Abstract
Keywords
Introduction
Nutrition is an important aspect of the treatment of adult patients in the intensive care unit (ICU). Enteral nutrition (EN) should be prioritised over parenteral nutrition (PN)1,2.There are different recommendations for regarding the most appropriate EN method. Although the most commonly used method is continuous infusion, intermittent and/or bolus nutrition administered during the day may be more practical and physiological.
3
Side effects such as hypoglycaemia, hyperglycaemia, high gastric residual volume (HGRV), intolerance, delayed reaching of target calories (TCs), or not reaching it at all have been reported during EN.3–8 Causes of hypoglycaemia and hyperglycaemia may include the inability to reach TC, overfeeding, inappropriate feeding rate (too high or too low), imbalance in metabolic processes, and development of insulin resistance.3,9,10 One of the most important barriers to reaching TC is the development of enteral feeding intolerance (FI), which have marker of HGRV. When studies comparing the effects of EN on the development of intolerance were examined, the continuous method was better tolerated because of the lower feeding rate, whereas bolus or intermittent feeding (IF) showed better outcomes.3,6,10–12 Sepsis is a condition with high mortality and morbidity and is frequently observed in critically ill patients, particularly those who are intubated. Nutrition is one of the vital part of the intensive care treatment of critically ill patients, especially those with sepsis.1,2 It is important to meet caloric needs appropriately. However, studies investigating the optimal route, composition, volume, and duration of feeding in critically ill patients with sepsis are lacking, and information on blood glucose level (BGL) and nutritional intolerance during feeding in such patients is insufficient.
13
Therefore, we designed this study to include patients with sepsis in the ICU. A few studies have demonstrated a direct relationship between EN and BGL
In addition to bolus and IF methods, continuous EN (CEN) has been reported to provide potential benefits in improving glycaemic control and maintaining stable nutrition in critically ill patients. Recent studies suggest that CEN can achieve more stable glycaemic control in patients with diabetes and in critically ill patients. Furthermore, the synergistic benefits of CEN combined with continuous insulin infusion may help minimise glucose variability and reduce gastrointestinal intolerance, which are crucial in the management of ICU patients with sepsis.14–16
The primary objective of this study was to compare the effects of bolus, intermittent, and CEN techniques on BGL in adult ICU patients with sepsis. The secondary objective was to compare their effects on EN intolerance.
Methods
This was a prospective, randomised, controlled study. Patients were enrolled after approval from the ethics committee, and signed consent was obtained from each patient's relatives. This study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. All patient details have been de-identified. The reporting of this study conforms to CONSORT guidelines. 17 The name of the ethics committee /review board is Health Science University Ümraniye Training and Research Hospital Ethics Committee (address: Health Science University Ümraniye Training and Research Hospital Adem Yavuz street, no 1, Istanbul, Turkey, approval number: B.10.1.TKH.4.34.H. GP.0.01/362; date of approval: 16/12/2021).
In our study, the qSOFA was used as a screening tool. When sepsis was suspected in a patient screened using the qSOFA, a blood culture was taken, and the patient was included in the study. If the culture result was negative during the study period, the patient was excluded.
Patients were not fed after 24:00 on the day of suspected sepsis, and were excluded if the intolerance was positive at 05:00.
Patients who received partial PN at any time during the study period were also excluded.
The amounts of TC and EN required were calculated using the Harris–Benedict (HB) formula. We did not use an indirect calorimetry device, which is recommended for critically ill patients in the ICU. In a meta-analysis, the using of HB formula was reported to have a 52% correlation and 50% agreement. In ESPEN's meta-analysis, it was noted that inflammatory status, body size, age, and minute volume appeared to be more strongly related to energy expenditure; however, further studies are needed. The protein target was calculated to be at least 1.2 g/kg (actual body weight was used as our patients had a BMI <30). Throughout the study, no patient received non-nutritional calories, such as propofol, dextrose infusion, or citrate dialysis. This is intendet by study protocol.
Patients were fed through a nasogastric catheter (No. 16–18), confirmed by PAAC radiography at initial insertion, and by auscultation before each feeding in a head-up position. Feeding was performed with the same EN formula at room temperature using a feeding pump.
