Abstract
The Coronavirus (COVID-19) pandemic had a huge impact on all sectors around the world. In particular, the healthcare system has been subject to an enormous pressure that has surpassed its ability in many instances. Additionally, the pandemic has called for a review of our daily medical practices, including our approach to colorectal cancer management where treatment puts patients at high risk of virus exposure. Given their higher median age, patients are at an increased risk for severe symptoms and complications in cases of infection, especially in the setting of immunosuppression. Therefore, a review of the routine colorectal cancer practices is needed to minimize risk of exposure. Oncologists should weigh risk of exposure versus the patient’s oncologic benefits when approaching management. In addition, treatment protocols should be modified to minimize hospital visits and admissions while maintaining the same treatment efficacy. In this review, we will focus on challenges that colorectal cancer patients face during the pandemic, while highlighting the priority in each case. We will also discuss the evidence for potential modifications to existing treatment plans that could reduce infectious exposure without compromising care. Finally, we will discuss the impact of the socio-economic difficulties faced by Lebanese patients due to a poor economy toppled by an unexpected pandemic.
Introduction
In December 2019, several patients presented with atypical pneumonia of unknown origin in Wuhan, China. 1 The pathogen was later identified as a novel enveloped RNA betacoronavirus, named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) due to its similarity to SARS-CoV.2,3 In March 2020, WHO declared the pneumonia outbreak of COVID-19 as a pandemic. 4 Presentations of COVID-19 range from asymptomatic, to severe pneumonia, acute respiratory distress syndrome (ARDS), and death. 5 Both upper and lower respiratory symptoms can be exhibited in cases of COVID-19 infections, owing to the virus’ ability to infect respiratory epithelial cells through angiotensin-converting enzyme 2 (ACE2), which is predominantly expressed in type II alveolar cells of the lung and several sites in the aerodigestive tract. 6 As of February 28, 2021 a total of 113,472 187 cumulative cases and 2,520,653 cumulative deaths have been reported across >15 countries. 7
On February 21, 2020, Lebanon had its first reported case of COVID-19. As of March 6, 2021, 390,070 cases have been confirmed with 4,971 deaths. 8 Unfortunately, Lebanon has a highly vulnerable population percentage with 30% being displaced persons and migrants living in underserved, crowded areas within the host community. 9 Additionally, Lebanon has a history of poor data collection, and data regarding how the pandemic affected cancer management in different hospitals in Lebanon is scarce.
While breakthroughs in COVID-19 prevention and management have been made especially with the advent of vaccines against COVID-19, the only approved treatment for severe COVID-19 is corticosteroids.10,11 The issue then becomes trying to balance the risk of acquiring COVID-19 infection versus the risk of cancer progression and death, prompting oncologists to choose between starting a treatment plan or, if delaying it, for how long. Colorectal cancer (CRC) ranks as the fifth most common cancer in Lebanon, and country that has the second highest incidence of this disease in the Middle East and North Africa (MENA) region, with more than 90% of colorectal cancer cases occurring in people aged 50 or older.12,13 This review will highlight the management of CRC and provides the available evidence for different approaches that can be adapted to the COVID-19 era, while also highlighting the socio-economic difficulties faced by Lebanese patients due to a poor economy made worse by the pandemic.
Colon cancer management
A critical component in the management of any pandemic is the care of vulnerable populations, such as cancer patients. Per the latest findings, populations who are at highest risk are individuals over the age of 60, immunosuppressed patients, and those with comorbidities such as cardiovascular diseases, diabetes and chronic respiratory insufficiency. 5 In times of a pandemic, risk versus benefit needs to be weighed on a case-by-case basis as to avoid exposing the patient to unnecessary treatment. We will attempt to address the optimal management plan for patients per their prognosis, keeping in mind that no plan can be all inclusive, and that extensive dialogue needs to occur between physician and patient.
Per recent ESMO guidelines, priorities are classified per the following (Figure 1):

ESMO guidelines for priority classification in CRC management. 14
Significant overall survival (OS) gain or substantial improvement in quality of life are indicators of high priority interventions. 14 Intuitively, clinically unstable patients should be considered part of the high priority group. Noncritical cases that can be delayed up to 6 weeks, but not for longer as to avoid potentially impacting overall outcome and benefit, qualify for the medium priority category. Stable patients in whom intervention would not affect survival outcome or quality of life are classified as low priority.
