Abstract
Vascular dementia (VaD) is recognized as the second most prevalent form of dementia after Alzheimer's disease, with its pathogenesis intricately associated with cerebrovascular pathologies, including chronic hypoperfusion, oxidative stress, and neuroinflammation. Recent studies have highlighted dysregulated iron metabolism as a pivotal factor in the pathogenesis of VaD; however, the precise mechanisms and therapeutic implications of this dysregulation remain insufficiently elucidated. In this article, we synthesize clinical and basic research to systematically explore the interaction between iron metabolism and VaD. Aberrant iron metabolism contributes to neuronal damage by exacerbating oxidative stress, lipid peroxidation, and ferroptosis, while cerebrovascular injuries, such as blood–brain barrier disruption and chronic inflammation, further compromise iron metabolism, thereby perpetuating a detrimental cycle. From a diagnostic standpoint, imaging modalities such as susceptibility-weighted imaging, alongside biomarkers like ferritin and soluble transferrin receptors, serve as effective instruments for detecting iron-related pathologies in VaD. At the therapeutic level, the use of chelating agents such as deferoxamine and deferiprone, in combination with antioxidants and innovative nanomaterials, has exhibited neuroprotective effects in animal models by alleviating iron-induced oxidative damage. Nevertheless, further validation is required to establish their clinical efficacy and safety. This is a narrative review. This article investigates the influence and significance of iron metabolism in the pathophysiology of VaD and explores potential therapeutic targets associated with iron metabolism, offering implications for future clinical applications.
Introduction
Vascular dementia (VaD) represents the second most prevalent form of dementia following Alzheimer's disease (AD), constituting approximately 15% to 20% of all dementia cases. 1 The primary etiological factors of VaD are cerebrovascular lesions, notably cerebral small vessel disease (SVD), lacunar infarcts, and multiple strokes, which result in inadequate cerebral blood flow and subsequently induce neurocognitive decline. 2 With the ageing global population, the prevalence and societal impact of VaD are escalating, posing a significant public health challenge. 3 As time progresses, our living environment increasingly accumulates various heavy metal ions due to a multitude of factors, with iron ions being particularly prevalent. Prolonged exposure to these ions can disrupt iron metabolism within the body, potentially contributing to the development of VaD.4,5
In recent years, a growing body of research has indicated that abnormalities in iron metabolism may significantly contribute to the pathogenesis of VaD. Iron, as a vital trace element, is integral to numerous physiological processes, including neurotransmitter synthesis, mitochondrial function maintenance, and antioxidant defense mechanisms. 6 Both iron overload and deficiency can adversely impact the brain. Excessive iron accumulation induces oxidative stress, which compromises the structural and functional integrity of neuronal cells, thereby expediting the progression of neurodegenerative disorders. 7 Dysregulation of iron deposition is closely linked to the development of SVD, poststroke cognitive impairment, and various other neurodegenerative conditions. 8
An imbalance in iron metabolism may significantly contribute to the pathological progression of VaD through multiple mechanisms. Excessive iron accumulation can induce oxidative stress and promote the generation of free radicals, thereby exacerbating damage to vascular endothelial cells and neurons. 9 Furthermore, abnormalities in iron metabolism may intensify the brain's chronic inflammatory state by activating inflammatory response pathways, which further contribute to cognitive decline. 10 Increasingly, research is concentrating on the role of iron metabolism in cerebrovascular pathology, particularly its association with microangiopathy and ischemic brain injury.
Despite advancements in understanding, the precise mechanisms connecting iron metabolism to VaD remain inadequately elucidated. Current research indicates that disorders in iron metabolism not only constitute a risk factor for VaD but may also serve as a potential therapeutic target. The advent of iron-modulating pharmaceuticals and iron chelators has shown promise in preclinical models, 11 suggesting novel avenues for clinical intervention. Nonetheless, empirical evidence supporting the therapeutic efficacy of iron metabolism interventions in VaD remains sparse, necessitating further investigation.
This narrative review systematically retrieves and organizes recent literature concerning the interaction between iron metabolism and VaD, utilizing databases such as PubMed and Web of Science. It conducts a comprehensive examination of the role of iron metabolism dysfunction in the pathogenesis of VaD and identifies potential therapeutic targets. By synthesizing existing clinical and basic research findings, this review aims to offer novel insights for future studies on VaD and potential treatment strategies.
Methods
A comprehensive systematic search of English-language literature was undertaken utilizing PubMed, Web of Science, Embase, and Scopus databases, covering publications up to 30 May 2025. Additionally, the reference lists of the included studies and reviews were meticulously examined, and supplementary sources such as books and conference proceedings were incorporated. The search strategy employed a combination of terms associated with VaD and keywords related to iron metabolism, oxidative stress, and iron chelators.
Pathophysiology of VaD
VaD is a neurodegenerative disorder resulting from cerebrovascular lesions, primarily characterized by a progressive deterioration in cognitive abilities, particularly affecting executive function, attention, memory, and language skills. 1 The pathogenesis of VaD is intricate, often closely associated with factors such as diminished cerebral blood flow, inadequate oxygen supply, neuronal damage, inflammatory responses, and oxidative stress (Figure 1).

Pathophysiology of vascular dementia.
Cerebrovascular injury
Cerebrovascular injury represents a principal etiological factor in the development of VaD. Specific cerebrovascular lesions, including cerebral SVD, lacunar infarctions, and strokes, contribute to a persistent reduction in cerebral blood flow, thereby adversely affecting the neurological functionality of various brain regions. This reduction in cerebral blood flow induces hypoxia, which subsequently disrupts normal neuronal function and metabolism. In critical brain regions, such as the frontal lobe and hippocampus, prolonged inadequate blood supply can lead to neuronal death and cognitive impairment. 12 The nature of the cerebrovascular lesion significantly influences cognitive outcomes. For instance, lacunar infarcts, which are localized injuries resulting from the occlusion of small blood vessels, typically cause nonspecific cognitive decline, whereas multiple strokes are associated with more severe cognitive deterioration. 13 Additionally, traumatic brain injuries (mTBIs), including spinal cord injury 14 and mTBI, are often associated with compromised cerebrovascular and cognitive functions. For instance, following mTBI, the integrity of cerebral white matter fiber tracts is disrupted, which correlates with impaired neurovascular blood flow regulation postinjury. 15 Although various types of vascular lesions result in distinct pathological changes, they share common characteristics, such as diminished cerebral blood flow and neuronal damage.
