Abstract
Objectives
To investigate the risk factors associated with thrombocytopenia in patients undergoing extracorporeal membrane oxygenation (ECMO) support.
Methods
This retrospective cohort study included 230 patients who received ECMO support during January 2018 to December 2024. Clinical data collected included age, gender, causes of ECMO, medical history, vital signs, laboratory test results, complications and outcomes. The patients were divided into the thrombocytopenia group and the control group based on whether the patient's platelet count was less than 150*109/L. The differences in clinical data between the two groups were compared. Moreover, risk factors associated with thrombocytopenia were identified using univariate and multivariate regression analyses.
Results
In this study, a total of 230 patients undergoing ECMO were involved. The mean age of patients was 52 ± 8 years with 119 (51.7%) males and 111 (48.3%) females. The age, sex distribution, reasons for ECMO, previous medical history, temperature, respiration rate, heart rate, mean arterial pressure, white blood cells, hemoglobin, C-reactive protein, platelets, total bilirubin, alanine aminotransferase, aspartate aminotransferase, prothrombin time, activated partial thromboplastin time, blood lactate between two groups showed no statistically significant differences (P > 0.05) while the APACHE II score (24(20,28) vs 21(17,24)) was higher in thrombocytopenia group (P < 0.05). The bleeding events were significantly higher (62.4% vs 15.7%), in-hospital survival (49.7% vs 62.9%) was lower, the duration of ICU stay (19(16,23) vs 18 (14,22)) was longer, and more platelet (30(10,40) vs 15 (10,20)) and plasma transfusions (600(400,800) vs 450 (200,600)) in thrombocytopenia group (P < 0.05). Logistic regression analysis showed that APACHE II score, rotational speed over the first 48 h, with sepsis, the heparin doseage before ECMO, use of continuous renal replacement therapy (CRRT) and intra-aortic balloon pump (IABP) during ECMO were risk factors associated with thrombocytopenia.
Conclusions
The incidence of thrombocytopenia in patients receiving ECMO is high and may be associated with poor prognosis. Patients with higher APACHE II score, higher rotational speed during the first 48 h, sepsis, higher heparin doseage before ECMO initiation, and the use of CRRT or IABP may at an increased risk of developing thrombocytopenia.
Introduction
Extracorporeal membrane oxygenation (ECMO) is an important cardiopulmonary support technique in critical care medicine. In recent years, the widespread use of ECMO has improved survival rates for many patients. However, like many technologies, it is inevitably associated with various risks and complications, among which bleeding is the most common. According to related reports, the incidence of bleeding during ECMO can be as high as 60%. 1
The pathophysiological mechanisms underlying bleeding during ECMO are complex and multifactorial. It involves the use of anticoagulant drugs, consumption of coagulation factors, platelet activation and dysfunction, and thrombocytopenia caused by platelet consumption.1–3 It is well known that platelets play a crucial role in maintaining hemostatic balance. After ECMO initiation, both the quantity and function of platelets undergo significant changes. Studies have shown that thrombocytopenia frequently occurs during ECMO support and is often severely underestimated.4,5 One study found that up to 95% of patients developed thrombocytopenia during ECMO support. 4
Thrombocytopenia increases the risk of bleeding and mortality, leading to poor patient outcomes.6–8 Thus, identifying the factors associated with thrombocytopenia is of great significance for its prevention and management, as well as for reducing bleeding risk and improving patient prognosis. Therefore, this study aims to investigate the risk factors associated with thrombocytopenia during ECMO support, in order to provide evidence that can inform clinical decision-making in this field.
Materials and methods
The study was approved by the ethics review board of Jinhua Municipal Central Hospital (approval No. 2024-102, approval date: 2024/05/14) and conducted in accordance with the Declaration of Helsinki (as revised in 2024). As the study was a retrospective design, informed consent was waived. In addition, the reporting of this study conforms to STROBE guidelines. 9
Subjects
This retrospective cohort study consecutively included clinical data from 230 patients who received ECMO support in Jinhua Municipal Central Hospital from January 2018 to December 2024.
Inclusion criteria: (1) Patients who received ECMO support for any cause; (2) The patients were over 18 years old.
Exclusion criteria: (1) Patients with more than 20% missing data; (2) Patients with ECMO support duration less than 72 h; (3) Patients with a history of hematological diseases; (4) Patients with hyperfunction of the spleen; (5) Patients with platelet count less than 150*109/L before ECMO support; (6) Patients with heparin induced thrombocytopenia (HIT).
The diagnosis of HIT is based on a combination of the 4Ts score and laboratory testing. If HIT is suspected, the initial evaluation should begin with the 4Ts score. A low probability score (≤3) makes HIT unlikely, and continued clinical observation is recommended. If the score indicates intermediate or high probability (≥4), HIT antibody testing should be performed. If the result is positive, the diagnosis should be confirmed by functional assays or in combination with clinical manifestations.
