Abstract
Objective
Sepsis remains a primary contributor to mortality among critically ill individuals, and the assessment of vascular stiffness, indicated by estimated pulse wave velocity (ePWV), could offer insights into patient outcomes. This retrospective cohort study examines the correlation between ePWV levels and all-cause mortality in individuals with sepsis.
Methods
A total of 22,166 sepsis patients from the MIMIC-IV database were included. Cox regression analysis, restricted cubic spline (RCS) analysis, and subgroup analyses were conducted to evaluate the association between ePWV and 30-day and 365-day mortality.
Results
The 30-day and 365-day mortality rates were 18.28% and 32.93%, respectively. Compared to the first quartile (Q1), patients in the highest ePWV quartile (Q4) had a 92% higher risk of 30-day mortality (HR: 1.92, 95% CI: 1.73–2.13, p < 0.001) and an 75% higher risk of 365-day mortality (HR: 1.75, 95% CI: 1.62–1.89, p < 0.001) in adjusted models. RCS analysis identified a nonlinear association between ePWV and mortality (p for non-linearity < 0.001) with inflection points at 9.04 m/s for 30-day mortality and 10.81 m/s for 365-day mortality. The nomogram model demonstrated good performance in predicting both 30-day and 365-day mortality, with area under the curve values of 0.736 and 0.775 in the training set, respectively.
Conclusions
This study demonstrates that higher ePWV is significantly associated with increased short- and long-term mortality risk in critically ill sepsis patients, with evidence of a nonlinear relationship. ePWV may serve as a valuable prognostic marker to identify sepsis patients at higher risk of mortality in the ICU.
Introduction
Sepsis, a severe condition characterized by organ dysfunction resulting from an abnormal host response to infection, is primarily initiated by bacterial pathogens. 1 It remains a critical global health challenge, affecting millions annually with a mortality rate exceeding 25%.2,3 Survivors often endure long-term complications, including cognitive impairment, while the associated multi-organ dysfunction imposes significant burdens on healthcare systems and families worldwide.4,5 Early identification and timely intervention are essential to improving outcomes and reducing this burden.
Biomarkers that reflect systemic and organ-specific dysfunction are critical for risk stratification in sepsis. Arterial stiffness, an established marker of vascular health, has demonstrated prognostic value in various disease.6,7 Increased arterial stiffness reflects endothelial damage and vascular dysfunction, which are hallmarks of the disease's pathophysiology. Pulse wave velocity (PWV), considered the gold standard for evaluating arterial stiffness, is linked to negative outcomes, such as increased mortality and organ failure.8,9 However, its clinical application is limited by the need for specialized equipment and expertise.
Estimated pulse wave velocity (ePWV), derived from routinely available clinical parameters such as age and blood pressure, offers a practical and accessible alternative.10,11 ePWV has been shown to correlate with prognosis in various diseases, including coronary heart disease, 12 subarachnoid hemorrhage, 13 chronic kidney disease (CKD), 14 and atherosclerotic heart disease, 14 suggesting its broad applicability as a vascular biomarker. Despite this potential, the role of ePWV in sepsis remains poorly understood. Given the profound cardiovascular and endothelial dysfunction characteristic of sepsis, ePWV could offer critical insights into vascular alterations and their association with disease progression, mortality, and organ dysfunction.
This study examines the association between ePWV and adverse outcomes in sepsis patients. The findings aim to enhance early risk stratification and prognostication, ultimately improving outcomes for patients with sepsis.
Material and methods
Data source
The data used in this retrospective cohort study were obtained from the Medical Information Mart for Intensive Care IV (MIMIC-IV 2.2) database, a publicly available critical care database. MIMIC-IV contains de-identified health-related data of over 60,000 ICU admissions at the Beth Israel Deaconess Medical Center between 2008 and 2019. The database includes comprehensive clinical data such as patient demographics, vital signs, laboratory test results, medications, and outcomes. Shucun Liu (ID: 59144650) completed the required training and was certified to access and collect data for this study. This study was conducted in accordance with the principles outlined in the Declaration of Helsinki, 15 ensuring that all research procedures comply with ethical standards. The methodology and reporting of this study adhere to the principles set forth in the Strengthening the Reporting of Observational Studies in Epidemiology guidelines for observational research. 16
Study population
From an initial cohort of 50,920 first ICU admissions in the MIMIC-IV database, 24,672 patients were identified as having sepsis, based on the Sepsis-3 criteria. 1 After applying exclusion criteria, 2506 patients were excluded: 2482 patients with an ICU stay < 24 h, 24 patients without available systolic (SBP) or diastolic blood pressure (DBP) measurements required for ePWV, and no patients were excluded for being under 18 years of age. 22,166 patients were included in the final analysis and grouped into quartiles according to their ePWV values: Q1 (ePWV < 8.24 m/s; n = 5527), Q2 (8.24 ≤ ePWV < 10.21 m/s; n = 5545), Q3 (10.21 ≤ ePWV < 12.47 m/s; n = 5532), and Q4 (ePWV ≥ 12.47 m/s; n = 5562). The patient inclusion process is summarized in Figure 1.