Oxidative stress
Oxidative stress plays a critical role in the pathogenesis of VaD. As cerebral blood flow diminishes, the brain experiences an inadequate oxygen supply, resulting in elevated levels of reactive oxygen species (ROS) within cells. This excessive accumulation of ROS inflicts damage on cell membranes, proteins, and DNA, ultimately promoting neuronal death. 16 Notably, the excessive accumulation of iron, a potent oxidant, facilitates the generation of free radicals, thereby exacerbating oxidative stress and further intensifying cerebrovascular damage and neuronal degeneration. 8 In the context of cerebrovascular disease, oxidative stress not only harms neuronal cells but may also impair the function of vascular endothelial cells. This impairment leads to alterations in vascular permeability, facilitating the infiltration of inflammatory cells and cytokines into brain tissue, thereby initiating a localized chronic inflammatory response. 10 The interplay between prolonged oxidative stress and inflammatory responses contributes to the progression of neurodegenerative lesions. 17
Inflammatory response
The inflammatory response constitutes a critical mechanism in the pathogenesis of VaD. Cerebrovascular lesions initiate a localized inflammatory response, resulting in the release of cytokines, chemokines, and other inflammatory mediators, primarily through the activation of microglia and astrocytes. These inflammatory mediators induce alterations in cerebrovascular permeability and amplify the immune response within the brain. 18 Microglia, in particular, play a significant role in VaD. A study conducted by Eunjin Sohn et al. demonstrated that VaD enhances microglial activation. 19 Microglia are not only integral to the local immune response to cerebrovascular lesions but also contribute to peripheral neuronal damage by releasing inflammatory factors. Prolonged chronic inflammatory responses further exacerbate neurological decline and facilitate the progression of cognitive impairment. 20 Moreover, cerebrovascular lesions can result in endothelial cell dysfunction and compromise the integrity of the blood–brain barrier (BBB), a continuous endothelial membrane within the cerebral microvasculature characterized by sealed cell-to-cell contacts and coverage by mural vascular cells. 21 The BBB, characterized by elevated trans-endothelial electrical resistance and reduced paracellular and transcellular permeability,22,23 serves to shield neurons from external influences. Consequently, cerebrovascular lesions that compromise the integrity of the BBB permit the infiltration of immune cells and inflammatory mediators, thereby exacerbating localized neurological damage and inflammatory responses. 24
Disruption of neuroprotective mechanisms
While oxidative stress and inflammatory responses are primary contributors to the pathogenesis of VaD, the impairment of neuroprotective mechanisms also plays a critical role. Under normal physiological conditions, the brain is equipped with a robust system of antioxidant defenses that preserve neuronal integrity. However, an insufficient cerebral blood flow can compromise these defenses, rendering neurons more vulnerable to oxidative damage. This vulnerability may be further intensified by the excessive accumulation of iron within the brain. Although iron is an essential element for nervous system function, its excessive accumulation can destabilize cell membranes and amplify neuronal damage. 25
Epidemiology of VaD and iron metabolism
Current epidemiology of VaD
VaD is the second most prevalent form of dementia following AD and represents a significant public health challenge globally. A national survey conducted in South Korea, which assessed 6141 individuals aged 65 years and older, identified several factors associated with an increased risk of dementia, including age, gender, education, smoking, head trauma, and a history of depression. Conversely, alcohol consumption and moderate exercise were linked to a reduced risk of dementia. The standardized prevalence of VaD in this cohort was determined to be 2.0%. 26 The prevalence of VaD exhibits regional variability. Certain studies have reported higher dementia prevalence rates in rural areas compared to urban areas. In a regional study conducted in Taiwan, a sample of 10,432 participants aged 65 years and older was surveyed. The findings indicated that the prevalence of dementia among older adults was 8.69% in rural areas, 6.63% in suburban areas, and 4.46% in urban areas. This disparity was observed across all age and gender groups, with the rural-to-urban prevalence ratio being more pronounced in women than in men. 27 Furthermore, the development of VaD is linked to various risk factors, including hypertension, diabetes mellitus, and cardiovascular disease. Variations in the distribution of these risk factors across different regions and populations may contribute to the observed differences in the epidemiological characteristics of VaD.
Investigations into the relationship between abnormal iron metabolism and VaD
An increasing corpus of research indicates a significant correlation between abnormal iron metabolism and VaD. 28 In a study focusing on individuals aged over 60 with nonalcoholic fatty liver disease, 54% of the participants were diagnosed with mild cognitive impairment (MCI). This cognitive impairment was significantly associated with elevated levels of soluble transferrin receptor (sTfR) (odds ratio = 2.565, 95% CI [1.334–4.934]; p = .005), thereby suggesting a notable association between indicators of iron metabolism and cognitive impairment. 29
Animal studies have also substantiated this association. Administering an iron-rich diet to rats resulted in cognitive impairments, elevated iron concentrations in the brain, increased oxidative stress, and disrupted neurotransmitter metabolism. Rats subjected to a high-iron diet exhibited prolonged escape latency and impaired spatial learning and memory in the Morris water maze experiment. Additionally, these rats showed elevated levels of malondialdehyde (MDA) in the brain and reduced superoxide dismutase activity, indicating that iron overload may damage neuronal cells by inducing oxidative stress, thereby affecting cognitive function. 30 Furthermore, aberrant iron metabolism may contribute to the pathogenesis of VaD by influencing neuroinflammatory responses, apoptosis, and other pathways.
Epidemiological variations in iron metabolism and VaD across different populations
There are notable epidemiological variations in iron metabolism and VaD among different populations. Research indicates a nonlinear relationship between dietary iron intake and the incidence of dementia. Both low (<10.93 mg/day) and high levels of iron intake are associated with an increased risk of all-cause dementia, with the risk associated with low iron intake being particularly significant among women without hypertension. 31 Regarding gender differences, several studies have suggested that women may be more vulnerable to the effects of abnormal iron metabolism on VaD. Hepcidin plays a crucial role in maintaining cerebral homeostasis, and its inhibition may partially mitigate brain damage. 32 In studies involving postmenopausal women, estrogen deficiency has been linked to increased cerebral iron deposition, potentially due to estrogen's modulatory effect on the iron-regulating hormone hepcidin. The research demonstrated that aged mice exhibited increased concentrations of hepcidin and ferritin within the brain and cerebral microvasculature relative to their younger counterparts. Furthermore, estrogen replacement therapy was observed to mitigate these elevated levels. This finding implies that a deficiency in estrogen may contribute to heightened cerebral iron accumulation by upregulating hepcidin expression, thereby potentially elevating the risk of VaD in women. 33
In a study conducted by Jiao Wang et al., involving 207,203 dementia-free participants aged 60 years or older who were followed for up to 16 years through the U.K. Biobank, anemia was found to be associated with a rapid decline in overall cognitive performance (β = −0.08, 95% CI [−0.14, −0.01]) and processing speed (β = −0.10, 95% CI [−0.19, −0.01]). Over the course of a median follow-up period of 9.76 years (interquartile range 7.55–11.39), 6272 participants developed dementia. The risk ratio for dementia was 1.57 (95% CI [1.38, 1.78]), and anemia was associated with an earlier onset of dementia by 1.53 years (95% CI [1.08, 1.97]). The risk of dementia was particularly elevated in individuals with anemia and high C-reactive protein levels (risk ratio = 1.89, 95% CI [1.60, 2.22]), indicating that anemia is linked to cognitive decline and an increased risk of dementia, particularly in populations with high inflammatory markers. 34
The association between iron metabolism and VaD exhibits variability across populations with distinct disease states. In individuals with diabetes, for instance, dysregulated iron metabolism is linked to an elevated risk of developing VaD. Diabetic patients frequently experience insulin resistance and disruptions in glucose metabolism, which can influence iron metabolism and distribution. This can result in iron accumulation in the brain, thereby exacerbating neurological damage. Empirical evidence indicates that increased serum ferritin (SF) levels in diabetic individuals correlate with cognitive decline, suggesting that aberrant iron metabolism may significantly contribute to the pathogenesis of diabetes-related VaD. 35 Furthermore, genetic factors significantly contribute to the interplay between iron homeostasis and VaD. Research conducted on individuals from Espírito Santo, Brazil, revealed variations in the distribution of HFE gene polymorphisms associated with hereditary hemochromatosis (C282Y, H63D, and S65C) among the general population, Pomeranian descendants, and individuals diagnosed with hemochromatosis. These findings imply a genetic influence on iron metabolism and its associated disorders. Although this study does not directly address VaD, it offers valuable insights into the genetic foundations of iron metabolism. 36 Furthermore, some studies suggest that populations with specific genetic susceptibilities—such as mutations in genes related to iron homeostasis or the presence of the APOE4 allele—may have an elevated risk of developing VaD. This association underscores the role of genetic regulation in iron metabolism and its implications in neuropathological processes. 37 Nonetheless, a comprehensive understanding of the specific mechanisms underpinning these genetic factors necessitates additional rigorous research. However, further research is required to elucidate these genetic influences.
