Abstract
Introduction
Plants usually contain chemical compounds known as secondary metabolites, which have wide range of biochemical and pharmacological activities ranging from antioxidation, antiinflammation, antiallergic to antithrombotic effects. 1 These metabolites may include saponins, tannins, oxalates, phytates, phenolic compounds, flavonoids and glycosides. Some of these metabolites may be used directly as medicinal agents. 2 Every medicinal plant species, in addition to having pharmacologically active compounds, possesses other nutritional phytochemicals that are essential for the physiological functions of human and animal body, thereby providing the energy requirements for metabolic processes. These nutrients include carbohydrates, fats, proteins, vitamins, antioxidants, 3 minerals, fatty acids and fibre. 4 Phytochemical analyses of edible plants and vegetables play an important role in assessing their nutritional and medicinal potentials.5,6
So far, there is little or no report on the phytochemicals, nutritional elements and toxicity studies of
In this study, the phytochemical compounds of the dried leaf powder of
Materials and methods
Plant materials
Fresh leaves of

Botanical identification
The plant was botanically identified at the IBS, UPM, by Dr. Mohd Firdaus Ismail, where the kingdom, family and species of
Extraction procedure
The extraction method described by9,23,24 was employed in this study with modifications. The dried powdered leaves were extracted using 70% ethanol (hydroethanolic solvent) at the ratio of 1:10, that is, 100 g of
Liquid chromatography-electrospray ion mass spectrometry (LC-ESI-MS/MS)
Preparation of samples
Samples were prepared according to the method described by Aliyu et al.
25
About 1000 mg of powdered MOHE was dissolved into 1 mL of LC-MS-grade methanol and sonicated for 5 min at 25°C by Bransonic Ultra sonic cleaner (2510E-DTH, USA). The samples were later filtered using
Materials and equipment
Standard compounds used for this experiment were LCMS grade acetonitrile (ACN), formic acid, methanol (MeOH) and LCMS grade water. The compounds were purchased from Fisher Scientific (Fair Lawn, USA).
High performance liquid chromatography (HPLC) separation was performed using a Thermo Scientific™ Dionex Ultimate 3000 LC system (Thermo Fisher Scientific, USA) with a Thermo Hypersil Gold aQ (1.9 µm, 100 mm × 2.1 diameter). A Thermo Scientific Q Exactive Focus (Thermo Fisher Scientific, USA) equipped with a pump: HGP-3200RS, Autosmpler: WPS3000TRS, column compartment: TCC3000RS a degasser: SRD3400, DAD (Diode Array Detector), Orbitrap mass analyser with a heated-electrospray ionization (H-ESI II), and software of Xcalibur and Chromaleon was used for LC–MS and LC–MS/MS detection.
LC-ESI-MS/MS for compounds identification
The bioactive compounds in crude extract of MOHE were determined according to the method described by Coppin et al. 26 with modifications. Mobile phase containing solvents B and C in linear gradient was used for the liquid chromatography (LC) separation. Solvent B was 0.1% formic acid (v/v) in acetonitrile, while C was 0.1% formic acid (v/v) in water. The gradient was set as follows: 10–30% B in 20 min and 30% B in 20–30 min at a flow rate of 0.2 mL/min. The injection volume was 10 µL and the UV detector was set at 254, 280 and 370 nm. The eluent was observed using a Thermo Scientific ion max API source (H-ESI II), under positive and negative ion modes and scanned from m/z 190 to 800. Needle voltages of 4.2 and 3.5 kV were respectively used for the conduction of (ESI) for positive and negative modes under optimum collision energy level of 30. Dry gas (99.999% high-purity nitrogen) was used at a capillary temperature of 320°C and a flow rate of 12 L/min. Nitrogen was used as nebulizer at 40 psi.
Ethical approval
Ethical approval for these studies (acute and sub-acute toxicity studies) was obtained from the Animal Ethic Committee (AEC) of Malaysian Agricultural Research and Development Institute (MARDI) (approval reference number: 20170717/R/MAEC00023).
Animal welfare
The present study followed institutional guides for humane animal treatment and complied with the relevant legislation, according to the guide for the care and use of laboratory animals of MARDI.
Acute and sub-acute toxicity studies of moringa oleifera hydroethanolic leaf extract
The acute and sub-acute toxicity studies were conducted according to the principles of Organisation for Economic Co-operation and Development (OECD) guidelines (OECD 425 and 407 respectively) with slight modifications. The studies were conducted at the Animal Metabolism, Toxicology and Reproductive Centre (AMTREC), MARDI, Serdang. Each mouse was placed in a polycarbonate plastic cage and acclimatised to the optimum housing conditions with temperature ranges between 22°C and 25°C, humidity within the range of 40%–70% and 12 h light/12 h dark cycle for 1 week. The cages were cleaned regularly, bedding and water were changed accordingly.
Ten female mice, at 8-week old were randomly divided into 2 groups A and B using randomised complete block design (RCBD), with 5 mice each. Group A served as the control group and was given distilled water, while mice in group B were fasted for about 3 h and administered with a single dose of 2000 mg/kg MOHE via oral gavage in the morning.
16
The weight of each mouse was monitored on day 0 (prior to the extract’s administration) and then repeated weekly for 14 days. Changes in the body weight of each mouse in the treatment group were measured and compared with that of the control group. The extract was freshly dissolved in 5% dimethyl sulfoxide (DMSO) before being administered orally to the mice. Subsequently, mice in groups A and B had free access to water and commercial chow
The sub-acute toxicity study was carried out according to OECD guidelines 407. Twenty five female 8-week-old mice were randomly divided using RCBD into five groups (A to E) of five mice each; group A was administered with distilled water and served as the control group, while groups B, C, D and E received 125, 250, 500 and 1000 mg/kg MOHE (dissolved in 5% DMSO) respectively. The extract was administered once daily in the morning for 28 days at a volume of 1 mL/100 g body weight. The mice were observed daily for any signs of toxicity and were sacrificed using CO2 chamber on the 29th day of the experiment. In both study, administration of the herb was through oral gavage (using stainless steel needle) to symbolise the normal consumption of the herb in animals or human.
Weekly body weight gain
The weekly body weight of the mice in each group was determined throughout the study period using electric weighing scale and recorded as described by Aliyu et al. 25 and Asyura et al. 27 The body weight gain was calculated weekly by deducting the average weight gain of each mouse in the preceding week from that of the present week. 25
Collection of samples
The mice from both acute and sub-acute toxicity studies were respectively sacrificed humanely on days 15 and 29 of the experiments, using CO2 chamber (Labquip Sdn. Bhd, Malaysia). Blood samples were collected using 26-gauge needle and 1 mL syringe, from the heart into clean bottles containing anticoagulant [ethylenediamine tetra acetic acid (EDTA)]. The blood samples were used for haematological and biochemical analysis.16,28
Necropsy was conducted on each mouse and tissue samples were collected from liver, kidneys, heart, brain, spleen, lungs and uterus. The weight of these organs was determined and recorded, afterwards the organs weight ratios were calculated as described by Aliyu et al. 25
Haematological analyses
The blood samples collected in anticoagulated (EDTA) tubes were transported in ice packs to the Haematology and Clinical Biochemistry Laboratory, Faculty of Veterinary Medicine, Universiti Putra Malaysia and analysed for complete blood count using an automated haematology analyser (ABC Vet®, ABX Diagnostics, France). The parameters investigated included total red blood cells, total white blood cell, platelet count, haemoglobin concentration, mean corpuscular volume (MCV) and mean corpuscular haemoglobin concentration (MCHC).29,30 Blood smears were prepared and stained with Wright stain and examined under a light microscope. Differential WBC count, the absolute values of each WBC type, packed cell volume (PCV) and plasma protein concentration were determined as described previously.25,27,29,31
Plasma biochemical analysis
After the ETDA blood was processed for analysis of hemogram, the blood was centrifuged using a benchtop centrifuge (Centrifuge S417R, Eppendorf, CA, USA) for 15 min at 3000 rpm to obtain plasma. The plasma was further analysed by a completely automated clinical chemistry analyser (BioLis 24i Chemistry Analyzer, Japan) for urea, creatinine, alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatinine kinase (CK), total protein (TP), albumin (ALB) and globulins.25,29,32
Histopathological analysis
The tissue samples collected from the liver and kidneys from each mouse at the end of the experiment were processed at the Histopathology Laboratory, Faculty of Veterinary Medicine, Universiti Putra Malaysia according to the methods described previously.25,31 The tissues processed on the slides were stained with haematoxylin and eosin (H&E) and examined under a light microscope at different magnifications of 40, 100, 200 and 400. 25
Lesion scoring
Lesions including acidophilic hepatocytes, pyknotic, karyolitic and karyorrhectic nuclei, activated Kupffer cells, none-obstructive sinusoidal dilatation, degenerative (cytoplasmic vacuolation), regenerative and inflammatory changes were scored for each stained section of liver from both groups of mice.25,27,31 In the kidney, acidophilic epithelial cells, pyknotic nuclei, degenerative change (hydropic swelling), hyaline and epithelial casts formation were scored for each mouse. Methods of scoring and determination of severity of lesions in liver and kidney is showed on Table 1.
Assessment for the severity of lesions seen in the liver and kidneys of female ICR-mice in acute toxicity study of

