Abstract
Objectives
To evaluate the association between systemic inflammatory response syndrome (SIRS) and 30-day mortality in critically ill cirrhosis patients with acute kidney injury (AKI).
Methods
This retrospective cohort study utilized the Medical Information Mart for Intensive Care-IV database. Multivariable Cox proportional hazard regression, with covariates selected by Least Absolute Shrinkage and Selection Operator regression, was employed to assess the association. Model performance was evaluated with the Brier score, and the additive predictive value of SIRS to the model for end-stage liver disease (MELD) and sequential organ failure assessment (SOFA) scores was compared using the DeLong test.
Results
Among the 1797 enrolled patients, the 30-day mortality rate was 38.23% (n = 687). A higher SIRS was independently associated with increased 30-day mortality (adjusted hazard ratio: 1.31; 95% confidence interval: 1.20–1.43). This association remained consistent across subgroups stratified by age, gender, albumin infusion, sepsis, AKI stage, and etiology (all p < 0.05). The predictive performance for mortality was significantly improved when SIRS was combined with MELD or SOFA scores compared to each score alone [areas under the curve: SIRS + MELD vs. MELD, 0.711 vs. 0.697; SIRS + SOFA vs. SOFA, 0.635 vs. 0.617].
Conclusion
An elevated SIRS score was associated with a higher risk of short-term mortality in critically ill cirrhosis patients with AKI. As an easily obtainable bedside metric, SIRS represents a valuable tool for identifying high-risk patients, potentially enabling timely clinical interventions.
Keywords
Introduction
Liver cirrhosis represents a terminal stage of chronic liver disease, characterized by fibrosis and regenerative nodules that disrupt hepatic architecture and function.1,2 A critical complication in these patients is acute kidney injury (AKI), with its prevalence ranging alarmingly from 20% to 60% among hospitalized individuals.3–6 The development of AKI signifies a drastic worsening of prognosis, with studies reporting a tenfold increase in 30-day mortality compared to cirrhotic patients without AKI. 7 This grim statistic underscores the urgent need for early identification of modifiable risk factors to improve stratify high-risk patients and guide timely interventions.
The pathogenesis of AKI in cirrhosis involves both hemodynamic alterations2,3 and systemic inflammation.8,9 Systemic inflammatory response syndrome (SIRS), a clinical phenotype reflecting dysregulated inflammation, is highly prevalent in this population. 10 Defined by readily available parameters like temperature, heart rate, respiratory rate, and white blood cell count (WBC).11,12 SIRS has been consistently associated with the development and progression of AKI in patients with cirrhosis 13 and predicts both AKI progression and 90-day mortality in severe alcoholic hepatitis, 14 indicating its central role in organ failure pathophysiology.
However, evidence regarding its impact on short-term mortality specifically in cirrhotic patients with established AKI remains limited—a critical knowledge gap driven this high-risk populations’ need for accurate prognostication. While current prognostic models like the model for end-stage liver disease (MELD) 15 and sequential organ failure assessment (SOFA) scores 16 are valuable, they may not fully capture systemic inflammation's contribution to mortality.
Notably, despite the emergence of qSOFA and Sepsis-3 criteria, SIRS retains relevance in the context of cirrhosis. The components of SIRS are objective, routinely measured, and avoid the confounding influence of hepatic encephalopathy on the neurological assessment required by qSOFA. SIRS continues to be a validated prognostic tool specifically in hepatology research, capturing the spectrum of systemic inflammation that portends poor outcomes in cirrhotic patients, irrespective of a confirmed infectious etiology.10,13,14 Furthermore, SIRS as a simple bedside assessment of acute inflammatory status 17 offers a potential complementary tool. Emerging evidence suggests its prognostic utility in hepatorenal syndrome,18,19 but its additive value to established scores like MELD and SOFA for predicting short-term mortality in a broad cohort of cirrhotic patients with AKI is not yet clearly defined. Filling this knowledge gap could significantly enhance risk stratification, allowing for more personalized and aggressive management in this vulnerable population. Therefore, this study aimed to: 1) definitively assess the association between SIRS and 30-day mortality in critically ill cirrhotic patients with AKI, and 2) evaluate whether integrating SIRS with traditional prognostic models (MELD and SOFA) improves the predictive accuracy for short-term mortality. By leveraging the comprehensive Medical Information Mart for Intensive Care-IV (MIMIC-IV) database, we sought to provide robust evidence for SIRS as a practical and valuable prognostic tool in this clinical setting.