In the cancer population, patients need to frequently leave their homes for follow-up, testing, and management of their disease, leaving them exposed to different people and environments and at high risk for both community and nosocomial infections. 15 Additionally, two main factors that put cancer patients at risk for severe COVID-19 infection are their immunosuppressed state and age, as most cancer patients are over the age of 60 years. 14 A 2020 Chinese-based study showed that the majority of patients (53.6%) infected by SARS-CoV-2 were aged 50 or older, which coincides with the predilection age of colorectal cancer, that is, colorectal cancer patients might be at higher risk of acquiring COVID-19 given their average age.16,17 While hospitals worldwide attempt to isolate COVID-19 patients to specialized hospital wards, many cases are under-recognized or received delayed diagnoses exposing patients in the general medicine ward. 18
CRC is the third most common tumor in men and the second in women. 19 With 880 792 deaths estimated each year, CRC is the third most common cancer-related cause of death in the world. 17 Although local failure rates are very low in colon cancer, systemic recurrence of the disease following surgery is frequent and ultimately leads to death. 20 The staging features of the TNM classification determine prognosis of the disease.
Per ESMO guidelines, adjuvant treatment is recommended for stage III and “high-risk” stage II patients. Stage III colon cancer is defined as any T, N1-N2, and M0. It can also be divided into low risk and high risk. Low risk refers to T3-N1, while high risk refers to T4 or N2. 21 High risk stage II colon cancer is defined as T4N0 or T3N0 with the following features: perineural invasion, lymphovascular invasion, obstruction, or perforation, less than 12 lymph nodes sampled, and poorly differentiated tumor. Standard treatment for stage II colon cancer is surgical resection and anastomosis, followed by adjuvant chemotherapy only in high-risk patients as defined above.
Surgical priority in colon cancer
Surgery is a major therapeutic approach in terms of cancer survival, especially in early stage CRC. Cancer related or intervention related complications and adverse events can require immediate surgery. High priority cases for surgical intervention are bowel obstruction, bowel perforation, peritonitis, massive gastrointestinal bleeding, anastomotic leak post-surgery, and post-colonoscopy perforation or bleed. 14
Medium priority cases are non-emergent but major delays in surgery should be avoided. Primary surgery of early-stage CRC should not be postponed for more than 6 weeks due to risk of cancer progression or related complications.
Reconstructive procedures are low priority and should therefore be postponed. Prophylactic surgeries for familial cases of CRC should also be postponed until the end of the pandemic. 14
Chemotherapy in early colon cancer
As previously mentioned, frequent hospital or clinic visits will put patients at risk for exposure. In a pandemic such as COVID-19, the risk of transmission to health care workers will only increase with time. When proper personal protective equipment (PPE) implemented, transmission risk is greatly lowered, but unfortunately, PPEs are not widely available in all hospitals in Lebanon as resources have become limited with the increased infection rates.22,23 One way to circumvent exposure when possible, would be adopting the use of telemedicine more widely.
However, as no remote alternative exists for administration of I.V. medication, clinicians can assess the possibility of choosing regimens that require less in-person visits to hospitals/clinics. Depending on patients’ tumor characteristics and staging, risk versus benefit needs to be evaluated for drug administration and infection risk. While immunosuppression can be completely avoided with delay of adjuvant chemotherapy, data shows that delaying initiation of chemotherapy results in severely decreased survival outcomes. 24 The choice and duration of adjuvant treatment depends on staging, risk assessment, and prognostic markers.
Several trials assessed the use of alternative chemotherapy regimens as compared to classical regimens, highlighting the same observed benefits while decreasing hospital exposure.
Stage II colon cancer
Per the IDEA collaboration, patients with high risk stage II colon cancer may be considered for 3 months of CAPOX (also known as XELOX: Capecitabine, Oxaliplatin), as the IDEA pooled analysis showed noninferiority of 3 months of CAPOX and inferiority of 3 months of FOLFOX when compared with 6 months of FOLFOX. 25 As such, patients with high-risk stage II colon cancer can be switched to CAPOX 3-months reducing chemotherapy exposure and duration of immunosuppression without losing survival benefit.
Moreover, a 10-year follow-up on the MOSAIC Trial showed that for low-risk patients with stage II disease, the addition of oxaliplatin to 5-FU does not provide a survival benefit. Analysis for the whole stage II colon cancer population showed no benefit of adding oxaliplatin to FU/LV in either disease free progression or OS. 24 Delaying oxaliplatin, if possible, while relying on single oral agent in high risk stage 2 patients could be incorporated in the treatment plan depending on the status of the pandemic. 26
Another approach that should be considered when treating colon cancer patients, is limiting chemotherapy to only those who would objectively benefit. In the case of stage II colon cancer, a retrospective cohort study performed by Koenig et al. showed that stage II colon cancer patients that were microsatellite stable (MSS) benefited from adjuvant chemotherapy, in contrast to those that were microsatellite instable (MSI). 27 This could help stratify which population would benefit more from chemotherapy, and which patient population could avoid unnecessary immunosuppression.
