Abstract
Objective
Immune–inflammatory dysregulation contributes to the pathophysiology of schizophrenia and other chronic disorders. Interleukin-6 (IL-6) and C-reactive protein (CRP) are established inflammatory biomarkers, yet their blood-based assessments are costly, time-consuming, and invasive. The Immune Status Questionnaire (ISQ), a brief self-report measure, may offer a noninvasive alternative for identifying individuals who require further immune evaluation. This study examined the associations among ISQ, IL-6, CRP, and physiological factors in patients with schizophrenia.
Methods
This retrospective chart review included 94 patients with schizophrenia, among whom 74 completed both blood sampling and the ISQ assessment. Hierarchical linear regression and structural equation modeling (SEM) were used to examine associations between immune markers and physiological variables.
Results
ISQ and cholesterol significantly predicted CRP levels, whereas IL-6 and cholesterol remained significant in the final regression model. SEM confirmed these pathways, showing that age, ISQ, and cholesterol influenced CRP through IL-6 mediation. Receiver operating characteristic (ROC) analyses identified an ISQ cutoff score of 10 and an IL-6 threshold of 3.0 pg/mL, yielding an area under the curve of 0.59 and a negative predictive value of 72.2%.
Conclusions
The ISQ demonstrates modest but practical potential as a noninvasive preliminary screening tool for immune dysfunction in schizophrenia. Given its accessibility and low cost, the ISQ may complement laboratory testing during early clinical evaluation. However, due to the limited sample size and retrospective design, further large-scale, community-based studies are warranted to validate its reliability, discriminative ability, and broader clinical applicability.
Keywords
Introduction
Immune–inflammatory functioning plays a central role in determining overall health status and contributes to the pathophysiology of many chronic diseases, including psychiatric disorders.1–3 Dysregulation of inflammatory processes has been implicated in depression, bipolar disorder, and particularly schizophrenia, where immune activation may influence both symptom severity and disease progression.4–8
Traditionally, immune status is evaluated through objective biomarkers such as cytokines, chemokines, and antibodies.9,10 However, such blood-based assessments are costly, invasive, and time-consuming, which limits their use for frequent monitoring or large-scale screening. To address this limitation, self-reported assessments that capture individuals’ perceived immune functioning have been proposed as an accessible alternative.11–14 The Immune Status Questionnaire (ISQ) is one such measure, reflecting recent fluctuations in immune-related symptoms and general well-being. 14 Although subjective, ISQ scores may indicate physiological immune changes triggered by stress, infection, or lifestyle factors.12–15
C-reactive protein (CRP) and interleukin-6 (IL-6) are well-established biomarkers of inflammation and are often elevated in schizophrenia.4,5,16–20 CRP, a nonspecific acute-phase protein, is secreted by hepatocytes and induced by cytokines—particularly IL-6.5,16,21–23 IL-6 is a multifunctional cytokine that regulates immune signaling, inflammation, and even neurotransmission.22,24,25 Elevated IL-6 and CRP levels have been associated with increased risk for a range of psychiatric and neurodevelopmental disorders, including depression, bipolar disorder, autism, and schizophrenia.24,26–30 These findings support the hypothesis that immune dysregulation contributes to the development and chronicity of psychiatric conditions.
Nevertheless, access to laboratory-based cytokine testing remains limited in many clinical settings, including Taiwan, because such tests are expensive and not routinely available. 14 Moreover, recent studies have highlighted that inflammatory activity may vary dynamically, not only as a stable trait marker but also in response to short-term stressors, sleep disruption, and metabolic fluctuations.31,32 Consequently, there is a clinical need for a simple, noninvasive, and cost-effective tool that can serve as an initial indicator of immune functioning before proceeding to blood-based biomarker testing.
Schizophrenia is increasingly recognized as a chronic inflammatory disorder characterized by fluctuating immune activity influenced by environmental stress, sleep disturbances, and metabolic factors.4,20,33–35 Therefore, using a short-term self-assessment such as the ISQ may still capture clinically relevant immune variability reflective of these fluctuations.