Methods
Sources of data
The data for this retrospective cohort study were obtained from the MIMIC-IV database (version 3.0; https://mimic.mit.edu/docs/iv/). MIMIC is a large, publicly available intensive care unit (ICU) database containing detailed clinical information for over 70,000 ICU patients admitted between 2008 and 2022. It includes demographics, laboratory results, medication records, vital signs, treatment details, and other clinical variables. Leveraging the comprehensive data available in MIMIC-IV, the investigators were able to accurately assess the SIRS status in cirrhosis patients with AKI and to quantify the 30-day mortality risk using the MELD, MELD combined with serum sodium serum sodium (MELD-Na), 20 and SOFA scores. The analysis of this de-identified public database was approved by the Institutional Review Board of the Second Affiliated Hospital of Harbin Medical University, which granted an exemption from full review and waived the requirement for individual patient informed consent. All procedures were conducted in accordance with the ethical standards of the Helsinki Declaration (1975, as revised in 2024). The reporting of this study follows the Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 21
Study population
Participants in this retrospective cohort study were enrolled in 2008 to 2022. Cases of cirrhosis was identified based on International Classification of Diseases (ICD)-9 22 (5712, 5715, and 5716) and ICD-10 (K703, K743, K744, K745, and K746). 23 Specifically, codes 5716, K743, K744, and K745 were classified as cholestatic cirrhosis; 5712 and K703 were classified as alcoholic cirrhosis; 5715 and K746 as other or unspecified cirrhosis. AKI was identified according to the criteria set by the Kidney Disease Improving Global Outcomes (KDIGO).5,24 Diagnosis required meeting at least one of the following: an increase in serum creatinine (SCr) by 0.3 mg/dL within 48 h, a rise to 1.5 times the baseline SCr level within the past 7 days, or a urine volume of less than 0.5 mL/kg/hour for 6 h.
For this study, patient records were extracted from the MIMIC-IV database using the following inclusion criteria: (1) age ≥18 years; (2) diagnosis of cirrhosis-associated AKI at ICU admission; (3) an initial ICU stay for no less than 24 h. Given the challenge of precisely differentiating the etiology of AKI at initial ICU admission and to reflect the real-world scenario of early risk assessment, our study included all cirrhotic patients with AKI meeting the KDIGO criteria. Patients were excluded if they had missing critical data [SIRS, international normalized ratio (INR), creatinine, bilirubin] or survival information. As the MIMIC-IV database contains de-identified health information, this analysis was deemed exempt from informed consent by the Institutional Review Board of the Second Affiliated Hospital of Harbin Medical University.
Covariates and definitions
In this study, a range of potential confounding variables were collected, including demographic information (age, gender, race, insurance, and marital status), clinical characteristics (ICU type, etiology, AKI stage, sepsis status, vasopressor use, mechanical ventilation, liver transplantation therapy, renal replacement therapy, antibiotic administration, use of diuretics, and albumin infusion), laboratory measurements [mean blood pressure (MBP, mmHg), hemoglobin (g/dL), platelet (K/uL), saturation of peripheral oxygen (SpO2, %), red cell distribution width (RDW, %), prothrombin time (PT, sec), blood urea nitrogen (mg/dL), estimated glomerular filtration rate (eGFR, mL/min/1.73 m2), glucose (mg/dL), anion gap (mEq/L), urine output (mL, defined as the cumulative urine output during the first 24 h following initial ICU admission), aspartate aminotransferase/alanine aminotransferase (AST/ALT), and alkaline phosphatase (ALP, IU/L)], and clinical score systems [SOFA, MELD-Na, and Charlson comorbidity index].
The SOFA score (ranging from 0 to 24) and SIRS status (ranging from 0 to 4) were determined based on physiological and laboratory parameters recorded during the initial 24 h following ICU admission. Antibiotic use was extracted from the “antibiotics” category within chart events, including agents such as ampicillin, nafcillin, oxacillin, penicillin, piperacillin, azithromycin, vancomycin, clindamycin, daptomycin, erythromycin, tobramycin, gentamycin, gentamicin, amikacin, ciprofloxacin, levofloxacin, moxifloxacin, amphotericin, doxorubicin, cefazolin, cefepime, ceftazidime, and ceftriaxone. Sepsis was defined as the presence of documented or suspected infection accompanied by an acute increase in total SOFA score of 2 points or more. Diuretics use was identified using item IDs 228340, 221794, and 229639. Albumin infusions were captured using item IDs 220863, 220861, 220862, and 220864. Liver transplantation was identified via ICD-10 procedure codes 0FY00Z0, 0FY00Z1, and 0FY00Z2.
