Abstract
Objective
The aim of this study is to reveal the relationship between TURKOVAC, CoronaVac, and BNT162b2 and deep vein thrombosis.
Methods
Between 2022 and 2024, a case-control study was planned with 400 patients included (deep vein thrombosis group (n = 200) and control group (n = 200). Patients were divided into four groups according to their vaccine types. Group 1 (n = 54): those who received only CoronaVac, Group 2 (n = 233): those who received only BNT162b2, Group 3 (n = 49): those who received CoronaVac for the first dose, and then BNT162b2, Group 4 (n = 15): those who received BNT162b2 for the first dose and then TURKOVAC. First, the deep vein thrombosis and control groups were compared regarding basic characteristics. Then, patients in the four groups were compared in terms of deep vein thrombosis.
Results
No difference was found in the frequency of COVID-19 disease in the deep vein thrombosis and control groups. The frequency of vaccination was lower in the deep vein thrombosis group (84%–91.5%). According to the multivariate regression test result, we found that COVID-19 vaccination was protective against deep vein thrombosis (p = 0.003, OR = 0.377 [95% CI, 0.197–0.722]). After being divided into four groups, the rates of deep vein thrombosis were 36 (66.7%), 96 (41.2%), 29 (59.2%), and 7 (46.7%), respectively. The frequency of deep vein thrombosis was significantly lower in Group 2 (p = 0.003). Although no statistically significant difference was found among other groups, the lowest deep vein thrombosis frequency was found in Group 4, Group 3, and Group 1, respectively.
Conclusion
The frequency of COVID-19 vaccination was lower in patients with deep vein thrombosis than in those without. Deep vein thrombosis was least common in patients who received only BNT162b2. Although the frequency of deep vein thrombosis was similar in the groups, we found that the lowest rate of deep vein thrombosis was in the group that received TURKOVAC after BNT162b2.
Introduction
COVID-19, which emerged in Wuhan, China in December 2019 and spread throughout the world and was declared a pandemic by the World Health Organization in March 2020, has caused more than 445 million confirmed cases and 6 million deaths worldwide, according to reports in March 2022.1,2 SARS-CoV-2, which causes COVID-19, is a single-stranded RNA coronavirus that enters human cells by binding to the angiotensin-converting enzyme 2 (ACE2) receptor, which is expressed mainly in alveoli, cardiac myocytes, and endothelial cells. 3
It has been shown to cause complex and multifactorial endothelial activation, widespread inflammation, progressive loss of anti-thrombotic factors, and local thrombosis, as well as pulmonary complications in patients infected with SARS-CoV-2. 4 Therefore, one of the major organ systems affected by COVID-19 is the vascular system. SARS-CoV-2 affects many mechanisms, such as disruption of the renin-angiotensin-aldosterone system (RAAS), oxidative stress, endothelial dysfunction, an increase in some cytokines such as IL-6, IL-1β, and activation of Von Willebrand factor (vWF).5,6
Studies have shown a correlation between COVID-19 and deep vein thrombosis (DVT), and DVT has been reported to occur in ∼14.8% of COVID-19 patients. 7 Another important point is the COVID-19 vaccines used during the pandemic, although their long-term effects are unknown. In a cohort study published in 2024 with 20 million people, it was shown that the COVID-19 vaccine reduces the risk of cardiac and thromboembolic outcomes after COVID-19. 8 Many countries have produced and used their vaccines for this purpose. However, there is no definitive information about which of these vaccine types is more protective against DVT. For this reason, we planned this study.
The aim of this study is to reveal the relationship between TURKOVAC (Kocak Farma Production Facilities, Tekirdag, Turkey), CoronaVac (Sinovac Life Sciences Co., Ltd, Beijing, China), and BNT162b2 (BioNTech-Pfizer), which are frequently applied COVID-19 vaccine types in Turkey, and DVT, and also to investigate which vaccine is more protective against DVT.