Values in the same row with different superscripts differ significantly (
Statistical analysis
The results were expressed as mean ± standard error of the mean (SEM) and were accordingly subjected to students’‘
Results
Dry matter and percentage yield
The total moisture content of
Liquid chromatography-mass spectrometry
The total ion chromatograms (TIC) of the bioactive compounds detected by positive and negative ion modes in of

Total ion chromatograms (TIC) of the compounds in MOHE (positive ion mode).

Total ion chromatograms (TIC) of the compounds in MOHE (negative ion mode).
Phytochemical compounds identified in


The MS spectra and structure of Glucomoringin.

The MS spectra and structure of Niaziminin A.
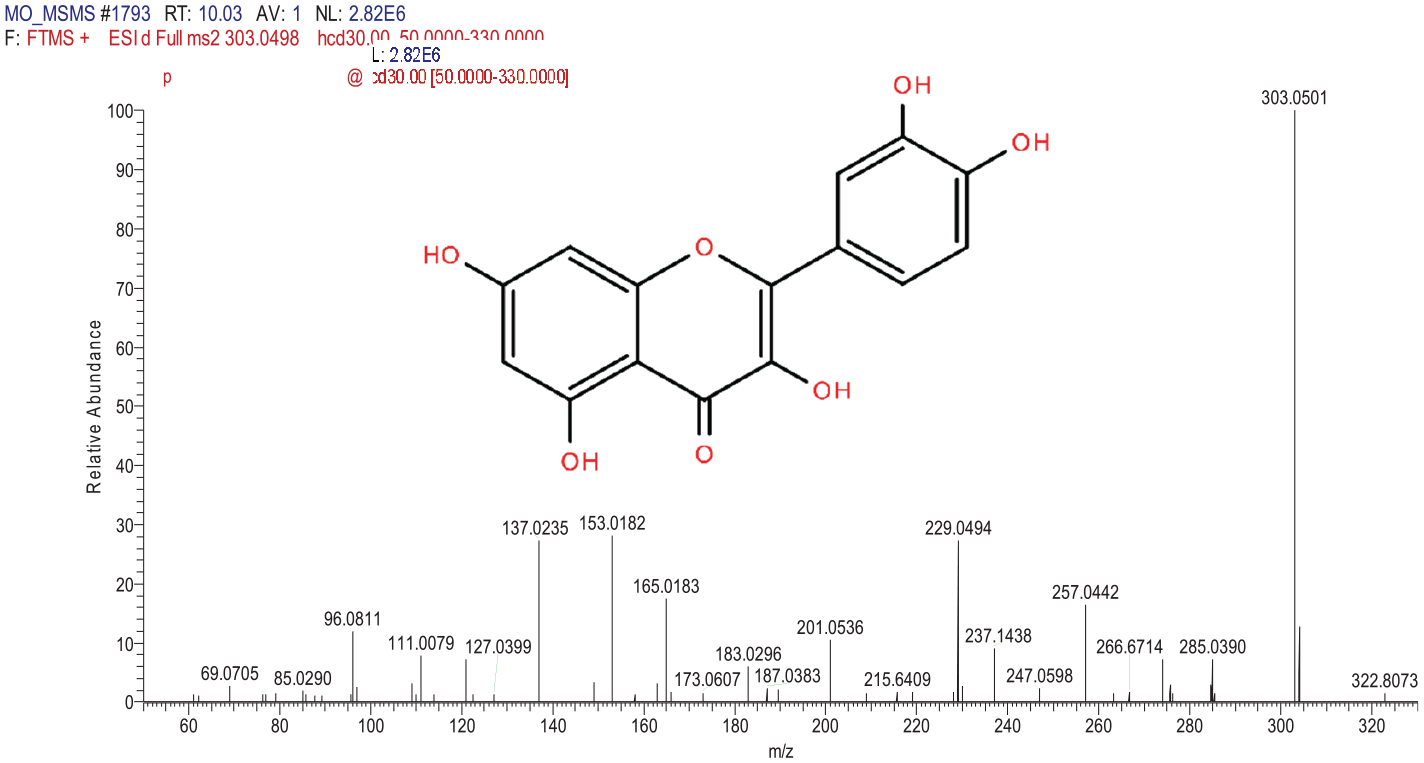
The MS spectra and structure of Quercetin.