All patient details have been de-identified. Clinical data including gender, age, reasons for ECMO, previous medical history, temperature, respiration rate, heart rate, mean arterial pressure, white blood cells, hemoglobin, C-reactive protein, platelet count, total bilirubin, alanine aminotransferase, aspartate aminotransferase, prothrombin time, activated partial thromboplastin time, blood lactate levels, history of antiplatelet drug use, acute physiology and chronic health evaluation II (APACHE II) score, ECMO mode, average speed during the first 48 h after ECMO, ECMO duration, whether sepsis was diagnosed during ECMO, total amount of heparin before ECMO, whether continuous renal replacement therapy (CRRT) was performed, whether intra-aortic balloon pump (IABP) was performed, as well as the patient's complications including bleeding after the onset of thrombocytopenia, thrombus (observed after ECMO removal) and outcomes including hospitalization duration, length of stay in ICU, survival and platelet and plasma transfusions were collected by reviewing electronic medical records and nursing notes. In our center, the platelet transfusion threshold is below 50*109/L. According to whether the patient's platelet count was less than 150*109/L, the patients were divided into the thrombocytopenia group and the control group. 5 The differences in relevant indicators between the two groups were compared. Univariate and multivariate regression analyses were performed to identify risk factors associated with thrombocytopenia.
ECMO cannulation and maintenance
ECMO was percutaneously cannulated by two or three experienced intensivists and two nurses under ultrasound guidance. Either a SORIN SCPC (London, United Kingdom) or MAQUET (Germany) or FRESENIUS (Germany) system was used. The venous cannulas were 21–26 Fr (MAQUET or Edwards) in diameter, while the arterial cannulas (return) were 15–18 Fr (MAQUET, Edwards, or Medtronic) in diameter. Intravenous unfractionated heparin was generally administered for anticoagulation with a target activated clotting time of 160–200 s.
Statistical analysis
Statistical analysis was performed using SPSS software (version 26, IBM Corp.) and R (version 4.4.3). Continuous variables with a normal distribution were expressed as mean ± standard deviation and comparisons between the two groups were conducted using independent samples t-test. If variances were unequal, the corrected t-test was applied. Continuous variables not following a normal distribution were presented as the median and interquartile range [M (Q1, Q3)] and comparisons between the two groups were conducted using the Mann–Whitney U test. Categorical variables were expressed as frequencies (percentages) and comparisons between groups were performed using the χ2 test. Univariate and multivariate logistic regression analyses were used to identify risk factors. Variables with a P value < 0.2 in univariate logistic regression were included in the multivariate logistic regression analysis using the Enter method. Possible interactions between risk factors were also tested. A two-tailed P value < 0.05 was considered statistically significant.
Results
A total of 230 patients receiving extracorporeal membrane oxygenation support were included. The mean age of patients was 52 ± 8 years with 119(51.7%) males and 111 (48.3%) females. The age, sex distribution, reasons for ECMO, previous medical history, temperature, respiration rate, heart rate, mean arterial pressure, white blood cells, hemoglobin, C-reactive protein, platelets, total bilirubin, alanine aminotransferase, aspartate aminotransferase, prothrombin time, activated partial thromboplastin time, blood lactate levels between the two groups showed no statistically significant differences (P > 0.05). The APACHE II score in the thrombocytopenia group was higher than the control group (P < 0.05). Patients’ basic characteristics are shown in Table 1.
Basic characteristics of patients.

Data are presented as n (%) or mean ± standard deviation. COPD, Chronic obstructive pulmonary disease; AST, Aspartate aminotransferase; ALT, Alanine aminotransferase; PT, Prothrombin time; APTT, Activated partial thromboplastin time; APACHE II, Acute physiology and chronic health.
Univariate logistic regression analysis showed that risk factors related to thrombocytopenia were dual antiplatelet treatment, APACHE II score, rotational speed over the first 48 h, presence of sepsis, the heparin doseage before ECMO, use of CRRT and IABP during ECMO (P < 0.2). Further multivariate logistic regression analysis showed that risk factors related to thrombocytopenia were APACHE II score, rotational speed over the first 48 h, presence of sepsis, the heparin doseage before ECMO, use of CRRT and IABP during ECMO (P < 0.05). The R2 of the regression model is 0.261 and P = 0.628. Univariate and multivariate logistic regression analyses are shown in Tables 2 and 3.
Univariate logistic regression analysis of risk factors related to thrombocytopenia.

APACHE II, Acute physiology and chronic health; ECMO, Extracorporeal membrane oxygenation; CRRT, continuous renal replacement therapy; IABP, intra-aortic balloon pump.