Thus, the present study aimed to evaluate whether the ISQ could serve as a preliminary indicator of immune status among patients with schizophrenia by comparing ISQ scores with serum IL-6 and CRP levels. Furthermore, this study investigated the interrelationships among ISQ, IL-6, CRP, and other physiological factors through hierarchical regression and structural equation modeling (SEM) to clarify the potential pathways linking subjective immune perception with objective inflammatory markers.
Methods
Ethical approval and sample size estimation
This retrospective chart review study was reviewed and approved by the Institutional Review Board of Kaohsiung Armed Forces General Hospital, Taiwan (KAFGH 106-046) on 21 May 2018. All procedures were conducted in accordance with the ethical standards of the Institutional Review Board and the Declaration of Helsinki (1975), as revised in 2024. The ethics approval was originally granted for the Cassia obtusifolia clinical trial, 36 from which the present secondary analysis data were derived. The reporting of this study also conforms to the Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 37 Using G*Power software (effect size = 0.17, α = 0.05, power = 0.8), the estimated sample size required was 76. Considering a potential dropout rate of approximately 20%, at least 92 participants were planned for recruitment.
Participants
Participants were originally recruited as part of a previously published randomized, double-blind, controlled clinical trial 36 entitled “Oral administration of processed Cassia obtusifolia L. seed powder may reduce body weight and cholesterol in overweight patients with schizophrenia: A 36-week randomized, double-blind, controlled trial of high and low doses.” The present analysis was conducted as a retrospective chart review and secondary analysis of data collected during that clinical trial. In addition to metabolic and clinical assessments, the ISQ was administered to evaluate participants’ perceived immune functioning.
A total of 94 patients with schizophrenia were consecutively recruited between July 2018 and January 2020 from both outpatient and inpatient psychiatric units in southern Taiwan. All diagnoses were made by board-certified psychiatrists according to the criteria of the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). 38 Eligible participants were required to be conscious, clinically stable, and not pregnant or lactating. Individuals with serious medical comorbidities such as cancer, congestive heart failure, or other systemic diseases were excluded. Among them, 74 participants completed both blood sampling for biomarker analysis and the ISQ self-report questionnaire. Demographic and clinical characteristics, including age, sex, education, and marital status, were recorded. All participant data were fully deidentified prior to analysis to ensure confidentiality and anonymity.
Materials
The ISQ was developed by Wilod Versprille et al. in 2019, 14 based on the 23-item Immune Fitness Questionnaire. 39 The ISQ comprises seven symptom items: “sudden high fever,” “diarrhea,” “headache,” “skin problems (e.g. acne or eczema),” “muscle and joint pain,” “common cold,” and “coughing.” Each item is rated on a five-point Likert scale from 0 (never) to 4 (always).
The ISQ was originally developed and validated under the Creative Commons Attribution (CC BY 4.0) license, 14 which permits unrestricted use, adaptation, and distribution for research purposes with proper citation. It is a self-report instrument designed to assess perceived immune functioning over the past 12 months and is publicly available as an open-access tool.
Additionally, two global rating questions assess perceived immune functioning and general health on a 0–10 scale (0 = very poor, 10 = excellent). Participants were also asked whether they currently experienced reduced immune functioning or had any chronic diseases. In the original validation, Cronbach's α ranged from 0.63 to 0.84. 14 In the present study, the ISQ demonstrated excellent internal consistency (Cronbach's α = 0.92), indicating strong reliability for clinical research.
Laboratory measures
Blood samples were collected by venipuncture in the morning after an overnight fast. Plasma was separated and analyzed for IL-6 and CRP levels. IL-6 concentrations were quantified using the BD™ Cytometric Bead Array Human IL-6 Flex Set (Cat. No. 558276; BD Biosciences, San Jose, CA, USA) according to the manufacturer's instructions. The assay had a detection range of 10.2–2500 pg/mL and a theoretical limit of detection of 1.6 pg/mL, analyzed on a BD FACS Array flow cytometer using FCAP Array™ Software.
