Abstract
Objective:
To explore the correlation between cardiac iron deposition and left ventricular function indicators, such as left ventricular end-diastolic volume index, left ventricular end-systolic volume index, and left ventricular ejection fraction, and to evaluate the accuracy of predicting patients with cardiac iron deposition by using left ventricular ejection fraction as an index.
Methods:
This quantitative cross-sectional study involved one hundred and fifty transfusion-dependent patients with β-thalassemia major who were evaluated by magnetic resonance imaging to obtain T2* values, left ventricular end-diastolic volume index, left ventricular end-systolic volume index, and left ventricular ejection fraction. The relationship between cardiac R2* values and left ventricular end-diastolic volume index, left ventricular end-systolic volume index, and left ventricular ejection fraction was analyzed.
Results:
Out of the 150 patients, cardiac iron overload was not observed for 92 patients, 42 patients exhibited mild to moderate cardiac iron overload, and 16 patients were severe cardiac iron overloaded. A linear correlation was not observed between the cardiac R2* values and left ventricular end-diastolic volume index, left ventricular end-systolic volume index, and left ventricular ejection fraction (P > 0.05). For the left ventricular ejection fraction index, the sensibility, specificity, positive predictive value, negative predictive value, and the agreement rate were 83.3%, 63.2%, 8.6%, 98.9%, and 64.0%, respectively; also, the area under the receiver operating characteristic curve was 0.348.
Conclusions:
A linear correlation was not observed between cardiac R2* values and left ventricular end-diastolic volume index, left ventricular end-systolic volume index, and left ventricular ejection fraction in patients with β-thalassemia major. Therefore, using left ventricular ejection fraction as an indirect index to predict cardiac iron deposition may be not reliable in clinical practice.
Introduction
Thalassemia is a group of hereditary hemolytic anemias caused by partial or complete inhibition of α- or β-globin chain synthesis. Studies indicate a high frequency of thalassemia in the population of southern China, mainly south of the Yangtze River, leading to high mortality; it is also the most common cause of secondary hemochromatosis. 1 Patients with β-thalassemia major require repeated blood transfusions to extend their lives after birth, leading to iron deposits in tissues and organs (liver, heart, endocrine glands, etc.) and causing damage. Cardiomyopathy, due to iron deposition left ventricular function abnormalities, and heart failure are the main causes of death in patients with thalassemia major. 2 Therefore, the early diagnosis of cardiac iron deposits is important in treating and reversing cardiomyopathy. This is emphasized by the fact that once the deposited iron reaches a critical level, leading to abnormal cardiac function, the prognosis is often poor, even if iron chelation therapy is performed.
Monitoring heart function, especially the changes in left ventricular function before heart failure, is a common method to assess the risk of cardiac decompensation in patients. 3 Because of the absence of direct methods to measure cardiac iron concentration, the relationship between cardiac iron deposition and left ventricular function is unknown. Since studies have indicated a linear relationship between magnetic resonance imaging (MRI) relaxation rate R2* (1/T2*) and cardiac iron concentration, the use of MRI to quantify cardiac iron deposition has gradually increased; also, in recent years, more studies have focused on the evaluation of the efficacy of iron removal drugs.4–8 Some studies conducted outside China have explored the relationship between cardiac T2* or R2* values and cardiac function, and found that left ventricular function indicators will change, especially left ventricular ejection fraction (LVEF) will decrease when cardiac T2* <10 ms in thalassemia patients. However, the MRI-quantitated iron deposition evaluation has not yet been fully popularized and applied in mainland China. Presently, few research reports exist on iron overload in thalassemia patients in China.
Therefore, this study aims to explore the correlation between cardiac iron deposition and left ventricular function indicators (such as left ventricular end-diastolic volume index (LVEDVI), left ventricular end-systolic volume index (LVESVI), and LVEF) in patients with β-thalassemia major through the application of MRI-T2* and cardiovascular magnetic resonance (CMR) examination methods. This study also aims to explore the accuracy of the clinical use of LVEF as an index to predict cardiac iron deposition.
