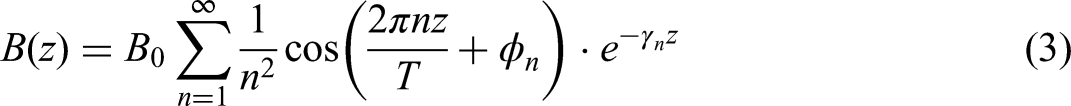Abstract
In pyrolysis effect tests, pyrolytic wires face challenges such as complex pyrolysis products, low particle separation accuracy, and signal susceptibility to interference. Traditional methods, lacking controllable pyrolysis and dynamic separation mechanisms, result in poor experimental reproducibility and weak data correlation. Current research often focuses on the intrinsic properties of materials while neglecting the integrated design of test equipment, urgently requiring the construction of high-precision, intelligent pyrolysis testing platforms. To address this, a pyrolysis particle separation test chamber based on a double helix thermal desorption particle separator is proposed, integrating controllable pyrolysis, dynamic particle separation, and closed-loop feedback testing functions. The pyrolysis reaction module uses dual-mode heating with induction/resistance, combined with a double helix separator to achieve particle sorting based on size, density, and charge differences through centrifugal force fields, pyroelectric effects, and external electromagnetic fields. Multi-material thermal stability tests show that high-temperature silica gel has optimal temperature stability during the initial thermal response phase, with a steady-state temperature drop of 0.3 °C; copper enameled wire has a thermal hysteresis coefficient of 0.99 and an energy dissipation rate of 0.22 W/m·K. Energy efficiency comparisons indicate that the input power fluctuation is only 2.0 kW, with total energy consumption of 62 kWh, reducing by 47.5% compared to traditional methods. The separation efficiency reaches 0.88 at a rotational speed of 3000 r/min. Under different parameter couplings, the pyroelectric signal peaks at 0.08 pC at an angle of 18° and an electric field strength of 300 V/m, verifying the multi-field collaborative optimization mechanism. The proposed scheme solves the problems of the uncontrollable pyrolysis process and low separation efficiency through intelligent regulation and modular coordination, and significantly reduces energy consumption. It provides a quantifiable and reproducible experimental platform for the performance research of pyroelectric materials, and promotes its engineering application in fire warning, cable fault diagnosis, and other fields.
Keywords
Introduction
With the development of modern industry and construction, the requirements for the safety, reliability, and durability of wires and cables are increasing. Especially in power systems, communication networks, and various electrical equipment, wires and cables, as key components for energy transmission and signal delivery, their quality directly affects the stable operation of the system and personal and property safety.1,2 Traditional testing methods for wires and cables often fail to fully simulate complex environments under actual usage conditions, such as extreme conditions like high temperatures, low temperatures, and humidity, which can affect the insulation materials of wires and cables. Therefore, there is a need for a testing device that can precisely control temperature and test the flame-retardant properties of wires and cables under different conditions.3,4 As environmental awareness grows and technology advances, research into more efficient and environmentally friendly testing methods continues to deepen, such as reducing harmful emissions and improving testing efficiency. These developments provide new research directions and impetus for the design of thermal release test chambers, leading more researchers to focus on the significant role thermal release testing plays in optimizing material performance. 5
Recent advances in pyrolysis-based materials and technologies have focused on addressing key challenges across multiple domains through controlled thermal processes, with particular attention to the pyroelectric effect—a fundamental property of certain materials that generate electric charge in response to temperature changes, forming the basis of pyroelectric sensing technology. The pyroelectric effect has gained significant traction in various applications, including fire detection, thermal imaging, and energy harvesting, due to its ability to directly convert thermal energy into electrical signals with high sensitivity. In pyroelectric sensing technology, the performance of sensors heavily depends on the precise control of material composition, microstructure, and thermal response characteristics, making it crucial to develop advanced testing platforms that can accurately characterize these properties under controlled conditions.
In the field of pyrolytic carbon materials, strategies to overcome limitations in energy storage performance and catalytic functionality have been explored through developing methods for the in-situ generation of metal/oxide composites via the catalytic pyrolysis of acetonitrile at 600 °C–700 °C. 6 MgCuAl/MgFeAl-type hydrous carbides were used as precursors to fabricate flake-like nitrogen-doped carbon structures. These structures have 62% micropores and a specific surface area of 1121 m²/g, featuring rich edge planes and slit mesopores that reveal the synergistic mechanism of microstructure regulation on multi-field coupling performance, offering new insights for energy storage and catalytic applications. In sensing technologies, the sensitivity, anti-interference capability, and cost-effectiveness of analytical methods were improved through pyrolysis-derived platforms by Bian et al. 7 Nano-gold modified electrochemical sensors were constructed on pyrolytic graphite substrates and demonstrated significant improvements in vitamin C detection. By optimizing pyrolysis temperature to prepare 20–50 nm gold particles/graphite composite electrodes and adjusting square wave voltammetry parameters (pH 5 acidic electrolyte and suitable scan rate), these sensors achieve linear responses over 5–100 μg/mL (R² = 0.999) with a detection limit as low as 1.86 μg/mL—three times more sensitive than conventional methods. This advancement highlights how the exposure of edge planes on pyrolytic graphite and the synergistic enhancement of electrocatalytic activity by gold nanoparticles create a new paradigm for interface regulation in trace nutrient detection.