CRP levels were measured using a latex agglutination turbidimetric assay (CRP Latex X2 “Seiken”; Denka Seiken Co., Ltd, Niigata, Japan), with an analytical range of 0.01–32 mg/dL. The reaction was measured photometrically at 800 nm in accordance with the manufacturer's protocol.
All assays were performed following the manufacturers’ recommendations, and quality control procedures were carried out prior to analysis.
Statistical analysis
Data were analyzed using SPSS 26.0 for Windows for descriptive and regression analyses. AMOS 26.0 was used for SEM to test hypothesized relationships among variables. SEM model fit was evaluated by the χ2 test, the adjusted goodness-of-fit index (AGFI > 0.90), and the root mean square error of approximation (RMSEA ≤ 0.10). 40
Results
A total of 74 patients with schizophrenia, including 52 males and 22 females, with a mean age of 48.51 years (SD = 9.03), were recruited. The demographic parameters distribution are shown in Table 1.
Basic demographic parameters distribution.

Note: IL-6: interleukin-6; CRP: C-reactive protein.
Hierarchical linear regression
Hierarchical linear regression analysis was conducted to identify predictors of CRP levels (Table 2). In Model 1, the variables age (β = 0.077, p = .758) and ISQ (β = 0.961, p = .202) were not statistically significant. After adding cholesterol in Model 2, ISQ (β = 1.143, p = .004) and cholesterol (β = 0.076, p < .001) became significant predictors of CRP. In Model 3, which further included IL-6, cholesterol (β = 0.061, p < .001) and IL-6 (β = 1.112, p < .001) remained significant.
Hierarchical linear regression analysis of predictors of CRP.

Note: IL-6: interleukin-6; CRP: C-reactive protein; ISQ: Immune Status Questionnaire.
DV: CRP.
**p < .01.
Structural equation modeling
A parsimonious SEM further confirmed the hierarchical regression model (Figure 1). The model demonstrated excellent fit indices (χ2 = n.s., p = .784; AGFI = 0.961; RMSEA = 0.000), suggesting that the hypothesized structure adequately represented the data.

The parsimonious model of the contributing factors for CRP.
Age showed a significant influence on ISQ (β = 0.31, p = .006), IL-6 (β = 0.54, p < .001), and CRP (β = 0.40, p < .001), whereas cholesterol also positively predicted IL-6 (β = 0.40, p < .001) and CRP (β = 0.67, p < .001). The model explained 45% of the variance in IL-6 levels and 83% in CRP levels.
Receiver operating characteristic and graphical analyses
To evaluate the diagnostic performance of the ISQ, the receiver operating characteristic (ROC) curve was plotted using IL-6 as reference biomarkers. The area under the curve (AUC) for the ISQ was 0.59 (95% CI [0.41–0.70]), indicating fair but limited discriminative ability (Figure 2).