Flow chart of the study population. ICU: intensive care unit; MIMIC: Medical Information Mart for Intensive Care IV; ePWV: estimated pulse wave velocity.
Data collection
Demographic data, including age, sex, and race, were collected for all patients. Vital signs recorded at ICU admission included heart rate, respiratory rate, and oxygen saturation (SpO₂). Comorbidities, including congestive heart failure (CHF), hypertension, diabetes, renal disease, malignant cancer, acute kidney injury (AKI), and septic shock were recorded. Disease severity at ICU admission was assessed using validated scoring systems: the Acute Physiology Score III (APSIII), 17 the Simplified Acute Physiology Score II (SAPS II), 18 SOFA score, 19 Glasgow Coma Scale (GCS), 20 and Charlson Index. 21 Laboratory data collected at ICU admission included hematological parameters such as white blood cell, red blood cell, hemoglobin, hematocrit, mean corpuscular volume (MCV), and platelet count. Biochemical indicators included blood urea nitrogen (BUN), creatinine, lactate, glucose, potassium, sodium, calcium, and anion gap. Treatment variables during ICU stay included the use of heparin, invasive mechanical ventilation, and continuous renal replacement therapy (CRRT). Additionally, considering that the use of vasoactive medications can influence arterial stiffness,22,23 we further extracted data on the administration and dosages of vasoactive drugs during the ICU stay. Clinical outcomes included ICU and hospital mortality, ICU and hospital length of stay, and 30- and 365-day mortality.
Exposure
The exposure was baseline ePWV, which was calculated as follows:24,25 ePWV=9.587−0.402×age+4.560 × 10−3×age2−2.621 × 10−5×age2×MAP+3.176 × 10−3×age × MAP−1.832 × 10−2×MAP3. Where mean arterial pressure (MAP) was calculated as DBP+0.4 × (SBP−DBP). 26
Statistical analysis
Baseline characteristics were compared across ePWV quartiles using appropriate statistical methods. Continuous variables were expressed as mean ± SD (for normal distribution) or median (IQR) for non-normal distribution, and compared by ANOVA or Kruskal–Wallis test, respectively. Categorical variables were presented as percentages and compared by chi-square test.
Least Absolute Shrinkage and Selection Operator (LASSO) regression analysis was employed to identify variables associated with 30-day outcomes. Cox proportional hazards models were used to examine the association between ePWV and 30- and 365-day all-cause mortality. Variance inflation factor (VIF) was calculated to assess multicollinearity among the selected variables, confirming that the covariates did not exhibit significant collinearity. Three models were constructed to adjust for confounders: Model 1: unadjusted; Model 2: adjusted for gender, race, respiratory rate, hypertension, AKI, septic shock, APSIII, GCS, CCI; Model 3: adjusted for Model 2 covariates plus source of infection, RDW, MCV, hematocrit, anion gap, lactate, vasoactive drug, vasoactive dose. Trend tests were performed across ePWV quartiles. Kaplan–Meier survival analysis was conducted to compare survival rates across quartiles.
Restricted cubic spline (RCS) analysis was performed to explore potential nonlinear relationships between ePWV and mortality risk. When non-linearity was observed, recursive algorithms were used to identify inflection points, and piecewise Cox models were applied to evaluate the associations on either side of the inflection points. Subgroup analyses were conducted based on sex, age (<65 or ≥65 years), hypertension, diabetes, renal disease, use of vasoactive drugs or heparin, and invasive mechanical ventilation. Missing data, as detailed in Table S1, were imputed using multiple imputation with the random forest algorithm (via the “mice” R package).
A nomogram model was constructed based on the top predictors identified through Boruta. The data were split into a training set (70%) and an internal validation set (30%). The model was then evaluated using receiver operating characteristic (ROC) curves.