In the realm of catalyst design and optimization, Li et al. 8 have developed innovative approaches to enhance stability and functionality through controlled pyrolysis. They utilized Schiff base ligand bridging to engineer Fe/Co heterometallic clusters, enabling in-situ reconstruction into MOx/CoFe@NC nanostructures via pyrolysis at 700 °C. This work revealed how the confinement effect of nitrogen-doped carbon layers and dynamic reconstruction of M–O–M bridge bonds optimize electron transfer pathways, addressing challenges in synergizing alloy engineering with bimetallic doping strategies. Zhang et al. 9 have focused on mitigating stability issues in Ni-based catalysts during pyrolysis aromatics reactions, where carbonization and amorphous carbon deposition often lead to performance degradation. By regulating the size of γ-Al₂O₃ supports, they achieved directional control of Ni particle pyrolysis behavior. Using a fixed-bed pyrolysis system (500 °C–800 °C) combined with in-situ characterization and density functional theory simulations, they analyzed the Ni⁰⁺ particle size-dependent carbon reconstruction mechanism, revealing that pyrolysis-driven growth of carbon nanotubes and intercalation energy barriers at the Ni³²C/Ni⁴C interface (90.2–116.0 kJ/mol) explain how large Ni particles extend catalytic life by reducing carbon atom doping rates. Additionally, in polymer insulation materials, Deng et al. 10 have developed magnetic and conducting Fe3O₄–cross-linked polyaniline nanoparticles with core–shell structure to investigate their thermal stability and catalytic behavior during polymer pyrolysis. These nanoparticles, synthesized via in-situ polymerization with an average diameter of 10–20 nm, exhibit enhanced thermal stability with a decomposition temperature increase of 40 °C–60 °C compared to pure polyaniline. The Fe3O₄ core provides magnetic responsiveness while the conducting polyaniline shell facilitates electron transfer, enabling controlled catalytic pyrolysis of polymer insulation materials commonly used in electrical wires and cables. This work reveals the synergistic effect between the magnetic core and conducting shell in regulating pyrolysis pathways, offering insights into the design of functional nanocomposites for wire insulation applications.
A spiral-separator process proposed by Yu et al. 11 offers an effective solution to the environmental pollution and carbon loss caused by coal-gasification fine slag: the concentrate obtained shows high carbon content and excellent combustibility, while the screened products are regarded as promising fuels and adsorbents. To overcome the poor heat-transfer efficiency of conventional cylindrical downer reactors, Lian et al. 12 introduced a vortex-type downer in which the feed stream is redirected from vertical to horizontal-tangential entry, creating a spiral particle strand that increases solid hold-up and markedly enhances heat transfer, thereby providing a theoretical basis for next-generation pyrolysis units. Employing computational fluid dynamics, Yuan et al. 13 investigated the separation behavior of viscous oil–vapor mixtures inside a spiral separator during in-situ pyrolysis of tar-rich coal; the study revealed that a volute cyclone separator achieves oil–vapor separation efficiencies above 98%, adapts well to light-oil-bearing tar, and enables the determination of optimal operating conditions.
A pyrolysis–physical-separation route introduced by Zhong et al. 14 tackles the challenge of comprehensively recycling spent Li-ion batteries: the process recovers 99.91% of the organic electrolyte and cleanly separates active materials from current collectors, while high-purity Al, Cu, and cathode powders are subsequently extracted from the pyrolytic residue; the reclaimed cathode material has already been validated for new-cell fabrication. To improve the poor quality of tar generated from oily-sludge pyrolysis and to valorize steel slag, Song et al. 15 proposed co-pyrolysis of oily sludge with steel slag followed by magnetic separation. Experimental runs showed that the slag catalytically enriches short-chain alkanes in the tar, delivering a tar yield of 10.63% at 550 °C, while magnetic separation recovers up to 55.03% of the slag, confirming the viability of the integrated route. Facing limited knowledge on the particulate classes and inorganic migration mechanisms in fast-pyrolysis bio-oil, Molinder et al. 16 employed scanning electron microscope and energy-dispersive X-ray spectroscope (SEM–EDS) to characterize filter-captured particles from oils derived from different feedstocks. Three particle categories—carbonaceous structures, spherical residues, and irregular fragments—were identified, with aerosol-phase particles exhibiting higher inorganic contents, thereby providing a basis for the design of experimental separation chambers.
The above studies have demonstrated certain advantages in pyrolysis particle separation and material property regulation. For instance, the spiral separation process proposed effectively addresses the environmental pollution and carbon loss issues of coal gasification fine slag; the vortex-type downflow reactor significantly enhances heat transfer efficiency; and the pyrolysis-physical separation route achieves high recovery rates of spent lithium-ion batteries. These methods have laid a foundation for technological progress in related fields. However, existing research still exhibits multiple limitations: First, most pyrolysis particle separation technologies focus on single separation mechanisms, such as relying solely on centrifugal force or magnetic field effects, lacking the synergistic design of multi-physical field coupling, which makes it difficult to meet the precise separation requirements of complex pyrolysis products. Second, the temperature control precision of traditional pyrolysis test equipment is generally insufficient, especially when dealing with the pyrolysis process of conductive circuits, making it challenging to dynamically regulate the pyrolysis rate and product characteristics, resulting in poor stability of particle morphology and composition. Third, existing pyrolysis product separation devices are mostly in batch processing mode, lacking continuous operation capability, which limits their efficiency in engineering applications. Additionally, although scholars have characterized particle categories in pyrolysis bio-oil using SEM–EDS, research on selective separation mechanisms for particles with different properties (such as conductivity and dielectric differences) remains insufficient, making it difficult to establish a quantitative relationship between particle properties and separation efficiency. Finally, existing equipment has obvious shortcomings in the integration of pyrolysis and separation processes, where secondary reactions easily occur during the transfer of pyrolysis products from the thermal zone to the separation zone, affecting the original properties of the particles and thereby reducing the reliability and relevance of experimental data.
To address the above issues, this study proposes a novel design of a pyrolysis test chamber based on a double helix thermal desorption particle separator, with its innovation mainly reflected in three aspects: First, a pyrolysis system integrating induction/resistance dual-mode heating and intelligent temperature control is constructed to achieve precise regulation of the conductive circuit pyrolysis process. Second, a double helix thermal desorption particle separator structure is designed, which realizes efficient separation of particles with different properties through the synergistic effect of centrifugal force field and electromagnetic homogenization field. Third, a pyroelectric particle separation test chamber (thermal decomposition particle separator chamber (TDPSC)) is developed, which reduces the occurrence of secondary reactions by optimizing the transfer path of pyrolysis products, providing a reliable experimental basis for establishing a quantitative relationship between particle properties and pyroelectric performance. The aim is to provide a quantifiable and reproducible experimental platform for performance analysis and engineering applications of pyroelectric materials in conductive wire systems, thereby promoting the development of new pyroelectric sensing technologies.