MBP was calculated by SBP +
Definition of systemic inflammatory response syndrome and outcome
The criteria for SIRS were as follows11,25: (1) tachycardia (heart rate >90 beats/min); (2) tachypnea or hyperventilation (respiratory rate >20 breaths/min or PaCO2 < 32 mmHg); (3) fever or hypothermia (a body temperature >38 or <36 °C); (4) leukocytosis, leukopenia or bandemia (WBC >12 × 109/L, <4 × 109/L or bandemia ≥10%).
The outcome of this study was 30-day mortality following ICU admission. Participants were tracked from the time of ICU entry until either death or the end of the 30-day follow-up period, whichever occurred first. Survival status was determined using hospital records and national social security database. For patients who died within 30 days, follow-up was terminated at the data of death.
We evaluated systemic inflammatory response using the SIRS diagnostic criteria and performed stratified analysis based on the number of fulfilled SIRS criteria, rather than treating them as a weighted scoring system. The SIRS assessment incorporated physiological and laboratory parameters collected during the initial 24-h ICU admission period. Patients were categorized by the number of SIRS criteria met to investigate associations with 30-day mortality. In statistical analysis, SIRS was incorporated as a stratified categorical variable within Cox proportional hazards regression to calculate hazard ratio (HR) for 30-day mortality, with adjustment for relevant confounding factors to ensure robust effect estimation.
Statistical analysis
Continuous variables with normal distribution were expressed as mean ± standard deviation (SD), and group comparisons were performed using independent samples t-test. Non-normally distributed continuous variables were summarized as median and interquartile range [M (Q1, Q3)], and the Mann–Whitney U test was applied for between-group comparisons. Categorical variables were reported as frequencies and percentages [n (%)], and differences among groups were assessed using the chi-square test or rank sum test, as appropriate.
Table S1 exhibited the proportion of missing data of all covariates considered. Variables with more than 5% missing values were excluded, while those with less than 5% missingness were imputed using predictive mean matching. 26 No substantial differences were observed before and after imputation; results of the imputation process are summarized in Table S2.
To avoid overfitting and to identify the most informative predictors, we employed a two-step variable selection process: first, covariates with a p-value <0.05 in the univariate Cox analysis (Table S3) were selected. Second, these variables were entered into a Least Absolute Shrinkage and Selection Operator (LASSO) Cox regression model. The optimal penalty parameter (λ) was determined via 10-fold cross-validation based on the minimum criteria. The variables retained by the LASSO algorithm were then included in the final multivariate Cox proportional hazards model (Model 2). The performance of the final prediction model was assessed using the Brier score. The variable selection process of the LASSO regression, including the coefficient profile and cross-validation, is visualized in Figure S1. The regression coefficients (Beta) for the variables retained in the final model are presented in Table S4. Model 1 was a crude model, and model 2 was adjusted for age, MELD-Na, RDW, PT, anion gap, urine output, vasopressor use, and liver transplantation. Subgroup analyses were further conducted based on age, gender, albumin infusion, antibiotic use, cirrhosis type, and AKI stage. HRs and 95% confidence intervals (CIs) were reported to quantify associations. The DeLong test was used to compare the areas under the curves (AUC) of different models to evaluate differences in their predictive abilities. 27 All statistical tests were two-sided, and a p-value <0.05 was considered statistically significant. Data cleaning, missing value handling, and statistical analysis were performed using SAS 9.4 (SAS Institute Inc., Cary, NC, USA). R version 4.3.2 was used for predictive model evaluation.