The MS spectra and structure of Kaempferol.
Acute toxicity study of MOHE
Behavioural changes
There were neither behavioural changes nor mortality observed in the treated mice throughout the period of observation (Table 3).
Behavioural observations for female ICR-mice in acute toxicity studies of MOHE in both control and treated groups (2000 mg/kg body weight MOHE).
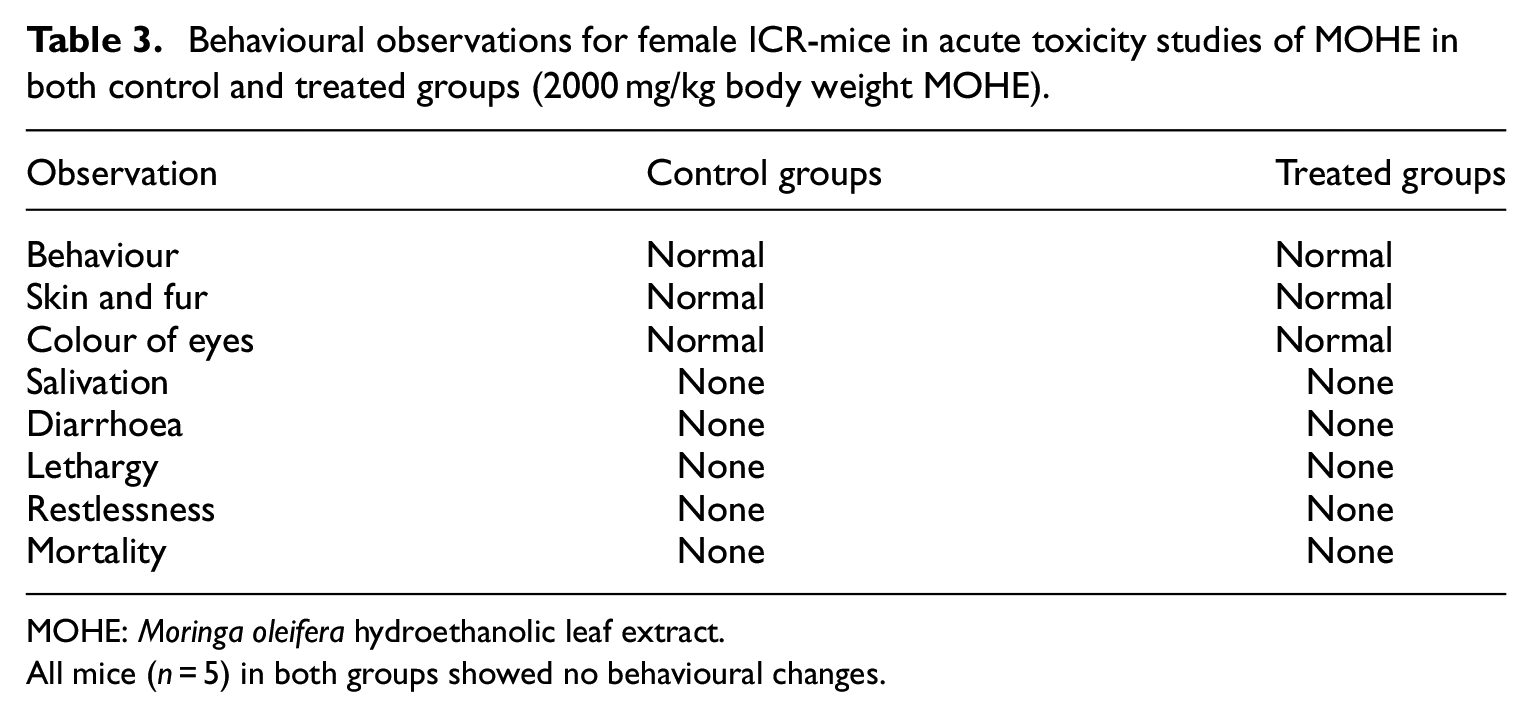
MOHE:
All mice (
Bodyweight
Figure 7 showed the effects of 2000 mg/kg MOHE on the average bodyweight gain of mice following single oral administration. There were no significant (