Stage III colon cancer
Alternatively, the IDEA trials also looked at stage III colon cancer and the use of 3 months adjuvant chemotherapy as compared to 6 months of treatment. 28 While it did not demonstrate noninferiority in patients receiving CAPOX, the efficacy was essentially the same as OS was 81.2% (3m) versus 82.1% (6m).28,29 Therefore, the loss of less than 1% in efficacy should be discussed with the patient, while highlighting the significant reduction in toxicity afforded by the 3 months CAPOX regimen. While similar data were obtained with FOLFOX, the IDEA Trial showed that FOLFOX was associated with a 51% rate of grade 3–5 toxicity. 25
As 60% of patients with stage III colon cancer are low risk, and 40% are high risk, risk versus benefit assessment still favors CAPOX 3m in both groups. In high-risk groups, the use of novel prognostic factors such as immunoscore might be considered to aid in the choice and intensity of adjuvant therapy. However this prognostic factor was not validated in prospective randomized trials and decisions should be made on a case by case basis. 30
Another problem during the pandemic, is the inability for patients to receive their I.V. treatment without presenting to the hospital or clinic, thereby increasing infection risk. The XELOX regimen has decreased hospital visit frequency to receive medication when compared to FOLFOX, as XELOX comprises oxaliplatin IV every 3 weeks and Xeloda (Capecitabine) tablets orally versus 5-FU/leucovorin and oxaliplatin IV every 2 weeks in FOLFOX regimen. The ability to take medication orally versus intravenously, and the decreased frequency of I.V. medication hold a major advantage in convenience and decreased infection risk, without losing treatment efficacy. The NO16968 trial, compared a three-weekly capecitabine and oxaliplatin regimen (XELOX) versus bolus 5-FU/LV (Mayo Clinic or Roswell Park regimen) for stage III colon cancer and showed a 3-year DFS improvement with XELOX vs. 5-FU/LV. Similarly, a significant improvement associated with XELOX in OS at 7 years was observed. 31 However, care should be taken when choosing the XELOX regimen as Xeloda has been known to have many side effects, mostly diarrhea and hand foot syndrome (HFS), with the latter a serious dose-limiting effect, potentially leading to increased hospital visits for treatment. 32
Reducing hospital visits for early colon cancer
Grade 3–4 toxicities usually require hospitalization. As one study found, dose reduction in chemotherapy due to side effects did not affect survival. While the study showed that dose modifications by as much as 25% be made proactively, particularly in the first few cycles, to ensure that grade 3–4 toxicities do not emerge in patients on intensive regimens, it is important to weigh the risk of tumor progression with the use of lower doses. 26 We can also add prophylactic G-CSF in patients with borderline neutrophil count, or to those with recurrent neutropenia to reduce the risk of febrile neutropenia, and subsequent hospitalization. 33
Severe complications from the administration of chemotherapy, surgery or radiation therapy are high priority issues. Patients with these conditions require hospitalization. However, regular outpatient visits should be avoided. One potential approach to decrease hospital visits is switching from flushing the portcath every 4 weeks to 6–8 weeks, an initiative that has been applied at Georgetown University in cases where patients were presenting solely for flushing of their catheter. 26 In addition, unless clinically indicated, weekly blood tests should be avoided, and when done, an attempt to perform them near a patient’s residence is preferred. Radiological evaluation should only be performed in cases of suspected relapse and progression, as benefits of changing treatment approach in cases of refractory cancer outweighs the risk of COVID-19 exposure. While most outpatient visits should be performed using virtual settings, several measures should be implemented in case of in-person visits to reduce the risk of infection. These include screening patients by phone for any fever or respiratory symptoms, tracing contact exposure, assessing travel history, mandating personal protective equipment and limiting the number of visitors or accompanying family members to one person only. 34
The suggested changes to the management of early colon cancer in the COVID-19 era are summarized below (Figure 2).

Suggested changes for the management of early stage colon cancer in the COVID-19 pandemic.
Chemotherapy in advanced colon cancer
Approximately 25% of CRC patients present with metastases at initial diagnosis contributing to the high mortality rates reported for CRC (11.6% and 13% in men and women respectively). 35
Management of metastatic colon cancer depends on degree of metastasis (oligo vs non-oligo metastatic), RAS status (mutant vs wildtype), microsatellite instability (proficient vs deficient), and tumor size.