Diagnostic techniques in VaD related to iron metabolism
Advances in imaging diagnostics
Imaging modalities are crucial in diagnosing VaD. The magnetic resonance imaging (MRI) technique known as susceptibility-weighted imaging (SWI) is particularly effective in detecting cerebral iron deposition. A study involving 35 patients with Subcortical Ischemic VaD (SIVD) and 35 healthy controls revealed significantly elevated phase shift values in the bilateral hippocampus, caudate nucleus, nucleus accumbens, right pallidum, and left substantia nigra among the SIVD patients. This finding indicates increased iron accumulation in these regions. Furthermore, the phase shift values in the left hippocampus and right caudate nucleus were strongly correlated with neuropsychological scores. These results suggest that SWI may serve as a valuable tool for detecting cerebral iron deposition and evaluating the progression of SIVD. 38
Furthermore, positron emission tomography (PET) serves as a valuable tool for evaluating brain metabolism and neurotransmitter function. In a study involving patients with VaD, PET imaging demonstrated decreased glucose metabolism in specific brain regions, correlating with cognitive impairment. Additionally, PET is instrumental in detecting amyloid-beta (Aβ) plaque deposition, facilitating early diagnosis and disease monitoring. For instance, an investigation utilizing 18F-florbetaben-PET to assess older adults with MCI and depressive symptoms revealed that approximately half of the participants exhibited cerebral amyloidosis. The group with cerebral amyloidosis showed poorer performance on the Word List Recall and Constructed Recall Tasks compared to the amyloidosis-negative group. These findings underscore the significant role of PET in the diagnosis and evaluation of VaD. 39
Studies on biomarkers associated with dysregulated iron metabolism
The identification of biomarkers indicative of abnormal iron metabolism is crucial for the diagnosis and monitoring of VaD. 40 In humans, SF serves as a marker of iron storage and plays a significant role in the regulation of iron metabolism. Some studies have reported alterations in SF levels in patients with VaD, with both sTfR and SF levels showing varying degrees of reduction in individuals with mild cognitive impairment. 29 Furthermore, alterations in hepcidin levels, a key hormone regulating iron metabolism, may also be linked to VaD. In patients with AD and Down syndrome dementia, elevated serum hepcidin levels have been observed, which are associated with hypoferritinemia, suggesting a potential involvement of hepcidin in neuroinflammation and iron metabolism dysregulation. 41 Consequently, the biomarkers examined in the aforementioned studies, including transferrin, sTfR, and hepcidin, are crucial in iron metabolism, and changes in their levels might indicate a link between iron metabolism disorders and VaD. However, further research is necessary to determine the precise significance of these biomarkers in diagnosing and treating VaD.
In a study involving patients with SIVD and AD, the integration of neuropsychological assessments and multimodal MRI scans demonstrated that individuals with SIVD exhibited deficits in information processing speed but performed relatively well in memory, language, and visuospatial tasks compared to those with AD. Furthermore, the composite cognitive score effectively discriminated between SIVD and AD patients, achieving an area under the curve of 0.727 (95% CI [0.62–0.84], p < .001). Additionally, the Auditory Verbal Learning Test recognition scores were inversely correlated with the total SVD scores, indicating that this integrated diagnostic approach can significantly enhance the diagnostic and differential diagnostic capabilities for VaD. 42
Furthermore, the concurrent utilization of biomarker testing alongside imaging modalities holds significant potential. For instance, integrating biomarker levels, such as SF and hepcidin, with brain iron deposition observed through MRI can enhance our comprehension of the interplay between iron metabolism abnormalities and VaD. This approach may also offer a more robust foundation for early diagnosis and therapeutic interventions. Nevertheless, the existing combined diagnostic methodologies encounter several challenges in clinical application, particularly concerning the standardization and integration of diverse detection techniques, which necessitate further investigation and refinement.
New advances in iron metabolism
Iron plays a crucial physiological role in the nervous system, being vital for its normal development and functional maintenance. During brain development, iron is integral to the proliferation, migration, and differentiation of neural progenitor cells. It is essential from early embryonic stages to ensure proper brain development, with the brain extensively expressing the TfR to maintain adequate iron supply during this period. 43 During neural development, iron significantly influences neuronal differentiation, migration, and synapse formation. As an essential trace element, iron is crucial for the normal functioning of the nervous system and plays a pivotal role in various physiological processes, including cellular respiration, neurotransmitter synthesis, and myelin sheath formation. Iron is integral to intracellular electron transfer processes and, more importantly, serves as a vital component of numerous enzymes, such as cytochrome oxidase and catalase, which are indispensable for sustaining energy metabolism in neurons. 44
