Abstract
Transjugular intrahepatic portosystemic shunt (TIPS) stands as a pivotal interventional therapy for managing portal hypertension (PH) and its associated complications. Distinguished from pharmacological treatments, endoscopic interventions, or surgical portocaval shunts, TIPS has achieved extensive clinical application attributed to its minimally invasive nature and efficacy. Notwithstanding, complications ensuing from TIPS, such as hepatic encephalopathy (HE) and shunt dysfunction, etc, significantly impact patient prognosis. Over the course of several decades, a diverse array of stent types and diameters have become available for TIPS procedures. Moreover, the stent puncture position and the initial stent position also exhibit variability. Our review aims to address this gap in knowledge by reviewing the research progress on stent types, diameters, puncture positions, and initial stent positions in the context of TIPS. It presents a comprehensive overview of the various stent options available for TIPS procedures and highlights the importance of individualized treatment approaches in reducing surgical complications and improving patient prognosis. The findings outlined in our review provide valuable insights for clinicians and researchers in the field.
Introduction
Transjugular intrahepatic portosystemic shunt (TIPS) refers to a minimally invasive interventional technique that involves puncturing the portal vein from the hepatic vein through the jugular vein approach to establish a portosystemic shunt between the hepatic vein and the portal vein, thereby reducing portal vein pressure and treating complications of portal hypertension (PH) such as esophageal-gastric variceal bleeding (VB), refractory ascites and so on. 1 Currently, this technique is only used to treat PH complications, but also be used for the secondary prevention of esophageal-gastric VB. In addition, Budd–Chiari syndrome, hepatorenal syndrome, hepatopulmonary syndrome, hepatic hydrothorax, portal hypertensive gastropathy, and hepatic veno-occlusive disease have also become recognized indications for TIPS. TIPS is highly effective in the early relief of PH, and it is generally superior to conventional medical and endoscopic therapy in the prevention of recurrent VB and in the treatment of acute VB, refractory ascites, and hepatic pleural effusion.2,3 But postoperative complications such as hepatic encephalopathy (HE) and shunt dysfunction limit its clinical application. Selecting the appropriate stent may effectively prevent TIPS complications while ensuring clinical efficacy, improving patient survival rates and shunt patency rates, which is crucial for improving patient prognosis.4,5 This review summarizes the research progress on stent types, diameters, puncture positions, and initial stent positions in TIPS (Figure 1). This review is a narrative review and is guided by the Scale for the Assessment of Narrative Review Articles (SANRA). 6

Research progress of TIPS stents.
Stent types
Bare stent
In the 1990s, there was no special stent, and bare metal stents were used instead, with the Wallstent and Palmaz stent being representative. It was a self-expanding memory alloy stent made by laser engraving. The control rate for esophageal-gastric VB that was unresponsive to drug and endoscopic treatment could reach over 90%. 7 However, a concurrent issue was that the stenosis rate of the shunt channel after TIPS with bare stents could be as high as 60% and 85% at 1 and 2 years, respectively, and the obstruction of the intrahepatic shunt channel had an adverse effect on the long-term efficacy of TIPS. 8 A randomized controlled trial (RCT) compared the efficacy of covered versus bare stents in TIPS for PH. The recurrence rate of gastrointestinal bleeding (18.3%) and refractory pleural effusion/ascites (6.9%) in the covered stent group were significantly lower than those in the bare stent group (33.9%, 16.5%). The 1 -, 2 -, 3 -, 4 -, and 5-year cumulative restenosis rates in the covered stent group were 6.9%, 11.5%, 19.1%, 26.0%, and 35.9%, respectively, which were significantly lower than those in the bare stent group (27.6%, 37.0%, 49.6%, 59.8%, and 74.8%) (P < 0.001). 9 Due to the obvious advantages of later TIPS-specific covered stents over bare stents in terms of improving the shunt patency, reducing rebleeding rates, controlling the incidence of postoperative HE, and increasing survival rates, bare stents have gradually been phased out in clinical practice.