All statistical analyses were performed using R software (version 4.4.1) and Free Statistics software (version 2.0). A two-tailed p-value < 0.05 was considered statistically significant.
Results
Baseline characteristics of included patients
Baseline characteristics are summarized in Table 1. The study included 22,166 sepsis patients, with a mean age of 66.56 ± 16.19 years, and 57.92% were male. Heart rate and respiratory rate increased, while SpO₂ decreased across higher ePWV quartiles (all p < 0.001). Comorbidities such as CHF, hypertension, diabetes, renal disease, and malignant cancer were more common in higher ePWV groups (all p < 0.001). Patients in higher ePWV quartiles also exhibited greater disease severity, with higher SAPS II, and CCI scores and lower GCS scores (all p < 0.001). Regarding the source of infection (p < 0.001), bloodstream infections were the most common (28.61%), with a slight increase in frequency across ePWV quartiles. Laboratory parameters such as RDW, BUN, creatinine, calcium, and anion gap were elevated in higher ePWV quartiles, while hemoglobin showed no significant difference (p = 0.23). Platelet count, hematocrit, glucose, sodium, potassium, and APTT also varied significantly across quartiles (all p < 0.001). Treatment approaches differed, with vasoactive drug use and invasive mechanical ventilation more frequent in lower ePWV groups, while heparin use increased slightly with higher ePWV (all p < 0.001). CRRT usage declined from 7.76% in Q1 to 3.22% in Q4 (p < 0.001). Clinical outcomes deteriorated with increasing ePWV. Hospital mortality rose from 11.87% in Q1 to 20.21% in Q4, while 30-day and 365-day mortality increased from 13.03% and 22.85% in Q1 to 27.33% and 48.67% in Q4, respectively (all p < 0.001).
Baseline characteristics according to ePWV quartiles.
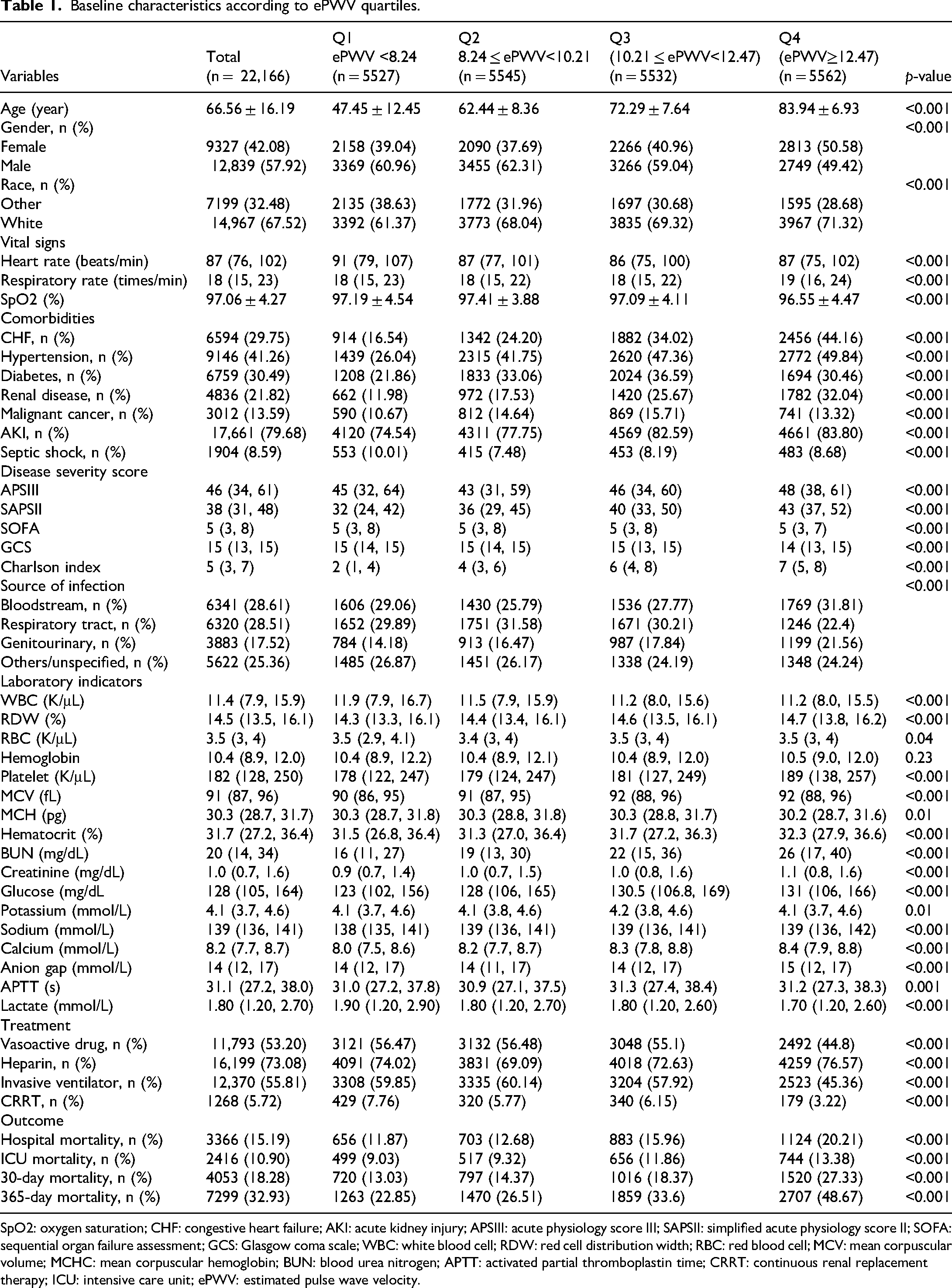
SpO2: oxygen saturation; CHF: congestive heart failure; AKI: acute kidney injury; APSIII: acute physiology score III; SAPSII: simplified acute physiology score II; SOFA: sequential organ failure assessment; GCS: Glasgow coma scale; WBC: white blood cell; RDW: red cell distribution width; RBC: red blood cell; MCV: mean corpuscular volume; MCHC: mean corpuscular hemoglobin; BUN: blood urea nitrogen; APTT: activated partial thromboplastin time; CRRT: continuous renal replacement therapy; ICU: intensive care unit; ePWV: estimated pulse wave velocity.
