Abstract
In recent years, the application of aspirin for coronary artery disease has become increasingly common, and aspirin-related enteropathy has gradually garnered more attention. It has increasingly been recognized that the related enteropathy after the long-term use of NSAIDs may have potentially fatal side effects. However, it remains controversial how to select alternative medications for these patients with coronary heart disease, particularly for those who have recently undergone stent implantation. Our case presented with recurrent chest pain and coronary angiography revealed he had severe stenosis in the left anterior descending artery, nearly 90% at the opening, 80% at the middle segments, and a total occlusion at the distal segment, accompanied by a TIMI blood flow grade of 2. However, after five years of taking aspirin, the patient often felt fatigued and was found have megaloblastic anemia and thrombocytopenia. We attempted to prescribe indobufen as an alternative to aspirin for antiplatelet therapy. Fortunately, after several months of follow-up, the patient's symptoms were alleviated, and his hemoglobin and platelet levels returned to normal. Indobufen might be a potential alternative medication for these patients in clinical practice in the future.
Keywords
Introduction
Antiplatelet drugs are an essential component following coronary stent implantation and are typically taken for life to prevent restenosis within the stent and additional coronary artery narrowing. However, in clinical practice, it has been observed that some patients develop related intestinal diseases after long-term use of aspirin, and in severe cases, life-threatening conditions such as severe anemia and thrombocytopenia may arise.1,2 Physicians encounter a dilemma when managing such cases, as discontinuing antiplatelet medication could result in blockages within the stent or other blood vessels. Our patient experienced this exact issue; he was diagnosed with coronary atherosclerotic heart disease via coronary angiography (CAG) and had been taking aspirin for five years, and he had to continue antiplatelet therapy following stent implantation in case of restenosis in stent despite of anemia and low-platelet count. We attempted to use indobufen as an alternative to aspirin for antiplatelet therapy in such situations and achieved quite positive clinical outcomes in the following months. It might be an alternative strategy for dealing with such conditions in clinical practice.
Case report
A patient in his late 70s was admitted to Zhejiang Provincial Hospital of Chinese Medicine in early November 2024, complaining of recurrent chest tightness for four days, which had worsened over the previous 7 h. The patient had experienced chest tightness without an obvious cause four days prior while out of town. He felt it slightly to the left behind the sternum, and it was relieved after rest. Consequently, he visited the emergency department of a local hospital. His vital signs were normal upon admission, with a temperature of 36.8°C, a pulse rate of 62 beats per minute, a respiratory rate of 14 breaths/min, and a blood pressure of 161/86 mmHg. His physical examination was unremarkable. An emergency examination of high-sensitivity troponin and electrocardiogram showed no obvious abnormalities at that time.
Unfortunately, the previous night, he suddenly felt chest tightness worsening, accompanied by profuse sweating for 2 h, and thus he went to the emergency department of our hospital. The instant ECG showed slight depression of the ST segment in leads V3, V4, V5, and V6 (Figures 1(a) and (b)). Additionally, troponin I was slightly higher than normal in the emergency room. He was diagnosed with acute coronary syndrome and was immediately sent to our cath lab for CAG. His past medical history included coronary heart disease (CAD) and hypertension. A recent computed tomography coronary angiogram taken in 2016 revealed multiple plaques in the coronary arteries and mild to moderate stenosis in the left anterior descending artery (LAD). Multiple mixed plaques and soft plaques had formed in the right coronary artery (RCA), especially in the proximal segment, with moderate to severe local lumen stenosis. He had been treated with clopidogrel (Plavix) tablets 75 mg daily, aspirin (Bayer) enteric-coated tablets 100 mg daily, and rosuvastatin calcium (Clotrimazole) tablets 10 mg every night since then. He had hypertension for more than 10 years, with the highest blood pressure recorded at 190/110 mmHg, and he took Norvasc 5 mg daily for hypertension. His family members were healthy and denied any history of malignant tumors and contagious diseases.

(a) Electrocardiogram (ECG) at admission. (b) ECG on admission with acute coronary syndrome.
The emergency CAG revealed that the left main coronary artery (LM) had an 80% stenosis in the distal segment. Diffuse calcified plaques were observed at the opening and along the entire length of the LAD, with 90% stenosis at the opening, 80% at the middle segments, and a total occlusion at the distal segment, accompanied by a TIMI blood flow grade of 2. The opening of the left circumflex artery exhibited 80% stenosis, with 40% in the proximal segment. The RCA showed 30% stenosis in the proximal segment, 40% in the middle, and distal segments, 60% stenosis in the posterior branch of the left ventricle, and 70% stenosis in the posterior descending branch. After discussing with his family, balloon PTCA was performed from the proximal to the middle segment of the LAD to the LM trunk lesion. A single 3.5 × 13 mm drug-eluting stent was implanted at that site.