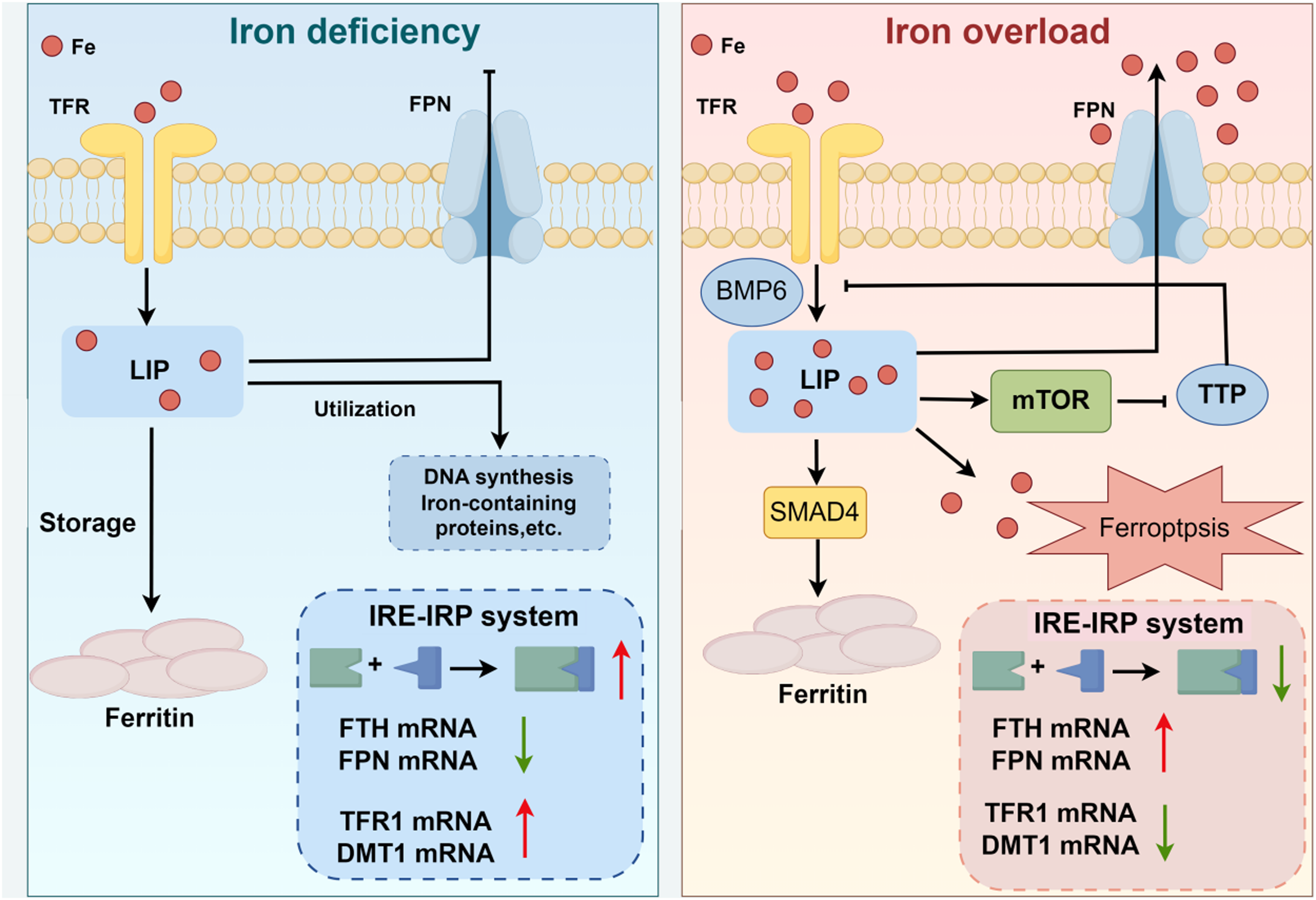
The absorption of iron is a multifaceted process influenced by various factors, intricately connected to the pathogenesis of VaD. Under normal physiological conditions, iron absorption predominantly occurs in the duodenum and upper jejunum, with transferrin receptor 1 (TfR1) and divalent metal transporter 1 (DMT1) playing crucial roles in its transmembrane transport. 45 Iron storage within the body is primarily mediated by molecules such as ferritin, and dysregulation in iron storage is significantly associated with the pathological mechanisms underlying VaD.
Under normal circumstances, iron exists in two forms: ferrous (Fe2+) and ferric (Fe3+) iron. Iron can act as a pro-oxidant by catalyzing theformation of ROS. The classical pro-oxidantreaction of iron, the Fenton reaction 46 :
Fe2+ + H2O2 → Fe3+ + HO•+HO− (Reaction 1)
Fe3+ + O2•− → Fe2+ + O2 (Reaction 2)
The sum of Reaction 1 (Fenton reaction) and Reaction 2 is commonly known as the Haber–Weiss reaction, with iron as the catalyst. The Haber–Weiss reaction demonstrates that, in the presence of catalytic quantities of redox-active aqueous low-molecular-weight iron—which increases in peripheral tissues under conditions of iron overload, or in the brain during various central nervous system pathologies—hydrogen peroxide (H2O2) can serve as a readily available source of the harmful hydroxyl radical. This occurs in the presence of ferrireductants, such as superoxide, which regenerate the reduced form of the metal. Iron catalysis leads to the production of highly reactive and detrimental ROS, and the resultant unstable iron-induced oxidative stress is a critical factor in the pathogenesis of central nervous system disorders, including AD and VaD. 8 In a separate study, it was observed that under certain pathological conditions, free heme can undergo oxidation to form heme, a process marked by the conversion of iron from its ferrous to ferric state, within the context of plaque hemorrhages in advanced lesions. The accumulation of erythrocytes within these plaques results in localized elevations of free heme and heme, which in turn facilitate the expansion of unstable iron pools, thereby accelerating the progression of atherosclerosis. This mechanism indirectly contributes to the pathogenesis of VaD. 47
In pathological states related to VaD, such as cerebral ischemia, the expression of molecules involved in iron storage undergoes alterations. For example, in a rat model of focal cerebral ischemia, the expression of ferritin-like protein (Fpn) in the hippocampal CA2 region demonstrates dynamic changes over time, with a peak observed 12 h postsurgery. This evidence indicates that ferroportin (Fpn) may alleviate ischemic brain injury through the regulation of iron storage, thereby holding significant potential for the prevention and treatment of VaD. 48 Intracerebral hemorrhage (ICH) constitutes a major risk factor for VaD, initiating a cascade of alterations associated with iron storage and neuronal injury. Research has demonstrated markedly elevated total iron concentrations in hematomas and adjacent tissues following ICH, which are accompanied by increased oxidative stress markers, such as aggregin. 49 This suggests that iron release may contribute to oxidative damage. Additionally, the disruption of the BBB subsequent to hemorrhage results in the upregulation of iron-related proteins, including the receptor for advanced glycation end-products (RAGE) and its ligand, high-mobility group box protein B1. The administration of RAGE-specific antagonists has been shown to attenuate BBB damage and cerebral edema, indicating that iron-related inflammatory responses play a pivotal role in the pathophysiology of VaD. 50
In experimental rat models of subarachnoid hemorrhage, there is an observed upregulation of hepcidin expression, accompanied by a concomitant downregulation of its downstream proteins, including ferroportin 1 (Fpn1) and ceruloplasmin. This dysregulation results in heightened iron-dependent oxidative damage, thereby highlighting the critical role of aberrant iron storage regulation in the pathophysiology of VaD. 51 Moreover, iron distribution within the brain demonstrates regional specificity, and alterations in this distribution are intricately linked to the onset and progression of VaD. Investigations employing MRI to assess iron distribution in the brains of patients with VaD have identified increased iron accumulation in specific regions, such as the hippocampus and basal ganglia.