Association between estimated pulse wave velocity and mortality
We identified variables with significant differences between the 30-day survival and mortality groups (Table S2) and selected 19 predictors, including ePWV, using LASSO regression (Figure S1). Univariate Cox regression identified variables with p < 0.05, which were included in the multivariable models. Table S3 shows the multicollinearity assessment using the GVIF, confirming no significant collinearity among the selected covariates. Results of the univariate and multivariable analyses are presented in Tables 2 and 3, respectively. For 30-day mortality, ePWV as a continuous variable was independently associated with an adjusted HR of 1.09 (95% CI: 1.08–1.11, p < 0.001). Patients in the highest ePWV quartile (Q4) had a 92% higher risk of 30-day mortality compared to Q1 (HR: 1.92, 95% CI: 1.73–2.13, p < 0.001). Similarly, for 365-day mortality, ePWV as a continuous variable showed an adjusted HR of 1.12 (95% CI: 1.04–1.21, p < 0.001), with patients in Q4 experiencing an 18% higher risk compared to Q1 (HR: 1.75, 95% CI: 1.62–1.89, p < 0.001). Kaplan–Meier survival curves (Figure 2) showed a significant decrease in survival probability across increasing ePWV quartiles (p < 0.001), with Q4 demonstrating the lowest survival probability for both 30-day and 365-day outcomes.

Kaplan–Meier survival analysis curves for all-cause mortality. Kaplan–Meier curves of 30-day (A) and 365-day (B) all-cause mortality stratified by ePWV. ePWV: estimated pulse wave velocity.
Univariate cox proportional analysis for 30-day mortality.

Abbreviations: ePWV: estimated pulse wave velocity; AKI: acute kidney injury; APSIII: acute physiology score III; GCS: Glasgow coma scale; CCI; RDW: red cell distribution width; MCV: mean corpuscular volume.
Association between ePWV and all-cause mortality in patients with sepsis.
