Abstract
Background
Inflammation is a key component in the pathophysiology of hypertension, in which proinflammatory cytokines interleukin (IL) 1β and IL6 and anti-inflammatory cytokines IL4 and IL10 are involved. Elevated remnant cholesterol (RC) level has emerged as an independent risk factor of incident hypertension, surpassing conventional risk factors based on its strength. Additionally, it has a higher penetration into the arterial wall than conventional lipids and induces arterial wall and systemic inflammations.
Objective
This study aimed to investigate the association between cerebrospinal fluid (CSF) ILs, blood pressure (BP), and RC and examine the moderation effects of RC on the relationship between ILs and BP.
Methods
This cross-sectional study included participants (mean age: 31.76 ± 10.22 years) who were divided into RC < 0.8 mmol/L (n = 81) and RC ≥ 0.8 mmol/L (n = 104) groups. BP was measured on the morning before CSF collection, and CSF IL levels were analyzed using ELISA. The linear regression models were constructed to estimate the moderation of RC on the relationship between ILs and BP.
Results
RC may play a moderation role in the relationship between IL10 and BP (R2 = 0.11, β = –0.31 for systolic BP [SBP] and R2 = 0.11, β = –0.28 for diastolic BP [DBP], both p < 0.001). BMI exhibited a positive correlation with the BP (R2 = 0.09, β = 0.28 for SBP and R2 = 0.05, β = 0.24 for DBP, both p < 0.05). CSF IL4 exhibited a negative correlation with triglycerides among males in the RC ≥ 0.8 mmol/L group (r = –0.25, p = 0.013).
Conclusions
This study demonstrated previously undocumented evidence regarding the potential protective effect of CSF IL10 on hypertension in individuals with RC ≥ 0.8 mmol/L. This indicates that elevated CSF IL10 levels may aid in maintaining BP for overall health, particularly in individuals with elevated total cholesterol levels.
Introduction
Hypertension affects over one billion adults globally, and approximately 45% of the adult population is diagnosed with the disease. 1 Hypertension is a disease and a significant risk factor for other diseases. Population studies reported an increasing incidence of cardiovascular events, including stroke and myocardial infarction, which are associated with elevated blood pressure (BP; systolic BP [SBP] ≥ 115 mmHg). This relationship is exponential and more pronounced for SBP than for diastolic BP (DBP). 2 However, the mechanisms of BP elevation have not been fully elucidated.
Increasing evidence reveals that hypertension is a chronic inflammatory condition. 3 Inflammation is a key component in the pathophysiology of hypertension. 4 Patients with hypertension exhibit elevated basal levels of inflammatory cytokines, including interleukin (IL) 1β and IL6. 5 IL1β modifies endothelial, immune, and central nervous system responses, thereby enhancing the development of hypertension. 6 Pharmacological blockade of IL1 signaling 7 and administration of an IL1β neutralizing antibody therapy 8 reduce BP. IL6 can induce hypertensive responses to angiotensin II infusion in normotensive individuals. 9 A previous Mendelian randomization study reported that IL6 causally influences the development of SBP rather than DBP or hypertension. 10 However, anti-inflammatory agents may have the opposite effect. The pleiotropic cytokine IL4 exhibits various effects. IL4 inhibits the production and secretion of IL1β and suppresses IL6 secretion and activities, 11 thereby exerting anti-inflammatory effects. 12 However, substantial evidence indicates that IL4 may promote inflammation 13 and has been associated with the development of pulmonary hypertension. 14 IL10 is a multifunctional cytokine with potent anti-inflammatory properties. 15 Endogenous IL10 may limit the pressor effect. 16 IL10 supplementation can lower BP in male hypertensive mice. 17 Despite extensive study, the etiology of human hypertension remains controversial. BP regulation is a complex process that has recently been indicated to involve central inflammatory mechanisms. Increased circulating inflammatory cells and cytokines in the brain can impair central BP regulation, causing hypertension, 18 especially cytokines released from brain cells. 19 A previous study reported that high cholesterol induces neuroinflammation, 20 while statins may exhibit anti-inflammatory properties by activating mechanisms responsible for brain protection. 21 Consequently, investigating the correlation between cholesterol, neuroinflammation, and BP is imperative.