The ‘closed envelope’ randomisation method was used. Patients who met the inclusion criteria were enrolled in the study within the specified period until the target number was reached.
In each group, 24 h was divided into four equal cycles, with start times at 06:00, 12:00, 18:00, and 24:00. By the third day, the TC was reached.

Time table. Blood glucose level (BGL) was measured four times a day at the same time before starting feeding in all three groups at 06:00 (T1), 12:00 (T2), 18:00 (T3) and 24:00 (T4). Subsequently, the designated feeding protocol for each group was initiated at 06:00 (T1), 12:00 (T2), 18:00 (T3), and 24:00 (T4).
The BGL was measured four times a day before nutrition started at 06:00 (T1), 12:00 (T2), 18:00 (T3), and 24:00 (T4) for 7 days in the biochemistry laboratory. The target range was accepted as 70–180 mg/dL.
In the case of hypoglycaemia (BGL < 70 mg/dL), 10% DXT 1 cc/kg was administered and recorded as the dextrose requirement. In the case of hyperglycaemia (BGL >180 mg/dL), crystallised insulin 1 U/10 mg/dL was administered and recorded as the insulin requirement. Administration was performed with hourly measurements until normoglycaemia (70 mg/dL < BGL < 180 mg/dL) was achieved. The number of administrations, dextrose requirement rate (number of dextrose requirements/number of patients administered dextrose) and insulin requirement rate (number of insulin requirements/number of patients administered insulin) were also recorded.
A target BGL target of 70–150 mg/dL is recommended for many ICU patients. However, interventions targeting this narrow range increase the risk of hypoglycaemia, which has a direct negative effect on cerebral function. To prevent this, we usually maintain a higher upper-limit target in the ICU. Therefore, in this study, we set our target as 70–180 mg/dL.
FI-HGRV assessment: Thirty minutes after stopping feeding, the N/G tube was drained, and the amount received was reassessed 30 min later. Gastric residual volume (GRV) was considered positive if more than 250 mL formula was returned with or without any other gastrointestinal symptoms (vomiting, diarrhoea, etc.). The same dose of treatment was administered. If the amount returned was less than 250 mL (negative GRV), the amount of formula was increased to the predetermined amount and continued.
The time to achieve TC was defined as the day on which 75% of the calculated calorie requirement was reached.
Outcome measures
Demographic data, NRS 2002 score, APACHE II, Reason for admission, qSOFA, TC, source of sepsis, comorbirty,
BGL (mg/dl) (four times a day, during seven days),
Hypoglycaemia,
Hyperglycaemia,
The number of administration of dextrose,
The number of administration of insulin,
Dextrose requirement rate (number of dextrose requirements/number of patients administered dextrose) and
İnsulin requirement rate (number of insulin requirements/number of patients administered insulin)
For secondry aim;
The time to achieve TC
FI-HGRV
Mean HGRV rate
were recorded.
These values were compared within and between groups to determine which group showed the least variability, including changes within the day, from day to day, and between groups.
Statistical analysis
For a power value of 0.80 and an alpha error probability of 0.05, the sample size per group was calculated as 29 (G*power 3.1, software). 6 However, we aimed to enrol at least 31 patients in each group to, increase the power of the study, and ensure equal numbers per arm.
The Number Cruncher Statistical System 2007 Statistical Software package (Utah, USA) was used. In addition to descriptive statistical methods (mean, standard deviation, median, interquartile range), the Shapiro-Wilk normality test was used to examine the distribution of variables. For variables showing a normal distribution, paired one-way analysis of variance was used for time comparisons, Newman–Keuls multiple comparison test for subgroup comparisons, one-way analysis of variance for intergroup comparisons, Tukey multiple comparison test for subgroup comparisons, Kruskal–Wallis test for intergroup comparisons of variables not showing a normal distribution, Dunn's multiple comparison test for subgroup comparisons, and chi-square test for comparisons of qualitative data. Statistical significance was set as p < 0.05.
Results
Of the 115 patients with sepsis who met the criteria, 22 were excluded (5 died, 8 were culture negative, and 9 met the exclusion criteria [1 started HDF, 6 had an increased APACHE II score, 1 developed shock, and 1 underwent GIS operation]) (Figure 2).
