Abstract
Objective
Chronic obstructive pulmonary disease is a leading cause of global mortality. Although exercise training improves lung function and quality of life in chronic obstructive pulmonary disease patients, its mechanisms remain unclear. This study aims to reveal the effects of exercise on chronic obstructive pulmonary disease and elucidate the underlying molecular mechanisms involved.
Methods
This pre-post observational cohort study included 11 chronic obstructive pulmonary disease patients who underwent a 12-week exercise training program to evaluate the effects of exercise intervention. A cigarette smoke-induced chronic obstructive pulmonary disease mouse model and a cigarette smoke extract-induced bronchial endothelial cell model were utilized to investigate the protective mechanisms of exercise. Next-generation sequencing, bioinformatics analyses, luciferase reporter assays, and RT-qPCR were employed to assess mRNA and miRNA expression levels and correlation. Bioinformatics analysis and luciferase reporter assays were performed to identify direct downstream targets of miR-124-3p.
Results
Exercise ameliorated lung inflammation in chronic obstructive pulmonary disease mouse lung tissue and human peripheral blood leucocytes. It inhibited M1 macrophage activation in response to cigarette smoke extract and led to the upregulation of miR-124-3p expression. Overexpression of miR-124-3p attenuated cigarette smoke extract-induced lung injury and inflammatory responses. Luciferase reporter assays identified endoplasmic reticulum to nucleus signaling 1 as a direct downstream target of miR-124-3p, and endoplasmic reticulum to nucleus signaling 1 overexpression reversed the anti-inflammatory effects mediated by miR-124-3p.
Conclusions
Exercise improved patient endurance and alleviated emphysema-related inflammation. It increased miR-124-3p expression, which inhibited the M1 macrophage phenotype and attenuated smoking-related lung injury by targeting endoplasmic reticulum to nucleus signaling 1.
Introduction
Chronic obstructive pulmonary disease (COPD) results in over 3 million deaths worldwide each year.1,2 It is characterized by persistent respiratory symptoms and a reduction in physical activity, which is reduced due to cachexia and airflow limitations. It is primarily caused by exposure to noxious gases and particles, with smoking being the leading risk factor. 3 However, despite the advancements in pharmacological treatments such as antibiotics, triple inhaled therapy, and alpha-1 antitrypsin augmentation therapy over recent decades, effective clinical treatment options for COPD remain lacking. 4 Therefore, it is imperative to develop additional complementary therapies to enhance the clinical outcomes of individuals with COPD.
An increasing number of studies have found that exercise serves to uphold and restore homeostasis across various levels of the organism, from tissues and cells to molecules. Its potential extends to the prevention or inhibition of a broad spectrum of illnesses, including COPD.5,6 Some studies suggest that exerkines, which are defined as humoral factors responsive to exercise, can improve physical capability, diaphragm function, and cognitive function in COPD patients,7,8 and may even reduce chronic inflammation associated with COPD through micro(mi)RNA-mediated microenvironmental regulation8–10. MiR-124 has been shown to be downregulated in lung cancer tissues, acute lung injury, and in patients with COPD combined with pulmonary arterial hypertension (PAH), and is associated with inflammation.11–13 Furthermore, it has been shown that miR-124-3p can suppress inflammatory responses, inhibit pulmonary fibrosis by regulating the TGF-β/Smad signaling pathway, and protect against traumatic acute lung injury.12,14,15 Exercise has been proposed as a potential modulator of miRNA expression, including miR-124, with implications for inflammatory responses and immune regulation. MicroRNAs such as miR-124 have diverse roles in biological processes. Exercise may protect against COPD through multiple pathways; here, we investigate miR-124's regulation of inflammation as a potential mechanism of exercise-induced protection.
In addition to its role in immune modulation, miR-124 has been shown to influence various cellular processes, such as differentiation, apoptosis, and tissue repair, in multiple contexts.16,17 Its ability to regulate macrophage polarization, shifting from the pro-inflammatory M1 macrophages (M1) to the anti-inflammatory M2 macrophages (M2), highlights its potential therapeutic value in inflammatory lung diseases like COPD. 18 Pulmonary macrophages play a crucial role in orchestrating lung tissue inflammation during COPD development by recruiting pro-inflammatory innate immune cells and activating adaptive immune cells. 19 Macrophage polarization, particularly the shift towards a pro-inflammatory M1 phenotype, is a key feature of COPD pathology. Studies have demonstrated that miR-124, known for its neuroprotective and anti-inflammatory properties, can regulate the expression of transcription factors such as NF-κB, IRF5, and STAT1, thereby inhibiting the polarization and inflammation of M1 macrophages.20,21 This regulation suggests that miR-124 could play a protective role in inflammatory diseases, including COPD, by modulating the immune response.