Design of TDPSC test chamber based on a double helix thermal desorption particle separator
This section details the comprehensive design of the TDPSC, focusing on its modular architecture and functional principles. The subsequent subsections elaborate on specific components and mechanisms critical to achieving controlled pyrolysis and efficient particle separation.
Overall design of the TDPSC pyrolysis wire thermal release test chamber
This subsection describes the integrated experimental system's architecture, including the pyrolysis reaction module, particle separation module, pyroelectric performance testing module, intelligent temperature control system, and data acquisition components. It explains how these modules collaborate to address technical challenges in thermal desorption testing of pyrolysis wires.
During the thermal desorption effect test of pyrolysis wires, a series of key technical challenges are faced, including complex pyrolysis product composition, wide particle size distribution, uncontrollable deposition behavior, and weak thermal desorption electrical signals that are easily disturbed. These issues make it difficult to achieve precise control over the pyrolysis process and effective separation of products using traditional experimental methods, thereby affecting the consistency and reproducibility of test results.17,18 To address these problems, a design proposal for an integrated experimental system based on a double helix thermal desorption particle separator is proposed. The aim is to achieve precise regulation of particle characteristics (such as size, density, and charge) through a combination of controlled pyrolysis, dynamic particle separation, and closed-loop feedback control, and to establish a quantitative relationship between these characteristics and thermal desorption performance. The entire system consists of five core units: pyrolysis reaction module, particle separation module, thermal desorption performance testing module, intelligent temperature control and energy management module, and data acquisition and feedback optimization module, forming an intelligent experimental platform with closed-loop control. The process flow of this scheme is shown in Figure 1.

The flowchart of the new pyrolysis wire thermal release test scheme.
The solution is composed of multiple functional modules, covering key aspects such as controllable pyrolysis of wire materials, particle separation, performance testing, and energy management. The pyrolysis reaction module, serving as the core at the system's front end, uses induction heating or resistance heating technology to perform high-temperature cracking on wire materials. By precisely controlling the temperature gradient and heating rate, it achieves efficient conversion of materials into gaseous and particulate states. The double helix particle separation module receives the pyrolysis products and, under the combined effects of centrifugal force and pyroelectric effect, dynamically sorts particles based on their size, density, and charge differences. Its separation efficiency is influenced by the rotational speed, channel inclination, and external electromagnetic field parameters, and it forms a coordinated adjustment mechanism with the pyrolysis temperature from the previous stage. The wire pyroelectric effect testing module characterizes the pyroelectric properties of particles of specific size and composition after deposition. By applying periodic temperature or stress excitation, it detects the charge release behavior and response time characteristics on the wire surface, providing feedback for optimizing particle separation parameters. The intelligent temperature control and energy management module integrates multi-zone temperature control units, combining PID regulation to achieve stable temperature control in each functional area, while also incorporating waste heat recovery devices to enhance system efficiency. The data acquisition and feedback optimization module monitors the system's operational status in real-time through a network of high-precision sensors, collecting multidimensional data including temperature, pressure, particle concentration, and charge quantity. It constructs a quantitative relationship model between particle characteristics and pyroelectric performance, thereby achieving closed-loop optimization and intelligent regulation throughout the process. In order to realize the controllable pyrolysis of wire materials in the thermal release effect test, a pyrolysis reaction module is designed as the key front-end unit of the system. Its structure and functional configuration directly affect the product generation characteristics and the stability of the subsequent separation test. The schematic diagram of its structure is shown in Figure 2.

Schematic diagram of the pyrolysis reaction module structure.
Figure 2 shows a schematic diagram of the pyrolysis reaction module, which serves as the core component at the front end of the system, responsible for the key function of controlled pyrolysis of wire materials. The main body of the module adopts a rectangular chamber structure, made of high-temperature resistant stainless steel (model SUS310S), with internal dimensions of 150 mm × 80 mm × 60 mm, ensuring good thermal stability and mechanical strength over the operating temperature range of 300 °C–800 °C. A pyrolysis particle collection tube is configured at the top, using a Φ8 mm quartz glass tube to connect the main reaction zone with the subsequent gas system, facilitating the directed transfer of particles and gas products. The heating system employs a nickel-based alloy heating plate (model Kanthal A1, thickness 1.0 mm), with parallel spacing set at 1.0 mm, forming a sandwich structure together with an alumina insulating layer (thickness 0.2 mm) to enhance thermal conductivity and prevent electromagnetic interference. The heating method supports dual-mode operation: induction heating (frequency 50–200 kHz, matching power supply model Ambrell EKO101) and resistance heating (power density 4 W/mm²). A K-type thermocouple (model Omega Engineering KMQSS-116-U-24) is embedded to achieve ±2 °C temperature control accuracy, meeting the temperature gradient regulation requirements for different experimental needs. The condensation system uses a counter-current heat exchange design, with fins made of copper-aluminum composite material (fin spacing 2 mm, height 15 mm), rapidly conducting latent heat of phase change through heat pipes. The control system integrates a programmable logic controller module (model Siemens SIMATIC S7-1200) with the controller's local area network bus (controller area network, CAN) communication interface to achieve data interaction with the dual-screw separation module. This establishes a dynamic temperature-speed matching matrix, optimizing the overall operational efficiency of the system. Based on the concept of the new pyrolysis wire thermal release test scheme, a pyrolysis wire thermal release test chamber has been designed and researched, as shown in Figure 3.

Design diagram of thermal decomposition particle separator chamber (TDPSC), pyrolysis wire thermal release test chamber.