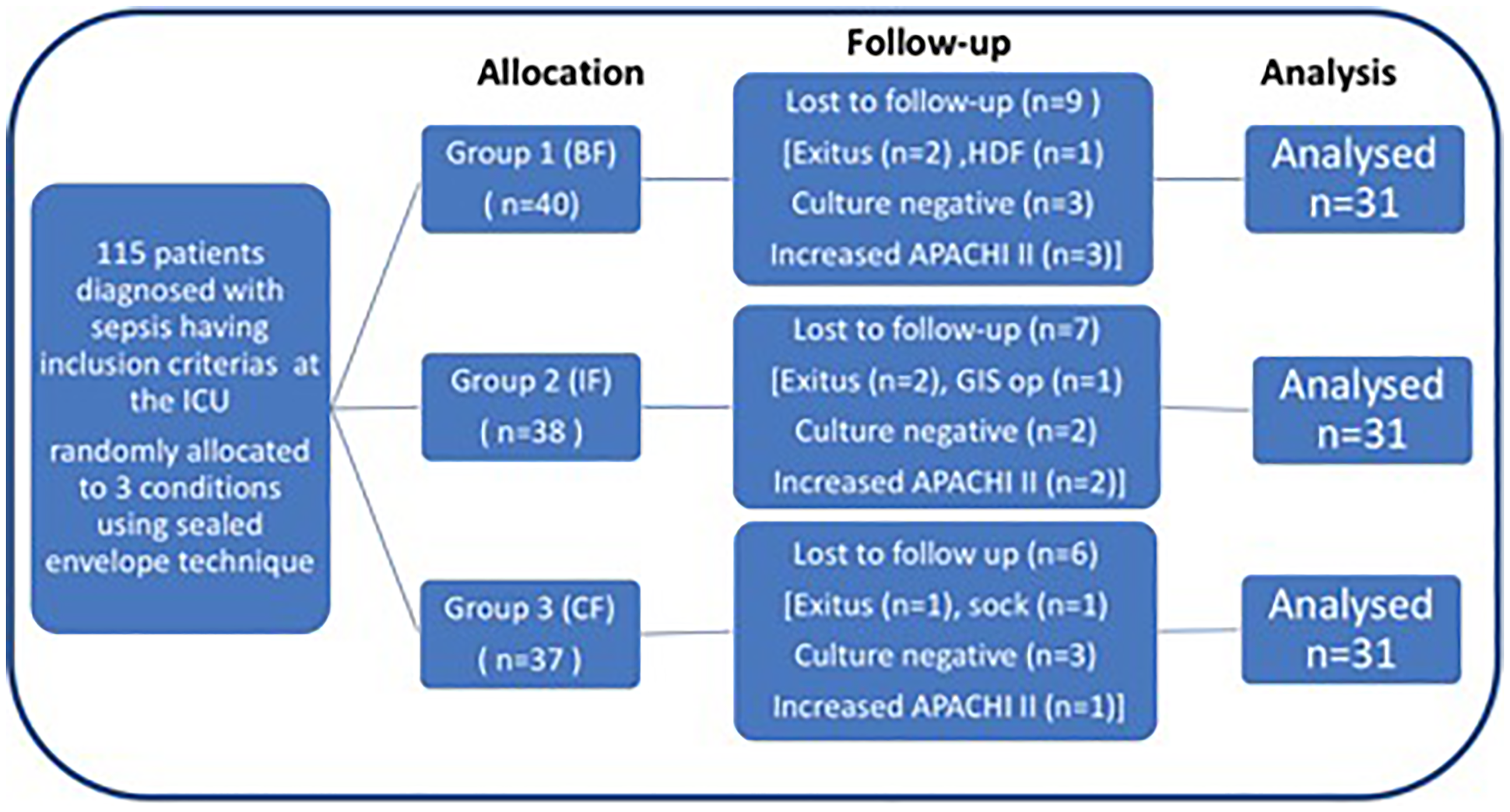
Flow diagram of the study.
