Abstract
Objective
Sepsis-associated encephalopathy (SAE) is a critical complication of sepsis with high mortality, yet reliable prognostic biomarkers remain limited. This study aimed to investigate the association between the lactate-to-albumin ratio (LAR) and short- and long-term mortality in SAE patients.
Methods
A retrospective cohort analysis was conducted using the MIMIC-IV database, including 874 adult SAE patients. LAR was calculated within 24 h of ICU admission. Participants were stratified into quartiles (Q1-Q4) based on LAR values. Cox proportional hazards models and restricted cubic spline (RCS) were employed to evaluate relationships between LAR and mortality at 30, 90, 180, and 365 days. The area under the receiver operating characteristic (ROC) curve (AUC) was employed to evaluate the predictive performance of LAR.
Results
Kaplan-Meier analysis demonstrated that, the Q2,Q3,Q4 group experienced significantly lower survival rate compared to the Q1 group for 30-day, 90-day, 180-day, and 365-day, with the Q4 group had the lowest survival rate. Cox proportional hazards regression revealed LAR as a robust independent predictor, demonstrating persistent prognostic significance across all observed time points. RCS analysis revealed a nonlinear association between LAR and the mortality of SAE. The ROC curves demonstrated robust predictive ability of LAR, with AUCs of 0.753 (30-day), 0.736 (90-day), 0.733 (180-day), and 0.725 (365-day).
Conclusions
LAR serves as an easily accessible, independent prognostic biomarker for predicting short- and long-term mortality in SAE patients, exhibiting a distinct nonlinear correlation with mortality risks over these periods. These results underscore its potential value in risk stratification and in guiding early clinical interventions. Additional prospective studies are needed to confirm its role in the management of SAE.
Keywords
Introduction
Sepsis-associated encephalopathy (SAE) is a common and serious complication in patients with sepsis, characterized by diffuse brain dysfunction in the absence of direct central nervous system infection. 1 It is estimated that up to 70% of sepsis patients are complicated by SAE, which not only significantly increases morbidity and mortality but also may lead to long-term cognitive impairment in survivors. 2 The progression of SAE is driven by multiple interrelated mechanisms, including systemic inflammation, blood–brain barrier disruption, neuronal impairment, and altered cerebral perfusion. 3 Emerging experimental studies have revealed that the JAK/STAT and JNK signaling pathways are critically involved in the neuroinflammatory and oxidative mechanisms underlying SAE. Pharmacological inhibition of these pathways has been shown to exert neuroprotective effects by alleviating neuronal damage and improving cognitive outcomes.4,5 Recent evidence has identified coagulase-negative staphylococci as significant risk factors for the development of SAE, although not directly associated with mortality. 6 These findings underscore the pivotal role of inflammatory and oxidative stress pathways in the pathogenesis of SAE and provide promising therapeutic targets.
Despite advances in understanding the underlying mechanisms and potential therapeutic targets, the lack of reliable prognostic biomarkers remains a major barrier to improving clinical outcomes in SAE. Therefore, there is an urgent need to identify accessible, cost-effective prognostic biomarkers based on routinely available laboratory parameters, to facilitate early detection and improve the management of patients with SAE.
Serum lactate levels are a well-recognized indicator of tissue hypoperfusion and have been shown to be associated with organ failure and death in a variety of critical conditions, including sepsis and trauma.7–9 As the end product of glycolytic metabolism, lactate is both an intercellular signaling molecule and an energy substrate for neuronal activity in the brain, and changes in its concentration have been associated with neuropsychiatric disorders.10,11 Lactate affects neuronal excitability, brain pH regulation, fluid homeostasis, neurovascular coupling and long-term memory formation. 12 Most studies have shown that elevated serum lactate levels in patients are associated with more severe injuries and poorer prognoses. However, serum lactate is influenced by various factors, including liver and kidney function. Albumin, a negative acute phase reactant (APR) produced by the liver, decreases in serum levels during inflammation. This decrease is due to both decreased production by the liver and increased protein hydrolysis, as the body redirects amino acids to produce positive APRs that aid in the fight against inflammation.13,14 In order to stabilize and improve the predictive value of serum lactate, a new index that combines the clinical significance of lactate and albumin has been developed–the lactate to albumin ratio (LAR). Elevated LAR has been evaluated in several studies and has been associated with a poorer prognosis in patients with a range of critical illnesses, including acute pancreatitis, sepsis, and cardiac arrest.15–17
To the best of our knowledge, there are no studies examining the clinical association between LAR and mortality in SAE. The aim of this study was to explore the association between LAR levels and all-cause mortality in patients with SAE admitted to the ICU using data from the Medical Information Mart for Intensive Care IV (MIMIC-IV) database This retrospective cohort study was designed to address this gap in knowledge, which has significant potential for understanding prognostic factors for SAE and may pave the way for the development of better predictive models and therapeutic strategies for the management of this complex disease.