Average (mean ± SEM) weekly body weight gain (
Relative organs weight
The effects of oral administration of 2000 mg/kg MOHE on relative organs weight (%) of mice is presented in Table 4. There were no significant (
Relative organ weights (mean ± SEM) of female ICR-mice in acute toxicity study of
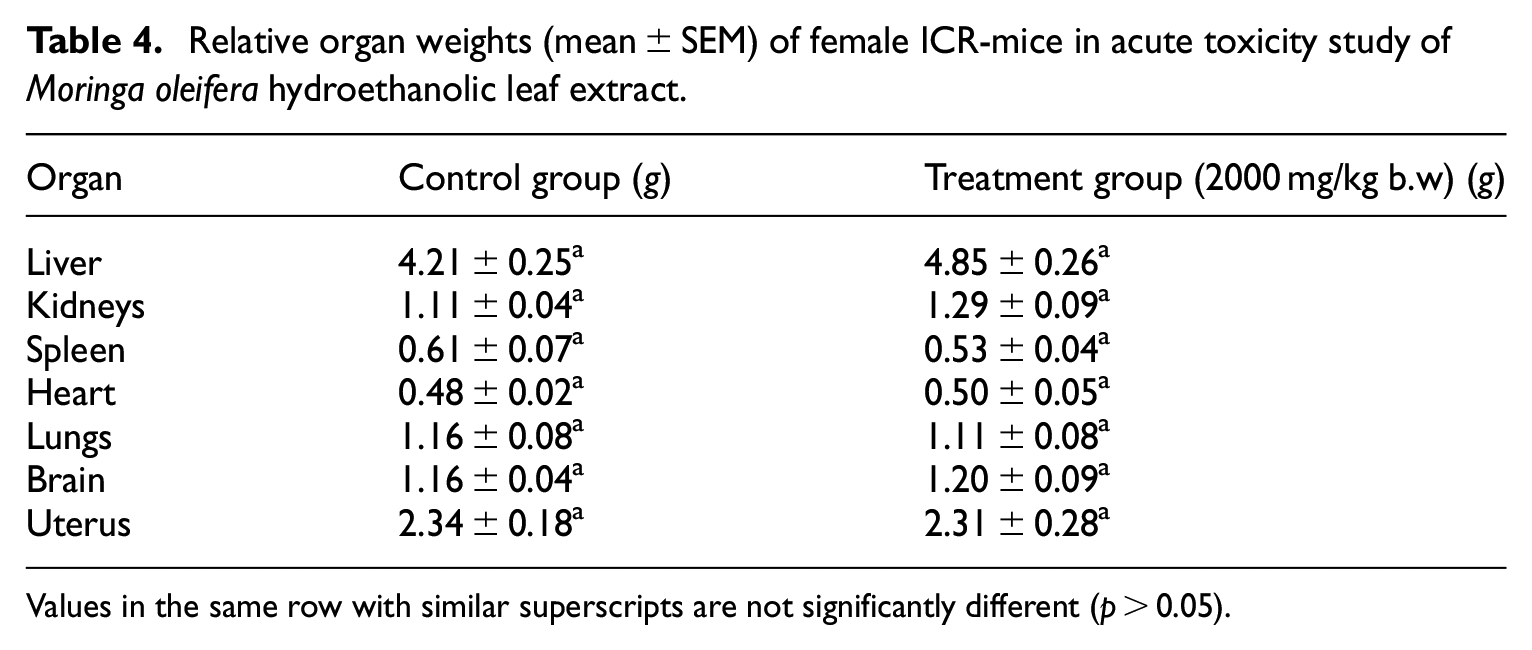
Values in the same row with similar superscripts are not significantly different (
Haematological parameters
Table 5 illustrated the effects of 2000 mg/kg MOHE on haematological parameters of mice following single oral administration. The values did not show any significant (
Haemogram (mean ± SEM) of female ICR-mice in acute toxicity study of

Values in the same row with similar superscripts are not significantly different (
indicate the vales are lower than control and/or reference range.
indicates the value is higher than control and reference range.
Plasma biochemical parameters
The effects of oral administration of 2000 mg/kg MOHE on plasma biochemical parameters of ICR-mice are shown on Table 6. There was 3.1% decrease (
Biochemical parameters (mean ± SEM) of female ICR-mice in acute toxicity study of

Values in the same row with asterisk differ significantly (
indicates the value is higher than the reference range.
indicates the value is lower than the reference range.
Histopathological evaluation of liver
Table 7 and Figure 8 showed the histopathological effects of single oral administration of 2000 mg/kg MOHE on the liver of ICR-mice. Mann-Whitney
Lesion scores (mean ± SEM) for histopathological changes seen in the liver of female ICR-mice in acute toxicity study of

indicate the values are significantly different (

Lesions seen in the liver of female ICR-mice orally administered with
Histopathological evaluation of kidney
Significant (
Lesion scores (mean ± SEM) of the kidney of female ICR-mice in acute toxicity study of
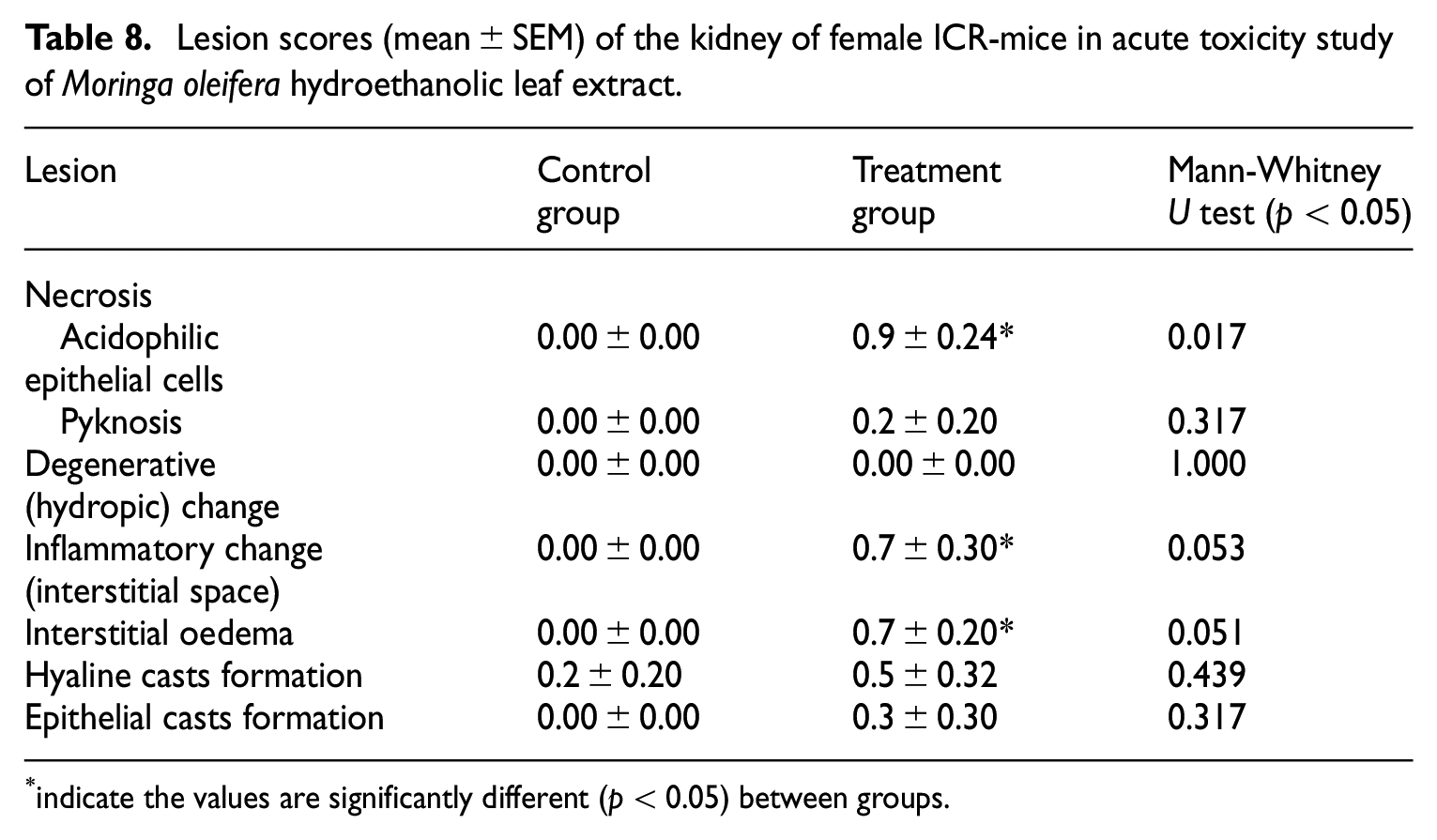
indicate the values are significantly different (
Lesions seen in the kidney of female ICR-mice orally administered with
Sub-acute toxicity study
Body weight gain
The average body weight gain of the treated mice was significantly (