The study was completed with 93 patients who were randomised into three groups, over a 6-month period.
There were no significant differences between the groups in terms of demographic and clinical characteristics, time to achieve the targeted calorie intake, or number of patients with HGRV (p > 0.05). The mean HGVR rate was higher in the BF group than in the IF group (Table 1(a) and (b); *p = 0.014, **p = 0.011).
(a) and (b) Demographic and clinical characteristics: The study was completed with a total of 93 patients randomised into 3 groups.
Table 1a.

Comparison of Time to achieve the targeted calorie intake and HGRV among groups.

BF: bolus feeding; IF: intermittent feeding; CF: continuous feeding; BMI: body mass index; HGRV: high gastric residual volume.
One way analysis of variance.
Kruskal–Wallis test + Chi-square test.
The group with the highest frequency of HGRV was the bolus group, while the intermittent group exhibited the lowest frequency. In Group BF, BGLs on the first day were higher than those in the other two groups but did not follow a stable course thereafter. They were more variable on a daily basis, and showed more high/low values (7/28 and 9/28, respectively) compared with the other two groups. In Group IF, almost half of the measurements (13/28) were lower than in the other groups, starting from the first day, and continuing to be lower, but these values remained closer to the target range (70–180 mg/dL). In Group CF, most measurements (15/28) were higher than those in the other two groups, and none were lower (Tables 2 and 3, Figure 3).

The variation of blood glucose values according to the time points during 7 days. Group BF; Group IF; Group CF. Blood glucose level (mg/dL) measurements: T1 at 06:00; T2 at 12:00; T3 at 18:00; T4 at 24:00. When analysing the course of T1, T2, T3 and T4 over seven days, it was found that the glucose values varied at all times in Group BF (p = 0.003, p = 0.002, p = 0.0001), at T2 and T4 in Group IF (p = 0.002, p = 0.0001) and at T3 and T4 in Group CF. (p = 0.0001, p = 0.008, POWAV, NKMCT, p < 0.05). BF: bolus feeding; IF: intermittent feeding; CF: continuous feeding; POWAV: paired one-way analysis of variance; NKMCT: Newman–Keuls multiple comparison test.
Comparison of mean blood glucose levels (mg/dL) among groups.

Comparison of mean blood glucose levels (mg/dl) among time points and among days.

BGL was measured four times daily at 06:00 (T1), 12:00 (T2), 18:00 (T3), and 24:00 (T4) for 7 days (4 multiplication by 7 = 28 time points, 28 multiplication by 3 groups = 84 mean BGLs obtained). BGL: blood glucose levels; BF: bolus feeding; IF: intermittent feeding; CF: continuous feeding.
Kruskal–Wallis (Mann–Whitney U test).
Difference with ¹bolus group p < 0.05, Difference with 2IF group p < 0.01, Difference with ³CF continious feeding group p < 0.05).
The intraday changes in BGLs were evaluated within the groups.
In Group BF, BGLs showed no significant difference on the first, second, and fourth days, progressively decreased on the third day, and fluctuated (high–low) on the following days.
In Group IF, there was no difference on day 1; levels were progressively lower on days 2, 3, 4, and 6, and fluctuated on days 5 and 7. Glucose levels were highest in the morning, progressively decreased during the day, and were lowest at night (24:00/fourth measurement) (p = 0.001, p = 0.005).
In Group CF, BGLs remained stable on the second, third, fifth, sixth, and seventh days. On the first day, they increased towards the evening, and on the fourth day, they showed a fluctuating pattern (Tables 2 and 3, Figure 3, p = 0.005, p = 0.026, p = 0.011).
The risk of developing day–night differences was assessed using the Kruskal–Wallis test Dunn's multiple comparison test. In general, morning glucose levels tended to decrease in the BF and CF groups, and particularly in Group BF. In Group IF, levels tended to increase, and this was significantly higher than that in the other two groups after 4 days. On the last day, the day–night difference was greater in Group CF than in Group BF (Tables 2 and 3, Figure 3).