Materials and methods
Research materials
In this quantitative cross-sectional study, thalassemia patients belonging to Guangxi, Guangdong, Hainan, Jiangxi, and Sichuan provinces from July 2017 to June 2021 were included based on the following criteria: (1) genetically diagnosed as β-thalassemia major; and (2) history of at least 50 units of red blood cell transfusion before the examination and receiving at least 10 units/year of red blood cell transfusion. The exclusion criteria were (1) patients with congenital heart disease and other diseases that are not related to cardiomyopathy caused by iron deposition; (2) patients who have claustrophobia for MR examination or cannot cooperate with MR examination; and (3) patients equipped with pacemakers, strong magnetic metal implants, and other devices that are not approved for safe use in MRI. A total of 175 patients were recruited, of which one case was diagnosed with atrial defect occlusion after one year, and 24 cases had respiratory artifacts due to failure to hold their breath as required during the examination. Finally, 150 patients (96 males and 54 females) aged 4–25 years and with a mean age of 9.4 ± 3.6 years met the criteria.
This study was performed in accordance with the Helsinki Declaration of 1975 as revised in 2013. The study was reviewed and approved by the Ethics Committee of the First Affiliated Hospital of Guangxi Medical University (Aug 30.2022/No: 2022-KY-E-297). All patients (patients under the age of 18 or those who cannot understand the contents of this study were represented by their legal guardians) signed an informed consent form before enrolling in this study. We have de-identified all patient details, so that they may not be identified in any way (Figure 1).

Flowchart of this study.
The reporting of this study conforms to STROBE guidelines. 9
MRI protocol
In this study, a 1.5 T MR scanner (MAGNETOM Avanto Fit, Siemens Healthcare, Erlangen, Germany), body coil, and retrospective electrocardiogram (ECG) gating were used. Before the MRI examination, the patient was repeatedly trained in inhalation, exhalation, and apnea; the patient was asked to calm down and hold his/her breath at the end of the expiration during the scan. Firstly, the chest cross-sectional localization scanning was performed with Turbo FLASH sequence to locate the cross-sectional image; the left ventricular long-axis image parallel to the ventricular septum was obtained, after which the left ventricular short-axis image perpendicular to the ventricular septum was obtained. Then, the long- and short-axis images of the left ventricle parallel and perpendicular to the ventricular septum were located, and a single-breath-hold cine sequence was scanned to obtain continuous left ventricular short-axis cine images from the left atrial ventricular junction to the apex of the heart. The left ventricular outflow tract cine images were obtained by locating the aortic plane perpendicular to the short axis of the left ventricle and the long axis of the left ventricle parallel to the ventricular septum. The papillary muscle in the middle of the left ventricular septum was scanned by a multi-echo gradient-echo (GRE) sequence to obtain images based on which the cardiac T2* values were measured quantitatively. (1) Single breath-hold cine pulse sequence was: TE 1.28 ms, TR 45.3 ms, FOV 380 mm, and layer thickness 8 mm. (2) GRE scanning sequence was: TE 2.97, 5.54, 8.23, 10.92, 13.61, 16.3, 18.99, and 21.68 ms, TR 138 ms, FOV 400 mm, reversal angle 20°, basic matrix 256 × 256, and layer thickness 10 mm.
Measurement methods of LVEDVI, LVESVI, LVEF, and cardiac T2* value
CMRtools software (Cardiovascular Imaging Solutions, London, UK) was installed to input the original Digital Imaging and Communications (DICOM) data of left ventricular short-axis cine images, left ventricular outflow tract cine images, and central ventricular septum images on the short-axis of the heart scanned by multi-echo GRE sequence into the computer for post-processing. (1) Measurement of left ventricular function: The contour of the left ventricular endocardium and epicardium were manually depicted layer by layer in cardiac short-axis cine images (papillary muscle does not include blood pool); the minimum value of left ventricular volume was taken as the end of systole and the maximum value as the end of diastole; the positions of the mitral valve and aortic valve root in the cine images of left ventricular outflow tract were marked; and the LVEDV, LVESV, and LVEF were automatically calculated by the software. The LVEDV and LVESV were divided by body surface area (BSA) to obtain LVEDVI (mL/m2) and LVESVI (mL/m2). BSA = 0.0061 × height (cm) + 0.0128 × weight (kg)-0.1528. (2) Measurement of cardiac T2* value: the whole ventricular septal section was drawn along the endocardium as the region of interest (ROI) on the middle layer of the ventricular septum on the short axis of the heart scanned by multi-echo GRE sequence. The software then measured the cardiac T2* value (ms) and R2* value (1/s) = 1/T2* × 1000.