Figure 3 shows the structural design of the pyrolysis wire thermal desorption test chamber. This device serves as the core load-bearing unit of the entire testing system, integrating functions such as sealing protection, environmental control, and electrical measurement, aiming to provide a stable experimental platform for controlled pyrolysis and particle deposition testing. The pyrolysis wire thermal desorption test chamber, as the main load-bearing structure of the system, integrates functions like sealing protection, environmental control, and electrical measurement, ensuring the safety and controllability of the experimental process. The chamber is made of transparent polycarbonate material, with dimensions of 390 mm × 290 mm × 480 mm. Although polycarbonate has good optical transparency, to prevent potential softening in high-temperature environments, an insulating layer is added to critical areas to enhance stability. The chamber uses a flange-sealed structure to prevent pyrolysis products from leaking and is equipped with a high-efficiency particulate air filter (HEPA) filtration system (pore size ≤ 0.3 μm) to ensure that the exhaust gas meets environmental standards. A built-in clean fan removes residual particles, improving experimental reproducibility; a pressure relief valve maintains the balance of internal and external pressure differences within the chamber, preventing structural deformation caused by pressure changes. The power system consists of a toroidal transformer and an electronic voltage regulator, offering a wide range of adjustable output power to meet various heating requirements. Electrical parameter monitoring employs high-precision instruments (LEM HX 100-P ammeters, Fluke 87 V voltmeters) in conjunction with multi-point temperature sensors (K-type thermocouples) to achieve synchronized acquisition of electrical and thermal parameters. The design of sampling holes and M6 stainless steel terminals ensures both the convenience of sample extraction and the reliability of electrical connections.
Design of a double helix thermal desorption particle separator
This subsection focuses on the innovative double helix thermal desorption particle separator, explaining its geometric configuration, working principles, and how it leverages centrifugal force fields and pyroelectric effects for dynamic particle sorting based on size, density, and charge differences.
The core of the pyrolysis wire test chamber lies in providing a closed, controllable, and highly reproducible experimental environment to ensure precise analysis of the controlled pyrolysis of wire materials and their products. However, in the face of the complexity and diversity of pyrolysis products, how to efficiently and accurately separate these particles becomes a key challenge for further research. To address this, an innovative solution has been proposed: designing a double helix thermal desorption particle separator. This separator utilizes the synergistic effect of centrifugal force fields and pyroelectric effects within the double helix structure, enabling dynamic sorting based on differences in particle size, density, and charge. By adjusting the rotational speed, inclination angle of the helical channels, and applying external magnetic or electric fields, selective separation of particles with different properties can be achieved. The unfolded diagram of the double helix thermal desorption particle separator blades is shown in Figure 4.

Schematic diagram of the double helix thermal desorption particle separator.
Figure 4 shows the blade deployment diagram of the double helix thermal desorption particle separator. The left image displays a side view of the double helix structure. Here, S represents pitch, which is the axial distance between adjacent turns of the spiral blades. B represents the thickness of the blade, which has a significant impact on structural strength and fluid dynamics characteristics. By adjusting these parameters, the centrifugal force field and pyroelectric effect can be optimized to achieve effective separation of particles with different sizes, charges, or densities. β is the unfolding angle of the spiral blade, which determines the rotation path and force direction of the particles during the separation process.
In equation (1),
In equation (2), m is the particle mass (kg), d is the instantaneous radial position of the particle (m), q is the particle charge (C), v is the tangential velocity of the particle (m/s),
In equation (3), z is the coordinate along the spiral axis (m), T is the magnetic field modulation period (m),

Simplified diagram of the circumferential section of the same helical double helix.
Figure 5 illustrates the structure and working principle of the double helix thermal desorption particle separator. Figure 5(a) shows the model of the double helix thermal desorption particle separator, which includes the housing, rotor, inlet side, and exhaust side. The housing forms the main body of the device, protecting the internal structure; the rotor rotates under the drive of the shaft, creating gas flow in the direction of suction, with the inlet side serving as the gas entry point and the exhaust side as the purified gas outlet. Additionally, the separator integrates an external electric heating system, achieving temperature gradient control within the chamber through PID temperature control, promoting the directional regulation of particle adsorption and desorption behavior. The exhaust side features an expansion section and baffle structure, utilizing Bernoulli's effect to further achieve efficient separation of gas-solid two-phase flow. Figure 5(b) is a top view of the axial cross-section in the same direction, presenting the cross-sectional shape of the double helix structure. Special flow channel designs guide gas and particle movement, providing a path foundation for the separation process. Figure 5(c) shows the blade deployment, displaying the distribution state of particles, where different colored particles represent their characteristic differences. The blades, through specific geometric shapes, facilitate particle separation under centrifugal force and other effects during flow. Between these elements, the housing and rotor form the physical space and power source, while the gas flow path from the inlet side to the exhaust side, combined with the double helix structure, extends the particle separation distance. The blades optimize separation efficiency through micro-effects, collectively achieving efficient separation of particles in the gas. The dual-rotor system is placed inside the cylindrical housing in a meshing configuration, forming symmetrically distributed spiral conveying channels. The kinetic equation for particle centrifugal separation is shown in the following equation:
In equation (4),

Schematic diagram of the structure of the double helix thermal desorption particle separator.
As shown in Figure 6, the main structure of the double helix thermal desorption particle separator is a long cylindrical double helical chamber, featuring an optimized geometric configuration with a high aspect ratio. The sides are mechanically supported by multi-pillar truss supports, which include axial restraint and radial damping structures to effectively reduce deformation stress caused by thermal expansion. The core working unit consists of two interlocking reverse helical rods, with the helix angle optimized for dynamics. Under the drive of motor 1, it can form a combined axial conveying and radial shearing motion. At the feed end, there is a beveled slot structure that connects to the drive unit via a variable diameter pipe, enabling continuous input and initial distribution control of pyrolysis particles. In the middle section of the separator, an external electric heating device is integrated, using ring-shaped resistance heating elements and a PID temperature control system to create a gradient temperature field on the surface of the helical chamber. This promotes phase change separation through heat conduction and convective coupling. The separated product is discharged through a vertical exhaust pipe at the right end, which includes expansion sections and baffle plates designed to utilize Bernoulli's effect for efficient gas–solid two-phase flow separation. Each functional module is sealed by flanges, and in terms of thermal parameters, the motor speed, heating power, and exhaust pipe diameter form a dynamic balance system to ensure that the residence time of pyrolysis particles within the chamber meets the thermodynamic conditions required for particle separation.
TDPSC performance test and application analysis
This section presents experimental results evaluating the TDPSC's performance across various parameters and applications. The following subsections provide detailed analyses of its separation efficiency, thermal stability, energy consumption, and practical applications in detecting wire degradation.