Oligometastatic colon cancer
Oligometastasis is defined as 1–3 or 1–5 metastases in one or few organs. 36 The liver is the most common site of colorectal metastasis with over half of all colorectal cancer patients developing colorectal hepatic metastases (HM).37,38 Surgical resection is the only potential curative modality for hepatic metastasis. Resectability is determined by three criteria: the ability to preserve two contiguous hepatic segments, preservation of adequate vascular flow as well as biliary drainage, and the ability to preserve adequate liver remnant. 39 Therefore, in patients with resectable live metastasis, surgery should be the first line treatment even in the COVID-19 pandemic, as it is curable. Additionally, perioperative chemotherapy has become a standard of care in many countries as the EORTC Intergroup trial 40983 showed that perioperative FOLFOX therapy in cases of resectable colorectal liver metastases showed an increase in progression free survival (PFS). 40 In contrast, there was no change in OS. 41 It is therefore reasonable to avoid perioperative chemotherapy when COVID-19 is still a risk, as we would be causing the patient immunosuppression, hospital exposure, and adverse effects when there is no proven data showing benefit for OS. When possible, combined colorectal and liver resection should be done as to avoid multiple operations and decrease exposure. A meta-analysis of 30 studies showed combined liver and colorectal resection was associated with shorter hospital stay without adversely affecting perioperative morbidity or long-term survival. 42
In addition, stereotactic body radiation therapy (SBRT) has shown efficacy in the treatment of oligometastatic colorectal cancer, forming a safe alternative during the COVID-19 pandemic. In a review done by Wild and Yamada, 43 SBRT has shown elevated 1 year local control rates in oligometastatic colorectal disease involving the liver (90%–100%), the lung (77%–100%) and spine (81%–95%).
Metastatic colon cancer
Metastatic CRC (mCRC) management requires a multidisciplinary approach. As most patients at the metastatic stage present with unresectable disease, the aim of the treatment in this group is to convert initially unresectable mCRC to resectable disease through a combination chemotherapy. 44 During this pandemic, we could recommend neoadjuvant chemotherapy in patients with mCRC who do not present with any surgical emergencies. While FOLFOXIRI is becoming the more popularly used treatment regimen, it is associated with myelosuppression and grade 3–4 neutropenia and anemia.44,45 This high-grade toxicity would result in frequent ER visits and hospitalizations, and therefore a higher risk of exposure to COVID. Several steps could be suggested to potentially reduce this risk. First, it is possible to recommend dose modifications by as much as 25% be made proactively, particularly in the first few cycles, to ensure that grade 3–4 toxicities do not emerge in patients on intensive regimens. 26 As one study found, dose reduction in chemotherapy due to side effects did not affect survival. 46 We can also add prophylactic G-CSF in patients with borderline neutrophil count, or to those with recurrent neutropenia to reduce the risk of febrile neutropenia, and subsequent hospitalization.14,26 Alternatively, we can opt to give doublet therapy with either FOLFOX or FOLFIRI to significantly reduce toxicity, but at the cost of reduced clinical efficacy as compared to FOLFOXIRI. 33 Again, treatment should be individualized and discussed on a case by case basis.
As with stages II and III of colon cancer, Capecitabine has been shown to be of equal or superior effectiveness in metastatic colon cancer to 5-FU. 47 Therefore, we can switch from I.V. 5-FU to oral Capecitabine and avoid hospital visits, keeping in mind the toxicity associated with Capecitabine. Additionally the 5-FU bolus portion from FOLFOX or FOLFIRI treatments adds significant toxicity in the form of myelosuppression, mucositis, and diarrhea, with no clear evidence of benefit. 48 Marshall et al. 26 thus propose dropping 5-FU bolus, and the discontinuation of leucovorin, in order to reduce the time in the infusion unit, and possibly decrease toxicity.
The choice of treatment regimen in mCRC also depends on RAS and BRAF mutations. Notably, the presence of activating mutations in RAS and BRAF are associated with poorer prognosis and are predictors of resistance to anti-EGFR monoclonal antibodies, cetuximab, and panitumumab, in mCRC.49,50 Since both cetuximab and panitumumab offer the same efficacy and have a similar side effects profile, either one can be used. 51 We can consider administering cetuximab every 2 weeks rather than weekly or, alternatively, panitumumab can be used as it is regularly administered every 2 weeks. Moreover, the approach to elderly patients is still controversial in regards to the intensity of treatment combination. 52 The PANDA trial, a randomized non-comparative phase II trial conducted on elderly patients above 70 years old with RAS and BRAF wildtype metastatic colorectal cancer, showed that the combination of 5-FU and leucovorin with panitumumab can form a reasonable alternative to the more aggressive combination of 5-FU, leucovorin and oxaliplatin with panitumumab, with a lower incidence of side effects related to oxaliplatin treatment. 53 As such, omitting oxaliplatin, especially in elderly, could potentially reduce the adverse events and their related hospitalizations without causing any disparities on treatment efficacy, a highly desirable finding in the setting of this pandemic.