However, the blood routine examination and C-reactive protein indicated megaloblastic anemia with thrombocytopenia. His results were as follows: neutrophil% 76.3% (increased), lymphocyte% 18.0% (decreased), monocyte% 2.8% (decreased), absolute number of lymphocytes 0.8 × 10^9/L (decreased), red blood cell count 1.89 × 10^12/L (decreased), hemoglobin 80 g/L (decreased), hematocrit 21.8% (decreased), mean corpuscular volume 115.5fl (increased), mean corpuscular hemoglobin content 42.1pg (increased), mean corpuscular hemoglobin concentration 364 g/L (increased), red blood cell volume distribution width 16.3% (increased), platelet count 43 × 10^9/L (decreased), platelet hematocrit 0.038% (decreased). We now faced a clinical dilemma regarding the subsequent antiplatelet plan, as his current CRUSADE score is 35 points, indicating a moderate risk of bleeding, and he recently required dual antiplatelet therapy after stent implantation. Therefore, we consulted the hematology department for their opinion, which was as follows: The patient had a history of thrombocytopenia for more than five years, with specific values unknown, and platelets had been transfused four times. A previous bone marrow biopsy pathology report suggested megaloblastic anemia (Figure 2). This time, he was admitted to the hospital for CAD. The current platelet count was 48 × 10^9/L, and there was no active bleeding. Due to treatment needs, anticoagulant treatment was currently required. It was recommended to monitor blood routine and coagulation function. If there were active bleeding, then aspirin should be discontinued. After careful consideration and obtain signed consent from our patient, we attempted to adjust the aspirin 100 mg daily to indobufen 100 mg every 12 h to inhibit platelet aggregation in addition to clopidogrel, as it is a selective, reversible cyclooxygenase inhibitor, and even if there were heavy bleeding, platelet activity would be restored promptly after discontinuation of indobufen. Additionally, we added folate 5 mg three times a day and vitamin B12 0.5 mg three times a day to the routine medication plan to improve megaloblastic anemia.

The morphology of the bone marrow biopsy is consistent with megaloblastic anemia.
His laboratory examination results were as follows (Figures 1 and 3; Table 1).

Comparison of platelet count and hemoglobin levels between aspirin and indobufen.
Laboratory test variation after treatment.
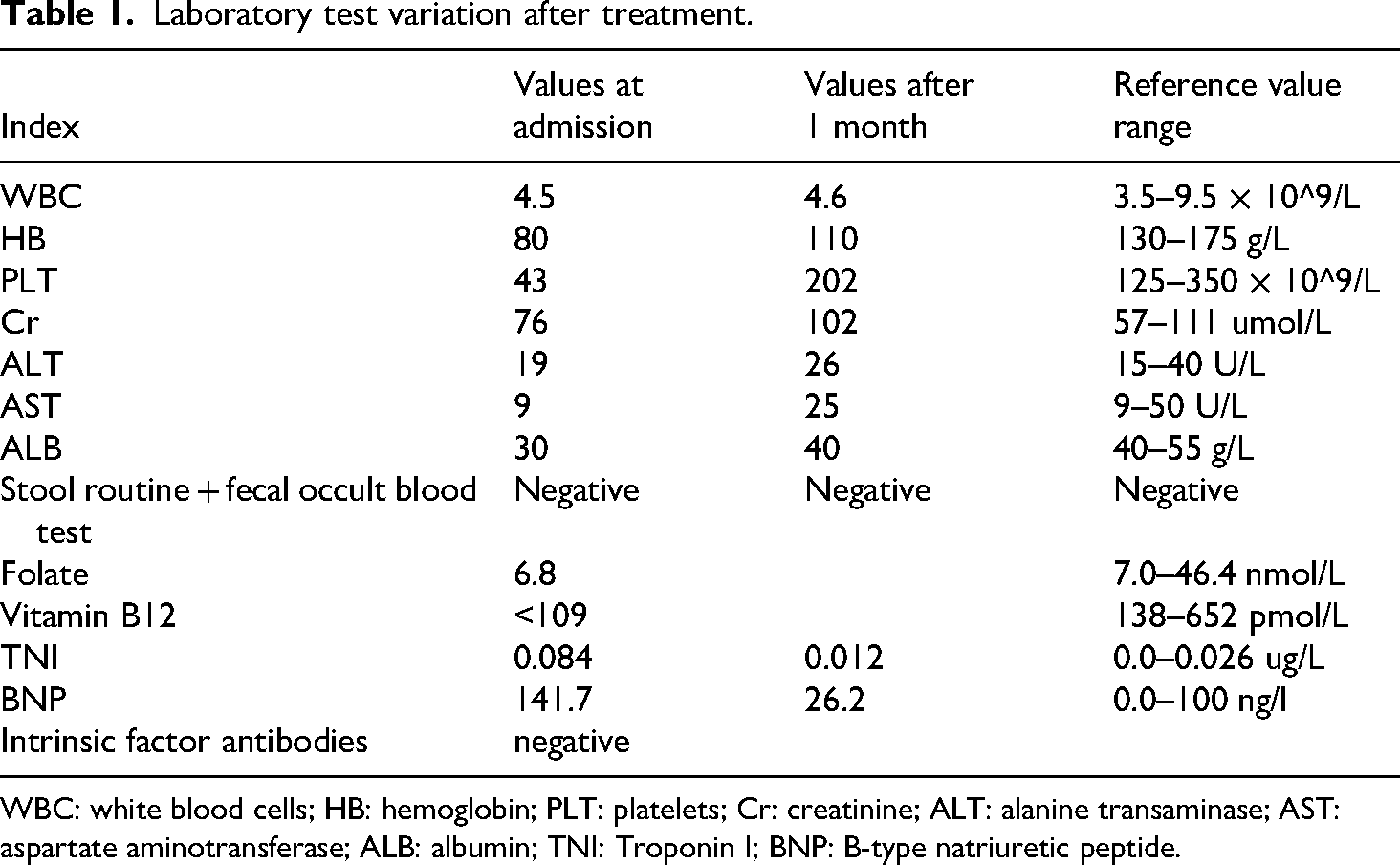
WBC: white blood cells; HB: hemoglobin; PLT: platelets; Cr: creatinine; ALT: alanine transaminase; AST: aspartate aminotransferase; ALB: albumin; TNI: Troponin I; BNP: B-type natriuretic peptide.
Discussion
Our case suggested that indobufen might replace aspirin in patients recently implanted with stents, who were experiencing severe aspirin-related enteropathy characterized by anemia and thrombocytopenia and we speculated that he was suffering from aspirin-related enteropathy based on clinical manifestations. In recent decades, with the increasing use of aspirin, aspirin-related enteropathy has gradually attracted physicians’ attention. As research on aspirin-related enteropathy has deepened, it has become apparent that chronic aspirin use-induced enteropathy is an important cause of small intestinal ulceration and affects the absorption of many vitamins, particularly vitamin B12 and folate. This can ultimately lead to megaloblastic anemia, as the vitamin B12–intrinsic-factor complex is absorbed in the terminal ileum and folic acid is readily absorbed in the jejunum areas that are significantly impacted by aspirin-related enteropathy. 3