Methods
This study was designed as a retrospective case-control study including a total of 400 patients. Patients over the age of 18 who were examined at the Cardiovascular Surgery Department of Istanbul Basaksehir Cam and Sakura City Hospital between January 2022 and January 2024 were included in the study. Those who had cardiac surgery and those with peripheral artery disease were excluded from the study. Patients were first divided into a case group and a control group. Case group (n = 200): patients diagnosed with DVT at the time of admission or followed up with the diagnosis of DVT. Control group (n = 200): consecutive patients who applied to the outpatient clinic were randomly selected without any selection criteria, and had no DVT diagnosis and no cardiac pathology. The patients were then divided into four subgroups according to their COVID-19 vaccine type, after excluding unvaccinated patients. Group 1 (n = 54): those who received only CoronaVac at least once, Group 2 (n = 233): those who received only BNT162b2 at least once, Group 3 (n = 49): those who received CoronaVac for the first dose and then BNT162b2, and Group 4 (n = 15): those who received BNT162b2 for the first dose and then TURKOVAC.
DVT was diagnosed by Doppler ultrasonography (DUS) or computed tomography (CT) venography. The initial diagnosis of DVT was based on direct visualization of the thrombus, absence of compressibility on the grayscale, and identification of a filling defect or thrombus continuing in the colored column of the vascular lumen or absence of flow on DUS.
All patients’ basic demographic characteristics, medical history, family history, COVID-19 history and COVID-19 vaccine information, and triggering factors for DVT (immobility, malignancy, travel history, trauma, surgery, rheumatic disease, oral contraceptive, and pregnancy) were recorded. First, the DVT and control groups were compared in terms of basic characteristics. Then, after excluding unvaccinated patients, patients in the four groups were compared within themselves in terms of DVT, and differences were investigated.
This study was approved by the Istanbul Basaksehir Cam and Sakura City Hospital Ethics Committee (Decision no: 2024-276). The study was conducted in accordance with the principles of the Declaration of Helsinki. The reporting of this study conforms to STROBE guidelines. 9
Statistics
Data were analyzed by using SPSS software version 20.0 (IBM, USA). Continuous variables in the study were presented as minimum, maximum, median, and interquartile range. Categorical variables were expressed as numbers and percentages. The normality of distribution was assessed by the Kolmogorov–Smirnov test. For numerical variables, differences between patients and controls were tested using t test for parametric data or the Mann–Whitney U test for non-parametric data. Categorical variables were analyzed using the Pearson χ2 test and Fisher's exact test. Pairwise comparisons were made using the Pearson χ2 test and evaluated using the Bonferroni correction. Multivariate regression analysis was conducted to identify the factors affecting DVT. The level of statistical significance was set at p < 0.05.
Results
Demographic data, comorbidities, COVID-19 vaccine, and history of the patients are shown in Table 1. No difference was found between the DVT and control groups in terms of gender, but age, height, and weight values were higher in the DVT group. No difference was found between the DVT and control groups in terms of chronic obstructive pulmonary disease (COPD) and smoking. However, diabetes mellitus, chronic renal failure, atrial fibrillation, and peripheral artery disease were more common in the DVT group than in the control group. Of all patients, 145 (36.2%) had COVID-19 disease, and 351 (87.7%) had received the COVID-19 vaccine at least once. No difference was found in the frequency of COVID-19 disease in the DVT and control groups. However, the frequency of COVID-19 vaccination was lower in the DVT group than in the control group (84% and 91.5%, respectively). The frequency of immobility, malignancy, travel history, trauma, history of surgery, rheumatic disease, oral contraceptive use, and pregnancy, which were investigated as triggering factors for DVT, were significantly higher in the DVT group than in the control group. The frequency of a family history of DVT and a proven genetic predisposition to thrombosis were also significantly higher in the DVT group than in the control group.
Patient's demographics, comorbid diseases, and COVID vaccine and history.

DVT: deep vein thrombosis; IQR: interquartile range; BSA: body surface area; BMI: body mass index; COPD: chronic obstructive pulmonary disease; PAD: peripheral arterial disease.
Bold value indicates statistical significance.