Methods
Data source and study population
This study extracted de-identified ICU patient records from the MIMIC-IV 3.1 database,18,19 a publicly accessible repository containing anonymized clinical data from Beth Israel Deaconess Medical Center (Boston, MA) spanning 2008–2022. Prior to data acquisition, the research team completed the NIH-certified “CITI Data or Specimens Only Research” training program. Author Fengzhen Huang holds valid authorization (Record IDs: 63858817, 63858818) to access this resource for academic investigation. This study was approved by the Massachusetts Institute of Technology and Beth Israel Deaconess Medical Center.
According to the Sepsis-3 criteria, sepsis was defined as a suspected infection with a SOFA score ≥2. 20 SAE was diagnosed in patients with sepsis who presented with delirium or a GCS score <15. 21 Eligible cases met all inclusion criteria: age ≥18 years, first-time ICU admission, and ICU hospitalization lasting ≥24 h. Exclusion criteria covered: (1) Primary neurological conditions (cerebrovascular disease, traumatic brain injury, intracranial infection or tumor, or epilepsy); (2) Metabolic or organ dysfunction-related encephalopathies (hepatic, renal, or hypertensive); (3) Severe metabolic abnormalities (sodium <120 or >150 mmol/L; glucose <54 or >180 mg/dL); (4) Substance use disorders or psychiatric/neurodegenerative conditions; (5) Incomplete essential laboratory data.
Clinical variables and outcomes
Clinical data extraction encompassed demographics, laboratory parameters, vital signs, severity scores, treatments, comorbidities, and discharge outcomes. Initial clinical assessments (laboratory results, vital measurements, and severity scoring) were obtained within the first 24 h of hospitalization or ICU admission. LAR was calculated as the ratio of serum lactate (mmol/L) to albumin (g/dL). 22 Participants were stratified into quartiles (Q1-Q4) based on LAR values. The primary outcome was 30-day mortality, while secondary outcomes included hospital and ICU stay durations, ICU mortality, and mortality rates assessed at 60, 90, 180, and 365 days post-admission.
Statistical analysis
Statistical analyses were performed in R (version 4.4.1). Baseline variables exhibited less than 10% missing data, addressed through random imputation via the “mice” package in R software. After conducting normality tests, it was determined that all continuous variables in our dataset were non-normally distributed. Consequently, these variables were analyzed using the Mann-Whitney U test and are presented as medians with interquartile ranges (IQR). Categorical variables were assessed using chi-square tests and reported as frequencies or percentages. Survival curves were generated via the Kaplan-Meier method, with between-group differences evaluated by log-rank tests. Cox proportional hazards models quantified the association between LAR and mortality risk, yielding hazard ratios (HR) and 95% confidence intervals (CI). Restricted cubic splines (RCS) explored relationships between LAR and mortality for 30-day, 90-day, 180-day, and 365-day. Subgroup analyses assessed outcome heterogeneity across demographic and clinical strata. The predictive performance of LAR for 30-day, 90-day, 180-day, and 365-day mortality was evaluated using receiver operating characteristic (ROC) curves, with discriminative capacity quantified by the area under the curve (AUC). Statistical significance was defined as a two-sided P < 0.05.
Results
Figure 1 outlines the participant selection flowchart. From the MIMIC-IV database, 18,649 initially identified SAE cases met first-time ICU admission criteria. After applying inclusion/exclusion filters, 874 individuals were included in the final cohort. The enrolled population comprised 543 males (62.1%) and 331 females (37.9%), with a median age of 70.64 years. Racial distribution showed 598 (68.4%) White participants and 276 (31.6%) from other ethnic backgrounds.