In cases of RAS mutation, either bevacizumab or aflibercept are used. 44 However, as aflibercept is associated with increased risk for diarrhea, and neutropenia when compared to bevacizumab, we suggest the use of the latter when possible. 47 However, when bevacizumab is used as part of the first line treatment in RAS mutated mCRC, it showed no consistent statistically significant improvement in OS. 54 On the other hand, a recent trial where FOLFOXIRI, instead of FOLFOX, with bevacizumab was used in KRAS mutated mCRC, showed significant benefit in both OS and PFS. 55 However, it had greater grade 3 toxicities, especially febrile neutropenia. We propose therefore, prophylactic initiation of G-CSF if this regimen is followed, especially in the setting of a previous episode of febrile neutropenia. 56 As with every drug, adverse effects need to be considered. 57 One study found that bevacizumab-related bowel perforations could be reduced by excluding patients with a history of bowel obstruction, or ileus. Perforation more likely occurred during the early dosing of bevacizumab, rather than during maintenance or off treatment, therefore stricter monitoring should be implemented with initial dosing. 58
Second- and third-line treatments
When first line treatment with FOLFOX fails, FOLFIRI with bevacizumab is commonly used, and has shown significant improvement in OS.59,60 While XELIRI can replace FOLFIRI without compromising efficacy, and the replacement of I.V. 5-FU with oral capecitabine is an attractive alternative to decrease hospital exposure, XELIRI has a much higher risk of diarrhea and vomiting when compared to FOLFIRI. 61 In this case, it is better to adhere to intravenous 5-FU, as severe diarrhea would likely increase emergency department visits and would be counterproductive to our objective.
Microsatellite instability (MSI) testing is also crucial for treatment decision. Prior to the results of Keynote 177, patient with MSI high (MSI-H) who failed prior chemotherapy were given immunotherapy. However, the Keynote 177 results presented in ASCO 2020 suggested that pembrolizumab might be considered even as a fist line treatment in the setting of MSI-H mCRC. In fact, The KEYNOTE-177 trial demonstrated that pembrolizumab almost doubled PFS in MSI-H/dMMR mCRC when compared to chemotherapy. 62
Concerning third line treatment, while Regorafenib and Trifluridine/Tipiracil are two options, OS benefit is small.63,64 Trifluridine/Tipiracil, specifically, has a high association with neutropenia. 64 We recommend in cases where first- and second-line treatments fail, to proceed to palliative care as the clinical benefits of third line are minimal compared to the higher health risk in the COVID-19 era. Testing for HER-2 in RAS wildtype patient could also offer a potential alternative in this setting, as targeting HER 2 has been shown to be a successful therapeutic strategy in the treatment of refractory mCRC.65–67 In cases of BRAF mutation, prognosis is especially poor in the setting of a well-known poor response to chemotherapy. We should therefore consider deferring surgery and considering treatment with Cetuximab and Encorafenib as second line treatment based on the results of phase III BEACON trial or even as first line treatment based on phase II ANCHOR trial that combined Cetuximab, Binimetinib and Encorafenib with promising results till date. 68 Both Encorafenib and Binimetinib are administered orally, and patients are therefore able to receive them at home. Even though Cetuximab is administered only through IV, it is received once weekly and would still minimize patient exposure. 69
In addition, a recent study showed significant differences in clinical characteristics, anatomic structure, and the genetic mutation profile between left- and right-sided CRC. 70 Retrospective analyses showed that RAS WT (wild type) right-sided mCRC, when compared to RAS WT left-sided mCRC, is associated with a poor prognosis, independent of treatment. 71 Depending on patient characteristics, and disease burden, we might suggest palliative care in patients with right sided mCRC as combination of chemotherapy and bevacizumab/panitumumab has been shown to have very limited efficiency and would expose patient to unnecessary myelosuppression and other adverse events, on top of COVID-19. Additionally, a retrospective study comparing the use of FOLFOXIRI versus doublet regimen in right sided mCRC, showed that triplet regimens may have potential detrimental effect on OS as they are associated with higher toxicity and early development of therapeutic resistance, which might impair the continuity of care, potentially influencing OS. Patients should therefore be carefully selected for FOLFOXIRI regimen. 72
Maintenance therapy
As for maintenance therapy, in the CAIRO-3 study, maintenance with capecitabine plus bevacizumab after six cycles of capecitabine, oxaliplatin, and bevacizumab (CAPOX-B) in patients with metastatic colorectal cancer was found to be effective without compromise quality of life. 73 Two OPTIMOX trials were also conducted. In OPTIMOX-1, continuous oxaliplatin, and fluorouracil were compared with a novel strategy of planned oxaliplatin breaks, but with continuous fluorouracil. 74 In OPTIMOX-2, the OPTIMOX-1 intermittent oxaliplatin strategy was compared with a complete chemotherapy-free interval strategy. 75 Neither trial showed a significant reduction in survival with intermittent therapy, and OPTIMOX-2 favored the continuation of fluorouracil during breaks. 75
Though 5-FU in combination with bevacizumab is commonly used in maintenance therapy, data showed increase in PFS, but no clear OS benefit. 76 We could therefore delay maintenance therapy in these patients. In addition, while standard maintenance therapy includes capecitabine with or without bevacizumab, we should consider single agent oral capecitabine especially in Ras wildtype patients. 77 This has been also suggested and implemented in several cases in China. 78 Finally, it is also advisable in the COVID-19 era to give maintenance therapy every three instead of two weeks given the long half-lives of the drugs. 77
Patients with poor prognosis are high priority for palliative management rather than aggressive treatment. Treatment and maintenance therapy should be delayed in patients with low disease burden and slow progression. Patients with several comorbidities should ideally be delayed treatment, similarly to those who had severe complications from treatment. As with early colon cancer, blood tests, when clinically indicated, should be performed near the patient’s home, and follow up through telemedicine is preferred.