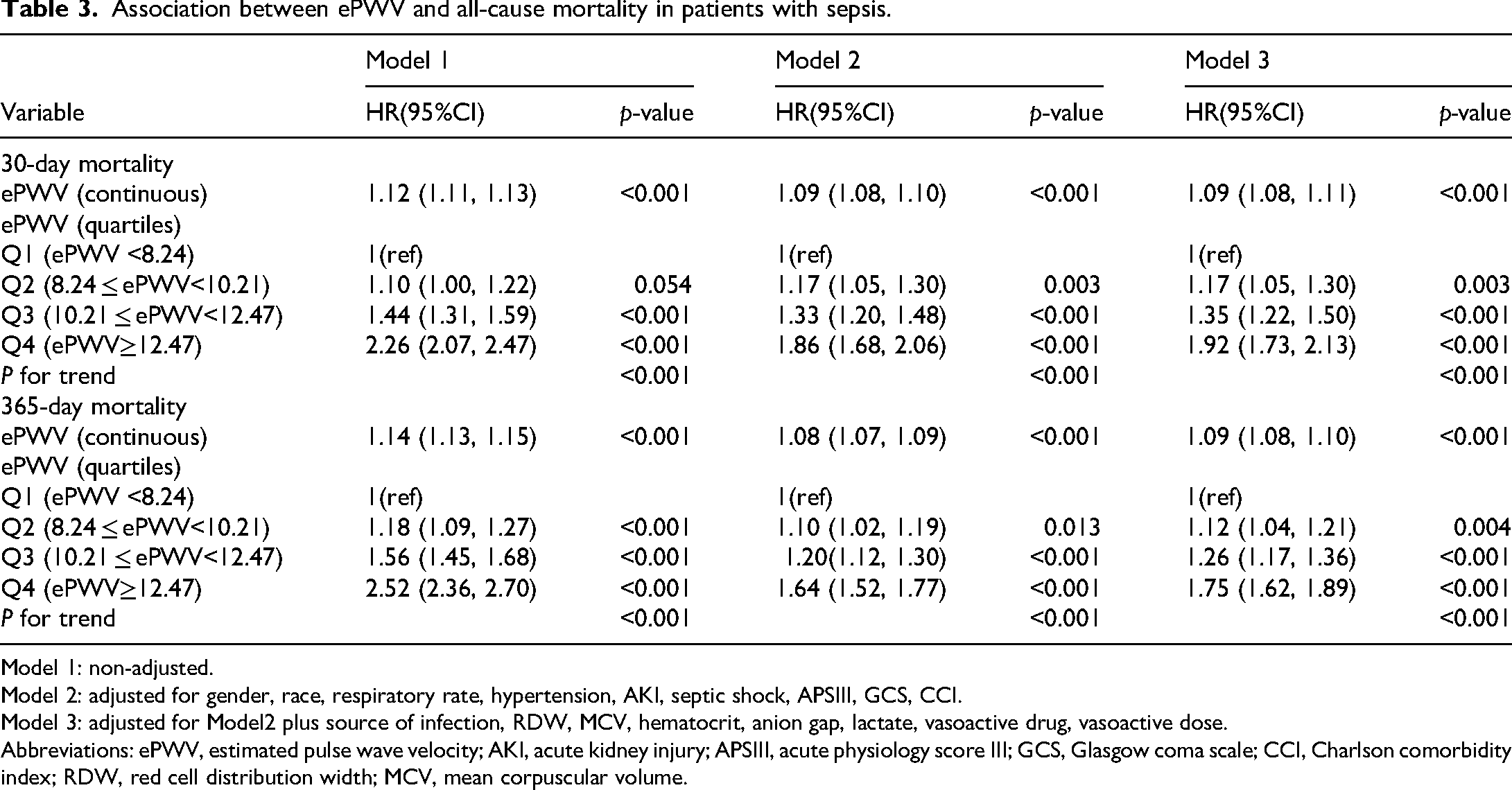
Model 1: non-adjusted.
Model 2: adjusted for gender, race, respiratory rate, hypertension, AKI, septic shock, APSIII, GCS, CCI.
Model 3: adjusted for Model2 plus source of infection, RDW, MCV, hematocrit, anion gap, lactate, vasoactive drug, vasoactive dose.
Abbreviations: ePWV, estimated pulse wave velocity; AKI, acute kidney injury; APSIII, acute physiology score III; GCS, Glasgow coma scale; CCI, Charlson comorbidity index; RDW, red cell distribution width; MCV, mean corpuscular volume.