Average (mean ± SEM) weekly body weight gain (
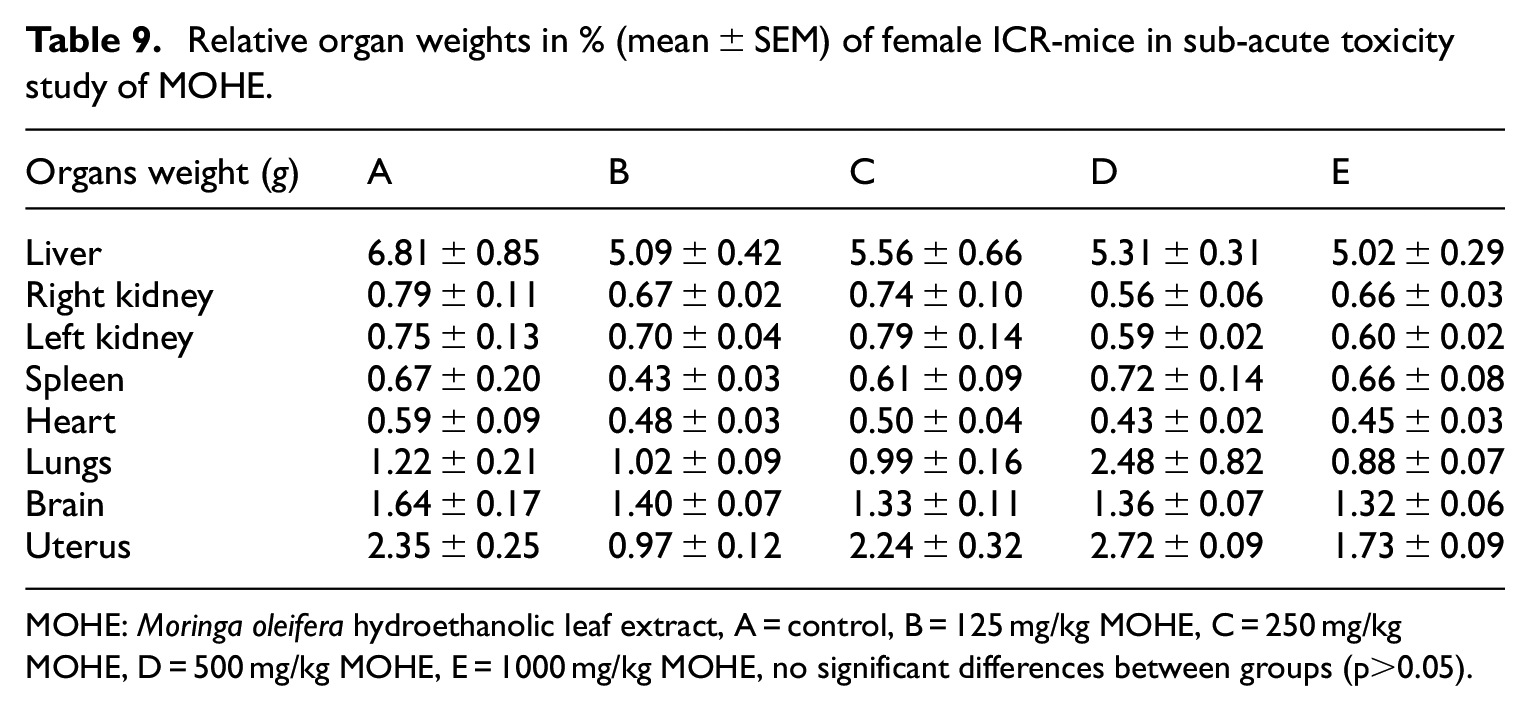
Relative organs weight
The relative organs weight of the treated mice was not significantly (
Relative organ weights in % (mean ± SEM) of female ICR-mice in sub-acute toxicity study of MOHE.
MOHE:
Haematological parameters
Table 10 shows the effects of repeated daily administrations of MOHE for 4 weeks on the haematological parameters of female ICR-mice. There was significant (
Haematological parameters (mean ± SEM) of female ICR-mice in sub-acute toxicity study of MOHE.

MOHE:
Plasma biochemical parameters
The biochemical parameters analysed in the plasma differed significantly (
Biochemical parameters (mean ± SEM) of female ICR-mice in sub-acute toxicity study of MOHE.

MOHE:
Histopathological evaluation of liver
There was significant (
Lesion scores (mean ± SEM) of the liver of female ICR-mice in sub-acute toxicity study of MOHE.