In our study, dextrose was administered 3 times in two patients in Group BF, 4 times in four patients in Group IF, and 2 times in two patients in Group CF. Insulin was administered 14 times in 11 patients in Group BF, 6 times in 5 patients in Group IF, and 26 times in 15 patients in Group CF. Insulin requirements were lower in Group IF than in the other two groups (Table 4; +p = 0.025, Table 5). The number of hypoglycaemia cases in Group IF, which appeared notable in Figure 2, was not statistically significant. There were no significant differences between the groups in terms of the dextrose requirement, dextrose requirement rate, or insulin requirement rate (Table 4; p > 0.05, Table 5).
Comparison of dextrose and insulin usage among groups.

BF: bolus feeding; IF: intermittent feeding; CF: continuous feeding.
Chi-square test.
One way analysis of variance.
Kruskal–Wallis test.
Comparison of number of dextrose and insulin administration among groups.

BF: bolus feeding; IF: intermittent feeding; CF: continuous feeding.
Difference with ¹bolus group p < 0.05, Difference with 2IF group p < 0.01, Difference with ³CF continious feeding group p < 0.05).
Discussion
In our prospective, randomised, controlled trial of ventilated patients with sepsis in the adult ICU, we compared the effects of bolus, intermittent, and CEN methods on BGL as the primary objective and intolerance risk as the secondary objective. We found that intermittent EN appeared superior to the other methods in maintaining normoglycaemia, as reflected by a higher proportion of BGLs within target limits, the lowest insulin use, and the least intolerance.
Most studies comparing EN methods assess the effects of multiple mediators on BGL; however, few have directly measured BGL. 12 Hormones such as ghrelin, glucagon-like peptide, insulin, C-peptide, GIS hormones, and markers of muscle protein synthesis have been studied, with varying results. Their release is influenced positively or negatively by the timing, method, and amount of EN.9,12,18
According to ESPEN, CEN is recommended in the ICU, but with low-level evidence due to heterogeneous study groups (B, 95% agreement). Three different findings have been reported: HGRV is higher and calorie delivery is better with BF3,6; the target is reached faster with CF, but GIS symptoms do not differ. 19 These techniques are therefore not considered superior to each other. 3 The only ICU study comparing BGLs in bolus and CF found no difference in insulin requirement or time to TC. 12 Based on these limited data, ESPEN concluded that bolus and CEN can both achieve nutritional goals without a differential increase in side effects. 3
Therefore, CEN is widely used. However, it may be interrupted during interventional procedures, prolonging the time required to reach the TC, reducing caloric intake, and potentially leading to hypoglycaemia. 9 In the long term, CF may also alter gastric hormone secretion and cause metabolic complications such as hyperglycaemia and insulin resistance.
The BGL is part of the physiological response to stress and haemostasis. A number of studies have reported an association between the severity of sepsis and the occurrence of hypoglycaemia or hyperglycaemia. 12 Therefore, in order to minimise confounding factors affecting BGLs, patients with diabetes, patients requiring steroids (>0.5 mg/kg), and those with baseline APACHE II scores that increased by more than 5 points during the study were excluded.
Gonzalez et al. reported that CF impairs hepatic insulin sensitivity, increasing insulin requirements to maintain euglycaemia, whereas IF may be more favourable in terms of endocrine and metabolic effects. 20 McNelly et al. found that IF may help establish a regular circadian rhythm, achieve higher nutritional targets than the CF regimen, and does not significantly increase hypoglycaemia incidence. 10 Although patients on IF required less exogenous insulin on days 8 and 9, cumulative insulin use did not differ between groups.
In this study, the CEN group had the highest insulin requirement, similar to Gonzalez's findings. However, the intermittent protocol used in their study was equivalent to the bolus protocol in this study. We observed the most variable glucose levels during and between days in the bolus group, that where insulin was required intermittently.
Daily glucose variability is an important parameter influencing mortality, especially in patients with sepsis.12,21,22 Even when the BGL is elevated, lower variability has been associated with reduced mortality. 12 Therefore, the EN method that minimises BGL fluctuations is recommended to lower mortality risk. In our study, BGLs were higher but appeared to be more stable in the continuous group. However, this was achieved through increased insulin use. Glucose levels were observed to decrease in the morning in both the continuous and bolus groups. In contrast, the intermittent group demonstrated lower, more stable BGLs within the target range, without increased insulin or dextrose requirements. Furthermore, levels exhibited a tendency to decrease progressively throughout the day. But the mortality rates were not evaluated in this study.