The nonlinear relationship between estimated pulse wave velocity and mortality
RCS analysis demonstrated a significant nonlinear association between ePWV and all-cause mortality for both 30-day and 365-day outcomes (Figure 3). Two-piecewise linear regression identified inflection points at ePWV values of 9.04 m/s for 30-day mortality and 10.81 m/s for 365-day mortality (Table 4). Below these inflection points, ePWV was not significantly associated with mortality (30-day: HR: 1.05, 95% CI: 0.99–1.11, p = 0.137; 365-day: HR: 1.04, 95% CI: 1.01–1.06, p = 0.006). However, above these thresholds, higher ePWV was significantly associated with increased mortality risk (30-day: HR: 1.12, 95% CI: 1.10–1.14, p < 0.001; 365-day: HR: 1.10, 95% CI: 1.10–1.14, p < 0.001). Likelihood ratio tests confirmed that the two-piecewise linear model provided a better fit compared to a single linear model (p < 0.001). To further illustrate these findings, Figure S2 shows Kaplan–Meier survival curves for 30-day and 365-day all-cause mortality, with higher mortality observed in the high ePWV group. In line with these results, Table S4 presents the association between ePWV inflection points and all-cause mortality. In Model 3, after adjusting for potential confounders, patients with ePWV ≥9.04 m/s had a significantly higher risk of 30-day mortality (HR: 1.38, 95% CI: 1.27–1.49, p < 0.001), and for 365-day mortality, the risk was also significantly higher for patients with ePWV ≥10.81 m/s (HR: 1.31, 95% CI: 1.23–1.38, p < 0.001).

Restricted cubic splines (RCSs) of ePWV and all-cause mortality. The RCS curves illustrate the association of ePWV with 30-day (A) and 365-day (B) all-cause mortality, using the median ePWV value (10.21 m/s) as the reference point. ePWV: estimated pulse wave velocity.
The nonlinear relationship between ePWV and all-cause mortality.

ePWV: estimated pulse wave velocity.
Subgroup analysis
The association between ePWV and all-cause mortality was consistent across most subgroups, including diabetes, renal disease, and invasive mechanical ventilation (Figure 4). Significant interactions were observed in certain subgroups. For both 30-day and 365-day mortality, the association was stronger in patients aged ≥65 years compared to those <65 years (both p for interaction1 < 0.001). For 365-day mortality, males showed a stronger association than females (p for interaction = 0.031), and patients without hypertension had a greater risk compared to those with hypertension (p for interaction = 0.003). Additionally, the use of vasoactive drugs amplified the association for 30-day mortality (p for interaction = 0.005).

Association between ePWV and all-cause mortality by baseline characteristics. Hazard ratios (HR) and 95% CIs for the association of ePWV with 30-day mortality (HR1) and 365-day mortality (HR2) are shown across subgroups. Analyses were adjusted for all covariates included in Model 3 from Table 2, except the stratification variable. ePWV: estimated pulse wave velocity.
Construction of nomogram model
We developed a nomogram model based on the top five prognostic variables identified through the Boruta feature selection method, including ePWV, age, SOFA score, RDW, and Charlson Index (Figure S3). Each variable contributed a weighted score, and the cumulative score provided an estimated mortality risk, offering clinicians a practical tool for assessing individual patient risk of mortality in sepsis (Figure 5). To assess the performance of the nomogram, we conducted ROC curve analysis in both the training set and internal validation set (Figure S4). In the training set, the ROC curve for 30-day all-cause mortality yielded an area under the curve (AUC) of 0.736 (95% CI: 0.714–0.758), while the AUC for 365-day all-cause mortality was 0.775 (95% CI: 0.758–0.792). In the internal validation set, the AUC for 30-day mortality was 0.714 (95% CI: 0.678–0.750), and for 365-day mortality, it was 0.765 (95% CI: 0.739–0.792).

Nomogram for estimating the risk of mortality in sepsis. Abbreviations: ePWV: estimated pulse wave velocity; SOFA: sequential organ failure assessment; RDW: red cell distribution width.
Discussion
Our study highlights that elevated ePWV is significantly associated with increased short- and long-term mortality in sepsis patients, exhibiting a nonlinear relationship. The identified inflection points of 9.04 m/s for 30-day mortality and 10.81 m/s for 365-day mortality suggest potential thresholds that could be clinically useful for risk stratification. These results emphasize the critical role of arterial stiffness as a prognostic marker in sepsis, suggesting that ePWV assessment could aid clinicians in identifying high-risk patients who may benefit from individualized monitoring and treatment strategies.