MOHE:
significantly different at

Effects of repeated oral administration of MOHE for 28 days on the histology of liver of female ICR-mice: (a) photomicrograph of liver section from a mouse in a control showing normal architecture of liver (H&E stain, ×100), (b) photomicrograph of liver section from a mouse in group E showing sinusoidal dilatation and hepatic degeneration characterised by cytoplasmic vacuolation (yellow encircled), hepatic necrosis characterised by eosinophilic cytoplasm (black encircled) and pyknosis (yellow arrow) of the hepatocytes (H&E stain, ×100), (c) photomicrograph of liver section from a mouse in group E showing pyknosis (yellow arrow) and hepatic degeneration characterised by cytoplasmic vacuolation (yellow encircled) (H&E stain, ×200) and (d) photomicrograph of liver section from a mouse in group E showing sinusoidal dilatation (blue encircled) and cellular infiltration (green arrow) (H&E stain, ×100).
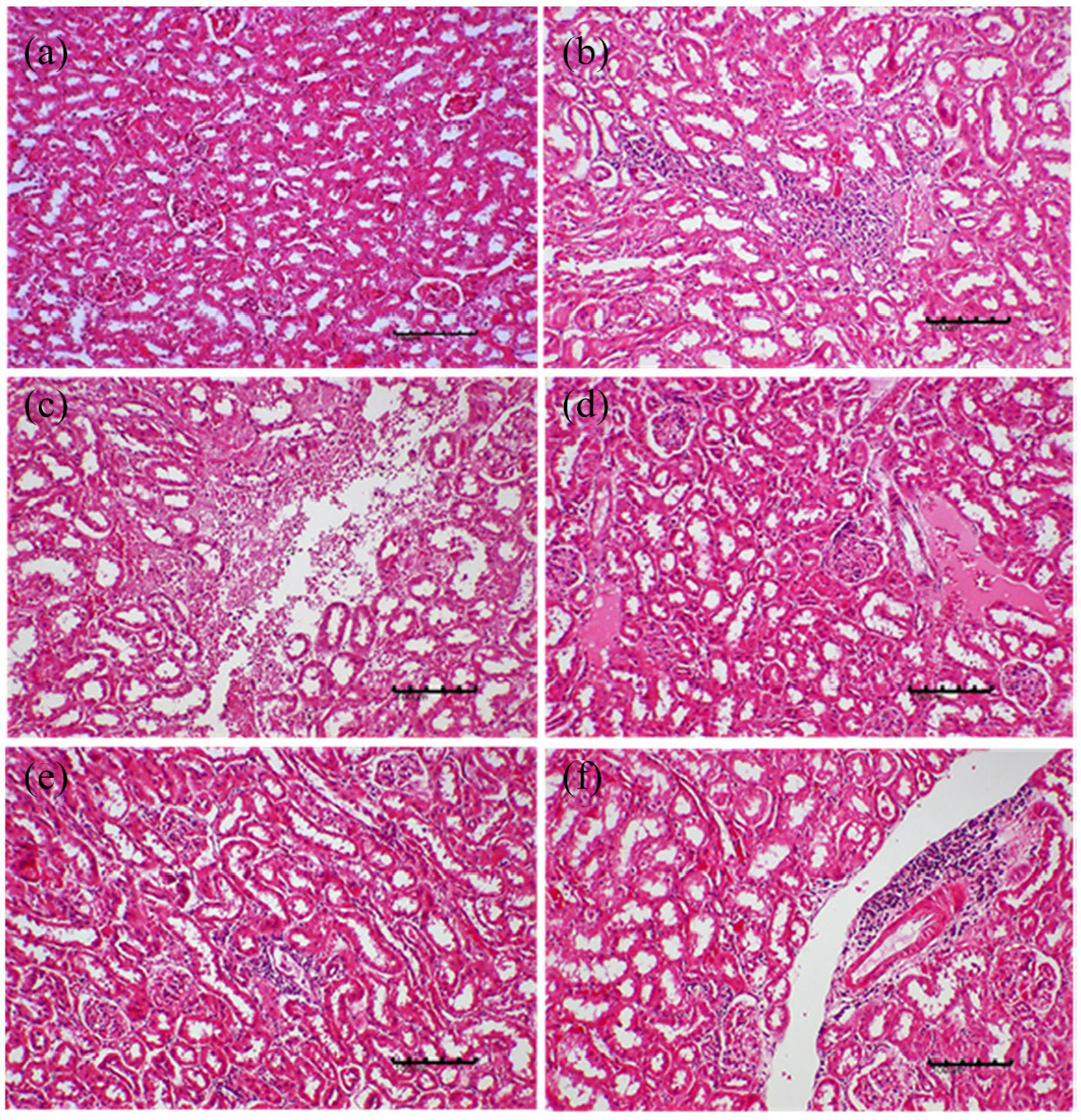
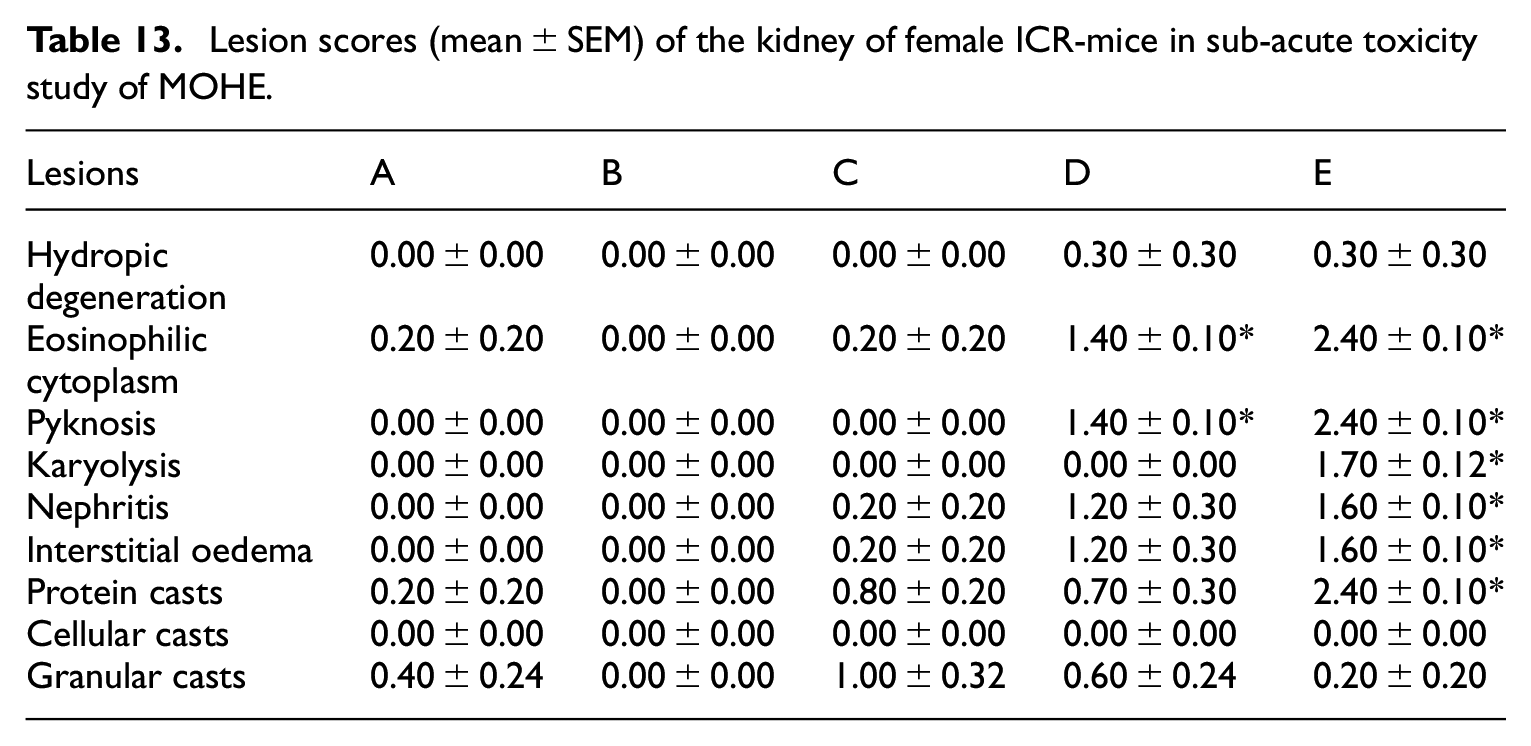
Histopathological evaluation of kidney
Significant (
Lesion scores (mean ± SEM) of the kidney of female ICR-mice in sub-acute toxicity study of MOHE.
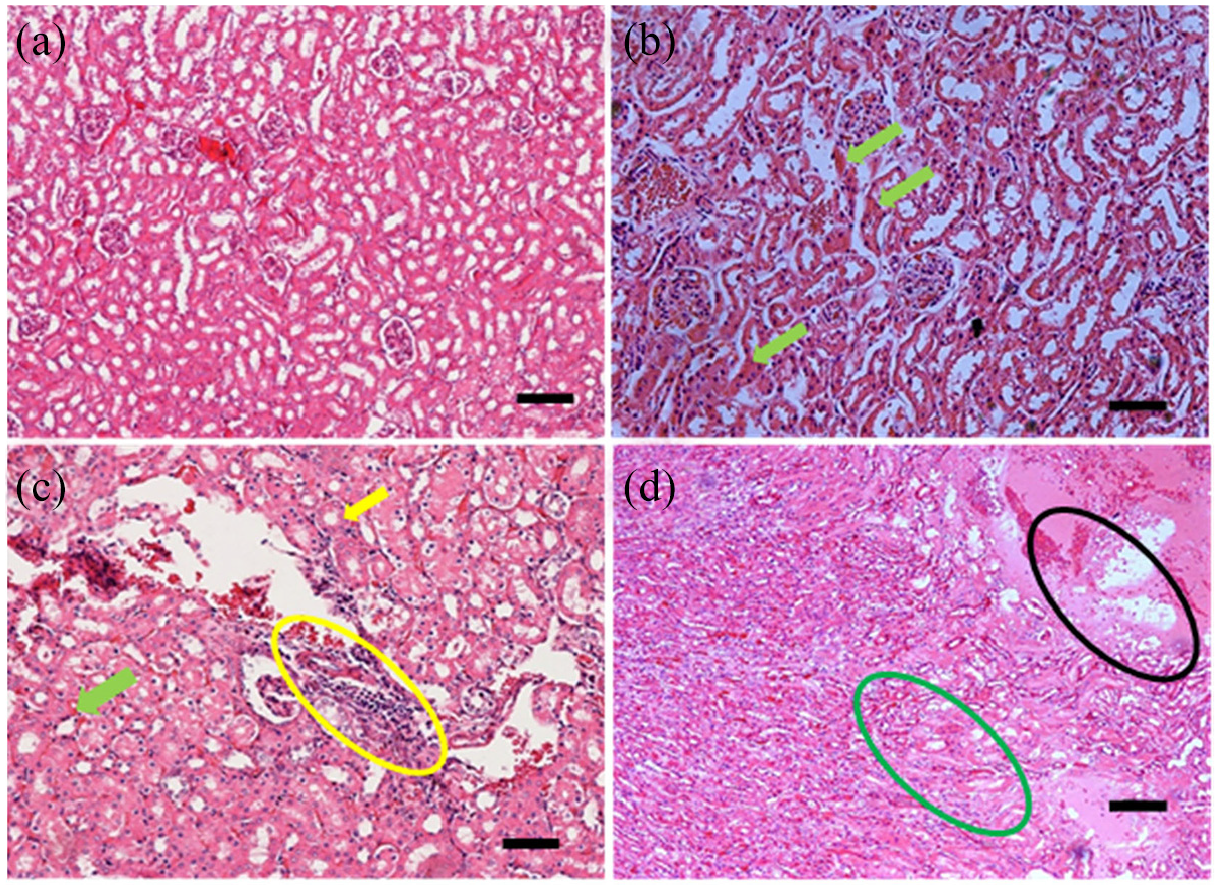
Effects of repeated oral administration of MOHE for 28 days on the histology of kidney of female ICR-mice: (a) photomicrograph of a kidney section (H&E stain, ×100) from a mouse in control showing normal architecture of kidney, (b) photomicrograph of a kidney section (H&E stain, ×200) from a mouse in group E (1000 mg/kg MOHE) showing renal necrosis characterised by eosinophilic cytoplasm (green arrow), (c) photomicrograph of a kidney section (H&E stain ×200) from a mouse in group E showing glomerulonephritis characterised by cellular infiltration (yellow encircled) and (d) photomicrograph of a kidney section (H&E stain, ×100) from a mouse in group E showing interstitial oedema (mainly at the top right).
Discussion
Results for the chemical profiling analysis of
As all the compounds possess a potential anticancer activity, it should not be forgotten that the compounds could also cause negative effects to healthy cells as what other chemotherapeutic drugs do. A few studies have been conducted to evaluate the safety level of the plant extracts.15–17,27,31,42,43 by determination of its lethal dose (LD). Basically, the smaller is the LD value, the more toxic is the substance, and vice versa. According to Hodge and Sterner 44 toxicity scale, LD50 value (administered through oral route) of 1 mg/kg body weight or less, falls under toxicity rating level 1, is considered extremely toxic, while 15,000 mg/kg body weight and above, falls under toxicity level 6, is considered relatively harmless, and the doses are equivalent to a drop and 1 L, respectively, for probable lethal dose for human. It should be remembered that LD50 only indicates that the substance is safe to be taken at single dose, when or where it would not cause acute death, but consumption at that particular dose over longer periods of time should be carefully evaluated before it is conclusively considered safe to be consumed for supplementary and medicinal purposes. Taken all the consideration, herbal toxicity studies are considered very important before determination of its medicinal potentials.
Acute toxicity study conducted by Adedapo et al.
17
revealed 17% and 33% of rats orally gavaged with 1,600 and 2,000 mg/kg body weight of
In this study, single oral administration of 2000 mg/kg