Performance test of the pyrolysis particle separation test chamber
This subsection reports on experiments investigating the TDPSC's pyrolysis efficiency, temperature response characteristics, and separation performance under different operating conditions, including temperature gradients, rotational speeds, and electromagnetic field parameters.
To provide a quantifiable and reproducible experimental platform for the performance analysis and engineering application of pyrolytic wire pyroelectric materials, a new pyrolytic wire pyroelectric test chamber based on a double helix thermal desorption particle separator was designed to study the pyrolysis efficiency, temperature response characteristics, and thermal conductivity stability of TDPSC. The research focuses on evaluating the optimization effect of the double helix structure on heat conduction by quantifying the dynamic changes in pyrolysis rate at different temperatures, clarifying the pyrolysis kinetic performance of the system over a wide temperature range (200 °C–800 °C). The experiment used four gradient temperatures (200 °C, 400 °C, 600 °C, and 800 °C) for comparative testing, with real-time monitoring of the pyrolysis rate changes in the wire from 0 to 35 minutes. The results are shown in Figure 7.
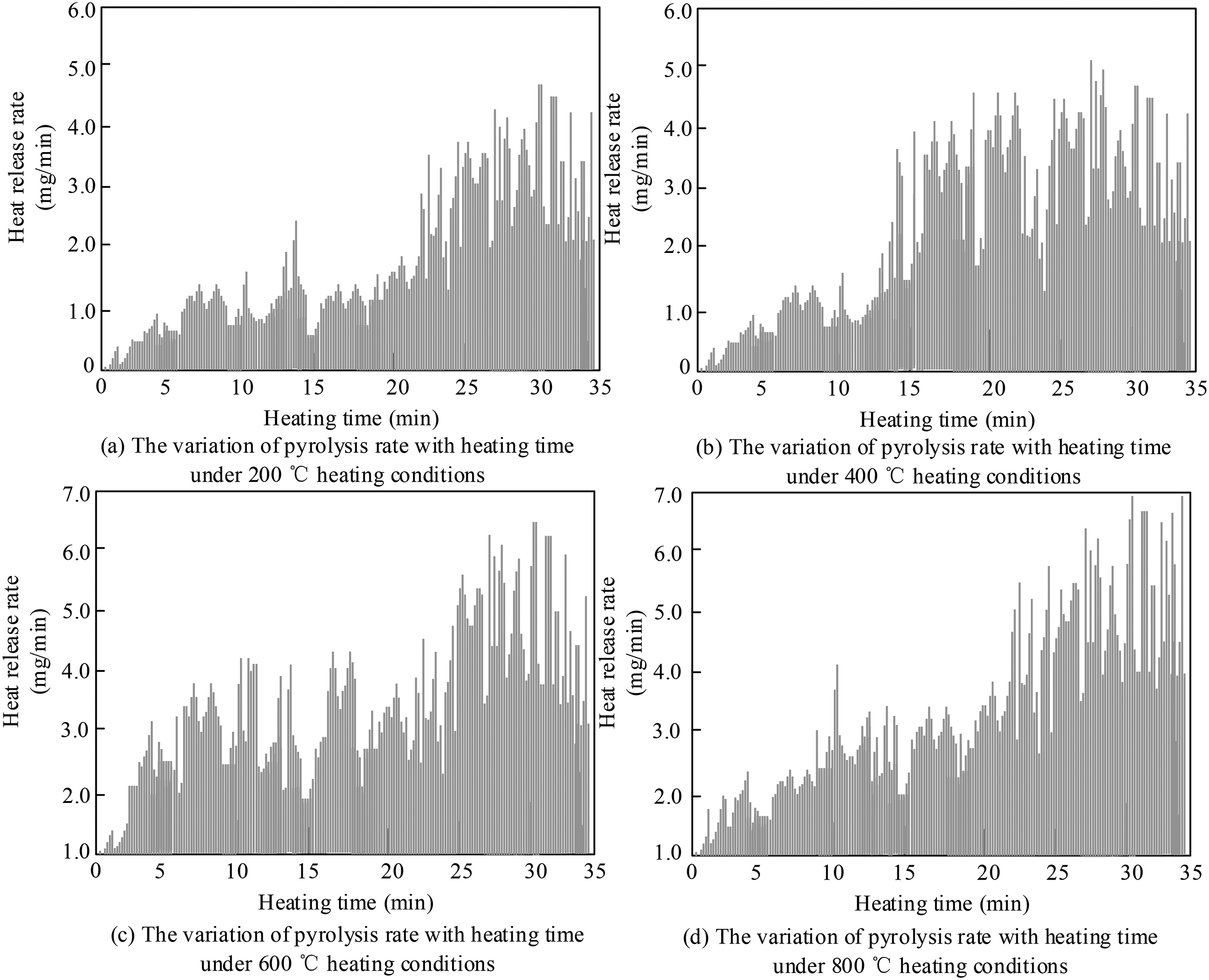
Variation of the pyrolysis rate of wire with time under different heating conditions.
From Figure 7(a), it can be seen that under the heating condition of 200 °C, the pyrolysis rate in the test chamber shows an increasing trend from 0 to 35 minutes, gradually rising from an initial rate of 0 mg/min to around 4.5 mg/min, with the maximum heat release occurring at 30 minutes. Figure 7(b) indicates that when the temperature is raised to 400 °C, the slope of the pyrolysis rate increases significantly, reaching 3.5 mg/min between 15 and 20 minutes, and peaking at 5.0 mg/min at 30 minutes. From Figure 7(c), it is evident that under high-temperature conditions of 600 °C, the system exhibits stronger thermal conductivity, with the pyrolysis rate surpassing the threshold of 4.0 mg/min at 15 minutes, and finally reaching 6.5 mg/min at 35 minutes. Figure 7(d) shows that the pyrolysis kinetics response is most pronounced at a temperature of 800 °C, with no significant improvement compared to the 600 °C condition. To investigate the effects of rotational speed, inclination angle, and magnetic field on the separation efficiency of the double helix thermal desorption particle separator, the study employed a controlled variable method, setting the rotational speeds at three gradients: 1000,2000, and 3000 r/min. The inclination angle and magnetic field parameters were changed at each rotational speed, and the corresponding separation efficiency data were measured to analyze the impact and trends of different rotational speeds, inclination angles, and magnetic fields on separation efficiency. The results are shown in Figure 8.