Diagnostic criteria
The diagnostic criteria for cardiac iron deposition on 1.5 T MRI was T2* < 20 ms. The T2* value of < 10 ms was considered to be the case with severe iron deposition and that of 10 ms ≤ T2* < 20 ms was mild to moderate iron deposition.4–6 The LVEF value of < 56% was clinically believed to have decreased left ventricular function. 8
Statistical methods
SPSS 20.0 statistical software package was used for statistical analysis. All patients were divided into three groups according to the cardiac T2* values: nil cardiac iron deposition, mild to moderate cardiac iron deposition, and severe cardiac iron deposition. According to the Kolmogorov-Smirnov test, the cardiac T2* value, LVEDVI, LVESVI, and LVEF of 150 patients did not accord with normal distribution; however, the cardiac T2* value, LVEDVI, LVESVI, and LVEF of each group accorded with normal distribution. Spearman's correlation analysis was used to explore the relationship between cardiac R2* value and LVEDVI, LVESVI, and LVEF in each group. One-way analysis of variance (ANOVA) was used to compare the differences of LVEDVI, LVESVI, and LVEF among the three groups of patients. The sensitivity, specificity, positive predictive value, negative predictive value, and diagnostic accuracy rate of the LVEF value < 56% for predicting cardiac iron deposition were calculated. Using T2* < 20 ms as the diagnostic criteria of cardiac iron deposition, the accuracy of predicting cardiac iron deposition with LVEF in 150 patients was evaluated, and the ROC curve was constructed. Statistical significance was defined as a P-value of < 0.05.
Results
Cardiac T2* value and the range of LVEDVI, LVESVI, and LVEF
The cardiac T2* and R2* values, and the distribution range of LVEDVI, LVESVI, and LVEF in 150 patients with thalassemia major are summarized in Table 1. Among them, 58 patients (38.7%) had cardiac iron deposition, and six patients (4.0%) had abnormal LVEF. Among the patients without cardiac iron deposition, one case (1/92) had abnormal LVEF. Whereas three cases (3/42) had abnormal LVEF in patients with mild to moderate cardiac iron deposition, also, among the patients with severe cardiac iron deposition, two cases (2/16) had abnormal LVEF. No significant difference was observed in LVEDVI, LVESVI, and LVEF among the three groups, that is, patients without cardiac iron deposition, mild to moderate cardiac iron deposition, and severe cardiac iron deposition (Table 1).
Cardiac T2*, R2* value and the range of LVEDVI, LVESVI, and LVEF in 150 patients.

Note: LVEDVI: left ventricular end-diastolic volume index; LVESVI: left ventricular end-systolic volume index; LVEF: left ventricular ejection fraction.
Relationship between cardiac R2* value and LVEDVI, LVESVI, and LVEF
A linear relationship or correlation was not observed between cardiac R2* value and LVEDVI, LVESVI, and LVEF in all the patients (Table 2, Figures 2 and 3).

(Panels A–C) Scatter plot of cardiac R2* value and LVEDVI, LVESVI, and LVEF in 150 patients, indicating the absence of a linear relationship between cardiac R2* value and LVEDVI, LVESVI, and LVEF.

(Panels A–C) The analysis results of a patient with β-thalassemia major: T2* = 8.9 ms (Panel A); left ventricular end-systolic volume = 25 mL (Panel B); and left ventricular end-diastolic volume = 79 mL (Panel C). (Panels D–F) The analysis results of the other patient with β-thalassemia major: T2* = 14.1 ms (Panel D); left ventricular end-systolic volume = 27 mL (Panel E); and left ventricular end-diastolic volume = 90 mL (Panel F).