The sub-acute toxicity study of MOHE in this study, showed significant reduction in the body weight gain of the mice treated with 1000 mg/kg MOHE daily for 28 days. This could be due to decreased feed intake or reduced fats deposition in the treated mice.
47
These reports were comparable to those reported by Kasolo et al.,
15
where rats treated with aqueous and ethanol extracts of
The repeated oral administration of MOHE for 28 days in this research resulted in some plasma biochemical alterations. The substantial decrease in the plasma level of urea in the group of mice treated with 1000 mg/kg MOHE may suggest that the sub-acute administration of the extract at this dose might have some degree of liver injury to the mice. This is because urea has been reported to decrease due to liver failure, low protein diet, anabolic steroids, or diabetes insipidus.
48
On the other hand, the creatinine level in the groups treated with 500 and 1000 mg/kg doses of the extract daily for 28 days was significantly elevated. This may indicate that the extract might have caused some degree of renal injury in the treated mice.49,50 This is because creatinine has been routinely used as marker for evaluation of renal function.
51
There was also a significant increase in the levels of ALT in the group treated with 500 and 1000 mg/kg MOHE daily for 28 days. At the same time, the AST level was significantly elevated at 1000 mg/kg, so also the CK level was remarkably elevated in the groups treated with 250, 500 and 1000 mg/kg doses of the extract. The significant increase in ALT and AST could indicate that the extract at 500 and 1000 mg/kg induced some degree of liver injury in the mice.48,52 This is because ALT is known to increase in liver injury particularly hepatic necrosis.
48
Significant increase in the serum levels of ALT and AST was reported previously in rats treated with 400 and 1600 mg/kg
The histological evaluation of liver and kidneys of the mice treated with different doses of MOHE daily for 28 days revealed some degrees of lesions. The moderate pyknosis and karyolysis as well as eosinophilic cytoplasm of the hepatocytes observed in the group treated with 500 and 1000 mg/kg of the extract daily for 28 days may suggest mild hepatic necrosis,27,31,54 and this corroborates the results of plasma biochemical analysis, especially that the mice administered with 1000 mg/kg showed significant hepatic karyolysis compared to the control and other treatment groups. This further suggested that the extract at 500 and 1000 mg/kg might have induced adverse effects on the liver of the treated mice.27,31,54 Similarly, the mild and moderate eosinophilic cytoplasm, pyknosis and karyolysis respectively observed in the renal tubules of the mice treated with 500 and 1000 mg/kg daily for 28 days suggest that the extract at these concentrations induced some degree of necrosis27,55,56 in the kidneys of the treated mice. The sub-acute adverse effects of the extract at these doses were further evidenced by the mild tubular and interstitial nephritis55,56 seen in group of mice treated with 500 and 1000 mg/kg MOHE, as well as the moderate tubular protein casts27,56 observed in group treated with 1000 mg/kg of the extract. Moreover, these findings supported the plasma biochemistry results that showed significant increase in the creatinine level at 1000 mg/kg dose.
The results of the histological evaluation of liver and kidney in this study have agreed with Olayemi et al.
53
who reported that Wistar rats treated with
Anaemia in mice treated with MOHE could be attributed to the presence of glucomoringin in the leaf extract. Glucomoringin, once hydrolysed, the compound is converted to isothiocyanate (ITC).