Multivariate logistic regression analysis of risk factors related to thrombocytopenia.
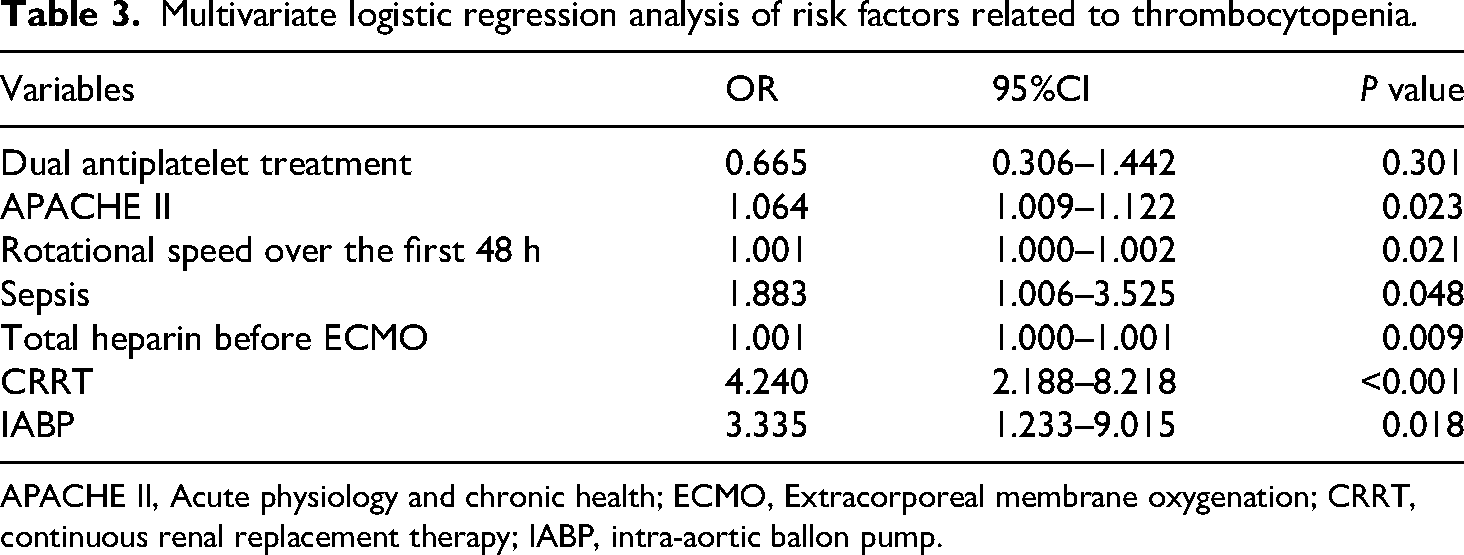
APACHE II, Acute physiology and chronic health; ECMO, Extracorporeal membrane oxygenation; CRRT, continuous renal replacement therapy; IABP, intra-aortic ballon pump.
The bleeding events were significantly higher in the thrombocytopenia group (62.4%) compared to the control group (15.7%) with the nasal cavity as the most common site (29.1%) (P < 0.05). The in-hospital survival was significantly lower in the thrombocytopenia group (49.7%) compared to the control group (62.9%) and the ICU stay was significantly longer (P < 0.05). The total platelet and plasma transfusions were much more in the thrombocytopenia group than in the control group (P < 0.05). Complications and outcomes of the patients are shown in Table 4.
Complications and outcomes.

Discussion
This study revealed that thrombocytopenia is common during ECMO support. Thrombocytopenia is associated with higher bleeding events, lower in-hospital survival, longer ICU stay, and more platelet and plasma transfusions. Independent risk factors associated with thrombocytopenia were higher APACHE II score, rotational speed over the first 48 h, presence of sepsis, the heparin doseage before ECMO, use of CRRT and IABP.