Arterial stiffness, characterized by reduced arterial wall elasticity, is often linked to aging, atherosclerosis, hypertension, and other cardiovascular conditions. 27 PWV, a well-established marker of arterial stiffness, measures the velocity of the pressure wave through the arteries and is predictive of cardiovascular events.28,29 The ePWV, derived from non-invasive parameters such as age and blood pressure, provides a practical and reliable alternative to direct PWV measurement and has demonstrated strong concordance with direct methods. 10 Its ease of use makes it suitable for both research and clinical practice. In patients with HFpEF, Xue et al. demonstrated that higher ePWV was linked to an elevated risk of both all-cause and cardiovascular mortality, with those in the highest quartile (ePWV ≥ 12.806 m/s) nearly doubling their mortality risk. 30 Li et al. further identified ePWV as an independent predictor of reduced survival in non-traumatic subarachnoid hemorrhage, with a linear association between higher ePWV and mortality risk. 31 Similarly, Cui et al. found that among patients with CKD, ePWV > 11.90 m/s was linked to significantly higher risks of in-hospital (HR 4.72, 95% CI 3.01–7.39) and 1-year mortality (HR 2.04, 95% CI 1.31–3.19). 14 Building on this evidence, our study demonstrates the prognostic relevance of ePWV in patients with sepsis, highlighting its potential as a valuable tool for risk stratification in this high-risk population. However, while our findings support the prognostic value of ePWV, a recent study by Markakis et al. did not observe a significant correlation between PWV and mortality in patients with septic and cardiogenic shock. 32 This discrepancy could be attributed to differences in the study populations. Our cohort consisted of critically ill sepsis patients with a broad spectrum of comorbidities and causes of sepsis, whereas Markakis et al. focused specifically on patients with septic and cardiogenic shock, a more homogeneous group. This difference in patient characteristics and the severity of shock could explain why arterial stiffness, as measured by PWV, was predictive of mortality in our study but not in theirs. Additionally, the sample size in our study (22,166 patients) provided greater statistical power, while the smaller sample size of 57 patients in the study by Markakis et al. might have limited their ability to detect significant associations, potentially contributing to the contrasting results.
The relationship between arterial stiffness and sepsis outcomes is complex, reflecting the interplay between vascular, cardiac, and systemic inflammatory factors. Elevated arterial stiffness impairs vascular compliance and increases cardiac afterload,33,34 which may exacerbate the hemodynamic instability characteristic of sepsis. This can result in reduced cardiac output, impaired tissue perfusion, and heightened susceptibility to AKI, myocardial dysfunction, and multi-organ failure. Moreover, arterial stiffness is associated with endothelial dysfunction,33,35 a hallmark of both atherosclerosis and sepsis. Endothelial dysfunction may amplify the inflammatory cascade, increasing vascular permeability, dysregulated coagulation, and systemic inflammation, thereby worsening outcomes in sepsis.36,37 From a hemodynamic perspective, arterial stiffness limits the vasculature's ability to accommodate changes in intravascular volume and blood pressure, compromising the effectiveness of fluid resuscitation and vasopressor therapy.8,38 As a result, patients with significant arterial stiffness may require more tailored resuscitation strategies to optimize perfusion and oxygen delivery.
In our study, while patients in the higher ePWV quartiles had worse outcomes, they did not show an increased need for vasoconstrictors or invasive ventilation compared to those with lower ePWV. This may seem counterintuitive, as higher arterial stiffness is typically associated with greater hemodynamic instability, which would suggest a higher demand for interventions. However, several factors may explain this discrepancy. First, in sepsis, hemodynamic instability is primarily driven by systemic vasodilation and inflammatory responses rather than arterial stiffness. 39 Therefore, patients with higher ePWV may not require more vasoconstrictors, as endothelial dysfunction and vasodilation are the main contributors to shock. Additionally, invasive ventilation is more often determined by respiratory failure or oxygenation issues, which are not directly linked to arterial stiffness. Second, patients with higher ePWV may have developed compensatory mechanisms, 40 such as increased sympathetic activation, helping to maintain vascular tone and perfusion without the need for higher doses of vasoconstrictors or mechanical ventilation. Lastly, these patients may have been in a chronic or compensated state of shock, where their cardiovascular systems had adapted to arterial stiffness over time, reducing the need for more aggressive interventions. Thus, while increased ePWV is associated with worse outcomes, its lack of association with increased intervention needs likely reflects the complex interactions between arterial stiffness, vasodilation, and the stage of shock.