Figure 3 summarizes the suggested guidelines for the treatment of advanced colon cancer in the COVID-19 era.
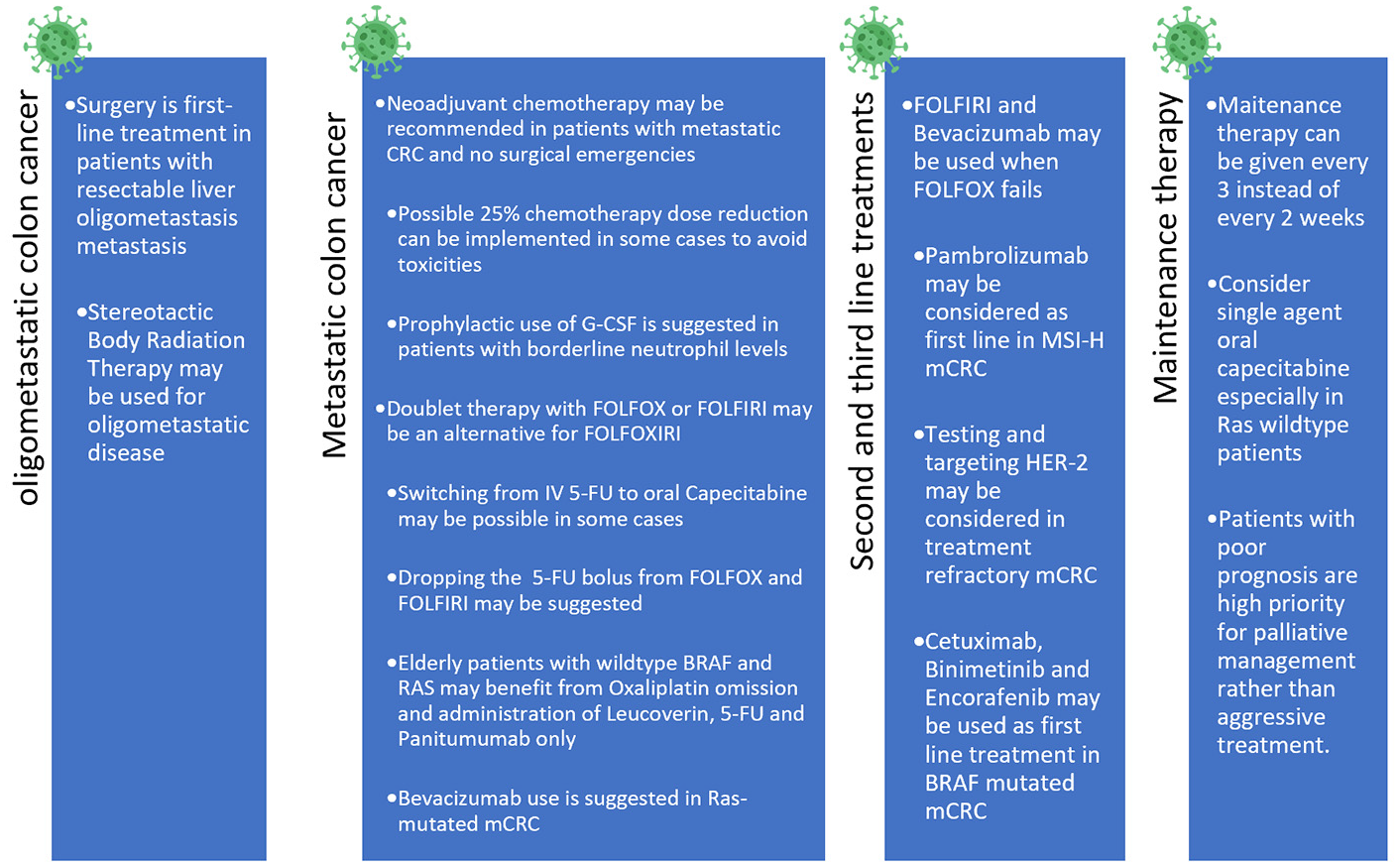
Suggested guidelines and changes for the treatment of metastatic colon cancer during the COVID-19 pandemic.
Rectal cancer management
Very early cT1N0, with low grade (G1/G2) rectal cancer should be treated with local excision. 79 Surgery should not be delayed as it is curative.