Results
General characteristics of participants
Figure 1 illustrates the selection process of the patients. Initially, 2411 participants were recruited from the MIMIC database. After applying the inclusion and exclusion criteria, 1797 were included in the final analysis. As summarized in Table 1, the mean age of the included patients was 58.09 ± 12.37 years. The majority of participants were male (65.28%), presented with AKI stage 2 or 3 (77.35%), and had alcoholic cirrhosis (53.98%). Among all enrolled participants, the M (Q1, Q3) of SIRS was 3.00 (2.00, 3.00), and the overall 30-day mortality was 38.23%. Significant differences were observed between the survival and non-survival groups at 30 days in several variables, including age, SIRS, MELD-Na, SOFA, AST/ALT, ALP, SpO2, eGFR, and glucose levels (all p < 0.05).

Flow chart of selecting eligible patients. AKI: acute kidney injury; ICU: intensive care unit; MIMIC-IV: Medical Information Mart for Intensive Care-IV; SIRS: systemic inflammatory response syndrome; INR: international normalized ratio.
Baseline characteristics of the study population stratified by 30-day mortality.
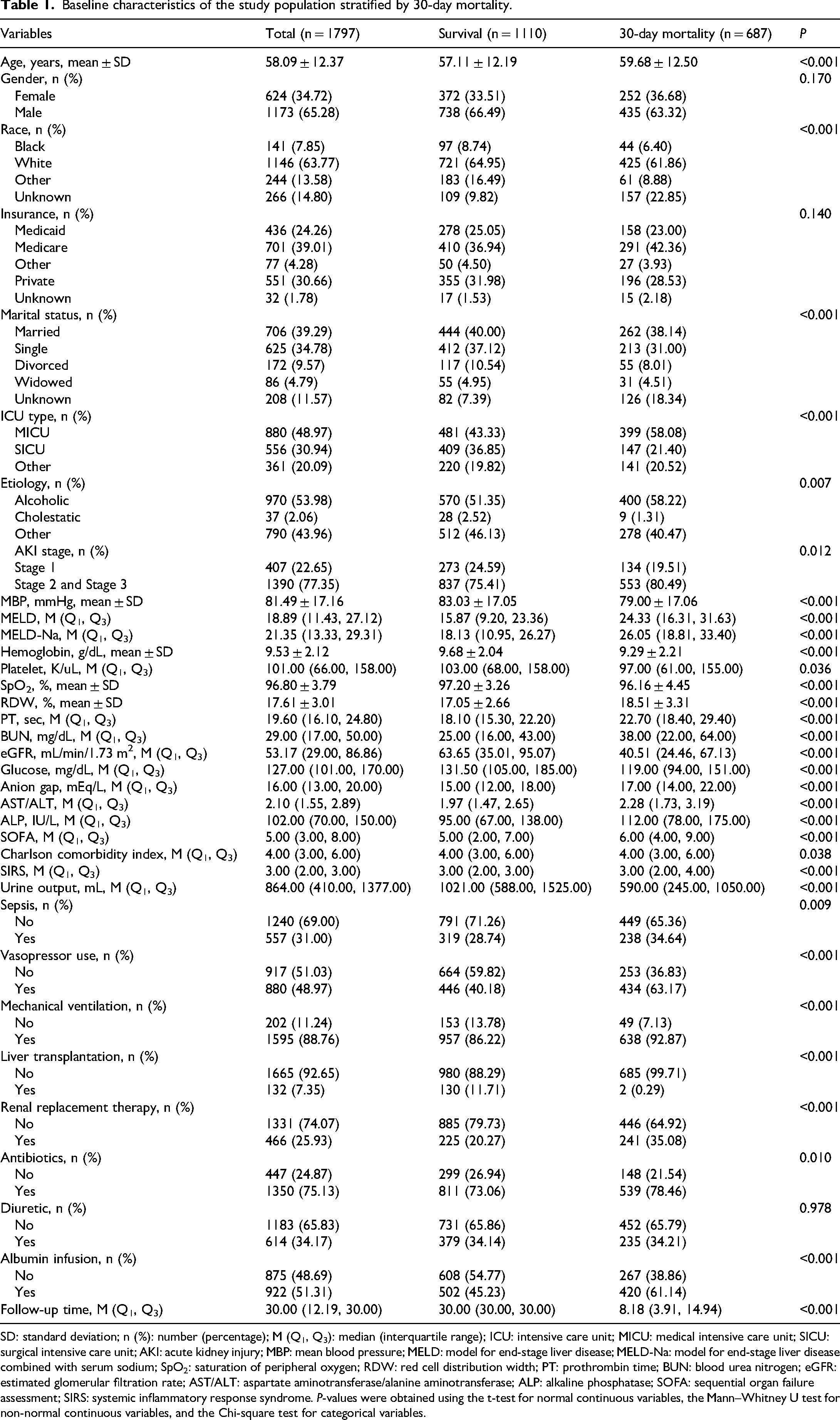
SD: standard deviation; n (%): number (percentage); M (Q1, Q3): median (interquartile range); ICU: intensive care unit; MICU: medical intensive care unit; SICU: surgical intensive care unit; AKI: acute kidney injury; MBP: mean blood pressure; MELD: model for end-stage liver disease; MELD-Na: model for end-stage liver disease combined with serum sodium; SpO2: saturation of peripheral oxygen; RDW: red cell distribution width; PT: prothrombin time; BUN: blood urea nitrogen; eGFR: estimated glomerular filtration rate; AST/ALT: aspartate aminotransferase/alanine aminotransferase; ALP: alkaline phosphatase; SOFA: sequential organ failure assessment; SIRS: systemic inflammatory response syndrome. P-values were obtained using the t-test for normal continuous variables, the Mann–Whitney U test for non-normal continuous variables, and the Chi-square test for categorical variables.