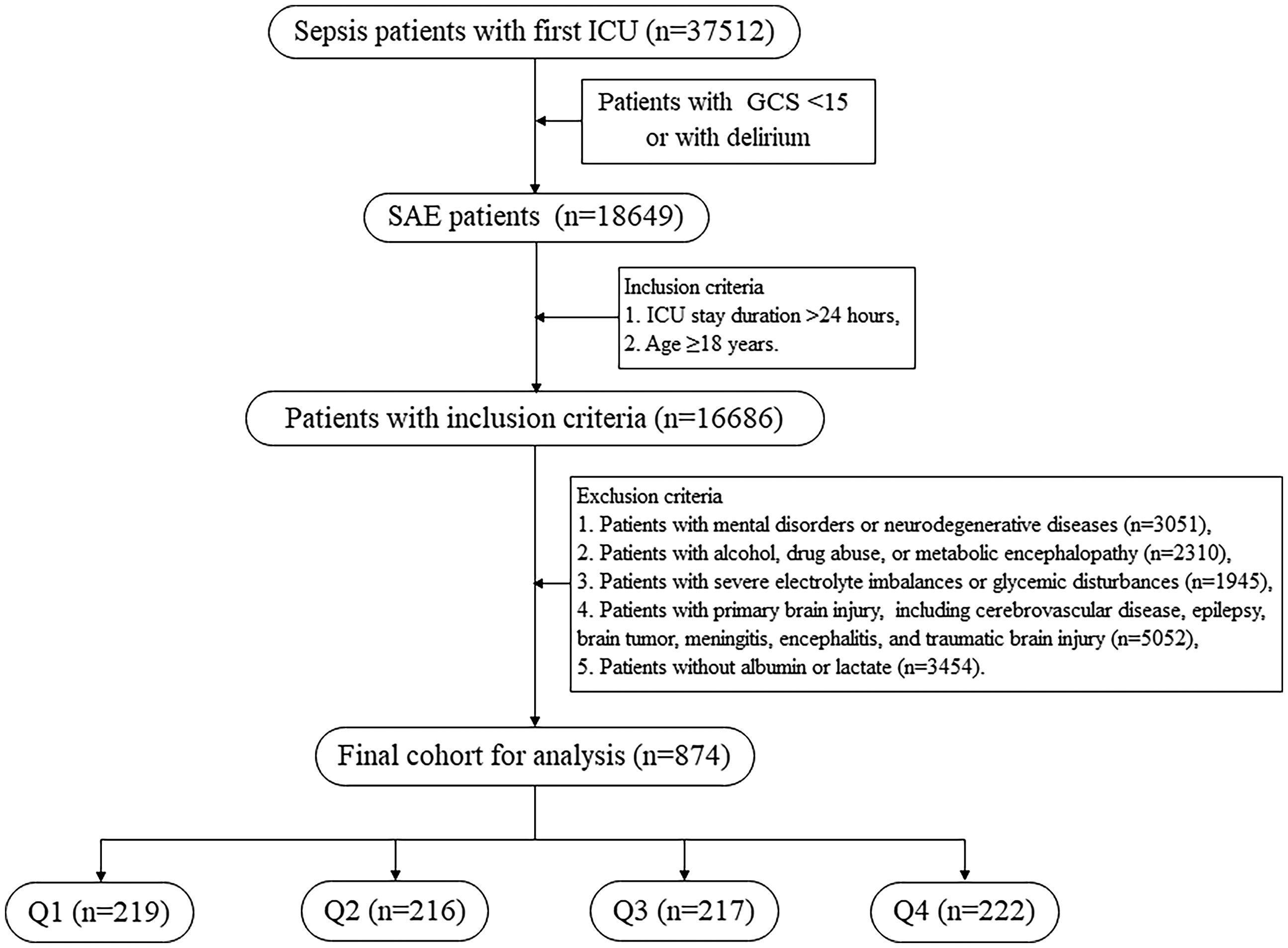
The flowchart for the selection of SAE patients.