After excluding unvaccinated patients, the comparison of vaccinated groups in terms of DVT after dividing into subtypes is shown in Figure 1 and Table 2. The number of patients in the groups was 54, 233, 49, and 15, respectively. The rates of DVT were 36 (66.7%), 96 (41.2%), 29 (59.2%), and 7 (46.7%), respectively. In the comparison made between the groups in terms of DVT frequency, it was found that the frequency of DVT was significantly lower in Group 2 (p = 0.003). Although no statistically significant difference was found among Group 1, Group 3, and Group 4 (p = 0.35), the lowest DVT frequency was found in Group 4, Group 3, and Group 1, respectively, (46.7%, 59.2%, and 66.7%, respectively).
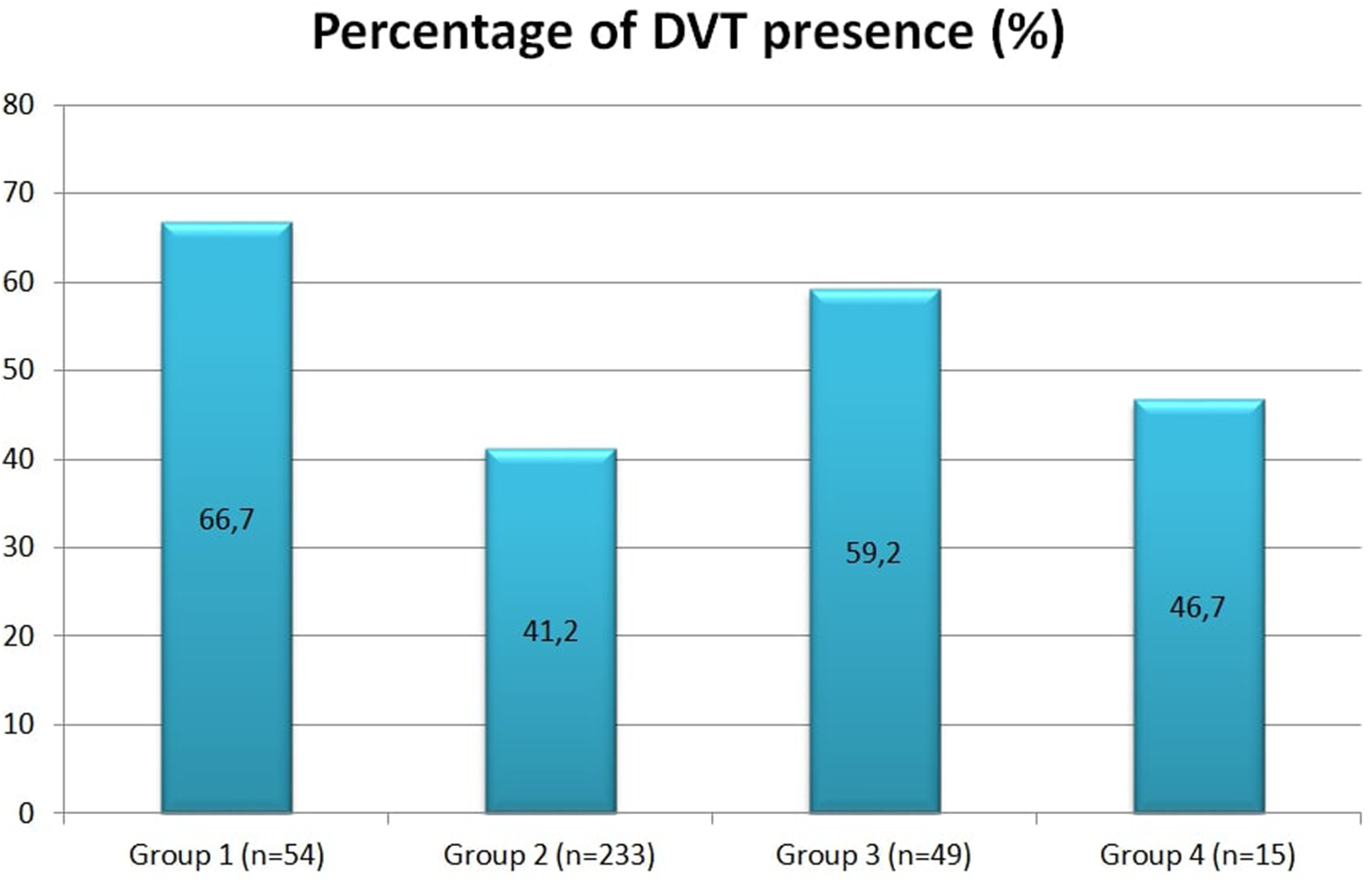
Comparison of the percentage of deep vein thrombosis between vaccination groups.