The current standard of care for locally advanced rectal cancer includes chemoradiation therapy, followed by total mesorectal excision and post-operative adjuvant chemotherapy. Despite improvement of local control, this treatment modality did not show any added benefit on OS, with a cumulative distant metastasis incidence of 30% in 10 years. 80 As a result, we suggest switching to total neoajuvent therapy (TNT), a regimen that consists of delivering short course radiotherapy and six cycles of chemotherapy in the preoperative setting followed by surgery. In fact, results of the RAPIDO trial presented in ASCO 2020 showed lower disease-related treatment failure, lower distant metastasis rate and doubling of pathologic complete response in TNT compared to standard of care. 81 TNT would, therefore, provide improvement in local control while allowing a delay in surgery. According to data recently reported by the OPRA trial in ASCO 2020, patients with clinical complete response documented by repeated digital rectal exam, flexible sigmoidoscopy with biopsy and MRI, can be offered a watch and wait (W&W) strategy, allowing for organ preservation and decreased hospital admissions and surgical procedures. 82 In fact, the International Watch & Wait Database (IWWD) reported 1009 cases who underwent “watch and wait” strategy after neoadjuvant treatment between 2015 and 2017 and found the 5-year overall and disease-free survival were 85% and 94%, respectively, and complete response was found in 80.7% of patients. 83 As such, we suggest the W&W approach versus TME in patients with CR or near CR. Per the NCCN Clinical Practice Guidelines, assessment for tumor response should be done at week 5–12 from the end of neoadjuvant treatment. 84 It is possible to delay the timing of the operation to 16–24 weeks after neoadjuvant therapy, provided a greater control of the pandemic is expected within that time frame. 85
The standard approach for preoperative radiation for rectal cancer is a treatment delivered daily over 5–6 weeks. Based on the Polish II trial and TROG 01.04 trials, no difference in local recurrence rates and 3-year OS were noted between short vs long course chemoradiation therapy in locally advanced rectal cancer.86,87 As a result, and when appropriate, short-course radiation use is recommended for neoadjuvant treatment of rectal cancer. Treatment in five fractions instead of 28 dramatically decreases the patient’s exposures and allows for better patient accommodation, when resources become constrained due healthcare workers becoming unavailable from illness and hospitals becoming overwhelmed.
Another area of controversy is whether patients with high rectal cancer, located 10 cm or more above the anal verge, would benefit from radiotherapy. Based on the results of the Dutch trial, no statistically significant difference in local recurrence rate was noted between radiotherapy followed by TME and TME alone in rectal cancer located 10 cm or more from the anal verge. 88 As a result, we can consider decreasing treatment duration and radiotherapy side effect in the middle of the pandemic by going directly to upfront surgery in high rectal tumors.
Figure 4 summarizes the suggested guidelines for the treatment of rectal cancer in the COVID-19 era.

Suggested guidelines for the treatment of rectal cancer during the COVID-19 pandemic.
Remote follow-up and telemedicine
Cancer patients are usually prone to develop complications related either to their primary disease or to their treatments. 89 These symptoms constitute a frequent cause of hospitalization and contribute to a decrease in survival outcome. 89 As such, frequent outpatient follow-up is required in order to reduce the frequency and severity of such complications. 90 On the other hand, outpatient visits are not encouraged during this pandemic in order to reduce patients’ exposure. 91 As such, the implementation of telemedicine has offered a successful solution for this dilemma, allowing patients to be frequently and remotely followed-up without breaching the required “social distancing”.26,91 An attempt to implement telemedicine at Georgetown University for CRC patient follow-up was met positively from both patients and their providers. 26 While some elements from face-to-face interactions cannot be replicated remotely, the protective benefits outweigh these losses. While this has reduced the infectious exposure, concerns were raised about the ability of remote follow-ups to effectively monitor laboratory results, vital signs as well as other clinical aspects as compared to outpatient visits. This is why we encourage the use of tools that respect social distancing while maintaining connectivity to the healthcare system and providing objective data for management. 92 For example, several electronic patient-reported outcomes (ePRO) in oncology patients were found to be successful in reducing the grade and severity of adverse events as well as in improving survival outcomes.93–95 These include electronic follow-up applications, telephone-based daily symptom reporting, wearable patches and many others.93,96
Palliative care
Palliative care provides essential support to patients with an emphasis on symptomatic control, and psychological and social support. Palliative care is provided through an interdisciplinary approach with good communication that enables the patient to be involved in any decision-making about care and treatment. 97 Providing palliative care during pandemics poses a unique challenge as patients who require palliative and hospice care during their end of life can surpass the capacity of palliative care teams. 98 The COVID-19 pandemic has introduced several challenges to the management of patients requiring palliative care, given the need to limit public exposure and risk of infection. Below are the challenges faced by patients in different clinical settings and suggested solutions (Table 1).99,100
Palliative care challenges for patients in the COVID-19 era.