Numerous studies have explored the relationship between iron and the nervous system from various perspectives. In cellular experiments, iron overload has been shown to induce mitochondrial dysfunction by activating the endoplasmic reticulum-mediated p-eIF2α/ATF4/CHOP pathway. This activation results in elevated ROS levels, increased expression of endoplasmic reticulum stress-related proteins, and apoptosis. 52 In animal studies, early iron deficiency results in diminished iron concentrations in specific regions of the porcine brain, alters the concentrations of gray and white matter, and compromises the integrity of white matter fibers, with these effects persisting despite subsequent iron supplementation. 53 Furthermore, in neurological disorders such as Parkinson's disease and AD, dysregulation of iron metabolism is evident, and iron accumulation may contribute to neuronal damage and disease progression through multiple mechanisms. 54
Mechanisms governing iron metabolism regulation
The regulatory mechanisms governing iron metabolism are intricate and finely balanced, involving numerous interactions and molecules. Iron overload influences iron metabolism by enhancing hepcidin synthesis through the activation of the BMP6/SMAD pathway. The regulation of iron metabolism by the mammalian target of rapamycin (mTOR) has garnered significant attention. mTOR modulates iron metabolism by regulating the expression of Tristetraprolin. As a third iron-regulatory system, the mTOR pathway, alongside the hepcidin-ferroportin hormone system and the cellular iron-responsive element (IRE)/ferroportin regulatory network, plays a crucial role in the regulation of systemic iron metabolism. 55 Furthermore, within the intracellular environment, iron-regulated proteins 1 and 2 (IRP1 and IRP2) serve as fundamental regulators of cellular iron metabolism. Under conditions of iron deficiency, these proteins interact with IREs to modulate the expression of proteins associated with iron metabolism. Conversely, in states of iron sufficiency, the SCFFBXL5 ubiquitin ligase targets the IRPs for ubiquitination, thereby inhibiting their ability to bind to IREs and regulating iron metabolism accordingly.56,57 Investigating biomarkers of abnormal iron metabolism is crucial for the diagnosis, treatment, and monitoring of related diseases. In a study involving patients with metabolic syndrome, it was observed that elevated SF levels were linked to impaired glucose homeostasis.
MicroRNAs play a crucial role in the regulation of cellular transcription by modulating mRNA expression. They significantly impact iron metabolism, particularly in the regulation of ferroptosis. MicroRNAs can be categorized into antiferroptotic and pro-ferroptotic types. Antiferroptotic microRNAs target mRNAs that encode proteins involved in promoting ferroptosis, such as ALOX15 and ACSL4, which are implicated in the production of phospholipid hydroperoxides. Additionally, miR-7-5p targets transferrin, thereby indirectly reducing labile iron pools and Fenton reactions, ultimately decreasing iron uptake. 58 This issue may emerge as a central focus for future research on the treatment of VaD. Pro-ferroptotic microRNAs exert their effects on the SLC7A11/GPX4 axis by promoting lipid peroxidation, thereby inducing ferroptosis. In addition to directly targeting GPX4, its expression is modulated by the ATF4 target gene HSPA5. Moreover, miRNAs influence iron storage and release by targeting FTH and FPN, respectively. 59 Simultaneously, Nrf2 functions as a principal antioxidant regulator, remaining inactive when sequestered by Keap1. Consequently, microRNAs that target Keap1 to activate Nrf2 may be classified as antiferroptotic.60,61 Thus, microRNAs play a pivotal role in regulating iron metabolism, ferroptosis, and oxidative stress through various pathways, all of which are critical in the pathogenesis of VaD.
Additionally, iron overload was associated with alterations in a specific subset of serum phosphatidylcholines, and pathways involving sarcosine and citrulline may play a role in iron-induced disruptions of glucose metabolism. 62 At present, there is a lack of targeted research on markers of abnormal iron metabolism specific to the nervous system, particularly in relation to VaD. It is anticipated that future scholarly investigations will delve deeper into this area to enhance the prevention, diagnosis, and detection of diseases associated with VaD.
The relationship between iron metabolism and neuroprotection
Iron plays a crucial role in brain health, with its metabolic balance in the brain being essential for preserving normal neurological function. Research indicates that iron status during pregnancy is correlated with neuropsychological outcomes in children. Specifically, adequate maternal SF levels and iron intake are associated with improved working memory and executive function scores in offspring. These findings suggest that maintaining optimal iron status during pregnancy is vital for child neurodevelopment. 63
In the context of cerebral ageing and neurodegenerative disorders, it has been observed that cerebral iron levels escalate with advancing age, and this accumulation is linked to various neurodegenerative conditions. For instance, a study involving cognitively normal adults demonstrated a negative correlation between cerebral iron content and myelin content, indicating that reduced myelin content corresponded with elevated iron levels. Furthermore, iron levels were found to increase significantly with age, exhibiting a tendency to be higher in males compared to females. 64 In certain animal models of neurological disorders, such as a murine model of AD, elevated cerebral iron is associated with cognitive deficits. Notably, physical exercise may enhance cognitive function and mitigate neuronal damage by modulating proteins involved in iron metabolism. 65 Additionally, in patients with Parkinson's disease, quantitative magnetization rate imaging studies have identified increased iron deposition in specific brain regions, such as the substantia nigra, which correlates with both motor and certain non-motor symptoms. 66