33
Two forms of ITC, which are allyl ITC (AITC) and benzyl ITC (BITC) are present in plants/vegetables. AITC is detected in cabbage, cauliflower, arugula, broccoli etc. A study conducted by Jenner et al.
59
indicated AITC oral LD50 is 340 mg/kg in rats with pronounce jaundice, while at 40 mg/kg via oral route, it induced acute toxicity on rats’ liver, thymus, kidney and blood.
60
Meanwhile, administration of AITC daily for 20 days at 50 mg/kg body weight in rats induced subacute stomach ulceration. For BITC, subacute daily administration at 200 mg/kg body weight in rats induced significant alterations in the haematological parameters.
61
In a study conducted by Adedapo et al.
17
on subacute toxicity evaluation of aqueous extract of
Toxicity results presented in this study are similar to our previous toxicity studies on other ethanolic extracts, which include
Limitations of the study
The current study is limited to the toxicity effects of single oral administration of high dose (2000 mg/kg) and short-term duration (4 weeks) of repeated lower doses (125, 250, 500 and 1000 mg/kg) of the extract (MOHE) in mouse model. However, administration of herbal extracts in traditional medicine, requires longer repeated administration, especially in the treatment of chronic illnesses, including cancers. Consequently, it is recommended to further evaluate the sub-chronic and chronic toxicity effects of the extract in mouse model. Moreover, the quantification of the bioactive compounds identified in the current study has not been reported. It is therefore, recommended that the quantity of each of the compounds identified in
Conclusions
It is concluded that MOHE cultivated in Selangor, Malaysia contains phenolic compounds and flavonoids. Its lethal dose (LD50) in female ICR-mice is higher than 2000 mg/kg. At that dose level the extract causes mild anaemia, stress leukogram and mild to moderate hepato-nephrotoxicity in female ICR-mice. Similarly, this study demonstrated that repeated daily oral administrations of MOHE for 28 days induced significant alterations in the parameters for injury markers in the liver, kidney and muscles at 500 and 1000 mg/kg in female ICR-mice. In addition, repeated oral administrations of MOHE daily for 28 days in female ICR mice induced moderate hepatic degeneration at 500 mg/kg and necrosis at 1000 mg/kg, as well as mild kidney necrosis at 1000 mg/kg. Therefore, the plant extract at 1000 mg/kg dose, is considered toxic and unsafe for repeated consumption as food supplementation and/or alternative medicine; however, lower doses and/or short time administration could be used safely for medicinal purposes.
Footnotes
Acknowledgements
The technical support from the staff of the Natural Product Laboratory, Institute of Bioscience, UPM (Mr Azizul Isa, Mrs. Zurina Zainal, Mr. Ahmad Fauzi Mokhtar), Histopathology Laboratory, Faculty of Veterinary Medicine, UPM (Mr. Mohd Jamil Samad, Mrs. Jamila Jahari, Mrs. Latifah Mohd Hanan, Ms. Zainatulaishah Abdul Manaf) and Haematology and Clinical Biochemistry Laboratory, Faculty of Veterinary Medicine, UPM (Mrs. Darulmuqamah Masud, Mrs. Noorain Azman, Mr. Abdullah Misron and Mr. Arman Addelan)) are highly acknowledged. The authors also wish to acknowledge the contributions of the Malaysian Agricultural Research Development Institute (MARDI).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the IPS Research Grant (IPS/2018/9594600) Universiti Putra Malaysia, and the Tertiary education trust fund (TETFUND) Nigeria.
