepsis, DLL1-Notch crosstalk governs monocyte/macrophage functional reprogramming, directly influencing the magnitude and temporal dynamics of systemic inflammatory responses. 40 Furthermore, emerging evidence highlights the vascular regulatory role of DLL1: the endothelial expression of DLL1 modulates vascular barrier integrity and inflammation-associated permeability through Notch-dependent mechanisms, suggesting a dual role in both immune coordination and tissue homeostasis maintenance during infection.17,41,42
This study has several limitations that warrant consideration. The single-centre cohort design may limit the generalizability of the findings across diverse clinical settings. The temporal dynamics of DLL1 levels (including their potential decline posttreatment and prognostic implications) remain unexplored. While subgroup analyses were conducted to mitigate biases from incomplete PCT data, the possibility of residual confounding cannot be fully excluded. Mechanistic validation through targeted experimental models is required to determine the exact pathophysiological role of DLL1 in BIs complicating decompensated cirrhosis.
Conclusion
In this study, serum DLL1 was established as a novel diagnostic biomarker for BIs in patients with decompensated cirrhosis. The combined CRP–DLL1 model demonstrated synergistic diagnostic performance improvement with cross-cohort validation robustness, significantly outperforming conventional biomarkers. The weak correlation between DLL1 and TBIL levels suggests a reduced susceptibility to liver-related confounding factors compared with existing markers.
Supplemental Material
sj-doc-1-sci-10.1177_00368504251358310 - Supplemental material for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study
Supplemental material, sj-doc-1-sci-10.1177_00368504251358310 for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study by Juanjun Huang, Luhu Yu, Debin Zeng, Yulin Wang, Zhi Wang, Jian Chen and Wei Zhu in Science Progress
Supplemental Material
sj-doc-2-sci-10.1177_00368504251358310 - Supplemental material for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study
Supplemental material, sj-doc-2-sci-10.1177_00368504251358310 for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study by Juanjun Huang, Luhu Yu, Debin Zeng, Yulin Wang, Zhi Wang, Jian Chen and Wei Zhu in Science Progress
Supplemental Material
sj-doc-3-sci-10.1177_00368504251358310 - Supplemental material for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study
Supplemental material, sj-doc-3-sci-10.1177_00368504251358310 for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study by Juanjun Huang, Luhu Yu, Debin Zeng, Yulin Wang, Zhi Wang, Jian Chen and Wei Zhu in Science Progress
Supplemental Material
sj-doc-4-sci-10.1177_00368504251358310 - Supplemental material for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study
Supplemental material, sj-doc-4-sci-10.1177_00368504251358310 for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study by Juanjun Huang, Luhu Yu, Debin Zeng, Yulin Wang, Zhi Wang, Jian Chen and Wei Zhu in Science Progress
Supplemental Material
sj-doc-5-sci-10.1177_00368504251358310 - Supplemental material for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study
Supplemental material, sj-doc-5-sci-10.1177_00368504251358310 for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study by Juanjun Huang, Luhu Yu, Debin Zeng, Yulin Wang, Zhi Wang, Jian Chen and Wei Zhu in Science Progress
Supplemental Material
sj-doc-6-sci-10.1177_00368504251358310 - Supplemental material for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study
Supplemental material, sj-doc-6-sci-10.1177_00368504251358310 for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study by Juanjun Huang, Luhu Yu, Debin Zeng, Yulin Wang, Zhi Wang, Jian Chen and Wei Zhu in Science Progress
Supplemental Material
sj-doc-7-sci-10.1177_00368504251358310 - Supplemental material for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study
Supplemental material, sj-doc-7-sci-10.1177_00368504251358310 for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study by Juanjun Huang, Luhu Yu, Debin Zeng, Yulin Wang, Zhi Wang, Jian Chen and Wei Zhu in Science Progress
Supplemental Material
sj-doc-8-sci-10.1177_00368504251358310 - Supplemental material for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study
Supplemental material, sj-doc-8-sci-10.1177_00368504251358310 for A Serum DLL1 and CRP dual-marker model for bacterial infection detection in patients with decompensated cirrhosis: A dual-cohort diagnostic study by Juanjun Huang, Luhu Yu, Debin Zeng, Yulin Wang, Zhi Wang, Jian Chen and Wei Zhu in Science Progress
Footnotes
Abbreviations
Acknowledgements
The authors would like to thank the American Journal Experts (AJE) for English language polishing of the paper.
Ethical considerations
This study was approved by the Clinical Research Ethics Subcommittee of Ganzhou Hospital-Nanfang Hospital, Southern Medical University (Ganzhou People's Hospital) (TY-ZKY2023-023-1).
Author contributions
JH and JC were involved in conceptualization and funding acquisition; JH in data curation; LY, WZ, and JH in formal analysis; LY, WZ, JC, DZ, YW, and ZW in investigation; WZ and JH in methodology; JH in project administration; WZ in software and visualization; JC and ZW in validation; JH and WZ in writing—original draft. All the authors contributed to writing—review & editing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ganzhou Science and Technology Program Project (grant numbers: 2022-YB1449, 2023LNS17536).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data are available upon reasonable request from the authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