Studies have found that both short-term and long-term use of aspirin can cause gastrointestinal damage. It is generally believed that continuous use of aspirin for more than one month increases the likelihood of NSAIDs-related enteropathy. 4 Aspirin-related enteropathy can affect any part of the gastrointestinal tract, from the esophagus to the rectum, with the stomach and small intestine being the most susceptible. Aspirin-related gastrointestinal mucosal damage may be a gradual process. Some studies indicate that aspirin affects gut microbiota dysbiosis and the intestinal homeostasis of Parabacteroids. goldsteinii and its metabolite 7-keto-LCA, which is a protective factor for intestinal tissue. 5 Other studies suggest that aspirin might cause intestinal injury by inhibiting FXR and activating ET-1. 6 Moreover, the acidic component of aspirin's structure can directly damage the phospholipid membranes of gastrointestinal epithelial cells, causing oxidative phosphorylation to uncouple and leading to mitochondrial damage. This results in cellular energy depletion and calcium overload, producing a large number of free radicals that destroy the integrity of the intercellular structure and increase the permeability of the gastrointestinal mucosa. This makes it more vulnerable to further damage by various gastrointestinal contents, including gastric acid, food, and various enzymes. Other studies have shown that immune mechanisms, microcirculatory disorders, or intestinal motility abnormalities also play significant roles in aspirin-induced enteropathy. 7 Meanwhile, individuals who frequently take aspirin often receive proton pump inhibitors and are susceptible to various bacterial infections, such as Clostridioides difficile, which can also make the intestinal tract more vulnerable to bleeding.8,9 Some studies have shown that aspirin might modulate bile acid metabolites and impair the self-renewal of intestinal stem cells, thereby disrupting intestinal homeostasis.10,11 Other research has revealed that aspirin could serve as the most common nonchelating drug causing iron deficiency anemia due to increased excretion of iron, a result of aspirin chelating metabolites, 12 All of the aforementioned mechanisms may damage various parts of the small intestine, particularly the terminal ileum or the duodenum and proximal jejunum, which are the primary sites for the absorption of vitamin B12 and folic acid. Vitamin B12 is primarily absorbed in the terminal ileum, whereas folic acid (B9) is primarily absorbed in the duodenum and proximal jejunum. 8 Vitamin B12 and folic acid play a crucial role in hematopoietic cell division, specifically DNA synthesis. DNA synthesis requires the enzyme 5-tetrahydrofolate-homocystine methyltransferase. Vitamin B12 serves as a coenzyme for this enzyme, facilitating the conversion of homocysteine to methionine which act as the primary source for S-adenosylmethionine, a crucial methyl group donor for DNA methylation. 9 Therefore, its deficiency can lead to impaired DNA synthesis in hematopoietic cells, causing megaloblastic changes in hematopoietic cells, with the most prominent changes observed in erythrocytes, leukocytes, and platelets.