Relationship between different degrees of cardiac iron deposition (R2* value) and LVEDVI, LVESVI, and LVEF.
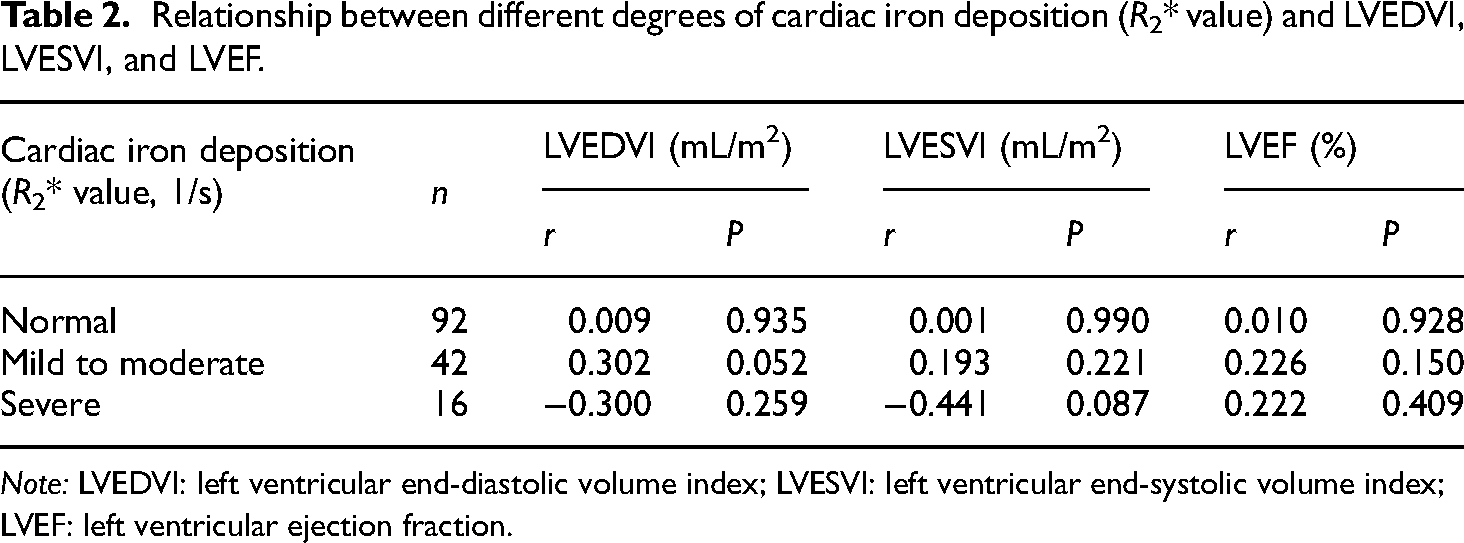
Note: LVEDVI: left ventricular end-diastolic volume index; LVESVI: left ventricular end-systolic volume index; LVEF: left ventricular ejection fraction.
The accuracy of using LVEF as an index to evaluate the cardiac iron deposition
When the LVEF value of < 56% was used to predict cardiac iron deposition, the sensitivity, specificity, positive predictive value, negative predictive value, and the accuracy rate were 83.3%, 63.2%, 8.6%, 98.9%, and 64.0%, respectively. The area under the ROC curve for predicting cardiac iron deposition based on LVEF was 0.348 (Figure 4).

ROC curve of LVEF predicting cardiac iron deposition in 150 patients.