Influence of different parameter settings on separation efficiency.
From Figure 8(a), it can be seen that when the rotational speed is 1000, the separation efficiency ranges roughly between 0.76 and 0.82, with the efficiency showing an initial increase followed by a decrease as the inclination angle increases. In Figure 8(b), when the rotational speed increases to 2000, the separation efficiency remains within the range of 0.76–0.86. Compared to Figure 8(a), the data points are more densely distributed, and the impact of inclination on efficiency has diminished. In Figure 8(c), at a rotational speed of 3000, the overall separation efficiency rises to the range of 0.80–0.88. The double helix thermal desorption particle separator relies on centrifugal force fields and pyroelectric effects to separate particles. At a rotational speed of 1000, the centrifugal force is relatively weak, and the inclination angle significantly affects the separation efficiency, leading to an initial increase followed by a decrease in efficiency as the inclination angle increases, within the range of 0.76–0.82. When the rotational speed increases to 2000, the centrifugal force becomes dominant, reducing the influence of inclination, resulting in more densely distributed data points and an expanded efficiency range to 0.76–0.86. At a rotational speed of 3000, the strong centrifugal force enables more effective particle separation, with overall efficiency rising to 0.80–0.88, demonstrating that the rotational speed influences separation efficiency by altering the intensity of centrifugal force, and the higher the rotational speed, the stronger the dominance of centrifugal force, leading to improved separation efficiency and reduced dependence on inclination. Additionally, the study optimized the comprehensive performance of the pyroelectric wire particle separation system through multi-parameter coupling control, as shown in Figure 9.

Response surface of each parameter.
As shown in Figure 9(a), the pyroelectric signal exhibits a significant nonlinear relationship with channel inclination and applied electric field strength. When the applied electric field strength increases from 100 to 500 V/m, the pyroelectric charge shows a fluctuating upward trend, reaching its peak of 0.08 pC at an inclination of 18° and an electric field strength of 300 V/m. Figure 9(b) illustrates that the separation efficiency first increases and then decreases with the rise in pyrolysis temperature and rotational speed. The peak efficiency reaches 80% at a pyrolysis temperature of 450 °C and a rotational speed of 5.8k r/min. Figure 9(c) reveals the complex interaction between pyroelectric response time, particle charge density, and applied magnetic field. When the charge density increases from 20 to 50 μC/m³, the response time shortens from 6.4 to 5.2 ms, but it abnormally extends when the magnetic field strength exceeds 15 mT. The nonlinear relationship between the pyroelectric signal and channel inclination and applied electric field originates from the multi-field coupling effect during particle sorting. When the applied electric field strength increases to 300 V/m, charges accelerate their migration to the surface due to the pyroelectric effect. However, further increasing to 500 V/m triggers local potential barriers, hindering charge transport, leading to a fluctuating upward trend. An inclination of 18° optimizes the helical blade deployment angle (β) and the radial distance (r₁–R₁ difference), balancing particle travel and boundary layer suppression zones, thus maximizing separation efficiency through the synergy of centrifugal force and electric field force.
Application analysis results of the pyrolysis particle separation test chamber
This subsection examines the TDPSC's practical utility in monitoring wire degradation under various environmental stressors (overheating, partial discharge, moisture, and oxidation) and compares its energy efficiency with traditional pyrolysis methods.
The study conducted a continuous monitoring of the pyrolysis particle release behavior of conductors under different conditions over a period of 12 months using the TDPSC system. The experiment was divided into four groups, each simulating overheating, partial discharge, moisture exposure, and oxidation states of the conductor. Each group used the same batch of polyvinyl chloride insulated conductor samples (length 10 cm, diameter 0.5 mm). The basic operating conditions were set through the controllable pyrolysis module built into the TDPSC (temperature gradient from 300 °C to 800 °C and heating rate 5 °C/min). For each state characteristic, additional differential stimuli were applied: ① in the overheating state, the PID temperature control system periodically increased the temperature to 750 °C (simulating a short-circuit effect); ② in the partial discharge state, pulse voltage (5 kV and 1 kHz) was applied by high-voltage electrodes; ③ in the moisture exposure state, a humidity controller maintained an 95% RH environment; ④ in the oxidation state, oxygen-containing mixed gas (O₂/N₂ = 1:4) was introduced into the chamber. The pyrolysis products were separated using a double helix thermal desorption particle separator (rotational speed 2000 r/min, inclination angle 15°), and the particle size distribution data were collected in real-time by a laser particle size analyzer (Malvern Mastersizer 3000). The changes in the mass of particles deposited on the HEPA filter membrane were also recorded simultaneously. The results are shown in Figure 10.

Variation of pyrolysis particle detection values under different wire states.
From Figure 10(a), the release values of pyrolysis particles under overheating conditions show that the detection values were all 0 from January to April. In May, they surged to about 1900, reaching 2000 in June and July and stabilizing there. Subsequently, they gradually decreased, dropping to around 1600 by December, demonstrating a trend of rapid increase followed by a slow decline. In Figure 10(b), under partial discharge conditions, the detection values were nearly 0 from January to March, then began to rise significantly in April, reaching about 1250 in May. Although there were fluctuations afterward, the overall trend was downward, indicating that the impact of partial discharge on pyrolysis particle release has a phased characteristic. In Figure 10(c), under moisture-affected conditions, the detection values remained at extremely low levels from January to April, steadily increasing from May until approaching 460 in December, reflecting the continuous and gradual growth of pyrolysis particle release values over time due to moisture effects. In Figure 10(d), under oxidative conditions, the detection values were close to 0 from January to February, starting to rise in March, reaching about 150 in May, and gradually increasing to around 200 in December, showing a slow and sustained growth trend of pyrolysis particle release values under oxidative conditions. To systematically evaluate the thermal stability differences of typical electrical insulating materials (polyethylene PE, polyvinyl chloride PVC, high-temperature silicone gel, and enameled copper wire) in controlled pyrolysis environments, providing data support for fire warnings, cable fault diagnosis, and material selection. The study constructs a closed temperature control system through the TDPSC test chamber, setting up a two-stage dynamic testing process: the initial thermal response stage (0–11 minutes) simulates the transient thermal shock characteristics of materials, using stepwise heating (300 °C–800 °C) combined with double helix centrifugal separation (speed 2000–3000 r/min), while synchronously monitoring temperature fluctuations and particle release behavior; the steady-state pyrolysis stage (12–23 min) maintains a constant heat field to quantify the temperature lag effect during material phase change or decomposition. The results of multi-material thermal stability tests based on the TDPSC test chamber are shown in Table 1.