The observed night–morning variability between groups may be related to modulators of BGL (e.g. cortisone, gut peptides, and ghrelin) secreted in circadian patterns, which are themselves affected by sepsis and/or EN methods. More detailed studies with larger cohorts are required to clarify these associations.
When a comparison is made between EN methods in terms of time to TC intake, a divergence of results is observed: Some studies favour CEN, others favour intermittent or BF, and some report no difference.7,11,12 Additionally, no differences in HGRV between EN methods have been noted.6,9
In the study by Lee et al., which used a feeding protocol different from ours (the intermittent group in their study resembled the bolus group in this study), CF significantly improved the achievement of TC. 11 In contrast, MacLeod et al. compared continuous drip feeding with IF in six parts every 4 h. They observed no significant difference in complications, such as intolerance. However, the intermittent feeding group demonstrated a higher rate of achieving target intake by day 7. They concluded that intermittent feeding was simpler and yielded results that were equivalent to CF. 7 In our trial, none of the three methods was found to be superior in terms of time to TC intake. In all groups, TC was reached on day 3. HGRV occurred the most frequently in the bolus group and least frequently in the intermittent group. These differing outcomes observed may be attributed to variations in study design, inclusion criteria, patient characteristics, sample sizes, feeding protocols, patterns, target glucose range, interruptions in CF, or initial feeding amounts.6,7,9,11,12
In our study, feeding was initiated, and BGL and HGRV were measured simultaneously for each group. The inclusion and exclusion criteria, as well as the incremental increases in nutritional intake, followed a strict protocol. Our study was conducted using an innovative, original, and analytical approach to a subject that still requires clarification in the literature.
We initially hypothesised that BGL would be more stable in the bolus-fed group. However, in critically ill patients with sepsis, split-meal feeding did not result in improved BGL, and this group demonstrated the highest intolerance. This may be attributable to the large volume of formulas delivered in a short period.
The better tolerance observed in our intermittent group, which received continuous infusion during the day and no feeding at night, may be explained by allowing the GIS to utilise the night period for digestion. Similar to healthy individuals, circadian rhythm-dependent hormonal and modulatory secretions were not adversely affected, and the side effects commonly associated with BF were absent.
Although numerous studies have investigated EN, only a few have compared three different feeding methods in patients with sepsis using the parameters applied in this study. We hope our findings will contribute to this area, which still requires further clarification.
Conclusıon
We recommend that intermittent EN be considered for ICU patients with sepsis to achieve better glycaemic control and reduced the risk of gastric intolerance.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251392606 - Supplemental material for Which is better for glycemic control? bolus, intermittent (nocturnal pause) or continuous enteral feeding in septic critically ill patients
Supplemental material, sj-docx-1-sci-10.1177_00368504251392606 for Which is better for glycemic control? bolus, intermittent (nocturnal pause) or continuous enteral feeding in septic critically ill patients by Senay Goksu and Zeliha Tuncel in Science Progress
Footnotes
Acknowledgements
The authors would like to express our gratitude to Yonca Yanli, MD and Prof. Dr Yalim Dikmen, MD.
Ethical approval
This prospective, randomised, controlled study was conducted in accordance with the Declaration of Helsinki (1975, revised 2024). The Health Science University Ümraniye Training and Research Hospital Ethics Committee approved the study (Approval No: B.10.1.TKH.4.34.H.GP.0.01/362; Date: 16/12/2021). The study is registered with ClinicalTrials.gov (Identifier: NCT05812664).
Consent for publication
No data and visuals identifying the individual person have been used. All patient data were de-identified.
Informed consent
Written informed consent was obtained from the relatives of all patients with sepsis prior to enrolment.
Author contribution
SG and ZT were critical in conceptualisation and design of the study, SG collected and analyzed the data, SG and ZT wrote the manuscript; performed the statistical analysis, revised and critically reviewed the manuscript; All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data supporting the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