Baseline characteristics
Table 1 summarizes baseline characteristics of SAE patients stratified by LAR quartiles (Q1: 0–0.29; Q2: 0.29–0.41; Q3: 0.41–0.67; Q4: > 0.67). We observed a significant increasing trend in the heart rate and respiratory rate as the LAR level increased (P < 0.001). In terms of disease severity scores, the sepsis-related organ failure assessment (SOFA), acute physiology score III (APSIII), Oxford acute severity of illness score (OASIS), and Charlson comorbidity index (CCI) increased significantly with the increase in LAR levels (P < 0.05). Regarding treatment, patients with higher LAR levels were more likely to receive continuous renal replacement therapy (CRRT; P = 0.001). Comorbidity analysis revealed a rising prevalence of septic shock alongside declining rates of hypertension in association with incremental LAR levels (P < 0.05).
Baseline characteristics of SAE patients.

Table 2 compares baseline characteristics between survivors and non-survivors at 30-day follow-up. Non-survivors were older and exhibited lower body weight compared to survivors (P < 0.05). Laboratory profiles demonstrated elevated WBC, BUN, Creatinine, and potassium levels alongside reduced RBC counts, hemoglobin, platelets, and sodium concentrations in non-survivors (P < 0.05). Disease severity metrics (SOFA, APSIII, OASIS, CCI) were significantly higher in the non-survivor cohort (P < 0.05). Clinically, non-survivors required more frequent interventions such as CRRT and mechanical ventilation (P < 0.05). Median LAR values were markedly elevated in non-survivors relative to survivors (0.70 vs. 0.38; P < 0.001). Comorbidity analysis revealed hypertension prevalence declined, while septic shock incidence rose significantly in non-survivors (P < 0.05).
Baseline characteristics between survivors and non-survivors.

Composite outcomes of different groups
We compared composite outcomes across groups with different levels of LAR (Table 3). When comparing the group with low levels of LAR (Q1), we observed that patients in Q2, Q3, Q4 groups had longer hospital stay and ICU time (P < 0.05). Moreover, the Q2, Q3, Q4 groups were associated with higher mortality rates for ICU, 30-day, 60-day, 90-day, 180-day, and 365-day (P < 0.05). The results suggested that elevated levels of LAR were linked to a greater risk of mortality over both short-term and long-term periods, highlighting their potential role as prognostic indicators in SAE patients.
Composite outcomes of SAE patients.

Associations between LAR and mortality
Kaplan-Meier analysis revealed a progressive decline in survival rates for 30-day, 90-day, 180-day, and 365-day follow-ups with ascending LAR quartiles (Q1–Q4), where Q4 exhibited the lowest survival probability (P < 0.001) (Figure 2). Cox proportional hazards regression demonstrated that continuous LAR values were associated with mortality risk at all time points, with unadjusted analyses confirming significantly higher mortality in Q2–Q4 compared to Q1. After multivariable adjustment (Model 3), the adjusted HRs (95% CIs) from LAR Q1 to Q4 categories were 1.00 (reference), 3.04 (1.29–7.15), 4.43 (1.94–10.12), and 5.11 (2.25–11.63), respectively, for 30-day mortality (P for trend < 0.001); 1.00 (reference), 2.11 (1.09–4.09), 2.93 (1.55–5.51), and 4.25 (2.27–7.95), respectively, for 60-day mortality (P for trend < 0.001); 1.00 (reference), 2.09 (1.15–3.79), 2.61 (1.46–4.64), and 3.73 (2.11–6.62), respectively, for 90-day mortality (P for trend < 0.001); 1.00 (reference), 2.15 (1.25–3.72), 2.61 (1.54–4.43), and 3.44 (2.03–5.85), respectively, for 180-day all-cause mortality (P for trend < 0.001); and 1.00 (reference), 1.77 (1.10–2.85), 2.23 (1.41–3.52), and 2.75 (1.74–4.37), respectively, for 365-day all-cause mortality (P for trend < 0.001). Similarly, the adjusted HRs (95% CIs) of LAR for 30-day, 60-day, 90-day, 180-day, and 365-day mortality in model 3 were 3.25 (1.85–5.72), 2.84 (1.84–4.39), 2.54 (1.71–3.79), 2.39 (1.65–3.46), and 2.08 (1.51–2.87), respectively (all P < 0.001) (Table 4).

Survival curves of SAE patients according to different LAR levels based on Kaplan-Meier survival analysis. (A) 30-day mortality; (B) 90-day mortality; (C) 180-day mortality; (D) 365-day mortality. Shaded areas represent the 95% confidence intervals for the survival estimates.
Association between LAR and mortality in SAE patients.

HR: Hazard ratio, CI: confidence interval.