Discussion
In thalassemia major, iron overload is a common result of multiple blood transfusions and an inappropriate increase in iron absorption associated with ineffective erythropoiesis. The outpouring of catabolic iron exceeded the iron-carrying capacity of transferrin, leading to the emergence of non-transferrin-bound iron (NTBI). NTBI is preferentially cleared by the liver and myocardium at a rate 200 times higher than transferrin iron. NTBI catalyzes the formation of free radicals, leading to oxidative stress and damage to mitochondria, lysosomes, lipid membranes, proteins, and DNA. Long-term iron toxicity can lead to cardiomyopathy. 10 Therefore, in thalassemia, both iron overload and chronic iron radical exposure are blamed in cardiomyopathy etiology. Since the strengthening of iron chelation therapy before cardiac dysfunction can reverse cardiomyopathy caused by early iron deposition, timely diagnosis of cardiac iron deposition in patients with thalassemia can prevent heart failure. The typical symptoms of heart failure appear in the late stage, so the clinical diagnosis is often delayed. In this context, quantitative cardiac iron deposition evaluation with MRI is an accurate, convenient, and non-invasive method. Also, its emergence challenges the clinical method of predicting cardiac iron deposition through the changes in cardiac function.
In our study, a correlation was not observed between the cardiac R2* value and LVEDVI, LVESVI, and LVEF of the patients. A previous study demonstrated nil correlation between the two in patients without cardiac iron deposition and a low to moderate correlation between the two when cardiac T2* < 20 ms, 11 which is different from the results of our study. In our study, no difference was observed in end-diastolic volume index (EDVI) and end-systolic volume index (ESVI) between patients without cardiac iron deposition and patients with different degrees of cardiac iron deposition; also, the degree of iron deposition had no linear relationship with EDVI and ESVI. These results suggest that abnormal left ventricular diastolic or systolic function cannot effectively reflect the iron load of the myocardium. The reason may be that iron deposition causes damage to cardiomyocyte function, and after a certain time, LVEDV and LVESV gradually appear abnormal, that is, iron deposition occurs earlier than the reduction in cardiac function. Therefore, these results do not indicate a parallel relationship between the two at this stage. For ischemic cardiomyopathy caused by iron deposition, the change of diastolic function is the most sensitive early indicator of left ventricular function. Since abnormal left ventricular diastolic function usually occurs before abnormal systolic function, 12 some studies believe that abnormal diastolic function can be used as an early indicator for the diagnosis of cardiac iron deposition to guide the clinical adjustment of iron chelation therapy. 13 However, in our study, the abnormal left ventricular diastolic function did not reflect the presence of iron deposition in the myocardium, thereby indicating that this index is not helpful for clinical diagnosis. For patients with cardiac iron deposition, the increase of LVESV is usually regarded as a precursor of subclinical left ventricular dysfunction. 14 Patients with chronic ischemic anemia are often in a state of high cardiac output, thereby increasing left ventricular volume; however, the left ventricular systolic function often drops suddenly, showing abnormal function, and it is difficult to reflect the degree of cardiac iron deposition. Since the abnormalities of LVEDV and LVESV occur later than cardiac iron deposition, an earlier diagnosis of cardiac iron deposition is possible by MRI-T2* than the clinical prediction of left ventricular dysfunction.
Studies conducted outside mainland China reported that the LVEF decreased with a decrease in cardiac T2* value, especially when the cardiac T2* value was < 10 ms.11,15–19 However, we did not observe any correlation between cardiac R2* and LVEF in thalassemia patients with or without cardiac iron deposition. The reason may be that when iron deposition in the myocardium (cardiac T2* value decreases) increases without causing cardiomyocyte injury, the cardiac ejection fraction remains normal. When increased iron deposition leads to cardiomyocyte injury and dysfunction, the patient gradually presents the clinical symptoms of heart failure. That is, when the cardiac T2* value is < 20 ms, it indicates that cardiac function damage has begun, while the decrease of LVEF appears in the late stage of the iron deposition only. Some studies have found that patients with thalassemia with normal LVEF might also have cardiac iron deposition, leading to heart failure if left untreated,20,21 which is in accordance with our study. For such patients, heart failure could be avoided if their iron chelation regimen was optimized.20,21 Results obtained in this study, like the majority of the patients with moderate and severe cardiac iron deposition, had normal LVEF (14/16) and that the degree of iron deposition was not related to LVEF are consistent with the research results of some Chinese scholars. 7 This may be related to the treatment status of patients with thalassemia in China. Due to economic reasons, most patients in China receive blood transfusion treatment only, without receiving regular iron chelation therapy. Therefore, the age of cardiac iron deposition is generally lower than that of patients in developed countries, and heavy iron overload in the myocardium occurs in a relatively short time. At this time, cardiomyocyte injury may not lead to abnormal function, so the increase in iron deposition is inconsistent with the decrease in LVEF. In our study, the area under the ROC curve for predicting cardiac iron deposition with LVEF was 0.348, and the correct rate of predicting cardiac iron deposition with LVEF < 56% was 64.0%, indicating that the diagnostic efficiency of LVEF is low. In patients with thalassemia major, LVEDV increases with a decrease in LVESV, leading to an increase in ejection fraction. It has been found that the lower limit of LVEF measured by MRI (male > 59% and female > 63%) in patients with thalassemia major without cardiac iron deposition was higher than that of the normal control group. 22 Therefore, the clinical use of 56% as the lower limit of abnormal ejection fraction to predict the presence of cardiac iron deposition may reduce the positive predictive value of diagnosis.