Comparison of groups in terms of deep vein thrombosis.

Bold value indicates statistical significance.
Multivariate regression analysis results are shown in Table 3. Accordingly, no significant association was found between diabetes mellitus (DM) and COPD or DVT. However, a positive significant association was found between advanced age, chronic renal failure, and DVT (p = 0.001, OR = 1.021 [95% CI, 1.008–1.034] and p = 0.008, OR = 15.923 [95% CI, 2.074–122.226]), respectively. A negative significant association was found between COVID vaccination and DVT (p = 0.003, OR = 0.377 [95% CI, 0.197–0.722]).
Multivariate logistic regression analysis.

COPD: chronic obstructive pulmonary disease; CI: confidence interval.
Discussion
COVID-19, which emerged in 2019 and affected the whole world, has caused millions of deaths. While the most important cause of death associated with COVID-19 is often pulmonary complications, the vascular system is also one of the most important systems affected and is a significant cause of morbidity and mortality. It has been shown that the risk of thrombosis increases significantly with COVID-19. 10
SARS-CoV-2 causes thrombus formation through several pathways. The first is RAAS disruption. SARS-CoV-2 can bind directly to ACE2, which leads to an imbalance in the conversion of ACE2 to ACE1. This causes an increase in ACE2 and a decrease in ACE1, which in turn aggravates inflammation, triggering oxidative stress, endothelial dysfunction, and coagulation. 5 SARS-CoV-2 can infect direct vascular endothelial cells, cause cell damage and apoptosis, and reduce the antithrombotic activity of the endothelium. 11 SARS-CoV-2 causes a cytokine storm, increasing proinflammatory cytokines such as TNF-α, IL-6, IL-1β, PAI-1, and MCP-1, and damages endothelial cells, causing thrombosis. 12 Another pathway is the activation of vWF, a procoagulant factor. 6 Additionally, NLRP3 inflammasome activation and an increase in NLRP3 inflammasome products such as IL-1β and IL-18 also cause proinflammatory effects. 13
Due to the sudden pandemic caused by COVID-19 and the difficulty in finding an effective treatment at that time, COVID-19 vaccines were produced and used under emergency conditions. Due to the high mortality rates of SARS-CoV-2, the vaccines were started to be used before the phase studies were fully completed, but research on their long-term effects is ongoing. Although the effect of COVID-19 on thrombosis is known, concerns have begun to arise that vaccines, especially mRNA vaccines, may cause thrombosis. In one study, a case of sudden massive DVT was reported in a young man with inferior vena cava anomaly 20 hours after the second dose of the mRNA vaccine for COVID-19. 14 Another study reported a patient who developed acute DVT in the right upper extremity 3 days after receiving the second dose of the Moderna vaccine. 15 A study with 20 million people showed that the COVID-19 vaccine reduced the risk of cardiac and thromboembolic outcomes after the disease. 8 Another review showed that existing mRNA vaccines are not associated with an increased risk of venous thromboembolism. 16 Another case-control study reported that the COVID-19 vaccination rate in the DVT group was lower than in the control group. 17 Considering that DVT is a common disease with a rate of 1.6 per 1000 people per year, 18 it should not be forgotten that some DVT cases may occur coincidentally after vaccination. Rather than case reports, larger studies involving millions of people, such as the study by Mercade-Besora et al., 8 provide more valuable information on this subject. Similarly, in our study, we found that the COVID-19 vaccination rate was significantly lower in those with DVT compared to the control group. According to the multivariate regression analysis, we also found that COVID-19 vaccination may be protective against DVT. These results suggest that the findings in our study are consistent with studies in the literature on large patient series, and we believe that COVID-19 vaccines may be useful in reducing the risk of DVT.