Association of systemic inflammatory response syndrome with 30-day mortality in cirrhosis patients with acute kidney injury
The LASSO Cox regression analysis identified eight predictors for the multivariate model: age, MELD-Na, RDW, PT, anion gap, urine output, vasopressor use, and liver transplantation. The process of variable selection is detailed in Figure S1, and the coefficients are listed in Tables S3 and S4.
As summarized in Table 2, a higher SIRS score was significantly associated with an increased risk of 30-day mortality after adjusting for these covariates identified by LASSO (Model 2: adjusted HRs = 1.31, 95%CI: 1.20–1.43, p < 0.001). The overall predictive performance of the Model 2 for 30-day mortality was assessed, yielding a Brier score of 0.1745, which indicates a good level of predictive accuracy. Consistent with these findings, Figure 2 demonstrates a graded increase in the risk of 30-day mortality with a higher SIRS level, further supporting its role as an independent predictor of poor outcome in this patient population.
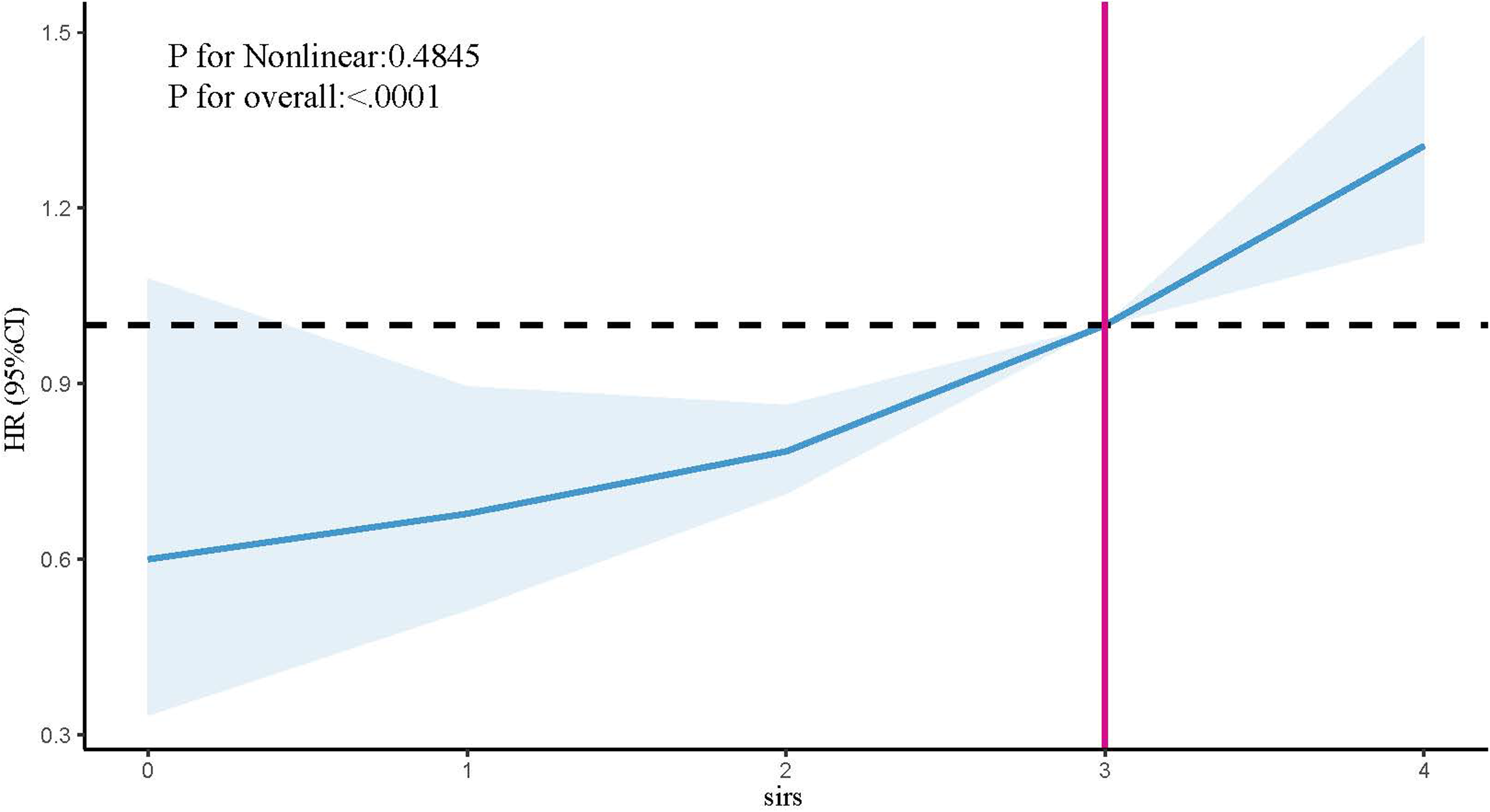
RSC of the association between SIRS and 30-day mortality in cirrhotic patients with AKI. RCS: restricted cubic splines; SIRS: systemic inflammatory response syndrome; AKI: acute kidney injury; HRs: hazard ratios; CI: confidence interval.
Association between SIRS score and 30-day mortality in critically ill cirrhosis patients with AKI.

Model 1: adjusted for none.
Model 2 (adjusted for variable selected by LASSO regression): adjusted for age, MELD-Na, RDW, PT, anion gap, urine output, vasopressin use, and liver transplantation.
HRs: hazard ratios; CIs: confidence intervals; SIRS: systemic inflammatory response syndrome; AKI: acute kidney injury; MELD: model for end-stage liver disease; RDW: red cell distribution width; PT; prothrombin time; LASSO: Least Absolute Shrinkage and Selection Operator.