Therefore, iron metabolism is intricately connected to the functioning of the nervous system (Figure 2). In the subsequent discussion, we will examine the associations and interactions between iron metabolism and VaD.
Disorder of iron metabolism.
Interactions between iron metabolism and VaD
Cells primarily affected by abnormal iron metabolism in VaD
In VaD, iron metabolism can impact a variety of cell types, with neurons being particularly vulnerable. Among these, hippocampal neurons exhibit heightened sensitivity to aberrant iron accumulation. Excessive iron can induce oxidative stress, disrupt autophagic processes, and elevate apoptosis rates in neurons, thereby impairing cognitive functions such as learning and memory. 45 Empirical evidence indicates that iron overload enhances ROS production and lipid peroxidation in neurons, compromising cell membrane integrity and functionality. This process also activates apoptosis-related proteins, such as Bax, culminating in neuronal apoptosis. Furthermore, iron influences neuronal energy metabolism and disrupts mitochondrial function, exacerbating neuronal damage. In addition to neurons, endothelial cells are profoundly influenced by iron metabolism. Dysregulation of iron metabolism can result in endothelial cell dysfunction, thereby compromising the normal physiological functions of blood vessels. Specifically, iron overload can inflict damage on endothelial cells, leading to increased vascular permeability and impaired vasodilation, which subsequently affects the functionality of neurovascular units and facilitates the progression of VaD. 67 Furthermore, microglial cells are integral to iron metabolism and the pathogenesis of VaD. Activated microglial cells have the capacity to uptake iron, and their functional status is modulated by iron levels; excessive iron accumulation may lead to hyperactivation of microglial cells, prompting the release of inflammatory mediators, triggering neuroinflammation, and causing further damage to neurons and neural tissues. 68
Iron metabolism abnormalities can cause VaD
The relationship between the pathological mechanisms of VaD and dysregulated iron metabolism is intricate. Numerous studies have indicated that aberrant iron metabolism may play a crucial role in the etiology of VaD. For instance, excessive iron accumulation has been associated with neuronal damage, heightened oxidative stress, and cognitive impairment in various animal models. In experiments where mice were subjected to a high-iron diet, a reduction in hippocampal neuron count was observed, along with increased oxidative stress markers and cognitive decline. These findings suggest that iron overload may induce neurological damage via oxidative stress and other pathways, thereby contributing to the onset of VaD. 30 Abnormal iron metabolism has been implicated in influencing the proliferation and migration of vascular smooth muscle cells, thereby exacerbating vascular sclerosis and SVD through modulation of the hydrogen sulfide (H₂S) signaling pathway.69,70 Furthermore, studies have identified that genetic mutations linked to iron metabolism disorders, such as those associated with hemochromatosis, substantially elevate the risk of VaD, particularly when combined with amyloid-beta (Aβ) deposition, leading to significant alterations in cerebrovascular pathology. 71
Furthermore, at the molecular level, iron metabolism may contribute to the pathogenesis of VaD by modulating intracellular signaling pathways. Research indicates that iron overload activates the AMPK/autophagy pathway, which is crucial for maintaining intracellular homeostasis and managing cellular stress. Under conditions of iron overload, there is an upregulation in the expression of molecules such as AMPK, Beclin1, and LC3, which facilitates the formation of autophagosomes. However, excessive autophagy can result in neuronal damage and death, potentially impairing cognitive function. 45 Iron metabolism may influence neuronal survival, proliferation, and apoptosis by modulating various signaling pathways, including the mitogen-activated protein kinase pathway and the phosphatidylinositol-3-kinase (PI3K)/protein kinase B (Akt) pathway. In the context of AD, aberrant iron accumulation can activate kinases such as glycogen synthase kinase-3β and cyclin-dependent kinase 5. This activation results in the abnormal phosphorylation of tau protein and amyloid precursor protein, thereby accelerating the formation of amyloid β plaques and neurofibrillary tangles. A similar mechanism may be implicated in VaD. 72
Abnormalities of iron metabolism can be secondary to and exacerbate VaD
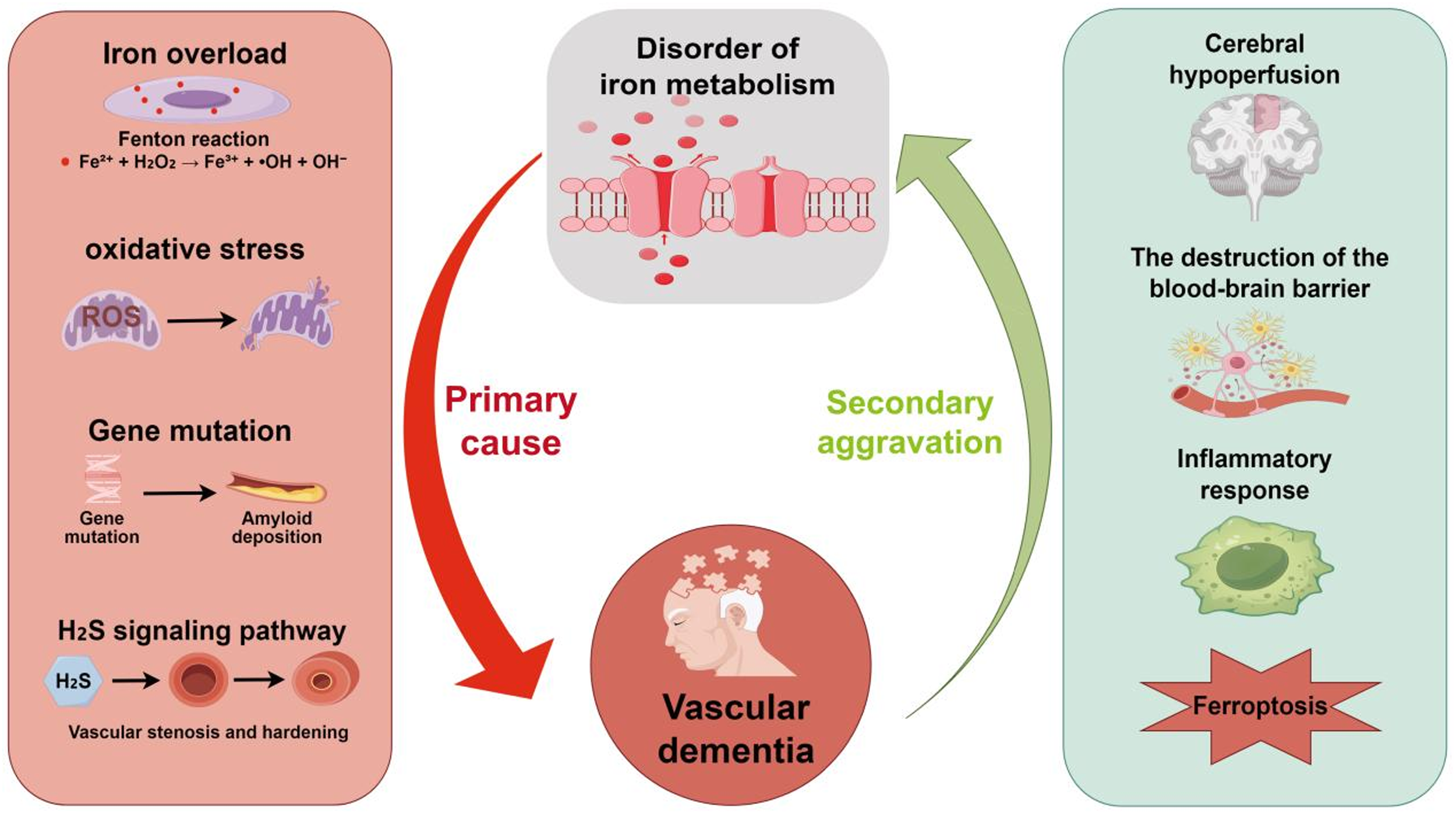
Conversely, there is a growing body of evidence suggesting that the pathological processes underlying VaD may contribute to disruptions in iron metabolism, rather than the latter directly initiating the disease. Patients with VaD frequently exhibit cerebrovascular lesions, which can impact iron transport and metabolism, resulting in abnormal iron accumulation in the brain.
Cerebral hypoperfusion and hypoxia-induced iron release
A key pathological aspect of VaD is reduced cerebral blood flow, such as chronic cerebral hypoperfusion (CCH), which is a significant cause of VaD. CCH severely impacts the brain's energy supply, leading to increased energy use, reduced ATP production, enhanced anaerobic fermentation, and the production of large amounts of lactic acid. 73 This lactic acidosis in tissues not only causes irreversible neurological damage but also speeds up free radical production and lipid peroxidation. 74 Furthermore, ischemia-reperfusion damage worsens oxidative stress and neuronal harm through mitochondrial dysfunction and increased ROS, 75 triggering the release of iron from iron-sulfur cluster proteins and forming free iron (labile iron).76,77 This release of iron results directly from vascular injury rather than a primary disorder of iron metabolism.