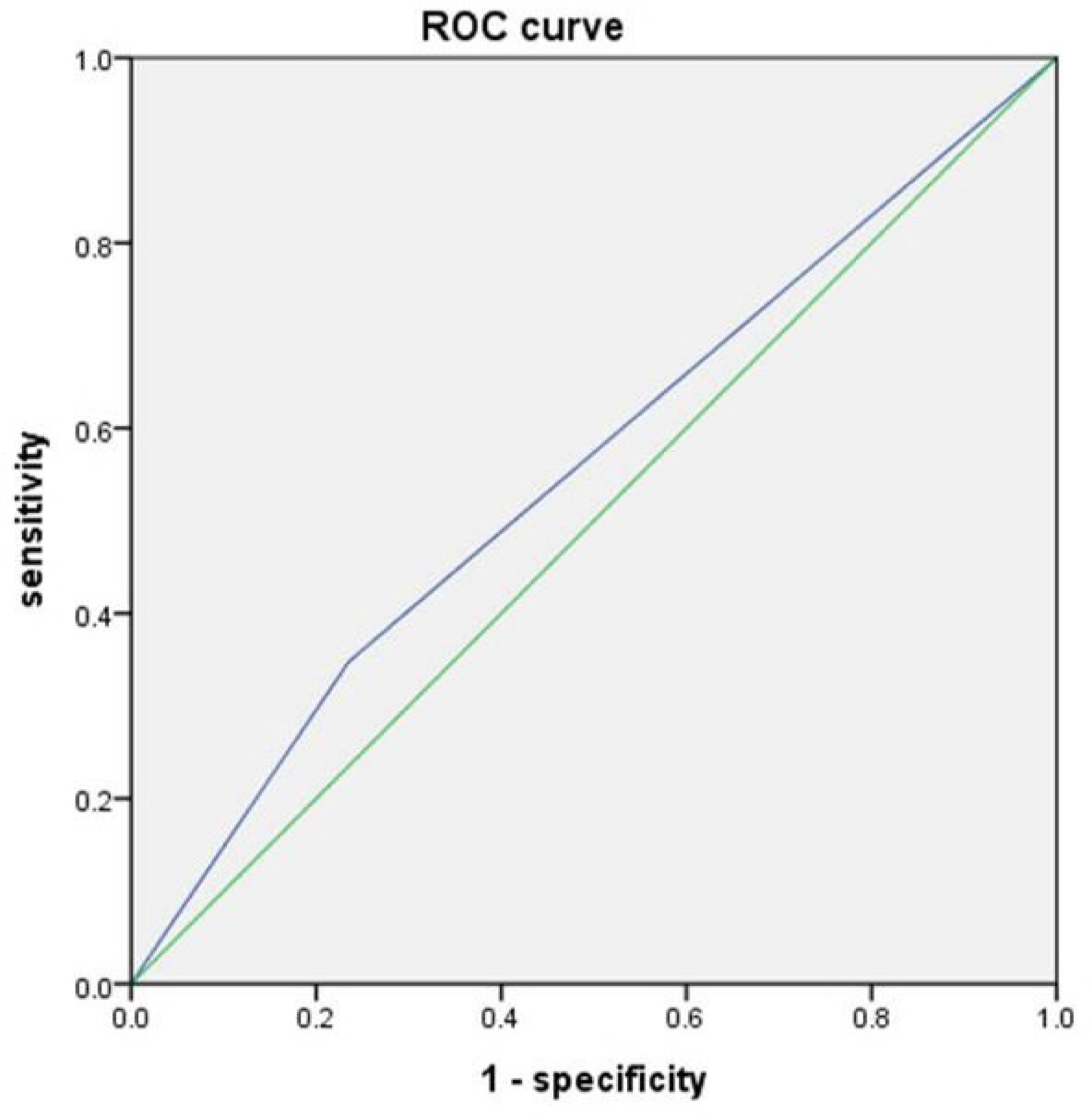
The receiver operating characteristics curve graph.
Because the sample size was relatively small and the population consisted of clinical schizophrenia patients, the optimal cutoff points for the ISQ and IL-6 were identified using the graphical method rather than the ROC-based Youden's index, by visually determining the intersection of sensitivity and specificity curves.
A cutoff score of 10 on the ISQ and 3.0 pg/mL on IL-6 were established as reference thresholds. At these cutoff points, the ISQ demonstrated a sensitivity of 34.78%, specificity of 76.47%, positive predictive value of 40%, and negative predictive value (NPV) of 72.22%.
Discussion
The ISQ is a brief tool designed to rapidly assess an individual's perceived immune status over the past 12 months. 14 It is believed that the ISQ can be applied as an alternative screening measure for immune function to determine whether further blood assessment is required. The biomarkers of inflammation, CRP and IL-6, are commonly used indicators of immune activation. 41 However, blood-based assessments are costly, time-consuming, and invasive. The present study demonstrated that the ISQ was strongly associated with CRP levels through the mediating role of IL-6, supporting its potential use as a noninvasive screening indicator.