Previous studies have reported that the total cholesterol (TC) level is positively correlated with BP in general populations and in patients with hypertension. 22 Emerging evidence indicates an association between remnant cholesterol (RC) and hypertension. 23 RC is a class of cholesterol content of triglyceride (TG)-rich lipoproteins. 24 RC exhibited higher penetration into the arterial wall than conventional lipids and induced arterial wall and systemic inflammation. 25 However, elevated RC has emerged as an independent risk factor of incident hypertension, surpassing the conventional risk factors. Monitoring RC levels and implementing interventions to lower RC are beneficial in preventing hypertension. 26 Despite the statement above, the impact of RC on the association between neuroinflammation and BP is unclear.
As a result, this study aimed to calculate RC using the literature 27 and measure IL levels in cerebrospinal fluid (CSF) to investigate the association between these ILs, RC, and BP and the moderation effect of RC on the transition from neuroinflammation to hypertension.
Materials and methods
Study design
This retrospective cross-sectional study followed the Strengthening the Reporting of Observational Studies in Epidemiology guidelines (https://www.strobe-statement.org/). 28
RC was calculated as TC−high-density lipoprotein (HDL)−low-density lipoprotein (LDL). 27 The cases comprised participants with RC ≥ 0.8 mmol/L, while the controls comprised individuals with RC < 0.8 mmol/L matched to the cases. 29
Participants
Although hypertension affects both sexes, young women are more often protected than age-matched men. 17 Therefore, this study consecutively recruited 185 Chinese male participants with a mean age of 31.76 ± 10.22 years who were scheduled for anterior cruciate ligament reconstruction surgery between September 2014 and January 2016. Of the 185 participants, 81 RC levels < 0.8 mmol/L, while 104 participants exhibited RC levels ≥ 0.8 mmol/L based on their fasting RC.
Sociodemographic data, including age, years of education, and body mass index (BMI), were collected from all participants. Clinical data, including a history of substance abuse and dependence, was obtained through self-report, which was subsequently confirmed by the next of kin and family members. Exclusion criteria included: (1) a family history of psychosis or neurological disorders; (2) the presence of systemic or central nervous system diseases and sleep disorders, as assessed by the Mini-International Neuropsychiatric Interview; (3) systemic inflammatory or autoimmune diseases; and (4) current use of anti-inflammatory medications.
All recruited participants had no history of substance abuse or dependence, except for cigarette smoking, and they had no other psychiatric disorders as defined by the criteria in the Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, before the publication of the 5th Chinese version. No participants reported receiving any antihypertensive medication.
This study was conducted in accordance with Declaration of Helsinki (1975), as revised in 2024 and was approved by the Institutional Review Board of Inner Mongolia Medical University (YKD2014031). All participants provided written informed consent, received no financial compensation for their participation, and all identifying information or data that could potentially reveal participant identity were removed.
Biological sample collection and laboratory tests
All participants submitted self-reported information on their smoking status, specifying whether they were nonsmokers or active smokers. Additionally, no participants disclosed any sleep disorders and had received a diagnosis of any sleep disorders.
Both peripheral blood sampling and lumbar puncture were performed in a fasting state, with a duration between the two procedures of less than 24 hours. Subsequently, each CSF sample was aliquoted into ten 0.5 mL tubes and immediately frozen at −80 °C. The optimal protocol for collecting and storing plasma/CSF samples involves dividing them into multiple aliquots for single use and storing them at −80 °C to prevent multiple freeze-thaw cycles that can lead to cytokine degradation. The duration between hospitalization and surgery did not exceed 5 days.
On the morning of the second hospital day following an overnight fasting, the BP was measured using a mercury column sphygmomanometer. Additionally, the levels of peripheral metabolic markers, including HDL, LDL, alanine aminotransferase (ALT), TC, TG, gamma-glutamyl transferase (GGT), and aspartate aminotransferase (AST), were measured using biochemistry analyzer (HITACH 7600, Hitachi Co., Tokyo, Japan).