Model1: Crude.
Model2: Adjust: age, weight, gender, race, insurance.
Model3: Adjust: age, gender, race, weight, insurance, hemoglobin, platelet, RBC, WBC, bun, creatinine, glucose, sodium, potassium, heart_rate, SBP, DBP, temperature, Resp_rate, Spo2, SOFA, GCS, APSIII, OASIS, CCI, CRRT, Vasoactive_agent, ventilation, hypertension, diabetes, COPD, Septic_shock, delirium.
Detection of nonlinear relationship between LAR and mortality
RCS regression was employed to evaluate relationship between LAR and mortality in SAE patients. Analyses demonstrated that LAR consistently exhibited a non-linear L-shaped relationship with mortality for 30-day, 90-day, 180-day, and 365-day periods, with both the overall P-values and the P-values for non-linearity being less than 0.05 (Figure 3). The spline curve illustrated a significant positive correlation, where elevating LAR levels corresponded to progressively higher mortality hazards. The curve is relatively steep before the threshold point, indicating that within this range, the impact of changes in LAR on the outcome is greater. The curve becomes flat after the threshold point, indicating that within this range, the impact of changes in LAR on the outcome is relatively smaller.

Restricted cubic spline curves of LAR for mortality in SAE patients. (A) 30-day mortality; (B) 90-day mortality; (C) 180-day mortality; (D) 365-day mortality.
Predictive value of mortality in SAE patients
The ROC curves evaluated the predictive performance of the LAR, lactate, and SOFA scores for mortality ranging from 30 to 365 days in patients with SAE (Figure 4). The AUC values consistently ranked LAR as the highest among individual predictors: 0.753 (30-day), 0.736 (90-day), 0.733 (180-day), and 0.725 (365-day), compared to lactate (30-day: 0.677; 90-day: 0.651; 180-day: 0.650; 365-day: 0.636) and SOFA scores (30-day: 0.575; 90-day: 0.563; 180-day: 0.544; 365-day: 0.540). Integrated models combining LAR and SOFA scores achieved the highest discriminative capacity (AUC: 0.794 at 30 days; 0.795 at 90 days; 0.792 at 180 days; 0.796 at 365 days), highlighting the enhanced accuracy of multivariable risk stratification over single biomarkers. These results underscore LAR's superior prognostic utility relative to both lactate and SOFA scores.