In the present study, the ISQ demonstrated excellent internal consistency (Cronbach's α = .92), indicating strong reliability for clinical research. Although the sample size was relatively small, it consisted exclusively of individuals with schizophrenia—a diagnostically specific and biologically homogeneous group—which may explain the high internal consistency observed. Schizophrenia has long been recognized as a disorder with a strong immune and inflammatory component, and increasing evidence supports its characterization as a neurodevelopmental and neuroimmune-related brain disorder rather than a purely functional psychiatric illness.42,43 This rationale underlies the choice of a schizophrenia sample for this preliminary validation study of the ISQ.
Both hierarchical linear regression and SEM revealed a clear pathway between ISQ, cholesterol, IL-6, and CRP. These findings confirmed that CRP acts as a downstream inflammatory biomarker in schizophrenia, consistent with previous evidence suggesting that high-sensitivity CRP (hsCRP) serves as a surrogate marker of upstream cytokine activity.27,44 Cytokines related to human atherosclerosis can be classified into proinflammatory and proatherogenic (e.g. IL-1, IL-6, TNF) versus antiinflammatory and antiatherogenic (e.g. IL-10, IL-1rA) categories.45–47 Only IL-6 was examined in this study, thus, future work should include other cytokines such as IL-1, IL-1β, IL-8, and IL-10 to clarify broader inflammatory pathways.
CRP, a nonspecific biomarker, is associated with increased infection susceptibility and immune dysregulation, and is tightly linked to IL-6. IL-6 plays a central role in mediating ISQ and CRP levels, consistent with prior studies. Higher ISQ scores predicted elevated IL-6 and subsequently higher CRP concentrations, suggesting that ISQ may serve as an early screening tool for inflammation in schizophrenia.
The NPV of 72.22% further indicates that approximately three-quarters of participants with IL-6 levels below 3.0 pg/mL were correctly identified by the ISQ as showing no apparent immune activation. This finding highlights the ISQ's potential as a practical first-line tool to rule out immune dysregulation before proceeding to more costly or invasive laboratory assessments.
A previous study showed that hsCRP levels predicted vascular risk more effectively than total HDL cholesterol. 27 Similarly, in this study, cholesterol significantly increased IL-6 and CRP levels, implying that metabolic factors may further amplify immune activation in schizophrenia.
Cholesterol also appeared to act as a moderating or confounding variable within the pathway linking immune status, IL-6, and CRP. Cholesterol metabolism has been increasingly recognized as an important regulator of inflammatory signaling and cytokine activity. 48 This suggests that lipid dysregulation may not only amplify immune responses but also be affected by antipsychotic-induced metabolic alterations—a relationship that warrants further longitudinal exploration.
Age was another significant factor associated with immune dysregulation. Older individuals are at greater risk of infection, chronic disease, and malignancy, which contribute to decreased immune functioning. 49 The present study found that age positively influenced ISQ scores, suggesting that older patients experienced greater immune dysfunction. Although sex differences have been reported in inflammatory patterns among schizophrenia patients, 34 no statistically significant differences were found between males and females in this study, likely due to the limited number of female participants (n = 22). Future studies with larger and more balanced samples are needed to confirm potential gender-related immune differences.
Although IL-6 was analyzed as a key mediator in this study, it should be acknowledged that IL-6 is a nonspecific proinflammatory cytokine involved in multiple chronic and psychiatric disorders, rather than a schizophrenia-specific marker. Recent research has highlighted additional, more specific alterations in immune-inflammatory pathways implicated in schizophrenia. For example, disturbances in kynurenine metabolism and microglial activation have been identified as key pathogenetic mechanisms linking peripheral inflammation with neuroprogression. 50 Moreover, dysregulation of the NLRP3 inflammasome and pyroptotic signaling cascades has been proposed to underlie aberrant immune activation and neural damage in psychotic disorders. 51 These findings collectively suggest that schizophrenia-related immune dysfunction extends beyond generalized cytokine elevation and may involve distinct molecular pathways influencing neuroimmune interactions.