Disruption of the BBB and peripheral iron leakage
Iron homeostasis in the brain is stringently regulated, with the BBB serving as the primary defense against iron overload. This barrier limits iron transport through highly regulated and selective transport systems. Under normal physiological conditions, the brain maintains iron equilibrium through three well-coordinated mechanisms: (A) iron transport into the brain, facilitated by TFR-mediated or non-TFR-mediated pathways across the BBB; (B) iron storage, primarily dependent on the availability of ferritin for cellular sequestration; and (C) iron efflux, the rate of which is modulated by cerebrospinal fluid flow and/or clearance mechanisms within the BBB. 78 Recent research suggests that abnormalities in iron metabolism may elevate lipoprotein levels in brain microvascular endothelial cells, consequently disrupting the integrity of the BBB. 79
Vascular lesions associated with VaD, such as SVD and endothelial dysfunction, can compromise the integrity of the BBB. This disruption facilitates the entry of iron from the peripheral circulation into the brain parenchyma through compromised vessels. Research indicates that damage to the vascular endothelium leads to increased expression of the TfR and ferroportin, 80 thereby promoting abnormal iron transport across the BBB. 81 For instance, in cases of subcortical VaD, MRI reveals reduced iron deposition in areas such as the bilateral hippocampus. This observation may reflect abnormal iron redistribution or extravasation following localized vascular injury. 12
Interaction between inflammation and iron metabolism
The vascular pathology associated with VaD is frequently accompanied by chronic neuroinflammation. In this context, activated microglia and astrocytes contribute to the upregulation of hepcidin expression through the release of pro-inflammatory cytokines such as interleukin-6 and tumor necrosis factor-alpha. This process inhibits the activity of the iron efflux protein ferroportin, resulting in the accumulation of intracellular iron.82,83 Moreover, inflammation induced by vascular injury enhances the expression of proteins related to iron uptake, such as DMT1, thereby further aggravating local iron overload. 84
Secondary involvement of iron death (ferroptosis)
Lipid peroxidation and glutathione depletion resulting from vascular injury may initiate the ferroptosis pathway.85,86 For instance, following ischemic stroke, ROS and free iron facilitate lipid peroxidation through the Fenton reaction, directly impairing vascular smooth muscle cells and neurons. This process has been proposed as a “downstream event” in the pathology of VaD. 87 Furthermore, animal models demonstrate that markers of ferroptosis, such as ACSL4 and PTGS2, are upregulated in cases of VaD, with this upregulation being positively correlated with the severity of vascular injury. 88
Regional abnormalities in brain iron distribution
Variations in the localization of vascular lesions can lead to disturbances in iron metabolism that are specific to certain brain regions. 89 For instance, bilateral reductions in iron levels within the hippocampus and thalamus in patients with subcortical VaD may indicate neuronal loss and iron depletion resulting from chronic ischemia. 90 Conversely, elevated iron levels in specific areas, such as the basal ganglia, may be associated with localized microbleeds or vascular leakage. These regional discrepancies imply that aberrant iron metabolism is a secondary consequence of vascular injury. 12
Vascular ageing and chronic disruption of iron homeostasis
Chronic vascular ageing, exemplified by conditions such as atherosclerosis and capillary thinning, may indirectly influence iron metabolism through mitochondrial dysfunction. In ageing vascular endothelial cells, abnormalities in mitochondrial iron metabolism result in elevated free radical production and accelerated iron accumulation in vascular smooth muscle cells. This process establishes a detrimental cycle characterized by “vascular injury-iron imbalance-oxidative stress.” 91 It is important to note that this disorder in iron metabolism arises as a consequence of accumulated vascular lesions, rather than serving as the primary etiological factor.
A review of the existing evidence indicates that the vascular pathology associated with VaD, such as ischemia, BBB disruption, and inflammation, significantly contributes to aberrant iron metabolism. Concurrently, disruptions in iron metabolism, including free iron accumulation and ferroptosis, exacerbate oxidative stress and neuronal degeneration, thereby initiating a pathological cascade. 92 Consequently, further research is warranted to elucidate the causal relationship between abnormal iron metabolism and VaD, with particular emphasis on exploring the intricate mechanisms underlying their interaction (Figure 3).
Interactions between iron metabolism and VaD.
Therapies for VaD linked to iron metabolism
Iron chelators in VaD
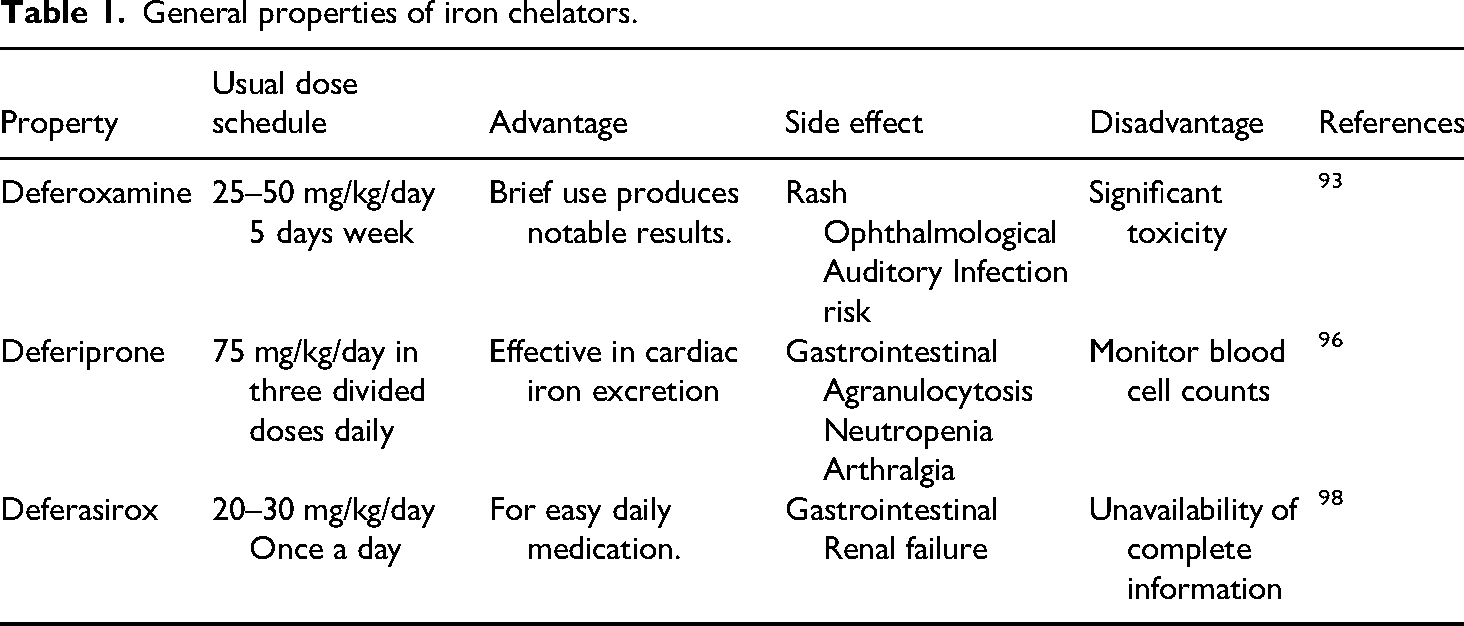
Iron chelators are employed as a therapeutic approach to mitigate excessive iron accumulation in the body and to alleviate neuronal damage resulting from iron overload. Deferoxamine (DFO), a high-affinity iron chelator, has demonstrated disease-modifying effects across a spectrum of neurodegenerative and neurovascular disorders when administered intranasally (IN). 93 In a study utilizing a rat model of streptozotocin-induced AD, IN DFO treatment was observed to enhance spatial memory, evidenced by a significant reduction in escape latency during the Morris water maze task. Additionally, there was a noted decrease in oxidative stress levels within the brain and an upregulation of insulin receptor expression. 94 Nonetheless, the long-term efficacy and safety of iron chelator interventions remain subjects of debate. While certain short-term studies indicate that iron chelators can effectively reduce bodily iron levels and ameliorate related symptoms, the long-term outcomes are yet to be conclusively determined. 95
Deferiprone, another iron chelator, has garnered attention in related research. In an investigation involving rabbits on a cholesterol-enriched diet, deferiprone was observed to significantly decrease levels of Aβ40 and Aβ42, as well as BACE1 expression and tau protein phosphorylation, while exhibiting no notable impact on ROS production. The study further demonstrated that deferiprone treatment led to reductions in plasma iron and cholesterol levels, although it did not affect brain iron levels, implying its potential to mitigate AD-like pathological changes by lowering plasma iron and cholesterol levels. 96 Nevertheless, the long-term use of iron chelators, such as deferasirox, may be associated with adverse effects, including the development of Fanconi's syndrome, necessitating careful monitoring in clinical applications. 97
Deferasirox is an oral iron chelator; however, its use is associated with significantadverse effects, including nephrotoxicity and disturbances in electrolyte balance 98 (Table 1).