The ROC curves illustrate the predictive performance of LAR alone, lactate alone, SOFA score alone, and the combined LAR-SOFA score for mortality in SAE patients. (A) 30-day mortality; (B) 90-day mortality; (C) 180-day mortality; (D) 365-day mortality.
Subgroup analysis and forest plots
Figure 5 illustrates the forest plot of the subgroup analysis examining the association between LAR and 30-day mortality in patients with SAE. In the subgroup analyses based on LAR quartiles as categorical variables, the p-values for interaction between LAR and each stratification factor were all greater than 0.05, supporting the robustness of the results.
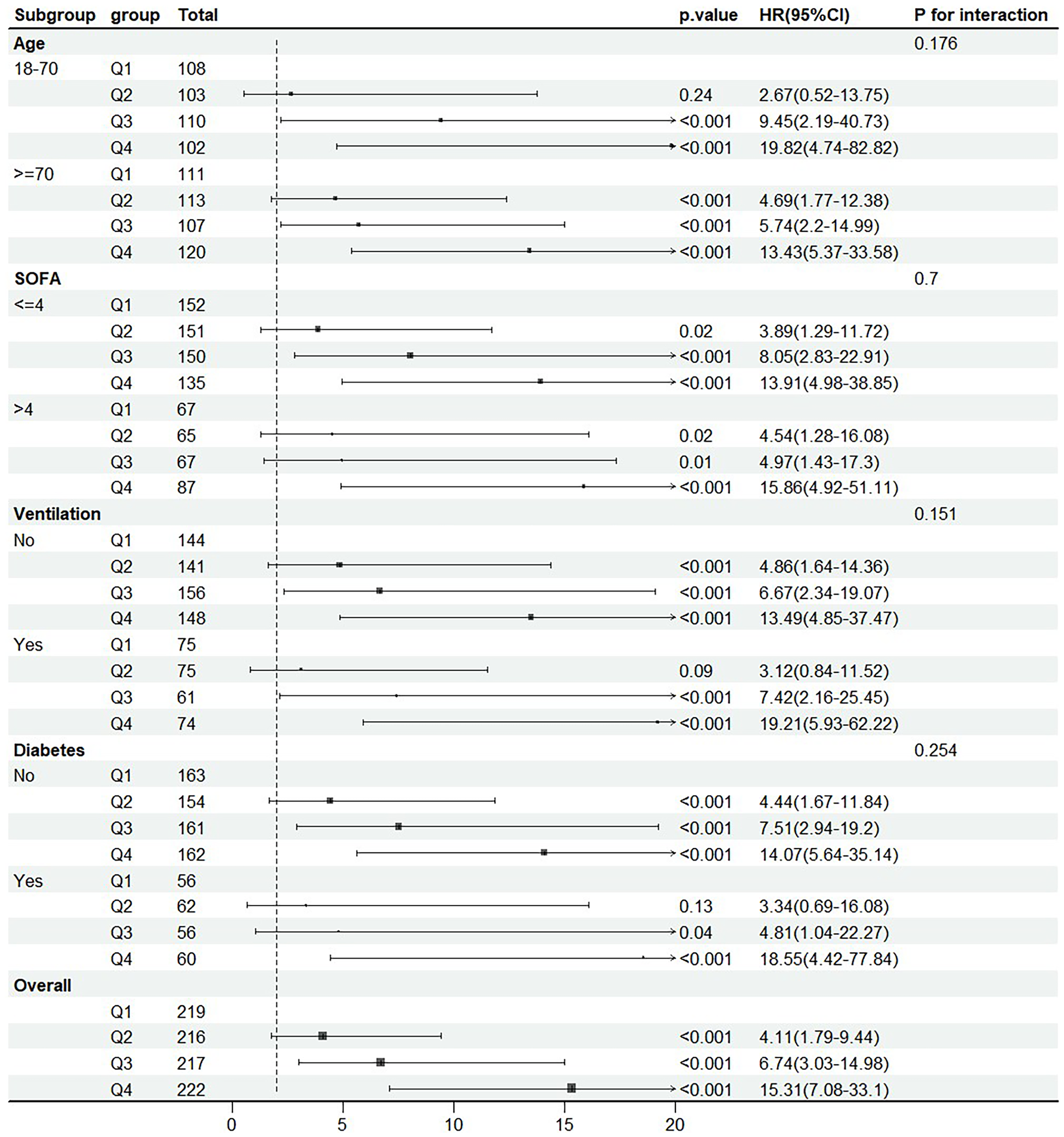
Forest plot for the primary outcome in different subgroups.
Discussion
As one of the most critical complications of sepsis, SAE has a complex pathological mechanism and lack of specific biomarkers, leading to significant limitations in clinical prognostic assessment. In this study, we systematically elucidated the value of LAR in the prediction of SAE mortality risk for the first time through a large-sample retrospective cohort analysis. Our findings indicated that 1) LAR was positively associated with SAE mortality; 2) RCS analysis revealed a nonlinear relationship between LAR levels and SAE mortality risk; and 3) the prediction model constructed using LAR in conjunction with SOFA demonstrated superior discriminative efficacy. These findings not only provide novel joint markers for the prognostic assessment of SAE, but also reveal the underlying pathological mechanisms of SAE from the perspective of metabolism-inflammation interaction, which is of great theoretical innovation and clinical translation significance.