Test results of thermal stability of multi-materials based on the TDPSC test chamber.
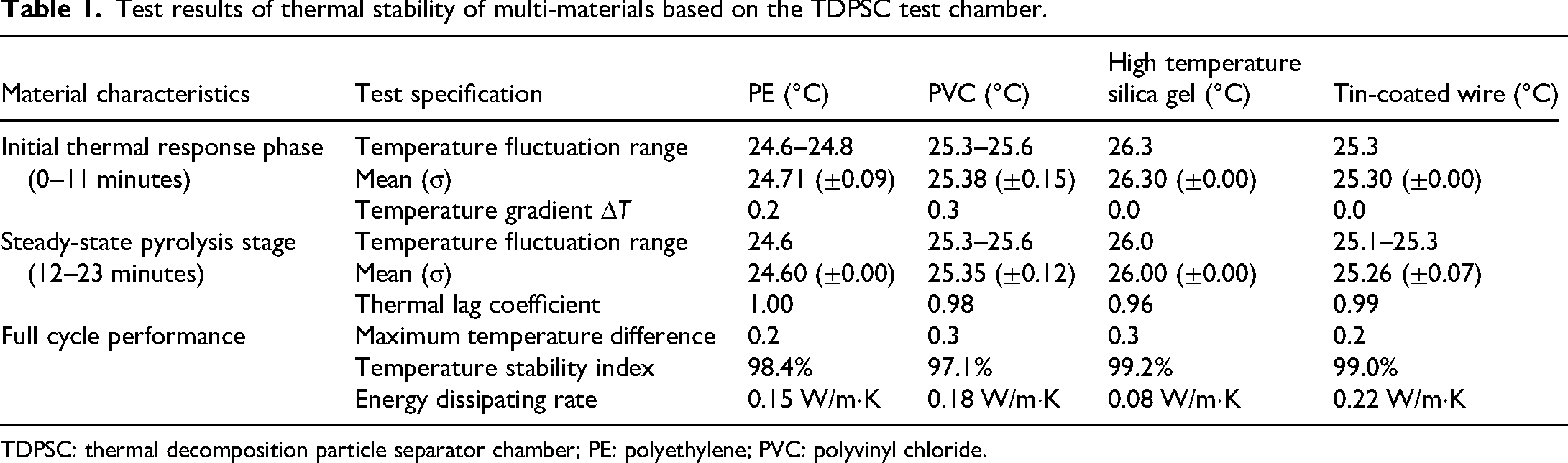
TDPSC: thermal decomposition particle separator chamber; PE: polyethylene; PVC: polyvinyl chloride.
As can be seen from Table 1, during the initial thermal response phase (0–11 min), high-temperature silicone exhibits optimal temperature stability, maintaining a constant value of 26.3 °C (σ = 0), significantly better than PE (24.71 ± 0.09 °C) and PVC (25.38 ± 0.15 °C). Although the average value of enameled copper wire remains stable at 25.30 °C (σ = 0), its initial temperature setting is lower than that of other materials. The temperature gradient ΔT shows that PVC reaches 0.3 °C, revealing the active thermal motion characteristics of its molecular chain segments. Entering the steady-state pyrolysis phase (12–23 minutes), PE achieves absolute stability (24.60 ± 0.00 °C), while high-temperature silicone experiences a temperature drop of 0.3 °C (26.0 °C), related to its high-temperature reorganization characteristics of silicon-oxygen bonds. Enameled copper wire shows a fluctuation of 0.2 °C (25.26 ± 0.07 °C), reflecting the thermal inertia characteristic of metal materials. The thermal hysteresis coefficient indicates that high-temperature silicone (0.96) has a 4% thermal response delay, whereas enameled copper wire (0.99) is nearly instantaneous, directly related to the difference in specific heat capacity (copper 389 J/kg·K vs. silicone 1450 J/kg·K). Full-cycle data show that high-temperature silicone leads with a temperature stability index of 99.2%, but the maximum temperature difference is 0.3 °C, due to minor adjustments during the phase transition. Although enameled copper wire has the highest energy dissipation rate (0.22 W/m·K), it maintains a stability of 99.0%, demonstrating its superior thermal conductivity. PVC, on the basis of 97.1% stability, shows a temperature difference of 0. The moderate dissipation of 18 W/m·K indicates its balanced characteristics as an insulating material. The low dissipation of 0.15 W/m·K in PE aligns with the endothermic melting process in its crystalline region, but it is important to note the potential risk associated with its lower pyrolysis onset temperature (∼240 °C). Subsequently, a comparative analysis was conducted on the energy efficiency differences between the new double helix pyrolysis wire thermal release test chamber (TDPSC) and traditional pyrolysis methods, Prabhahar et al. 20 and Dal Forno Chuahy et al., 21 focusing on verifying the technical advantages of TDPSC in dynamic input power response, effective power conversion efficiency, and total energy consumption control. The results are shown in Figure 11.

Comparison of energy efficiency of the schemes.