As the main limiting factor, in-stent restenosis after bare stent implantation is more common in the liver parenchyma and the hepatic vein end of the shunt. The causes may be as follows: (1) proliferation of pseudomembrane: mechanical injury of intrahepatic bile duct caused by the metal end of the stent, bile leakage into the shunt, resulting in excessive proliferation of inflammatory pseudomembrane in the stent. 10 (2) In-stent thrombosis: the process of intraoperative puncture and balloon dilatation can cause damage to hepatic parenchymal cells, intrahepatic bile duct injury, and bile leakage into the puncture tract, leading to inflammatory reaction, starting the coagulation system, and then leading to thrombosis. 11 (3) Mechanical damage to the hepatic vein intima caused by the operation, leading to excessive proliferation of the hepatic vein intima. (4) Inaccurate positioning during stent placement, resulting in stent displacement after stenting. 12
Covered stent
Covered stents not only retain the support function of bare metal stents, but also improve the long-term patency of the shunt by using the membrane to block the shunt. Since 1995, there have been a variety of covered material TIPS stents, such as polyethylene terephthalate (PET), polytetrafluoroethylene (PTFE), polyurethanes (PUs), silicone, etc. 13 Finally, through animal experimental research and clinical research, PTFE was a kind of material with good biological compatibility, the probability of rejection reaction was very small, and its unique micron porous structure could well reduce the penetration of bile, and then played a good control role in the proliferation of pseudointima, which can effectively maintain the patency of the shunt. Therefore, it has become the first choice of film covered material. 14 At present, Fluency stent and Viatorr stent are the two most commonly used PTFE-covered stents for TIPS in clinical practice.
The body of Fluency stent is composed of a Nitinol alloy stent, which is covered with an ultrathin double layer of expanded PTFE membrane, and the two ends of the metal bare area are 2 mm long. The inner membrane contains a carbon coating that can inhibit platelet aggregation, and the release method of this stent is in a backward form, which can achieve segmental release effect. It is mainly used in peripheral vascular surgery. 15 Nonetheless, there are some problems in its use. If the stent is placed too deep in the portal vein, it is easy to cause an excessively large angle with the blood vessels after its release, so that the blood flow in the stent can not pass through, which is easy to cause thrombosis. If the stent is constricted inside the liver or on the “upper wall” of the hepatic vein during placement, it will lead to the occurrence of “capping.” 16
The Viatorr stent was developed as a special covered stent for TIPS, which solved some of the problems of Fluency stent. It used self-expandable Nitinol alloy as the main material and was divided into two parts: covered segment and bare segment. The anterior segment of the stent was a 2 cm bare metal segment for easy insertion into the portal vein. In the posterior segment, it was enclosed by three layers of ultra-thin expanded PTFE membrane with different pore sizes, which was applied to the intrahepatic shunt placement and completely blocked the bile leakage in the shunt. Compared with Fluency stent, its bare metal segment design reduced the risk of hepatic blood flow and liver injury. 17 According to the study, compared with patients using Fluency stent alone, the stent patency rate after TIPS using Viatorr stent was significantly improved. 16 Before the Viatorr stent officially came into clinical application in China in 2015, double-stent technology, namely bare stent + Fluency stent, was used to simulate the effect of Viatorr stent in domestic clinical practice. Although the results suggested that the shunt patency rate, quality of life and survival rate were similar between the two groups, 18 further research is needed to confirm. What is clear is that the economic costs were lower in patients with double-stent than in those with Viatorr covered stent. In 2024, HepaFlow (MircoPort), a TIPS-specific covered stent independently developed in China, was officially approved for review by the National Medical Products Administration. Based on the anatomical characteristics of blood vessels in Chinese patients, it has been designed with as many as 25 size specifications, which can better achieve precise treatment. Furthermore, a study has confirmed the feasibility and effectiveness of TIPS using covered stents in children with PH. 19
Controlled diameter stent