ELISA kits were used to measure CSF levels of IL1β, IL4, IL6, and IL10 (Cat. SEA073Hu, SEA077Hu, SEA079Hu, and SEA056Hu, Cloud-clone Corp., Katy, TX, USA) by laboratory technicians anonymized to the clinical data of the participants.
Statistical analysis
In the descriptive analysis, continuous variables were presented as means ± standard deviations (SD), and categorical variables as frequencies and percentages. The Shapiro–Wilk and Levene tests were used to assess the normality of distribution and homogeneity of variances, respectively. Variables meeting these assumptions were analyzed using Analysis of Covariance (ANCOVA), while those that did not were evaluated using the Wilcoxon rank-sum test. Categorical variables were compared using the chi-square test. Partial correlation analyses were conducted stratified by RC groups (< 0.8 or ≥ 0.8 mmol/L), with age, BMI, and smoking status included as covariates to examine associations among continuous variables. The calculation method of moderation effect was same as the literature. 30
Three linear regression models were utilized to estimate the adjusted R-square of IL10 associated with BP. Model 1 was adjusted for age, BMI, and smoking status (nonsmoker or active smoker). Model 2 included additional adjustments for IL10 and the RC groups (< 0.8 or ≥ 0.8 mmol/L). Model 3 served as a complete model by further adjusting for the interaction between IL10 and the RC groups.
In the current study, part of CSF IL1β, IL4, IL6, and IL10 data was previously reported. 31 It is important to note that the current analysis employs a different methodological approach, which has led to the detection of new positive results. Therefore, we would like to acknowledge the use of this variable's data from the previous study and clarify that the present findings are based on an independent and distinct analytical methodology.
Statistical Package for the Social Sciences software for Windows (version 22.0; IBM Corp., Armonk, NY, USA) was used for statistical analysis. GraphPad Prism software (version 9; GraphPad Software Inc.) was used to generate figures. All tests were two-sided, and the significance threshold was set at p < 0.05.
Results
Demographic and clinical characteristics
The normality of all the variables was performed using the Kolmogorov–Smirnov test, and only the distribution of SBP and DBP was normally distributed (all p > 0.05). Therefore, the Mann–Whitney rank sum test was used.
Compared to individuals who exhibited < 0.8 mmol/L, those with ≥ 0.8 mmol/L exhibited significantly higher BMI (24.17 ± 3.75 vs. 26.25 ± 3.78, p < 0.001) and higher LDL, ALT, TC, TG, GGT, and AST levels (2.45 ± 0.58 vs. 2.82 ± 0.66, 26.20 ± 20.51 vs. 33.92 ± 23.54, 4.35 ± 0.67 vs. 5.11 ± 0.79, 1.28 ± 0.58 vs. 2.18 ± 1.19, 35.98 ± 34.81 vs. 48.73 ± 40.32, and 19.32 ± 6.40 vs. 22.45 ± 9.72, all p < 0.05) and significantly lower HDL (1.30 ± 0.29 vs. 1.21 ± 0.22, p = 0.034). No significant differences were observed between the two groups based on age, education, glucose, and smoking status (Table 1).
Differences in demographic and clinical characteristics.