Colorectal cancer management in other countries
Almost a year after the pandemic, a study in England looked at the impact of COVID-19 on the diagnosis and management of CRC. 101 The authors found a sustained reduction in the number of people referred, diagnosed, and treated for CRC, with major changes in the delivery of treatment. The number of surgical interventions fell sharply, and surgical methods were adapted to minimize COVID-19 risk. At the same time, a higher-than-expected number of patients received stomas as a temporary solution which could have a detrimental impact on long-term quality of life. Laparoscopic procedures were also lower than in previous years. In rectal cancer, surgical interventions were also decreased along with a significant increase in the use of short-course radiotherapy, potentially lowering quality of life as radiotherapy is associated with greater morbidity when combined with radical surgery without influencing survival rates. 101
In Tunisia, management of CRC was adapted to decrease infection risk and treatment was done only when benefit outweighed the risk of death from COVID-19. 102 Localized colon cancer was surgically treated in the same timeframe as before the COVID-19 pandemic. For adjuvant chemotherapy, protocols with oral chemotherapy were preferred such as CAPOX instead of FOLFOX to decrease hospital admission duration and hospital visits as compared to infusional 5-FU-based regimens. The period between two courses was also increased from 3 weeks to 4 weeks for capecitabine-based regimens. Management of stage III colon cancer involved eight cycles of CAPOX, while high-risk stage II patients received eight cycles of oral capecitabine only. 102 In locally advanced cases, NAT with the CAPOX regimen instead of FOLFOX was favored, with a delay in surgery until after the pandemic period. 102 For rectal cancer, a pre-operative short course radiotherapy without chemotherapy followed by four cycles of CAPOX and a delayed surgery were the preferred approach as to reduce the number of hospital visits. 102 Short course radiotherapy with delayed surgery was also favored in elderly patients (>75 years) with locally advanced disease. Home based care was reserved for patients unable to withstand intensive chemotherapy and with poor performance status. For metastatic CRC, CAPOX was the preferred regimen. For patients on regimens with high risk of neutropenia, they were given G-CSF with possibility of maintenance therapy or break in therapy if they lived far from the hospital. 102 Single oral capecitabine was the preferred approach in patients with significant comorbidities. 102
In China, changes in management of CRC were also implemented at the start of the COVID-19 pandemic (Table 2). 103
Recommended treatment strategies for colorectal cancer cases during the COVID-19 pandemic in a Chinese institute. 103

Socio-economic obstacles in Lebanon
The Lebanese economic instability started in November 2019, leading to a state of total national economic blockage by January 2020. The Lebanese currency started losing up to 10 times its value, leaving many Lebanese citizens below the poverty line, unable to afford basic food and health services.
The economic crisis was only accentuated by the COVID-19 pandemic, which led to a country wide shut down. The fear from the pandemic, and the inability to cover health care costs led to some patients delaying their clinic/hospital visits until they worsened enough to need an emergency department visit.
The financial difficulties in Lebanon left some patients unable to receive their cancer treatments, and if they did, were at risk of COVID-19 infection from hospital exposure. Healthcare service have been overwhelmed by the extraordinary influx of patients, often requiring high intensity of care. In addition, Lebanon has the highest number of refugees per capita in the world which further strains the Lebanese public healthcare system that is struggling to accommodate the shifted load of medical care from the private sector amid the economic crisis.104,105 Additionally, the severe infrastructural damage caused to several cancer care centers after the devastating Beirut blast on the 4th of August, has delayed the treatment of many cancer patients. 106 Amidst all the chaos, social distancing and hygiene measures have lost priority among the Lebanese people, further increasing exposure risk. 106 The Lebanese Society of Medical Oncology (LSMO) has issued several recommendations for cancer patient care, stressing the necessity to prioritize patients on a case-by-case scenario favoring screening for infections, telemedicine, oral rather than intravenous and curative rather than palliative therapy. 107 Unfortunately, Lebanese hospitals were not able to fully implement those guidelines for multiple reasons, including the reluctance of patients in delaying or modifying their treatments plans. 34 While the Pfizer/Biontech vaccine has entered the country, the vaccination rate in Lebanon has been slow with only 46,971 people vaccinated as of February 27, 2021. 108
Conclusion
The approach to CRC management during the COVID-19 pandemic necessitates modification and adaptation to the current situation as to limit infection risk. Risk versus benefit need to be weighed on a case-by-case basis, and multidisciplinary approach should be integrated for optimal management. Prognosis and quality of life dictate priority. As the pandemic is yet to end, governments need to take measures to prevent spread, protect hospitals from becoming overwhelmed, and provide uncompromised care for all patients. In Lebanon control has been poor with a recent spike in cases and increase in the number of deaths. Stricter measures need to be implemented; a task that has been proven to be hard due to unprecedented economic difficulties facing the country.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