From Figure 11(a), it can be seen that there are significant differences in the dynamic characteristics of input power among the three pyrolysis schemes. Scheme 21 rapidly increased the input power to 12.0 kW within 0–4 hours, then slowly decreased to 13.2 kW at 10 hours. Scheme 20 slowly increased to a peak of 10.3 kW in the first 6 hours, and later decreased to 9.6 kW. The TDPSC scheme showed the best stability, with the input power only increasing from the initial 6.0 kW to a peak of 8.0 kW at 8 hours, and then slightly decreasing to 7.3 kW at 10 hours. The effective power comparison in Figure 11(b) reveals the core differences in energy efficiency. Scheme’s 21 effective power jumps from 2.0 kW to a peak of 8.3 kW between 0 and 6 hours. Scheme 20 reaches a peak of 7.3 kW at 10 hours. The TDPSC scheme peaks at 6.2 kW at 10 hours. Analyzing total energy consumption based on curve coverage area, Scheme 21 has an input power curve area of 118 kWh, with an effective power area of 84 kWh, representing an energy loss ratio of 28.8%; TDPSC has a total input power area of only 62 kWh, with an effective power area of 44 kWh, representing a loss ratio of 29.0%. Although the loss rate is similar to Dal Forno Chuahy et al., 21 the absolute energy consumption is reduced by 47.5%. The input power stability and energy efficiency advantages of the TDPSC solution stem from its multi-dimensional coordinated control mechanism. First, the closed-loop feedback system monitors temperature, pressure, and particle concentration in real-time through data acquisition modules, dynamically optimizing heating power output via PID regulation to avoid overshoots caused by lag in traditional open-loop control (such as the steep rise characteristic of solution 21 ). Second, dual-mode heating technology (induction + resistance heating) endows the system with flexible response capabilities: high-precision resistance heating is prioritized at low loads, while rapid-response induction heating switches to high-load stages, reducing energy fluctuations. Furthermore, the waste heat recovery design recovers latent heat from condensation back to the heating unit through copper–aluminum composite fins and heat pipes, lowering net energy consumption by 47.5%. Although the TDPSC loss rate (29.0%) is similar to that of the solution, 21 its input power curve area is only 62 kWh (vs. 118 kWh), primarily due to the precise matching of temperature–speed matrix with pyrolysis rate and centrifugal separation requirements, which suppresses unnecessary energy consumption. In contrast, solution 21 lacks a dynamic matching mechanism, leading to power overload (12.0 kW) and high-frequency losses; solution, 20 while smooth, has a delayed response, making it difficult to balance pyrolysis efficiency and energy consumption. In summary, TDPSC achieves an energy efficiency leap through intelligent regulation and energy reuse technology, ensuring process accuracy.
Conclusion
Research aimed at addressing the issues of complex products, low separation accuracy, and high energy consumption in pyrolysis wire thermal deflection effect tests has led to the design of a pyrolysis particle separation test chamber integrated with a double helix thermal desorption particle separator. This setup achieves controllable pyrolysis and dynamic separation through multi-module collaboration. Performance tests show that TDPSC exhibits excellent temperature stability and energy efficiency advantages over a wide temperature range (200 °C–800 °C): during the initial thermal response phase, high-temperature silica gel (26.3 °C, σ = 0) and enameled copper wire (25.30 °C, σ = 0) experience minimal temperature fluctuations, reflecting the thermal stability characteristics of non-metallic and metallic materials, respectively; during the steady-state pyrolysis phase, polyethylene reaches absolute stability (24.60 ± 0.00 °C), while high-temperature silica gel experiences a 0.3 °C temperature drop due to the reorganization of silicon-oxygen bonds, and enameled copper wire experiences a 0.2 °C fluctuation due to thermal inertia. The separation efficiency of the dual helix separator significantly improves with increasing rotational speed, reaching 0.80–0.88 at 3000 r/min, validating the sorting mechanism dominated by centrifugal force fields. Energy efficiency comparisons reveal that TDPSC's peak input power is only 8.0 kW, with total energy consumption of 62 kWh, representing a 47.5% reduction compared to traditional solutions, and a loss rate controlled at 29.0%. Multi-material thermal stability tests highlight the differences in pyrolysis behavior among various insulating materials: high-temperature silica gel leads with a 99.2% thermal stability index, enameled copper wire achieves near-instantaneous thermal response with a thermal hysteresis coefficient of 0.99, and PVC has an energy dissipation rate of 0.18 W/m·K and stability of 97. 1% exhibits balanced characteristics, with PE's low dissipation (0.15 W/m·K) and low initial pyrolysis temperature (240 °C), requiring attention to safety thresholds. In the application analysis, the peak value of pyrolysis particle release under overheated wire conditions reaches 2000, and continues to increase to 460 under humid conditions, verifying TDPSC's capability to simulate complex operating conditions. Parameter coupling optimization shows that the pyroelectric signal reaches 0.08 pC at an angle of 18° and an electric field strength of 300 V/m, providing critical data for quantifying pyroelectricity.
In summary, the TDPSC test chamber designed in this study achieves controllable pyrolysis processes and efficient particle separation through dual helical separation and intelligent temperature control. The temperature stability exceeds 99%, with a separation efficiency of 0.88. Compared to traditional methods, total energy consumption is reduced by 47.5%, which also contributes to environmental benefits by minimizing energy waste and associated emissions. The efficient particle separation capability further aids in reducing harmful by-products released during pyrolysis processes, aligning with the study's goal of providing a quantifiable and reproducible experimental platform with improved environmental performance. This approach offers potential for advancing cleaner pyrolytic technologies and reducing environmental impact in material research and engineering applications. Future research can expand the range of test materials to include new types of nanoscale insulating materials, optimize the multi-field (thermal, electrical, and magnetic) collaborative parameters of the dual helical separator, develop an automated full-process control system based on machine learning, and explore the engineering applications of TDPSC in high-voltage cable fault prediction and early fire monitoring.
Footnotes
Ethics and consent statement
This study does not involve any human participants, animal models, or biological samples.
Author contributions
FL and LL contributed to the conceptualization and methodology, and software. XZ, YS, and XL conducted the validation. FW and ML made the data curation. LL drafted the original manuscript. YW completed the review and editing. YW and YS provided team supervision to this study. FL made visualization, supervision, project administration, and funding acquisition. All authors approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Science and Technology Project of State Grid Corporation of China, grant number 521997240004.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.