In 2017, Viatorr CX (VCX), a new generation of PTFE covered stent with controllable expansion, was launched in the United States. It added a balloon expandable cannula, the surgeon controlled the balloon expansion externally, and adjusted the diameter of the stent. The stent diameter could be adjusted from 8 to 10 mm and maintained at a constant diameter for precise control of postoperative portal venous pressure gradient (PPG). 20 A study has found 21 that the clinical symptoms of refractory HE can be improved by reducing the diameter of the VCX stent from 10 mm to 8 mm. The median PPG reduction was 10 mmHg in the VCX group and 12 mmHg in the conventional covered stent group (P = .02). Some scholars believe that the emergence of VCX solve the passive expansion characteristics of nickel-titanium memory alloys. A cohort research of 75 cirrhotic patients with TIPS using VCX stents was evaluated by CT to determine whether the 8-mm balloon-expanded VCX stent maintained its diameter in vivo. 22 The results showed that there was no correlation between the diameter of the stent and time after interventional treatment, and there was no significant change in the diameter of the stent over time. In the case of insufficient shunt with the VCX stent, the diameter of stent could be further expanded to increase the blood flow during the secondary interventional procedure. Notwithstanding, this study included a small number of cases and a short follow-up time, and its conclusions still need to be further verified. At present, there are few clinical data on VCX stents, and VCX stents with a diameter of 6–12 mm may become available in the future.
The latest study in the United States 23 evaluated the feasibility and effect of the new Liverty TIPS stent (BD) in a porcine model. It has been shown that the Liverty stent has advantages in delivery and positioning, and can create a smaller caliber shunt to meet the needs of different patients. However, further prospective human evaluation is needed to validate the findings.
Stent diameters
The optimal shunt diameter of TIPS stent is still controversial. Insufficient shunt may lead to shunt dysfunction and fail to reduce portal vein pressure, while excessive shunt may significantly reduce liver perfusion, leading to an increased risk of HE and liver function deterioration. 24 Therefore, the choice of shunt diameter should balance the needs of portal vein decompression and maintenance of liver perfusion. Whether an 8 mm or 10 mm stent can achieve this delicate balance remains controversial.
Recurrence rate of VB
A large RCT demonstrated that 25 8 mm covered stents in TIPS effectively prevent VB in patients with Child-Pugh grade A/B cirrhosis, showing better outcomes than pharmacological interventions. A study 26 revealed no statistically significant difference in postoperative VB recurrence rates between 8 mm and 10 mm stents. In summary, the above studies indicated that the 8 mm stent does not reduce the hemodynamic success rate or the efficacy of VB prevention.
Recurrence rate of ascites
A retrospective study comparing the treatment of refractory ascites with covered stents of different diameters showed 27 that more patients needed abdominal paracentesis for drainage of ascites after operation with 8 mm stents than with 10 mm stents (58% vs. 31%, P = .003). The higher recurrence rate of ascites in the 8 mm stent in the study may be associated with higher PPG.
Survival rate
A study have shown that both 8 mm and 10 mm stents have no significant effect on the survival rate of VB patients. 26 Nonetheless, one study 28 included 53 patients with VB who were implanted with 8 mm stents and 132 patients with other indications were implanted with 10 mm stents. The results showed that 8 mm stents could prolong the survival time of patients compared with 10 mm stents, and multivariate analysis showed that 1-year mortality was associated with complete stent expansion to 10 mm, and the risk was increased by 2 times. These results indicated that the 8 mm stent is expected to improve the survival rate of patients with VB and refractory ascites treated with TIPS.
Incidence of shunt dysfunction
A study 29 compared the shunt patency rate of VB patients implanted with 8 mm stent and 10 mm stent during the follow-up period, and the results showed that there was no significant difference. However, several studies26,30 showed that more patients with cirrhosis implanted with 8 mm stent needed secondary interventional treatment than those implanted with 10 mm stent (P < .05), but the median interval between secondary interventional treatment with 8 mm stent was longer regardless of whether the 10 mm stent was fully dilated or not (P < .05). Which diameter stent is superior in terms of shunt patency rate and patency time needs to be further confirmed by more high-quality multicenter RCTs.