Elevated blood lactate acts as a biomarker for compromised oxygen delivery and impaired tissue perfusion. Under hypoxic conditions, lactate dehydrogenase diverts pyruvate from mitochondrial oxidation to anaerobic glycolysis, facilitating lactate production via oxygen-independent pathways. Previous studies have shown that lactate levels are elevated in brain-injured patients. 23 However, interpreting serum lactate levels is often challenging. Patients suffering from liver disease may exhibit abnormal lactate metabolism, and certain medications, including salbutamol and metformin, can also increase lactate levels. Furthermore, some critically ill patients might present with low venous blood lactate levels, which diminishes the reliability of lactate levels as a sole predictor of a patient's prognosis. 24 Albumin is an important nutrient synthesized mainly by the liver and plays an important role in stabilizing plasma colloid osmotic pressure and is an important carrier of substances transported within the bloodstream. 25 Serum albumin levels are influenced by the nutritional status of the patient and are closely related to the inflammatory response. Albumin promotes tissue repair and disease containment by catalyzing the production of anti-inflammatory molecules, such as lipoxins, lysins, and protectins, thereby accelerating injury regeneration and suppressing pathological progression. 26 This pathophysiological mechanism results in significant albumin utilization, with its depletion rate clinically correlating with the magnitude of the underlying inflammatory burden.27,28 Previous studies have shown that the albumin level correlates with the short- and long-term prognosis of sepsis. 29 Albumin homeostasis is regulated by various physiological factors, such as acute-phase reactions, nutritional interventions, and chronic comorbidities. However, the lactate-albumin ratio, or LAR, stands out as a multifaceted biomarker in the management of SAE. This parameter synergistically integrates the counterregulatory dynamics of inflammation (represented by albumin) and tissue hypoperfusion (indicated by lactate), effectively neutralizing confounding variables through the compensatory interaction between two distinct metabolic pathways, thereby improving prognostic accuracy in critical care settings.
Notably, exogenous lactate supplementation has been shown to attenuate death and cognitive impairment due to a variety of brain injuries.30,31 In our study, elevated serum lactate helped predict mortality in patients with SAE. The translational significance of blood lactate in neurological injury lies in the crucial distinction between pathophysiologically generated hyperlactatemia versus therapeutically administered lactate. Experimental evidence reveals a striking metabolic dissociation, wherein cerebral lactate uptake constitutes a minimal fraction of total circulating lactate. 32 Thus, the initial fluctuations of serum lactate levels in SAE are attributable primarily to pathophysiologic changes throughout the body, not limited to the brain. The results of previous studies consistently demonstrate a dose-dependent relationship between circulating lactate concentrations and SOFA scores—a validated metric quantifying multiorgan dysfunction in heterogeneous ICU populations. 33 Serum lactate acts as a dual prognostic biomarker, quantitatively stratifying the severity of multiorgan dysfunction syndrome and the magnitude of primary neurological injury by monitoring systemic metabolic decompensation. Elevated serum lactate during exogenous supplementation does not indicate inadequate initial tissue perfusion and organ failure, but merely implies that more alternative energy fuels are available to the damaged brain tissue. Although higher serum lactate levels are beneficial for cerebral energy supplementation, the prognostic impact of adverse pathophysiologic conditions suggested by higher serum lactate levels may outweigh the relatively temporary and minor effects of energy supplementation.
In recent studies, LAR has been increasingly recognized as a valuable biomarker for assessing disease severity and predicting adverse outcomes across a wide range of clinical conditions. In cases of coronary heart disease, LAR has been demonstrated to correlate with 28-day mortality; a higher LAR is associated with a greater mortality rate. 34 Among individuals with severe burns, LAR has been reported as a reliable marker for predicting mortality and complications, reflecting the systemic metabolic response to extensive tissue injury. 35 Similarly, in acute pancreatitis and acute kidney injury, elevated LAR levels have been linked to worse clinical outcomes and may aid in early risk stratification.15,36 In patients with septic shock, LAR has demonstrated superior prognostic value compared to lactate or lactate clearance, reflecting both tissue hypoperfusion and impaired nutritional or inflammatory status. 37 Furthermore, LAR has been associated with the severity of the disease and a poor prognosis in various conditions, such as liver failure, respiratory failure, trauma, and others.38–42 Importantly, our study is the first to demonstrate the prognostic value of LAR in patients with SAE. This novel finding highlights the potential of LAR as a simple and accessible marker to identify high-risk SAE patients early in their clinical course, which may help guide timely interventions and improve outcomes. Therefore, our study adds meaningful new insight into the application of LAR in sepsis-related complications.