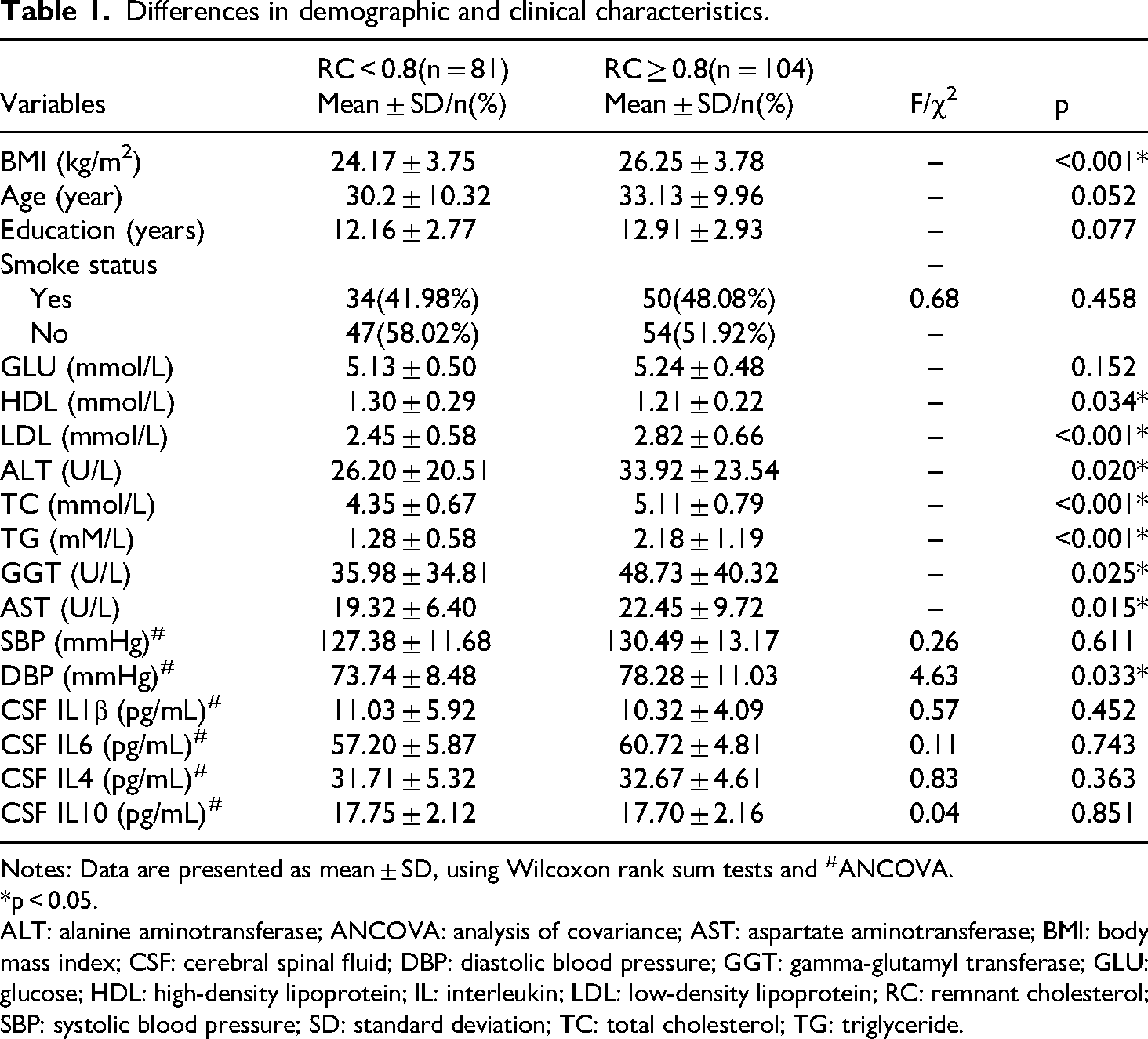
Notes: Data are presented as mean ± SD, using Wilcoxon rank sum tests and #ANCOVA.
*p < 0.05.
ALT: alanine aminotransferase; ANCOVA: analysis of covariance; AST: aspartate aminotransferase; BMI: body mass index; CSF: cerebral spinal fluid; DBP: diastolic blood pressure; GGT: gamma-glutamyl transferase; GLU: glucose; HDL: high-density lipoprotein; IL: interleukin; LDL: low-density lipoprotein; RC: remnant cholesterol; SBP: systolic blood pressure; SD: standard deviation; TC: total cholesterol; TG: triglyceride.
Differences and correlation analysis of CSF ILs
CSF IL levels and BP were compared as continuous variables using ANCOVA between individuals who exhibited < 0.8 mmol/L and those who exhibited ≥ 0.8 mmol/L, with age, BMI, and smoking status as covariates (all p > 0.05). Additionally, the ≥ 0.8 mmol/L group exhibited higher DBP than the ≤ 0.8 mmol/L group (73.74 ± 8.48 vs. 78.28 ± 11.03, p = 0.033; Table 1).
Partial correlation analysis was performed to examine the correlation between CSF ILs and BP, with age, BMI, and smoking status as covariates.