Another important point is which type of vaccine may be more protective against DVT, which is the main purpose of this study. Many vaccines have been produced from different countries, and some studies investigate their effects on DVT separately. However, there is insufficient information on which of these vaccine types can reduce the risk of DVT. In our study, we found that DVT was less frequent in the group vaccinated with only BNT162b2 (Group 2), and this was significantly lower than in the other groups (p = 0.003). mRNA vaccines were associated with lower levels of MCP-3, MMP-1, TNF-α, and CRP, and a decrease in cytokine release, and it has been reported that they may reduce the risk of thrombosis. 19 We can say that the result of our study is the clinical reflection of this pathophysiological feature. Chui et al. reported that in their study of ∼8.6 million people with BNT162b2 and CoronaVac, no statistically significant risk of thromboembolic events was observed for either vaccine. 20 TURKOVAC has been reported to have acceptable safety and tolerability in healthy adults aged < 65 years, based on findings from Phase 1 and two trials. 21 There is insufficient evidence in the literature regarding the DVT and thrombosis-reducing effect of this vaccine. Since it was authorized for emergency use as of December 2021, it was used less in our study compared to other vaccines. Although the number of patients was small, the DVT rates in the patient group receiving TURKOVAC (Group 4) were found to be statistically similar to the other groups. Moreover, although there was no statistical difference, the frequency of DVT was found to be lower than those receiving only CoronaVac (Group 1). However, to make a definitive comment on this issue, studies with larger patient numbers comparing these two vaccines are needed. Nevertheless, this study provides valuable information regarding the positive results of TURKOVAC in terms of DVT.
Limitations
The first limitation of the study is its retrospective nature and the low number of cases compared to many studies conducted on vaccine studies. Second, the DVT group was not divided into acute, subacute, and chronic, so the group was heterogeneously distributed. Third, due to reasons such as differences in the production times of the vaccines and accessibility, patients received multiple doses of more than one vaccine instead of a single type. In addition, the number of doses of the vaccines was not the same in all patients. This makes it difficult to compare the vaccine groups one-to-one. In this study, the types of vaccines in patients who applied to our clinic were evaluated, but vaccines such as ChAdOx1 (Oxford/AstraZeneca), Ad26.COV2.S (Janssen) mRNA-1273 (Moderna), which is frequently used in other countries, was not evaluated. Finally, it was observed that there were many differences in demographic characteristics, comorbid diseases, and triggering factors of patients in the DVT and control groups. However, it is expected that the control group represents a partially normal part of society, while the DVT group includes a higher-risk group in terms of both comorbid diseases and triggering factors. Therefore, we think that this difference is normal.
Conclusion
The frequency of COVID-19 vaccination was lower in patients with DVT than in those without. Among vaccinated patients, we found that DVT was least common in patients who received only BNT162b2. Although the frequency of DVT was statistically similar in the groups that received only CoronaVac, BNT162b2 after CoronaVac, and TURKOVAC after BNT162b2, we found that the lowest rate of DVT was in the group that received TURKOVAC after BNT162b2. However, future studies with larger patient numbers are required on this subject.
Footnotes
Acknowledgements
Not applicable.
Ethical approval
This study was approved by the Istanbul Basaksehir Cam and Sakura City Hospital Ethics Committee (Decision no: 2024-276).
Informed consent
Informed consent for patient information to be published in this article was not obtained because “Because the study was designed as a retrospective study, an ethics committee decision was made regarding this.”
Authors’ contributions
Suleyman Yazici: conceptualisation, data curation, formal analysis, methodology, supervision, writing–original draft, and writing–review and editing. Osman Fehmi Beyazal: conceptualisation, data curation, formal analysis, methodology, supervision, writing–original draft, and writing–review and editing. Hasan Tezcan: conceptualisation, data curation, formal analysis, methodology, supervision, writing–original draft, and writing–review and editing.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The dataset used and analysed during the study is available from the corresponding author on reasonable request and with ethical permission.