Systemic inflammatory response syndrome and 30-day mortality in cirrhosis patients with acute kidney injury in different groups
Subgroup analyses were conducted to assess the consistency of the association between SIRS and 30-day mortality across various patient characteristics. As shown in Figure 3, a significant and positive association was consistently observed in most predefined subgroups, including those stratified by age, gender, albumin infusion, sepsis status, AKI stage, and etiology of cirrhosis (all p < 0.05).

Subgroup analyses for the association of SIRS with 30-day mortality in cirrhotic patients with AKI. SIRS: systemic inflammatory response syndrome; AKI: acute kidney injury.
Notably, a significant association was found for antibiotic use (p < 0.05). The positive association between SIRS and 30-day mortality was significant in the subgroup that received antibiotics (HRs: 1.29; 95%CIs, 1.16–1.43).
The predictive value of systemic inflammatory response syndrome for 30-day mortality in cirrhosis patients with acute kidney injury
Table 3 indicates that the combination of SIRS with MELD or SOFA was associated with a statistically significant, albeit modest, improvement in predicting 30-day mortality compared to each score alone (all p < 0.05; SIRS + MELD vs. MELD, 0.711 vs. 0.697; SIRS + SOFA vs. SOFA, 0.635 vs. 0.617; SISR + MELD vs. MELD-Na, 0.711 vs. 0.691.
The predictive value of different models on 30-day mortality in cirrhotic patients with AKI.

AKI: acute kidney injury; Ref.: reference; SIRS: systemic inflammatory response syndrome; MELD: model for end-stage liver disease; MELD-Na: model for end-stage liver disease combined with serum sodium; SOFA: sequential organ failure assessment; AUC: areas under the curve.