Incidence of HE
A study has shown 21 that 8 mm stent placement does not have any advantage in reducing the incidence and severity of HE in patients with refractory ascites. Notwithstanding, a study 27 found that the incidence of HE after 8 mm stent implantation in VB patients was significantly lower than that of 10 mm stent (P = .034). Other related study has also confirmed 31 that the 8 mm stent implantation is beneficial to reduce the incidence of HE in patients with VB, and the risk rate is reduced by 47%, and the dominant HE rate can be reduced, which is lower than the average level (20%–31%). Therefore, reducing stent diameter may reduce the incidence of postoperative HE in patients with VB.
Stent puncture positions and initial stent positions
In-stent restenosis after TIPS is associated with multiple factors. Among them, the position of puncture into the liver from the hepatic vein, the position of puncture into the portal vein from the liver and the position of stent are closely related to the patency of stent after TIPS.32,33 Generally, the position of puncture into the liver is called point A, and the position of puncture out of the liver is called point B, with the shunt channel between these two points forming the initial stent position. Sufficient inflow blood flow, short, smooth and straight shunt and unobstructed outflow tract are the essential conditions to ensure the patency of TIPS stents. 34
Choice of point A
It is challenging to perform TIPS via the left hepatic vein due to the left hepatic vein has small diameter and near-vertical orientation relative to the inferior vena cava. 35 In the early TIPS, the right hepatic vein was selected as point A, and the right branch of portal vein was selected as point B, and the shunt was mostly in the shape of “C.” However, the relatively smaller diameter of the right hepatic vein led to a sharp increase in blood flow post-shunting, creating localized vortices and turbulence. Combined with puncture-related injury and mechanical stimulation of the stent, these leaded to dysfunction after shunt. Studies have shown that point A is selected 1–2 cm distal to the hepatic vein-inferior vena cava confluence with a better patency rate.36,37 In recent years, many scholars 38 used the retrohepatic inferior vena cava as point A to perform direct intrahepatic portocaval shunt (DIPS) between the inferior vena cava and portal vein branches. DIPS was mainly used in patients with cirrhosis who have atrophy of the hepatic vein or liver, and when portal vein occlusion makes it difficult to implement traditional TIPS. DIPS was indicated for patients with partial or all of the retrohepatic inferior vena cava was surrounded by the caudate lobe of the liver, and the large vessel diameter at this point allowed free selection of the puncture kit to find the best point B. Related anatomical and imaging studies have also confirmed the safety and feasibility of DIPS.3,39
Choice of point B
Historically, the right branch of portal vein was mostly selected as point B, and the success rate of operation was high. With the continuous improvement of technology, more and more scholars began to consider which of the left and right branches of portal vein puncture is more beneficial to patients. Studies have been conducted on the puncture of intrahepatic bifurcation, right branch and left branch of portal vein respectively.36,40 The results showed that the puncture of left branch of portal vein could significantly reduce the incidence of primary in-stent restenosis and HE, and the position of portal vein puncture is the influencing factor of HE after TIPS. A recent study have found that the incidence of HE in the short term after TIPS in patients who chose the left branch of portal vein puncture was lower than that in patients who chose the right branch of portal vein puncture, but there was no significant difference in the incidence of HE in the long term. 15 This is because the blood from the left branch of the portal vein mainly supplies the smaller left lobe of the liver, and even if the blood is cut off, only about a quarter of the liver function is impaired, while the blood from the right branch of the portal vein supplies most of the liver, and if it is diverted, about three quarters of the liver function is impaired. It directly enters the inferior vena cava through the shunt and causes hyperammonemia. In addition, right liver failure leads to HE after TIPS. The left branch of portal vein mainly comes from the splenic vein, so the left branch of portal vein puncture for TIPS will reduce the incidence of HE. In addition, the left branch of the portal vein is a continuation of the main portal vein, and the shunt from the left branch of the portal vein to the hepatic vein is straight, which is not easy to turbulent flow and cause platelet accumulation and thrombosis. Whereas, the shunt established by the right branch of the portal vein is close to an acute angle, which is prone to intimal hyperplasia in the stent and then cause shunt stenosis.32,41,42 Therefore, choosing the origin of the left branch of the portal vein as point B may not only improve the stent patency rate, but also reduce the occurrence of HE and in-stent restenosis.