ROC curve analysis showed that LAR has a better discriminatory ability in predicting SAE mortality. Compared with lactate or SOFA alone, LAR may reflect the pathophysiologic status of patients more comprehensively and improve the accuracy of mortality risk assessment. Therefore, LAR can be used as an important tool for early risk assessment in patients with SAE and provide strong support for clinical decision-making. Although the SOFA score is widely used for sepsis prognostic assessment, it relies on the assessment of multi-organ function and is complex to calculate. In contrast, LAR requires only two routine laboratory indicators and is more suitable for rapid decision-making in the emergency department or at the ICU bedside. Furthermore, this study found that the incorporation of LAR into the SOFA score resulted in higher AUCs for the prediction models of 30–, 90–, 180–, and 365-day mortality compared with SOFA and lactate alone, suggesting that LAR may be effective in supplementing the existing scoring system. Although emerging biomarkers such as S100B, neurofilament light chain (NfL), and neuron-specific enolase (NSE) have shown potential for prognostic assessment in SAE, their clinical use is limited by high costs, low availability, and insufficient specificity.43–45 In contrast, LAR is a readily accessible, cost-effective marker derived from routine laboratory tests, offering a practical advantage for early risk stratification in critically ill patients.
It is worth noting that a nonlinear relationship between LAR levels and SAE mortality was found in this study, suggesting that there may be a threshold effect of LAR on the prognosis of SAE, i.e. within a certain range, the elevation of LAR may significantly increase the risk of death, whereas above a certain threshold, the effect may level off. Our findings are consistent with those of Ma et al., indicating that LAR has a nonlinear relationship with all-cause mortality from sepsis in cirrhotic patients. 46 This nonlinear feature suggests a two-stage pathogenesis: at low LAR, lactate accumulation and albumin decline may mainly reflect reversible metabolic compensation, at which time prompt interventions (e.g. optimization of perfusion, anti-inflammatory therapies) may be effective in blocking progression of the disease; whereas once LAR exceeds the threshold, it signals the onset of severe and irreversible organ damage, at which time the effects of conventional therapies may be limited. However, it is also important to acknowledge that this plateau may reflect the inherent limitations of LAR in capturing the full complexity of mortality risk in SAE. Other unmeasured factors, such as individual comorbidities, treatment strategies, or additional biomarkers, may contribute to mortality but are not reflected in the LAR value alone. This observation underscores the need for further investigation and validation of LAR, ideally in combination with other prognostic indicators, to improve clinical decision-making in SAE.
Although this study found an important role for LAR in the prediction of mortality in SAE patients, there are still some limitations. Firstly, this study is a retrospective analysis based on the MIMIC-IV database and is therefore subject to inherent limitations, including potential selection and information bias. Unmeasured confounders—such as nutritional status or treatment strategies—may have influenced lactate and albumin levels, potentially affecting the association between LAR and SAE prognosis. Secondly, this study did not investigate the effect of fluctuations in LAR levels on patient outcomes. Dynamic monitoring of LAR could more accurately depict disease progression. Thirdly, external validation in prospective cohorts is needed. Future studies should also explore the prognostic performance of LAR in combination with other biomarkers to enhance predictive accuracy and clinical utility.
Conclusion
LAR is positively associated with mortality in patients with SAE, with its non-linear relationship confirmed by RCS analysis. The combination of LAR and SOFA score enhances the accuracy of mortality prediction. As a readily accessible biomarker, LAR holds significant potential for clinical application in SAE management. Nonetheless, this retrospective study has inherent limitations, and further multicenter prospective studies are warranted to validate these findings and to investigate the prognostic utility of LAR in conjunction with other clinical indicators.
Footnotes
Acknowledgements
Not applicable.
Ethics approval
The MIMC-IV database was developed in cooperation with Privacert (Cambridge, MA), which certifies that re-identification of risk is in compliance with the safe harbor standards (HIPAA Certification No. 1031219-2). Because the database is HIPAA compliant and in compliance with the Declaration of Helsinki, the need for ethics approval was waived. The data used in this study are publicly available.
Consent to participate
Because all personal information was listed anonymously, informed consent was waived.
Consent for publication
Not applicable.
Author contributions
Xiaojing Li and Xiaoxiang Gong were responsible for the study design and prepared the figures. Fengzhen Huang, Xiaojing Li, and Jiping Yi contributed to the data analysis and examination. Xiaoxiang Gong and Tieqiao Zhou drafted the initial manuscript.
Funding
This study was financially supported by Natural Science Foundation of Hunan Province (2025JJ70556, to Fengzhen Huang; 2023JJ50382, to Jiping Yi), Scientific research project of Hunan Provincial Health Commission (D202303077714, to Jiping Yi), Scientific research project of Xiangnan University (2020XJ88, to Jiping Yi) and Department of Science and Technology of Hunan Province in China (2023SK4018, to Liqun Liu).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