Our study analyzed the correlation between cardiac iron deposition and a few indexes of left ventricular function in patients with thalassemia major and the accuracy of the clinical use of LVEF to predict cardiac iron deposition. Our study has some limitations. First, our results showed that the number of patients with severe cardiac iron deposition is relatively small compared with the other two groups, and there may be an imbalance between groups. Second, our study did not conduct subgroup analyses based on different iron chelation therapy methods. This is because the patients we studied did not use the same medication alone for a long period of time during their long-term iron chelation therapy, and were treated with deferiprone, deferoxamine, and deferasirox at different stages. Some patients were treated irregularly, and there may be an insufficient dosage of iron-chelating drugs. We believe that with the further improvement of China's economy in the future, iron chelation therapy for thalassemia patients will be more standardized. In our future study, we will conduct subgroup analyses according to different iron chelation therapy methods. Third, our results are horizontal studies of patients conducted at the same time point. Therefore, the relationship between the changes in LVEF and cardiac iron deposition in the same patient at different time points needs to be further explored.
In conclusion, cardiac iron deposition in patients with thalassemia may not be related to left ventricular function indicators, such as LVEDVI, LVESVI, and LVEF. Also, LVEF may not be an effective predictor of cardiac iron deposition. Quantitative cardiac iron deposition evaluation with MRI is an accurate, noninvasive, and reproducible method. Since effective removal of excessive iron in the myocardium strengthens the line of treatment, measuring cardiac T2* value by MRI may help diagnose cardiac iron overload early and provide the basis for timely clinical treatment. It is suggested that MRI quantification should be included in the routine care of thalassemia patients.
Footnotes
Acknowledgements
We sincerely thank all the participants of our study. We thank Cheng Tang, Wenmei Li, and Junli Liang for their technical support in MRI scanning. We thank Gaohui Yang for his guidance on writing and his contribution. We thank the Department of Hematology and Pediatrics of the First Affiliated Hospital of Guangxi Medical University for their support.
Author contributions
Peng Peng contributed to the study's conception and design. Material preparation and data collection were performed by Shuai Peng, Fengming Xu, Guangxin Yang, Yanyan Zhang, Qin Tang, and Rongrong Liu. Data analyses were performed by Shuai Peng, Fengming Xu, and Guangxin Yang. The first draft of the article was written by Shuai Peng, and all authors commented on previous versions of the article. All authors read and approved the final article.
Consent to participate
All patients (or parents/guardians) gave written informed consent to participate in the study.
Consent to publish
Data availability statement
The dataset used in support of the findings of this study is available from the corresponding author at email address upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This study was performed in accordance with the Helsinki Declaration of 1975 as revised in 2013. The study was reviewed and approved by the Ethics Committee of the First Affiliated Hospital of Guangxi Medical University (Aug 30.2022/No: 2022-KY-E-297). All patients (patients under the age of 18 or those who cannot understand the contents of this study were represented by their legal guardians) signed an informed consent form before enrolling in this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundations of China (Nos. 81760305 and 81641066), advanced Innovation Teams and Xinghu Scholars Program of Guangxi Medical University, and NHC Key Laboratory of Thalassemia Medicine and Guangxi Key Laboratory of Thalassemia Research.