Our subgroup analyses showed that the association between ePWV and mortality was consistent across most subgroups, including patients with diabetes, renal disease, and those receiving invasive mechanical ventilation (p for interaction > 0.05). This consistency indicates that the relationship between ePWV and mortality is robust across various clinical conditions. However, significant interactions were observed in certain subgroups, suggesting that some patient characteristics may modify the impact of ePWV on mortality risk. For both 30-day and 365-day mortality, the association between ePWV and mortality was stronger in patients aged ≥65 years compared to those <65 years (both p for interaction < 0.001). This finding aligns with the understanding that arterial stiffness increases with age, making older individuals more susceptible to the adverse effects of vascular dysfunction during sepsis. Gender differences were also observed, with males showing a stronger association between ePWV and 365-day mortality compared to females (p for interaction = 0.031). This may be attributed to differences in vascular physiology between males and females, with males generally exhibiting higher arterial stiffness, which could contribute to worse outcomes in the context of sepsis. 41 Interestingly, patients without hypertension had a greater risk associated with elevated ePWV compared to those with hypertension (p for interaction = 0.003). One possible explanation is that patients with hypertension are more likely to be on antihypertensive therapies, which may have a protective effect on the vasculature, thereby mitigating the impact of elevated ePWV. In contrast, patients without a history of hypertension may not receive such treatment, leaving them more vulnerable to the adverse effects of increased arterial stiffness. The use of vasoactive drugs amplified the association between ePWV and 30-day mortality (p for interaction = 0.005). This suggests that patients with elevated ePWV who require vasoactive support may represent a particularly high-risk group. Vasoactive drugs, while necessary to maintain hemodynamic stability, can increase cardiac workload and impair tissue perfusion, which may be further exacerbated by underlying arterial stiffness. Careful management of vasoactive therapy in patients with high ePWV could be crucial in reducing mortality risk.
The clinical implications of these findings are substantial. The identified ePWV thresholds (9.04 m/s for 30-day and 10.81 m/s for 365-day mortality) provide a basis for early risk stratification in sepsis. Patients exceeding these thresholds may benefit from more aggressive interventions, including enhanced hemodynamic monitoring and targeted therapies to mitigate endothelial dysfunction and systemic inflammation. Routine ePWV assessment could facilitate personalized care strategies, ultimately improving outcomes in critically ill patients.
However, several limitations should be acknowledged. First, the cohort was derived from a single-center database, which may limit the generalizability of our findings to other populations and healthcare systems with different demographic and clinical characteristics. Second, ePWV was estimated using a formula based on age and MAP, rather than directly measured, which may introduce measurement errors, even though it has been validated in previous studies as a reliable proxy for arterial stiffness. Third, the calculation of ePWV depends on age and MAP, but critically ill sepsis patients often experience marked blood pressure fluctuations that may not accurately represent true arterial stiffness. While we addressed this potential bias by using the average blood pressure during the first 24 h of ICU admission, ePWV may still not fully reflect arterial stiffness. Fourth, this study did not adjust for the specific type of infection, the pathogens isolated, or the type of antibiotics administered, which could all influence patient outcomes. Different pathogens can affect disease severity, immune responses, and treatment effectiveness, while delayed or inadequate antibiotic treatment is a known risk factor for poor outcomes in sepsis. As a result, failure to account for these factors may have introduced residual confounding, limiting the ability to draw firm conclusions about the relationship between ePWV and mortality. Lastly, the nonlinear associations and identified inflection points for ePWV in relation to mortality require validation in prospective multicenter studies before they can be implemented in clinical practice. Future research should address these limitations and further explore the clinical utility of ePWV in sepsis management.
In conclusion, our study underscores the prognostic value of ePWV in sepsis, offering insights into its potential role in guiding clinical decision-making. Future research should focus on exploring the integration of ePWV into sepsis management protocols to enhance patient care.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251414251 - Supplemental material for Association between estimated pulse wave velocity and the risk of mortality in patients with sepsis patients: A cohort study
Supplemental material, sj-docx-1-sci-10.1177_00368504251414251 for Association between estimated pulse wave velocity and the risk of mortality in patients with sepsis patients: A cohort study by Shucun Liu, Miao Zhang, Wei Ye, Pingping Li, Xing Tang and Jiaqiong Li in Science Progress
Footnotes
Abbreviations
Ethics approval and consent to participate
This study made use of the publicly available MIMIC-IV database, in which patient information has been anonymized to ensure privacy. The construction of the MIMIC-IV database was approved by the Institutional Review Boards of the Beth Israel Deaconess Medical Center (Boston, MA, USA), with a waiver of informed consent. This study adheres to ethical standards, and the use of data strictly complies with the database's access requirements.
Author contributions
SL was responsible for data curation and drafting the initial manuscript. MZ, PL, XT, and WY conducted the formal analysis and applied software tools. JL provided critical review and editing of the manuscript. All authors have read and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the 2018 Municipal Science and Technology Bureau Key Research and Development(Social development), (grant number No. KC18189).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