General properties of iron chelators.
Impact of antioxidant treatment on iron metabolism
Antioxidant therapy, designed to mitigate oxidative stress damage caused by iron overload, may also influence iron metabolism. Numerous studies have investigated the modulatory effects of antioxidants on biomarkers associated with iron metabolism. For instance, in a study involving patients with β-thalassemia (β-TM), the administration of α-lipoic acid (ALA) over an 8-week period led to a statistically significant reduction in SF levels (p = .004), alongside a decrease in MDA levels and an increase in high-density lipoprotein cholesterol levels. These findings suggest that ALA may enhance iron metabolism through its antioxidant properties. 99
Comparable results have been observed in animal studies. The administration of naringenin, a natural flavonoid, to iron-overloaded rats resulted in improvements in anxiety-like behavior, a reduction in the production of ROS in the brain, and the restoration of iron overload-induced changes in acetylcholinesterase expression levels, mitochondrial membrane potential, and the activity of mitochondrial complexes. Furthermore, it regulated the activity and expression of ectonucleotidase, indicating that naringenin may exert neuroprotective effects through antioxidant mechanisms and pathways related to the regulation of iron metabolism. 100 Additionally, antioxidants such as vitamin C have been shown to influence cellular iron uptake and metabolism, including the stimulation of ferritin synthesis and the inhibition of lysosomal ferritin degradation. 101 However, the specific effects of these antioxidants in the treatment of VaD require further investigation (Table 2).
General properties of antioxidant.

Note: ALA: α-lipoic acid.
Advances in clinical trials for combined treatment approaches
Integrated treatment strategies that combine multiple therapeutic approaches are anticipated to be more efficacious in the management of VaD. Current clinical trials are investigating integrative treatments for this condition. Some studies have explored the combination of pharmacotherapy with lifestyle interventions. For instance, in one study, patients with VaD received drug therapy alongside lifestyle modifications, including cognitive training and physical exercise, resulting in notable improvements in cognitive function and daily living abilities. Additionally, research has been conducted on drug combination therapies. A meta-analysis revealed that the concurrent administration of donepezil hydrochloride and nimodipine in patients with VaD significantly enhanced MMSE, ADL, and CDR scores compared to monotherapy. The most pronounced improvements were observed after 12 weeks of intervention, indicating that combination pharmacotherapy may represent a more effective treatment strategy.27,102 Nevertheless, the strategy of combination therapy continues to encounter challenges in clinical trials, including the determination of optimal combinations and dosages of various treatments, as well as the evaluation of the synergistic effects of multiple therapeutic factors on patients. These issues necessitate further optimization through ongoing research and clinical practice.
Therapeutic uses of green nanomaterials
Green nanomaterials show promise in VaD research by overcoming the BBB to deliver drugs effectively. Chitosan-based nanoparticles, known for their biocompatibility, can encapsulate neuroprotective drugs like curcumin using ion-gelation methods. These nanocarriers enhance drug stability, provide sustained release, and target neuronal receptors, boosting brain drug accumulation and therapeutic efficacy. 102 Additionally, advanced systems like pH-responsive nanocarriers offer precise drug release in the brain's mildly acidic lesion sites. 103
Green nanomaterials are vital for neuroprotection in VaD, as they help reduce neuronal damage. Some, like epigallocatechin gallate (EGCG) from green tea, have antioxidant and antiinflammatory properties that lower oxidative stress and inflammation in the brain. Synthesized into nanoparticles, EGCG becomes more stable and bioavailable, effectively scavenging ROS and reducing neuronal oxidative damage and inflammation. 104
Certain nanomaterials activate neuroprotective pathways like PI3K/Akt, inhibiting apoptosis and supporting neuron survival. They also encourage neural stem cell growth and differentiation, aiding neural tissue repair. For example, green-synthesized nano-hydroxyapatite promotes neural stem cell differentiation into neurons, presenting new treatments for VaD. 105
Clinical trials using green nanomaterials for VaD are still in early stages, with some initial studies assessing their safety and efficacy. For example, a trial using green-synthesized nanocarriers for gene therapy showed good biocompatibility and some symptom improvement. However, larger sample sizes and long-term studies are needed to confirm these findings.
Analysis of result variations across studies
In research examining VaD and iron metabolism, variability in study outcomes is evident. This heterogeneity can be attributed to multiple factors. Primarily, variations in study populations contribute to differing results. Participants across studies vary in age, gender, underlying conditions, and disease severity, all of which can impact the observed relationship between iron metabolism and VaD. For instance, in studies involving older populations, the link between abnormal iron metabolism and VaD may be more pronounced due to age-related physiological changes. Conversely, in younger cohorts or those with distinct comorbidities, the findings may differ.
Secondly, variations in study methodologies significantly contribute to heterogeneity. These variations encompass differences in testing methods, diagnostic criteria, and treatment protocols across studies. For instance, in the detection of brain iron deposition, disparate imaging techniques may exhibit varying sensitivities and specificities, resulting in inconsistent findings. Similarly, when assessing biomarkers associated with iron metabolism, the use of different test kits and assays can lead to divergent outcomes. Furthermore, variations in study design, such as sample size and duration of follow-up, may also influence the consistency of study results. Consequently, it is imperative to thoroughly consider these heterogeneous factors when synthesizing and analyzing the findings of related studies to more accurately elucidate the relationship between VaD and iron metabolism.
Conclusion
The pathophysiological mechanisms underlying VaD are intricate, characterized by multifactorial interactions involving cerebrovascular injury, oxidative stress, neuroinflammation, and disturbances in iron metabolism. This review systematically elucidates the dual roles of iron homeostasis imbalance in VaD. Firstly, iron overload exacerbates neuronal injury by catalyzing free radical generation and activating the ferroptosis pathway. Secondly, vasculogenic pathologies, such as chronic hypoperfusion and disruption of the BBB, contribute to aberrations in iron metabolism, thereby perpetuating a vicious cycle of “vascular injury-iron accumulation-neurodegeneration.” Recent advancements in imaging techniques and biomarkers have introduced novel tools for the early diagnosis of VaD. Concurrently, iron chelators, antioxidants, and combined therapeutic strategies have demonstrated potential efficacy in preclinical models; however, their translation into clinical practice presents significant challenges. Ultimately, this article organizes and describes the relationship between VaD and iron metabolism abnormalities. The regulation of iron metabolism offers a novel perspective for the prevention and treatment of VaD, yet its clinical application necessitates interdisciplinary collaboration and technological innovation. To provide new insights and directions for future treatment whenever possible.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/ or publication of this article.