Initial stent positions
Studies have compared the clinical outcomes of TIPS stents at different implantation positions. Studies revealed that when the distal end of a TIPS stent is positioned at the hepatic vein-inferior vena cava confluence, it demonstrated higher patency rates and longer patency times compared to positioned at the hepatic vein.43,44 The latest study 45 also recommended that the distal end of the stent should reach the confluence of the hepatic vein and the inferior vena cava. The prima reason was that if the distal end of the stent does not completely cover the hepatic vein, the local blood flow at the outlet of the shunt will increase, the pressure will increase, and it is easy to produce vortex or turbulence. Combined with the mechanical stimulation of the hepatic vein wall by the stent, these may eventually cause vascular intimal damage and thrombosis over time. Additionally, the tip of the stent should be parallel to the portal vein wall as much as possible to avoid angulation, so as to reduce the impact of the main portal vein blood flow on the angulation and avoid shunt stenosis. Therefore, a study recommended the left branch of the portal vein should be chosen for puncture, and the distal end of the stent should reach the confluence of the hepatic vein and the inferior vena cava. 43 Currently, the definition of optimal initial stent position needs to meet two criteria: (1) the distal end of the stent extends to the hepatic veno-inferior vena cava confluence and extends into the inferior vena cava about 1 cm (avoid contact with the opposite side of the inferior vena cava); and (2) the proximal end of the stent was parallel to the portal vein wall.
Studies have shown that DIPS creates a smoother shunt.46–48 The average distance between the starting parts of the left or right branches of the portal vein, and the inferior vena cava in the posterior hepatic segment at the same level was 22.14–49.40 mm. Therefore, the shunt length of DIPS is generally within 50 mm, so that the short, smooth and straight shunt of DIPS has a better long-term patency rate. However, for patients with a large amount of ascites, the ascites compression of the liver causes the liver to move down, and the original retrohepatic inferior vena cava is exposed in the abdominal cavity. At this time, DIPS will increase the risk of surgery, so the puncture should be performed from the hepatic vein as far as possible. If it is necessary to puncture from the retrohepatic inferior vena cava, careful evaluation should be performed. Ensure that there is liver tissue surrounding point A.49,50
Conclusion
In clinical practice, individualized treatment plans should be made according to the specific conditions of patients to maximize their benefits. In conclusion, in practice, the small diameter PTFE covered stent is more inclined to be selected, 1–2 cm distal to the hepatic vein-inferior vena cava confluence is selected as point A, and the left branch of portal vein is selected as point B, and the stent is implanted to the best initial position (Figure 2). The VCX stent can be selected in hospitals where conditions permit, and expansion to 8 mm is encouraged as routine, with further modification and expansion if necessary (Table 1). DIPS may be used as an alternative scheme when TIPS cannot be implemented (Table 2). In the future, new TIPS stents with better biocompatibility and antithrombosis ability can be developed to improve the patency rate of stents and reduce complications. The innovation of this review is to comprehensively sort out the related research progress of the application of stents in TIPS, so as to provide a reference for clinicians to choose the appropriate TIPS stent. There are also limitations, most evidence arises from retrospective single-center cohorts, larger scale multicenter RCTs should be carried out to verify the differences in effects of different stent types in the future.