Negative correlations were observed between CSF IL10 and DBP (r = –0.27, p = 0.006), CSF, IL4, and TG (r = –0.27, p = 0.006) among individuals who exhibited RC ≥ 0.8. However, no correlation was found among those who exhibited < 0.8 mmol/L between CSF ILs and other continuous variables (all p > 0.05, Table 2).
Correlation analysis between baseline characteristics.

Note: All the data are reported as partial correlation analysis, with BMI, age, and smoke status as covariates.
*p < 0.05.
ALT: alanine aminotransferase; AST: aspartate aminotransferase; BMI: body mass index; CSF: cerebral spinal fluid; DBP: diastolic blood pressure; GGT: gamma-glutamyl transferase; GLU: glucose; HDL: high-density lipoprotein; IL: interleukin; LDL: low-density lipoprotein; RC: remnant cholesterol; SBP: systolic blood pressure; TC: total cholesterol; TG: triglyceride.
Moderation analysis
Moderation models were employed to examine the association among RC, ILs, and BP through regression analyses. This was aimed to determine if RC moderated the effects of CSF ILs on BP. Three separate moderation models were analyzed, and the results indicated the moderation effect of RC on the association between CSF IL10 and SBP/DBP (Tables 3 and 4).
Linear regression table for moderation analysis of SBP.

Note: Model 1: adjusted for age, BMI, and smoke status. Model 2: Model 1 plus adjustment for RC group and CSF IL10. Model 3: Model 2 plus adjustment for RC group and CSF IL10 interactions. All data are presented as moderation analysis. *
BMI: body mass index; CI: confidence interval; CSF: cerebral spinal fluid; IL10: interleukin10; RC: remnant cholesterol; SBP: systolic blood pressure.
Linear regression table for moderation analysis of DBP.

Note: Model 1: adjusted for age, BMI, and smoke status. Model 2: Model 1 plus adjustment for RC group and CSF IL10. Model 3: Model 2 plus adjustment for RC group and CSF IL10 interactions. All data are presented as moderation analysis. *p < 0.05.
BMI: body mass index; CI: confidence interval; CSF: cerebral spinal fluid; DBP: diastolic blood pressure; IL10: interleukin10; RC: remnant cholesterol.
Initially, we assessed the direct effect of bedtime on BP while controlling for age, smoking status, and BMI. The linear regression analysis revealed a significant positive correlation between BMI and BP (R2 = 0.09, β = 0.28, t = 4.18, p < 0.001 for SBP and R2 = 0.05, β = 0.24, t = 3.39, p = 0.005 for DBP), indicating that higher BMI correlates with higher SBP and DBP. Subsequently, we examined the impact of IL10 on BP while adjusting for age, BMI, smoking status, RC, and CSF IL10 levels. The linear regression analysis revealed that BMI significantly affected SBP (R2 = 0.09, β = 0.27, t = 3.89, p < 0.001), while BMI, RC, and CSF IL10 collectively had a significant effect on DBP (R2 = 0.09, β = –0.15, t = –2.13, p = 0.001).
We implemented moderation variables to assess the moderation effect of IL10 on BP through the moderation of neurotrophic factors. The linear regression analysis revealed a significant moderation effect of RC (R2 = 0.11, β = –0.31, t = –2.28, p < 0.001 for SBP and R2 = 0.11, β = –0.28, t = –2.04, p < 0.001 for DBP; Figure 1(A–B)).

(A) Simple slope analysis for the moderation effect of remnant cholesterol (RC) on the association between cerebral spinal fluid (CSF), interleukin (IL) 10, and systolic blood pressure (SBP). (B) Simple slope analysis for the moderation effect of RC on the association between CSF, IL10, and diastolic blood pressure (DBP).
Discussion
This is the first cross-sectional study that used CSF to investigate the association between ILs, RC, and BP. CSF IL10 exhibited a protective influence on SBP and DBP in individuals with RC ≥ 0.8 mmol/L, as indicated by the positive correlation between IL10 × RC and the BP of those with ≥ 0.8 mmol/L RC. This finding reinforces the idea that IL10 serves as a protective factor against hypertension, especially in individuals with high levels of RC.