Despite the identification of various molecules and pathways involved in pulmonary macrophage dysfunction in COPD pathogenesis, the specific effects of exercise training on modulating macrophage phenotype remain unclear. Therefore, the present study aims to investigate the relationship between exercise capacity and inflammatory biomarkers following exercise training in COPD patients, as well as to elucidate the miRNA regulatory mechanisms involved in the disease.
Materials and methods
Patients and exercise training protocol
Eleven COPD patients were recruited consecutively from a clinical cohort study conducted at Pudong New Area Gongli Hospital (registration number: ChiCTR2100053232, 11-16-2021). COPD diagnosis was made based on the Global Initiative for Chronic Obstructive Lung Disease (GOLD) criteria. Participants exhibited no significant cardiac dysfunction, active infections (e.g. hepatitis and tuberculosis), or contraindications to exercise such as neurological or psychiatric disorders. This prospective observational cohort study was approved by the Ethics Committee of Shanghai Pudong New Area Gongli Hospital (Approval No. 2021 [028]), and all participants provided written informed consent for the use of their blood samples for scientific purposes, in accordance with the Helsinki Declaration of 1975 as revised in 2024. All patient details were fully de-identified to protect privacy and ensure confidentiality. The reporting of this study conforms to the STROBE guidelines for observational studies. 22
Patients underwent training on a Cycle Ergometer (Qianjing 20003, China) over 3 days, with varied adaptive loads determined by cardiopulmonary exercise testing (CPET) and continuous functional assessment. Following the adaptive phase, exercise sessions began on the fourth day and continued for 12 weeks. Exercise sessions lasted 1 hour per day, 5 days per week, with the 1-hour duration divided into 3 to 4 sessions per day, depending on the patient's tolerance. The exercise intensity was individualized to a moderate level, quantitatively calculated based on CPET parameters (Δ50% load ±10 W). The formula9,22 for Δ50% load was as follows: Δ50% load = (load at anaerobic threshold–increasing load per minute × 0.75/2 + (peak load–increasing load per minute × 0.75)/2.
Calculation of the emphysema CT score
The calculation of the emphysema CT score is performed using ITK-SNAP for image segmentation and quantitative analysis. First, import the patient's pre- and post-exercise CT scan data (DICOM format) and use ITK-SNAP's manual or semi-automatic segmentation tools to label the emphysema regions. In manual segmentation mode, use the Brush Tool or Polygon Tool to label each slice. In semi-automatic mode, apply the Active Contour Segmentation method to optimize the selection. After segmentation, use ITK-SNAP's Statistics tool to extract the area (A
i
) of the emphysema region in each slice and calculate the total volume. Finally, calculate the CT score using the following formula: Score =
Animal experiments and treadmill aerobic training
C57BL/6 male mice (4 weeks old, 18–20 g) were purchased from Shanghai Jihui Laboratory Animal Care Co., Ltd (Shanghai, China) and maintained under pathogen-free conditions at 23 °C ± 2 °C under a 12-h light/dark cycle. The mice were fed a standard diet with free access to water. A total of 10 mice were used, with five mice per group. Due to mortality during the modeling process, 3–4 mice per group were included in the final analysis. All efforts were made to minimize the number of animals used and to reduce their pain and distress, according to the Guide for the Care and Use of Laboratory Animals (eighth edition).