This study has several limitations. First, the relatively small sample size, particularly the limited number of female participants, may have influenced the results. Second, only patients with schizophrenia were included. Given that schizophrenia is associated with chronic neuroinflammation, 52 their immune dysfunction may differ from that observed in healthy or other psychiatric population. Because of the limited sample size and the clinical nature of the study population, ROC-based estimation of cutoff points may not provide stable results. Therefore, the graphical method was adopted to determine the ISQ and IL-6 thresholds, offering a more intuitive and robust estimation approach for this exploratory setting. Future studies should include both healthy controls and other psychiatric groups, such as patients with major depressive disorder,26,53 to validate the generalizability of these findings.
Moreover, recent research has identified other inflammatory mediators—such as IL-1, IL-8, IL-10, IL-1β, TNF-α,20,27,30,45–47 gasdermin D (GSDMD), Casp-1, and astrocytic NOD-like receptor protein 3 (NLRP3)54–56—as potential contributors to psychiatric disorders. Further studies should incorporate these biomarkers to clarify their roles in the immune mechanisms underlying schizophrenia. In addition, a healthy control group was not included in this study because the primary objective was to examine the internal pathways linking subjective immune perception and objective inflammatory markers within schizophrenia, rather than to establish between-group differences. Nevertheless, future studies should recruit healthy and community control groups to provide broader external validation of the ISQ and its clinical utility.
Conclusion
The ISQ may be considered a practical and noninvasive preliminary screening tool for assessing immune dysfunction, offering an accessible alternative to blood-based testing during the early evaluation. Although the discriminative ability of the ISQ was modest (AUC = 0.59), this preliminary finding provides an initial indication of its potential role in identifying patients with immune-related alterations in schizophrenia. Given the limited sample size and the focus on individuals with schizophrenia, these results should be interpreted with caution and do not yet support the ISQ as a definitive diagnostic marker. Future studies with larger and more diverse samples, including individuals with depression, healthy controls, and community populations, are warranted to validate its clinical applicability and refine the optimal threshold.
Nonetheless, based on the current data, a cutoff score of 10 on the ISQ may serve as a tentative reference for preliminary screening and for guiding decisions on further laboratory assessments. With the increasing use of telemedicine following the COVID-19 pandemic, the ISQ's simplicity and low cost make it particularly valuable in community mental health settings and remote or resource-limited regions. Future research should continue to explore its reliability, construct validity, and integration into broader immune-monitoring frameworks for psychiatric disorders.
Footnotes
Acknowledgments
We sincerely thank Ms Mei-Chu Hou for her assistance in organizing and managing the study data. The authors also wish to acknowledge the developers of the ISQ (Wilod Versprille et al., 2019) for making the instrument publicly available under the Creative Commons Attribution (CC BY 4.0) license, which permits unrestricted use, distribution, and reproduction in any medium, provided that the original work is properly cited.
Ethics statement
This retrospective chart review study was reviewed and approved by the Institutional Review Board (IRB) of Kaohsiung Armed Forces General Hospital, Taiwan (KAFGH 106-046), and was conducted in accordance with the Declaration of Helsinki. The ethics approval was originally granted for the Cassia obtusifolia clinical trial, from which the present secondary analysis data were derived. All participants in the original trial provided written informed consent prior to enrollment. The IRB approved the use of these anonymized data for secondary analysis without requiring additional consent.
Author contributions
J.Y. performed experiments and wrote the manuscript; Y.H. helped with the trial process; C.L. and F.W. designed the protocol, recruited participants, performed the statistical analysis of the data, and revised the manuscript. All authors have reviewed the manuscript and agreed to its submission.
Funding
This work was financially supported by China Medical University Hospital (Grant No. DMR-111-183) and the Chinese Medicine Research Center, China Medical University, under the Featured Areas Research Center Program within the Higher Education Sprout Project framework of the Ministry of Education in Taiwan (Grant No. CMRC-CENTER-0).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data in this study are available to other researchers upon request.