Clinicians largely base their diagnosis on medication history, clinical manifestations, and related examinations.10–12 After taking NSAIDs, symptoms such as gastrointestinal bleeding and indigestion may appear. If no obvious abnormalities are found during gastroscopy, colonoscopy, small intestine endoscopy, or capsule endoscopy, but mucosal lesions such as inflammation, erythema, erosion, or ulcer are indicated, and if symptoms significantly improve after discontinuing aspirin, aspirin-related enteropathy can be diagnosed. 13
In this case, it is speculated that the patient's long-term use of aspirin resulted in multiple ulcers at the ileum's terminus, causing occult bleeding within the digestive system. Concurrently, because vitamin B12 is primarily absorbed at the ileum's end, aspirin might impair its absorption, leading to megaloblastic anemia. Following the cessation of aspirin, the patient's condition significantly improved after one month, as evidenced during the follow-up postdischarge, with the supplementation of vitamin B12 and folic acid, and the administration of an oral proton pump inhibitor.
The optimal treatment for aspirin-related enteropathy is to discontinue aspirin; however, this becomes complex when dealing with patients with acute coronary syndrome who require long-term dual antiplatelet therapy following stent placement, as was the case with our patient. In our scenario, we attempted to substitute aspirin with indobufen. This approach could potentially reduce the risk of microvessel bleeding and might also prevent stent restenosis due to its antiplatelet properties. Indobufen is associated with less gastrointestinal tract damage, has a shorter half-life, and offers reversible antiplatelet aggregation effects. Additionally, some studies indicate that indobufen is noninferior to aspirin after a 12-month follow-up in such patients. 14 Under close monitoring, this strategy might prove beneficial in treating coronary artery disease complicated by NSAID-related enteropathy in future. Our patient had no objections to receiving indobufen as an alternative to aspirin, and his symptoms significantly improved following the relevant treatment.
In this case, we attempted to prescribe indobufen for our patient as an alternative to aspirin for its antiplatelet effect, particularly in the presence of possible enteropathy. During close monitoring, the patient's condition improved, with hemoglobin and platelet counts rising to normal levels. This outcome may validate our clinical hypothesis and offer a potential medication strategy for patients with aspirin-related enteropathy who require long-term antiplatelet therapy following stent implantation.
However, there are several limitations in this case. First, our patient has been diagnosed with NSTEMI, and while indobufen could be beneficial in preventing platelet aggregation, there is no solid evidence that it is as effective as aspirin in treating STEMI. Additionally, indobufen is more expensive than aspirin, and many patients cannot afford it in the long term. Furthermore, patients must take it twice a day due to its short half-life, and many might miss an evening dose, which could lead to potential risks. Moreover, our case cannot directly prove that indobufen was less harmful in enteropathy, as we also added folate and vitamin B12 to the long-term medication plan.
Conclusion
Aspirin-induced enteropathy is receiving increased attention in clinical practice, yet it remains a challenging issue for physicians to manage, particularly for those who have recently undergone stent implantation. The challenge lies in the risk of stent restenosis if antiplatelet drugs are discontinued, or the potential for continued gastric tract injury, which could lead to severe megaloblastic anemia. In this scenario, we attempted to prescribe indobufen as a replacement for aspirin to prevent excessive platelet aggregation. Over time, the patient's condition gradually improved. For such patients, indobufen might serve as an alternative medication in clinical practice.
Footnotes
Acknowledgments
The author would like to express deep appreciation for my supervisors in our department, for their invaluable guidance and support throughout this project, and would also like to thank my family and friends for their unwavering encouragement and understanding.
Author contributions
Bingxu Chen and Xu Yi-fei have collected the clinic data during this time and finished the important part of the article; Wang Zhi-jun, Qiu Yuan-gang, and Chen Shen-jie have provided related article and collected relevant references; and Huang Shu-wei, Zhu-min, and Yang Bing-sheng formulated the design of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data are available from the corresponding author on reasonable request.
Disclosures
Written informed consent was obtained from the patient to publish their anonymyzed information in this study, and have deidentified all patient details. The reporting of this study conforms to CARE guidelines. 15