The pathogenesis of primary hypertension is multifactorial and complex. Nonetheless, increased sympathetic nerve activity is strongly associated with elevated BP. Central angiotensin II (Ang II) plays a pivotal role by activating sympathetic outflow and initiating inflammatory cascades critical for BP regulation. 32 Specifically, sympathetic activation induces nuclear factor kappa B (NF-κB)-mediated release of IL6, 33 while Ang II-driven nicotinamide adenine dinucleotide phosphate oxidase generates reactive oxygen species (ROS). 34 Excessive ROS reduces nitric oxide bioavailability, resulting in endothelial dysfunction and sustained vascular inflammation in hypertension. 35
During this process, IL10 serves as a key regulatory cytokine with multiple protective effects. In the brain, IL10 pretreatment exerts neuroprotective effects by inhibiting the release of IL6 and IL1β, while upregulating brain-derived neurotrophic factor (BDNF), thereby preserving neuronal function within cardiovascular control centers. 36 In addition, IL10 modulates microglial activation, attenuates sympathetic overactivity, 37 and suppresses central ROS production. 34 These central anti-inflammatory effects further contribute to improved peripheral endothelial function through descending autonomic pathways. 38 Consistent with these mechanisms, IL10 has been shown to lower BP in males, and in this study, IL10 levels were negatively correlated with both systolic and diastolic BP.
Recent literature revealed that elevated cholesterol may induce neuroinflammation 20 and RC accumulation in the arterial wall, potentially propagating local and systemic inflammation. 39 RC denotes the cholesterol content in remnants. These remnants are a subset of lipoproteins that are rich in TGs. RC particles are larger and more abundant, posing a higher risk to arterial endothelium. 40 A previous study reported that RC is an independent predictor of endothelial dysfunction in the general population. 41 The strong association between RC and hypertension may be associated with compromised vascular endothelial function and arterial elasticity. Chinese populations have demonstrated that increased RC concentrations are associated with increased BP and the development of hypertension. 23 Additionally, RC is associated with hypertension independent of LDL cholesterol in the general adult population. 42 Under physiological conditions, IL10 helps maintain vascular homeostasis by suppressing proinflammatory cytokine production and promoting endothelial repair. 43 However, the protective effect of IL10 on BP appears to depend on the underlying inflammatory burden. In individuals with low RC, the baseline inflammatory state is minimal, and basal IL10 activity may be sufficient to preserve vascular integrity, making additional IL10 effects difficult to detect. In contrast, when RC levels are elevated (≥ 0.8 mmol/L), the heightened proinflammatory environment increases the demand for IL10’ s anti-inflammatory actions. Under these conditions, elevated CSF IL10 levels become crucial for counteracting RC-induced endothelial dysfunction and vascular inflammation, thereby exerting a measurable protective effect on BP regulation.
The secondary outcomes of our study demonstrated a negative correlation between CSF IL4 and TG in individuals with RC ≥ 0.8 mmol/L. Elevated TG levels may directly contribute to a systemic proinflammatory state
44
and are typically associated with increased TC.
45
However, the pleiotropic cytokine IL4 exhibits varied effects. IL4 inhibits the production and secretion of IL1β, suppressing IL6 secretion and activities,
11
thereby exerting an anti-inflammatory effect.
12
IL4, as an anti-inflammatory cytokine, prevents insulin resistance, glucose intolerance, and weight gain by activating lipid droplet fragmentation, lipolysis, and thermogenesis in mature adipocytes while enhancing TG turnover.
46
IL4 possesses prolipolytic properties by inhibiting adipocyte differentiation and lipid accumulation while facilitating lipolysis in mature adipocytes.