CS exposures were conducted using the CSM-100C inhalation exposure apparatus (TOW-int tech, Shanghai, China) with Huangshan Brand cigarettes (China Tobacco Anhui Industrial Co., Ltd, Bengbu, Anhui, China), exposing both exercised and non-exercised control mice to CS (equivalent to 18 cigarettes/day). Mimicking human smoking habits, the exposures were administered once daily in 1-hour sessions, 5 times per week, over a 12-week period. All animal procedures were approved by the Animal Ethics Committee of Shanghai University of Medicine & Health Sciences (Shanghai, China) (Approval No. 2022-SJK-18-6501021970010 21622, 1 January 2022) and were conducted in accordance with institutional guidelines. This study was reported in accordance with the ARRIVE 2.0 guidelines. 23
All male mice in the exercise + smoking group were subjected to treadmill exercise using a motor-driven treadmill (TMS-8B; Melquest, Toyama, Japan). Following a 5-minute preparation period during which the speed gradually increased, the mice underwent treadmill exercise according to the same protocol each day, consisting of a 30-minute session at a speed of 18 m/min with a 0° incline, repeated 5 times per week. Treadmill aerobic training was conducted for 12 weeks prior to exposure to cigarette smoke. The exercise protocol was performed as described previously. 24 Mice were anesthetized with 1.5%–2.0% isoflurane and euthanized by cervical dislocation. Death was confirmed by the absence of respiration and reflexes. The procedure was performed by Dr Jin (Shanghai Qingjin Biotechnology Co., Ltd). We would like to express our gratitude to him.
Strand-specific RNA-Seq library construction and high-throughput RNA-Seq
Peripheral blood (3 mL) was collected from COPD patients and healthy controls before and after exercise. Leukocytes were isolated within 2 hours using Pancoll density gradient centrifugation from EDTA tubes. Total RNA was extracted from leukocytes using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). A total of 3 μg of RNA from each sample was used for RNA library construction using the KAPA Stranded RNA-Seq Library Prep Kit (Roche, Basel, Switzerland), followed by deep sequencing on the Illumina HiSeq 4000 platform at Aksomics, Inc. Human small RNA sequencing and analysis were performed by OE Biotech Co., Ltd (Shanghai, China).
Preparation of CS extract (CSE)
For CSE preparation, cigarettes were burned slowly, applying a constant speed vacuum pump to pull the cigarette smoke through 10 mL of RPMI 1640 medium(37 °C), which was then passed through a 0.22-μm filter for sterilization(Schleicher & Schuell GmbH, Dassel, Germany). The obtained CSE solution (considered as 100% CSE) was diluted with a medium to a working solution of a desired concentration, and used for experiments within 1 hour.
RAW 264.7 culture
The murine macrophage cell line RAW 264.7 (Chinese Academy of Sciences, Shanghai, China) was cultured in RPMI 1640 medium supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin, then incubated in 5% CO2 at 37 °C. For COPD induce, RAW 264.7 were cultured in RPMI-1640 medium and exposed to 4% CSE for 48 hours.
Dual-luciferase reporter assay
The 3′ untranslated region (3′-UTR) sequence of the gene encoding endoplasmic reticulum to nucleus signaling 1 (ERN1) was inserted into the pMIR-REPORT luciferase reporter vector. We performed RAW 264.7 cell co-transfections with 0.1 μg luciferase reporter vector and miR-124-3p mimic or negative control RNA (NC) using Lipofectamine 2000 (Invitrogen). The Dual-Luciferase Reporter Assay System (Promega, Madison, WI, USA) was employed to explore relative luciferase activity by normalizing firefly luminescence to Renilla luminescence at 48 h post-transfection.
Western blot assay
Lysed cells were centrifuged at 12,000 r/min at 4 °C after adding a protease inhibitor. Then, protein concentrations were determined using a Pierce bicinchoninic acid assay kit. The proteins were resolved by 10% SDS-PAGE before transferring them to PVDF membranes. To assay protein expression, primary antibodies against iNOS (Santa Cruz Biotechnology, Dallas, TX, USA) and β-actin (Sigma-Aldrich, St Louis, MO, USA) were applied before adding horseradish peroxidase-conjugated secondary antibody (Abcam, Cambridge, UK). Finally, an ECL chemiluminescent kit (Millipore, Billerica, MA, USA) was utilized to visualize the protein bands. Western blotting was performed with three independent biological replicates, and representative blots are shown.