Discussion
In this retrospective cohort study, we observed a significant association between higher levels of SIRS and increased 30-day mortality among cirrhosis patients with AKI. Our findings confirm and extend previous observations by establishing SIRS may be an independent predictor of mortality in a large, broad cohort of critically ill cirrhotic patients with AKI. Furthermore, we suggested that SIRS provides statistically significant incremental prognostic value to established scores like MELD and SOFA, offering a readily available tool for enhancing early risk stratification in this vulnerable population.
As one of the most common and serious complications in patients with cirrhosis, AKI is associated with a high mortality rate.5,12,28,29 The degree of inflammation has been reported to be critical in the pathogenesis and short-term mortality of cirrhotic organ failure, including renal failure.17,30 Cirrhosis patients exhibit increased susceptibility to SIRS, likely due to exacerbated inflammation during acute injury from both infectious and non-infectious (e.g. alcoholic hepatitis) insults, coupled with impaired immune responses.31–33 Recently, a growing number of studies have shown that SIRS is associated with the development of AKI and poor prognosis in patients with cirrhosis. Maiwall et al. 14 investigated 365 SAH cases, finding that baseline SIRS on admission can predict the development of AKI and 90-day mortality. In this study, we observed a correlation between SIRS and 30-day mortality. Consistent with our results, Wong et al. 13 analyzed data from 2297 cirrhosis ascites patients, finding that the development of stage 2 or 3 AKI was associated with systemic inflammatory response at admission and correlated with reduced 30-day transplant-free survival. Another study assessed outcomes in acute chronic liver failure, identifying SIRS as a predictor of persistent AKI, which is associated with higher in-hospital mortality. 34 In a study conducted in 84 hepatorenal syndrome-AKI (HRS-AKI) patients treated with terlipressin-albumin, the presence of SIRS enhanced the renal response to terlipressin, leading to improved short-term graft-free survival and reduced need for renal replacement therapy. 18 These findings suggest that SIRS can serve as a key prognostic factor for cirrhosis patients with renal diseases (particularly AKI), especially in terms of short-term mortality. Effectively preventing and treating SIRS may serve as a viable strategy to reduce mortality in cirrhosis patients with AKI. 35
In addition, subgroup analyses of patients with different characteristics in our study showed that higher SIRS was associated with higher 30-day mortality in various subgroups, including age, sex, albumin perfusion, sepsis, AKI stage, and etiology. The consistent results of subgroup analyses also demonstrated that SIRS and 30-day mortality were similarly associated across most populations, emphasizing the stability and reliability of the association between SIRS and 30-day mortality. However, when stratified by antibiotic use, this association was only observed in the subgroup receiving antibiotics. Although existing literature has reported a correlation between antibiotic use and mortality, 36 the primary objective of our study was not to investigate the relationship between antibiotic use and SIRS-mortality relationships, we could not comprehensively explore potential confounding factors. Notably, variables such as infection types, antibiotic types, and administration timing might critically influence this association. Therefore, we recommend focused investigations to elucidate potential mechanisms and clinical implications underlying this observation in future research.
Utilizing the SIRS criteria in our cohort was based on the following considerations: first, the components of SIRS are objective, routinely collected, and devoid of the confounders that affect tools like qSOFA in cirrhosis (e.g. hepatic encephalopathy); second, a substantial body of evidence in hepatology continues to support the prognostic value of SIRS in capturing the deleterious systemic inflammatory state that drives outcomes in cirrhotic patients, with or without overt infection.10,13,14 It is crucial to note that SIRS is a measure of the systemic inflammatory response itself, which can be triggered by both infectious and non-infectious processes. In the context of cirrhosis, severe alcoholic hepatitis is a classic example of a potent non-infectious driver of SIRS. Therefore, SIRS is best interpreted as a marker of the deleterious systemic inflammatory state that drives outcomes in cirrhotic patients, with or without overt infection. This non-specificity is, in fact, advantageous for our purpose of broadly capturing the overall inflammatory burden, which is a key determinant of mortality. Moreover, our study introduces a simple method to enhance the prediction of mortality risk in cirrhosis patients with AKI. It is important to acknowledge that the increase in AUC observed when adding SIRS to established scores (MELD and SOFA), while statistically significant, was modest. However, the clinical relevance of a prediction toll is not solely determined by the magnitude of the AUC improvement. The SISR score is derived from routine, readily available bedside parameters that incur no additional cost or time. Therefore, even a modest gain in predictive accuracy, achieved without the need for novel biomarkers or complex calculations, may be non-trivial. It could aid in reinforcing clinical intuition and improving risk stratification, particularly in environments with limited resources where advanced testing is not immediately available. Future research should focus on identifying specific patient subgroups or clinical scenarios where the addition of SIRS provides the greatest practical utility.