47
Furthermore, evidence from
Besides, this study found that BMI has a positive effect on SBP and DBP of individuals with RC ≥ 0.8 mmol/L, consistent with the findings of previous studies involving children and adults. 49 Furthermore, high BMI is a significant modifiable determinant of high BP, as demonstrated by meta-analyses of observational studies 50 and weight-loss interventions. 51 A significant association was identified for BMI even after adjusting for other covariates, indicating that overweight and obesity may directly contribute to the onset of hypertension and are implicated in its pathogenesis. 52 A 30-year prospective cohort study reported that long-term burden and increasing trends of BMI are associated with adult hypertension through the TG-glucose index, indicating that early weight management can potentially reduce hypertension burden stemming from childhood BMI increases. 53
Previous studies demonstrated that RC and IL10 contribute to cardiovascular regulation primarily through peripheral mechanisms.54–56 However, these investigations have largely overlooked central mechanisms, despite BP being ultimately regulated by central autonomic nuclei such as the paraventricular nucleus,57,58 where neuroinflammation characterized by decreased IL10 levels occurs during hypertension. 59 Our findings indicate that elevated RC may trigger peripheral-to-central inflammatory signaling via blood–brain barrier disruption by inflammatory mediators, leading to increased neuroinflammation within cardiovascular control centers. Under these conditions of heightened central inflammatory burden, the demand for IL10's anti-inflammatory actions increases substantially, making the protective effects of CSF IL10 on BP regulation detectable and clinically significant.
These findings identify CSF IL10 as a protective biomarker for BP regulation in individuals with elevated RC (≥ 0.8 mmol/L). Furthermore, given that current therapeutic strategies predominantly target peripheral mechanisms, these results suggest that resistant hypertension in dyslipidemic patients may reflect central neuroinflammation, warranting combined peripheral and central therapeutic strategies. Future studies should explore the underlying molecular mechanisms and assess whether cholesterol-modifying interventions can enhance IL10 function and improve hypertension management.
However, this study has certain limitations. First, the study population predominantly consisted of young male patients undergoing anterior cruciate ligament reconstruction surgery, which limits the generalizability of our findings to broader populations, including women, individuals from other age groups, and nonsurgical cohorts. Future studies should include more diverse demographics to establish the generalizability of these findings. Notably, while patients undergoing anterior cruciate ligament reconstruction surgery often experience acute inflammatory responses from the injury itself, 60 the inflammation is generally confined to the local area and has not been consistently associated with measurable changes in cerebrospinal fluid biochemical markers or BP alterations in many studies.61,62 Second, cholesterol-lowering drugs (statins) have not been recorded to reduce inflammation and significantly decrease IL10 expression. 20 However, this would not compromise the findings of this study. Third, this study did not exclude active smokers whose neuroinflammation level may be affected despite the prevalence of cigarette use among Chinese adults surpassing the global average. 63 Finally, BP was measured only once, which may not fully account for potential influences such as stress, circadian variation, or the white-coat effect. 64 However, because all measurements were conducted under uniform and standardized conditions, this limitation is unlikely to have differentially affected the study groups or compromised the validity of our comparative results.
Conclusion
This is the first study to demonstrate a potential protective effect of CSF IL10 on hypertension in young males with remnant cholesterol ≥ 0.8 mmol/L, suggesting that remnant cholesterol modulates the association between cerebral IL10 levels and BP regulation within this specific population.
Footnotes
Abbreviations
Acknowledgements
We thank all participants for their willingness to participate in the study and the time that they devoted to the study.
Ethics approval declaration
This study was conducted in accordance with the Declaration of Helsinki 2008 and was approved by the Institutional Review Board of Inner Mongolia Medical University (YKD2014031).
Author contributions
FW, XH, and YL designed the study. XY, LS, and JF led the drafting of the manuscript. YC finished the statistical analyses. LS collected the clinical data. YK, YC, and ZY input the data. XL responded to reviewer comments and polished the language. FW and XH secured funding for the study. All authors approved the final manuscript for submission.
Funding
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This work was supported by the following grants: the Technology Support Project of Xinjiang (grant number 2017E0267), the 10th Inner Mongolia Autonomous Region ‘Prairie excellence’ Project, Natural Science Foundation of Xinjiang Province (2018D01C228), Xinjiang Outstanding Youth Science Grant (2017Q007), Beijing Natural Science Foundation (7152074), Natural Science Foundation of Inner Mongolia (2016D01A022 and 2022MS08068), Youth project of Inner Mongolia Medical University (YKD2024QN012), and Urumqi Municipal Health Commission Science and Technology Plan Project (202236).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding authors. Requests to access these datasets should be directed to Fan Wang.