Through this study, we identified risk factors associated with thrombocytopenia during ECMO support. This provides a theoretical basis for subsequent clinical prevention and intervention. The quantity of platelets during ECMO operation plays a crucial role in maintaining the patient's coagulation function. During the ECMO operation, many factors can affect platelets. The first is the severity of the patient's condition. The APACHE II score is a common scoring system used in the field of critical care medicine to assess the severity of the patient's condition. It covers critical illness physiology, organ dysfunction, and direct and indirect indicators reflecting inflammation. In other studies, higher severity of critical illness, organ dysfunction, and sepsis have been proven to be independent predictors of thrombocytopenia in critically ill patients.10,11 Platelets are relatively fragile blood cells, and in critically ill patients, the likelihood of their being affected increases. Therefore, assessing the severity of critical illness and baseline platelet counts at the initiation of ECMO support may help clinicians anticipate which patients are at higher risk of developing severe thrombocytopenia and bleeding, thereby facilitating early intervention and management. Secondly, the high shear stress caused by ECMO rotation speed may also be a contributing factor. Thrombocytopenia commonly occurs during extracorporeal circulation due to factors such as shear stress, hypothermia, and platelet aggregation induced by contact with artificial surfaces.12,13 A recent study demonstrated that platelet counts decrease in a time-dependent manner within the first 1, 24, and 48 h after ECMO initiation. 14 The high shear stress during ECMO support can lead to the shedding of glycoprotein Ibα (GPIbα) from the platelet surface, which accelerates the clearance of GPIbα-negative platelets, thereby contributes to thrombocytopenia. 14 High blood flow rates during ECMO, which are essential for many patients, particularly those with severe cardiopulmonary failure, result in increased shear stress. For patients with cardiac arrest, especially in the early stages after resuscitation, a high level of flow support is often necessary to provide adequate oxygenation and perfusion.15,16
The proper functioning of ECMO relies heavily on effective anticoagulation. Heparin is currently the most widely used anticoagulant; however, its use may disrupt the delicate balance of a patient's coagulation system. One of the most severe and potentially fatal complications is HIT.3,17 Our study found that increased heparin doseage was associated with a higher risk of thrombocytopenia, which may be attributed to HIT. In addition, many critically ill patients in the intensive care unit develop thrombocytopenia due to infection or inflammation-induced bone marrow suppression, leading to decreased platelet production or increased destruction.18,19 Sepsis, which is a condition characterized by organ dysfunction resulting from infection, is also associated with thrombocytopenia during ECMO support. Furthermore, the use of CRRT and IABP during ECMO are another important factor influencing platelet levels. Both CRRT and IABP are extracorporeal support techniques that increase the duration of blood contact with artificial membranes and alter hemodynamic conditions,13,20,21 thereby further contributing to platelet consumption and increasing the risk of thrombocytopenia. In our study, the ECMO duration was not significantly associated with the risk of thrombocytopenia, which is consistent with the findings of Darryl et al. 12 It maybe that patients in the early phase of ECMO often require higher flow rates, a demand that tends to decrease in the later phase. Moreover, there are multiple causes of thrombocytopenia, and these factors may mask the effect of ECMO duration.
Thrombocytopenia can result from a variety of mechanisms, and its development becomes even more complex in the setting of ECMO support. During ECMO, contact between blood and the extracorporeal circuit, including the membrane oxygenator, can enhance platelet activation and increase platelet consumption. In addition, activation of inflammatory and coagulation cascades during ECMO further contributes to platelet loss. 5 Heparin, the most commonly used anticoagulant during ECMO, may also cause thrombocytopenia through HIT, an immune-mediated complication. 3 Regardless of its underlying cause, thrombocytopenia is associated with adverse outcomes, including decreased in-hospital survival and prolonged ICU stays. This may be attributable to an increased risk of organ dysfunction from bleeding events following thrombocytopenia or to impaired immune function caused by thrombocytopenia. 22 Therefore, identifying risk factors for thrombocytopenia and implementing timely monitoring and intervention are of considerable clinical importance.
This study has several limitations. Firstly, this study was retrospectively designed with a long inclusion period, advancements in ECMO technology and equipment over time may have influenced patient outcomes. Secondly, there were not enough HIT cases to include in the analysis. If enough patients had been included as a subgroup for analysis, the results might be more representative.
Conclusion
This study found that thrombocytopenia is common and associated with hemorrhagic events and transfusion requirements during ECMO support. It may lead to poor patient outcomes, including reduced in-hospital survival and prolonged ICU stays. Patients with higher APACHE II scores, elevated ECMO rotational speeds, sepsis, and increased use of heparin, CRRT, and IABP may more likely to develop thrombocytopenia. Therefore, appropriate preventive and interventional strategies targeting these factors would seem necessary to reduce the incidence of thrombocytopenia.
Footnotes
Acknowledgments
None.
Ethical approval
The study was approved by the ethics review board of Jinhua Municipal Central Hospital (approval No. 2024-102, approval date:2024/05/14).
Consent
Informed consent was waived due to the retrospective design.
Authors contributions
Hongjie Tong, Jiali Yao and Feiyan Pan were the major contributor in writing the manuscript. Kaixuan Wang and Jin Jinjin collected and analyzed the data. Qianqian Wang and Kun Chen designed the study. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jinhua Science and Technology Research Program, Zhejiang Provincial Medical Science Foundation, Provincial Key Clinical Specialty of Zhejiang, he Provincial medical and Health Science and Technology Program, provincial and Ministry co-construction project in China, (grant number 2022-3-060, 2023-4-074, 2024KY435, 2024-ZJZK-002, WKJ-ZJ-2303).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The authors are willing to share their data. If needed, please contact the corresponding author by email.