It must be recognized that there are still some limitations. First, the admission diagnoses documented in the MIMIC-IV database encompass diverse and clinically complex presentations. To mitigate potential bias arising from incorporating and analyzing these heterogeneous admission reasons, they were intentionally not subjected to detailed categorization. Subsequent research could explore the influence of admission etiologies on clinical outcomes, particularly in evaluating their prognostic significance for mortality risk prediction and enhancing risk stratification strategies. Secondly, our study is limited by the lack of specific inflammatory biomarkers. As detailed in Table S1, procalcitonin was not recorded, and CRP was unavailable for over 98% of our cohort. Although lactate was more frequently measured (83.79% completeness), its inclusion alone would not have sufficed for a comprehensive adjustment of the inflammatory state. This absence prevents us from comparing the prognostic value of SIRS against these biomarkers or from understanding the precise inflammatory milieu. Consequently, SIRS, as a composite of readily available clinical signs, should be interpreted as a practical but non-specific index for systemic inflammation in this context. The robust association we observed between SIRS and mortality, despite this limitation, mya underscore its utility in real-world settings where such specialized tests are not immediately available. Future prospective studies designed to include these biomarkers are essential to delineate their specific roles and interactions with the clinical SIRS criteria. Third, although we adjusted for a wide range of confounders and performed subgroup analyses by cirrhosis etiology, we could not differentiate the specific types of AKI [e.g. prerenal azotemia, HRS-AKI, acute tubular necrosis] due to the limitations of the database. The prognosis and response to therapy may differ among these subtypes. Future studies with more detailed clinical data are warranted to validate the prognostic values of SIRS within each specific AKI subtype. Fourth, this study has limitations inherent to its retrospective design. The data for SIRS and AKI were assessed at ICU admission. Therefore, while we demonstrate a significant and independent association, we cannot definitively establish the exact temporal sequence or a causal relationship between SIRS and AKI from our data. It remains possible that the severity of AKI influences the SIRS status, or that both are driven by a common underlying precipitant. However, our findings suggest that the SIRS score, easily obtainable at the bedside within the first 24 h of ICU care, serves as a powerful early prognostic marker for subsequent 30-day mortality in this critically ill population. Finally, although we used LASSO regression to enhance model stability and provided a Brier score for calibration, external validation of our predictive model in an independent cohort is still warranted. Therefore, future studies should integrate more databases with complete inflammatory indicators to further validate the role of these parameters in short-term mortality prediction for patients with cirrhosis complicated by AKI, thereby enhancing the comprehensiveness and clinical applicability of research findings.
Conclusion
In this retrospective cohort study, an elevated SIRS was associated with an increased risk of short-term mortality in critically ill cirrhosis patients with AKI. Our findings suggest that SIRS score may severe as a simple and practical prognostic tool. Furthermore, integrating the SIRS score with established scoring systems like MELD or SOFA may improve risk stratification for 30-day mortality in this patient population. These results highlight the potential clinical relevance of systemic inflammation and warrant further investigation into whether targeting these pathways could improve outcomes.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251387229 - Supplemental material for The association of systemic inflammatory response syndrome with 30-day mortality in critically ill cirrhosis patients with acute kidney injury: An analysis of the Medical Information Mart for Intensive Care-IV database
Supplemental material, sj-docx-1-sci-10.1177_00368504251387229 for The association of systemic inflammatory response syndrome with 30-day mortality in critically ill cirrhosis patients with acute kidney injury: An analysis of the Medical Information Mart for Intensive Care-IV database by Jia Tian, Ting Zhou, Rui Cui, Jing Sun and Xuanyi Du in Science Progress
Footnotes
Acknowledgements
None.
Authors’ contributions
XD designed the study; JT wrote the manuscript; JT, TZ, and RC collected, analyzed and interpreted the data; XD critically reviewed, edited the manuscript, all authors read and approved the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