Enzyme-linked immunosorbent assay (ELISA)
Expression of inflammatory factors interleukin (IL)-1β, IL-6, and tumor necrosis factor (TNF)-α in serum or in RAW 264.7 cell supernatants was tested utilizing ELISA kits (Sen-Xiong Company, Shanghai, China) following the manufacturer's protocols.
miRNA isolation and real-time PCR
Total RNA, including miRNA, was isolated from cells or tissues using RNAprep Pure Kit (DP443, TIANGEN, Beijing) according to the manufacturer's protocol. RNA concentration and purity were assessed using a NanoDrop spectrophotometer (Thermo Fisher Scientific, USA). For cDNA synthesis, 1 μg of total RNA was reverse transcribed using the Mir-X miRNA First-Strand Synthesis Kit (Takara, 638313) for miR-124, and a reverse transcription kit (Promega, Madison, WI, USA) for mRNA. Real-time PCR was performed using a SLAN-96S PCR System(Bio-) with 2×SYBR Green PCR Master Mix (Solarbio, Beijing, China). U6 snRNA and GAPDH were used as internal controls for miR-124 and mRNA, respectively. The relative expression levels of miRNAs and mRNAs were calculated using the 2−ΔΔCt method. All experiments were performed in quadruplicate to ensure reproducibility. Primer sequences are listed in Supplemental Table S1.
Immunofluorescence analysis
Immunofluorescence staining was performed as previously described. Lung tissue specimens were fixed in a 4% paraformaldehyde solution and subsequently embedded in paraffin. Sections (5 μm) were incubated overnight with primary antibodies against iNOS and F4/80 at 4 °C, followed by incubation with secondary antibodies (Abcam) for 1 hour at 37 °C. Staining was visualized using 3,3'-diaminobenzidine (DAB), and sections were photographed using a digital camera. Apoptosis was detected using a TUNEL assay kit to quantify apoptotic cells. The sections were examined with an Axiophot light microscope (Zeiss, Oberkochen, Germany) and photographed with a digital camera.
Statistical analysis
Quantitative data are presented as means ± standard deviation (SD) or medians with interquartile range [IQR], while qualitative data are reported as absolute frequencies. Statistical analyses were performed using GraphPad Prism (La Jolla, CA, USA) to evaluate differences between groups. For comparisons between two groups, a paired two-tailed Student's t-test was applied, whereas one-way ANOVA followed by a Bonferroni post-hoc test was used for analyses involving three or more groups. Spearman correlation analysis was conducted to assess associations between continuous variables. A p-value ≤ 0.05 was considered statistically significant. All statistical tests were two-sided.