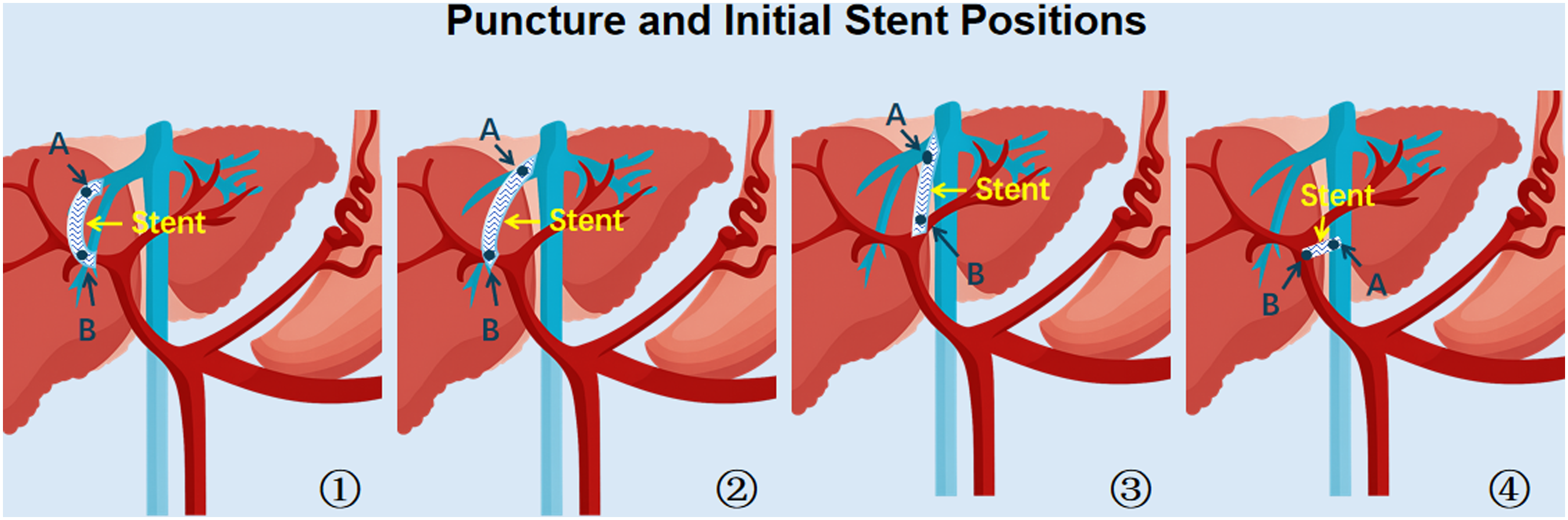
①: Point A is in the right hepatic vein, point B is in the right portal vein, and the shunt is in the shape of “C.” ②: Point A is in the 1–2 cm distal to the hepatic vein-inferior vena cava confluence, point B is in the right portal vein. ③: Point A is in 1–2 cm distal to the hepatic vein-inferior vena cava confluence, point B is in the left portal vein. ④: DIPS, point A is in the retrohepatic inferior vena cava, point B is in the portal vein branch.
Table for the comparative analysis of postoperative outcomes of different stent types.

Table for the comparative analysis of different point B.

Footnotes
Abbreviations
Acknowledgments
The authors are grateful to all the research participants and the staff that were involved in the study.
Ethics approval
This study, which is a review article, does not include any studies with human or animal participants.
Author contributions
Conceptualization: Lei Qi. Data curation: Yihao Wang. Resources, supervision, funding acquisition: Yihao Wang and Jian Zhou. Formal analysis: Yihao Wang, Wen Li, and Shuangshuang Li. Investigation, methodology: Lei Qi, Yihao Wang, and Wen Li. Project administration: Lei Qi, Yihao Wang, and Jian Zhou. Writing—original draft: Lei Qi. Writing—review & editing: Lei Qi and Shuangshuang Li.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Project of Integration Team of Tengfei Engineering [TF2024YZRH01].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