Bioinformatic analyses were performed using R software (version 4.3.3; The R Foundation for Statistical Computing, https://www.r-project.org/). Differential expression analysis of miRNAs was conducted using the DESeq2 package. Genes with a |log2 fold change| ≥ 1 and adjusted p-value < 0.05 were considered significantly differentially expressed.
Results
Exercise training enhances exercise tolerance and circulatory function in COPD patients
Accumulating studies have demonstrated that exercise can enhance physical performance, alleviate shortness of breath, and improve the quality of life in patients with COPD by reducing inflammation and oxidative stress.23,24 In our study, we recruited 11 COPD patients and conducted a 12-week exercise training program. The patients’ lung function and baseline clinical characteristics are summarized in Table 1.
Clinical information of patients before and after exercise.
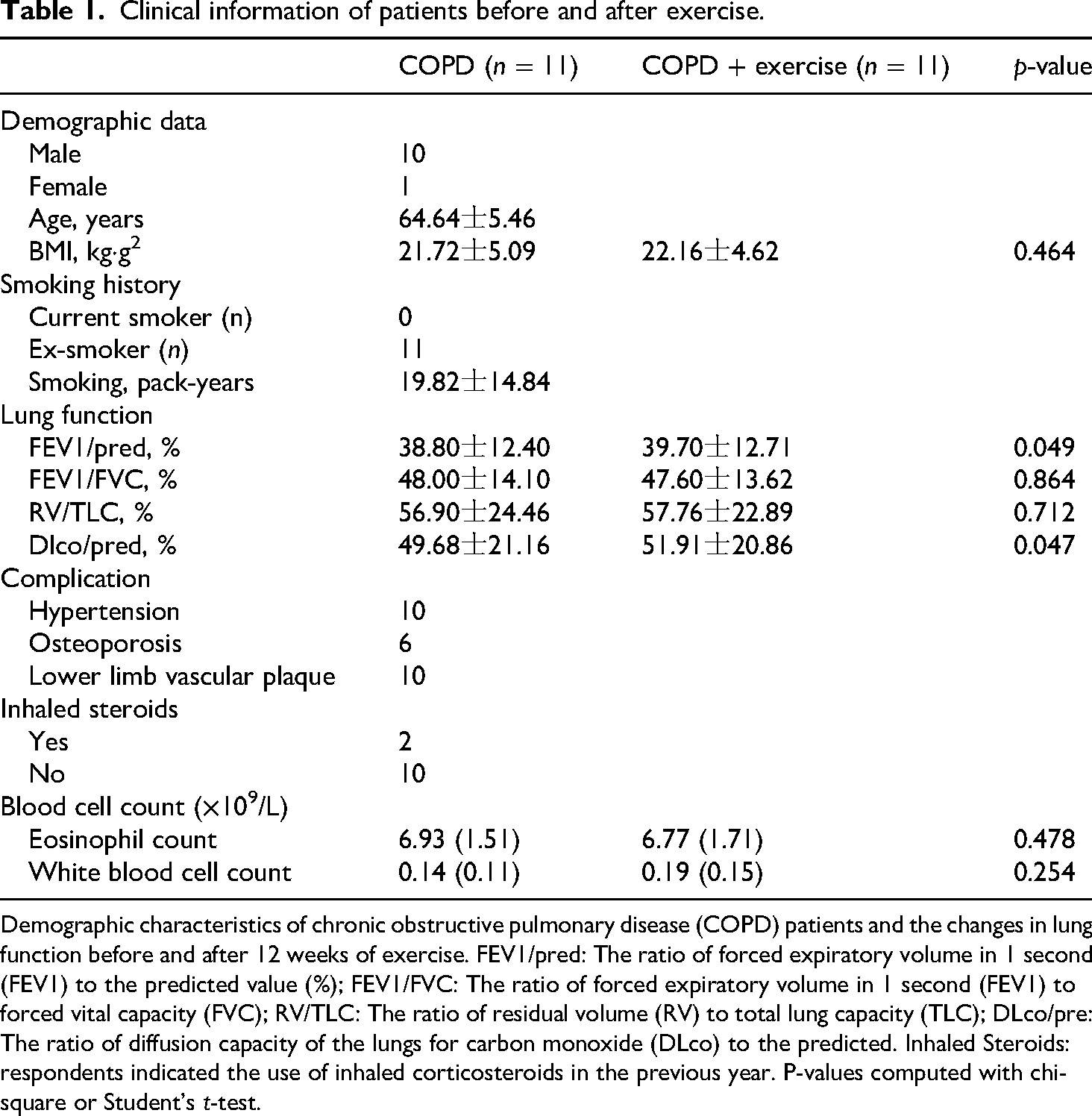
Demographic characteristics of chronic obstructive pulmonary disease (COPD) patients and the changes in lung function before and after 12 weeks of exercise. FEV1/pred: The ratio of forced expiratory volume in 1 second (FEV1) to the predicted value (%); FEV1/FVC: The ratio of forced expiratory volume in 1 second (FEV1) to forced vital capacity (FVC); RV/TLC: The ratio of residual volume (RV) to total lung capacity (TLC); DLco/pre: The ratio of diffusion capacity of the lungs for carbon monoxide (DLco) to the predicted. Inhaled Steroids: respondents indicated the use of inhaled corticosteroids in the previous year. P-values computed with chi-square or Student's t-test.
Following the exercise training, although pulmonary ventilation function showed no significant improvement (Figure 1(a) to (c)), patients exhibited a notable enhancement in exercise endurance. The improvement in VO₂ was 114.36 ± 45.3 mL/min, and both exercise duration and maximum load capacity also showed notable enhancements (Figure 1(d) to (f)). The average respiratory exchange ratio (RER) at the anaerobic threshold was 0.84, with a slight increase post-exercise, although this change was not statistically significant. In terms of circulatory function, most participants exhibited an upward trend in oxygen pulse after exercise (Figure 1(g) to (i)). The pre- and post-exercise CT scan images of a representative patient are shown in Figure 1(j) and (k). Radiological assessment by a specialist indicated an improvement in localized inflammation after exercise. The CT score of COPD patients significantly improved after exercise, decreasing from a pre-exercise mean of 17.16 to a post-exercise mean of 15.41 (p < 0.05). These results suggest that exercise training can improve both exercise tolerance and clinical imaging findings in COPD patients.
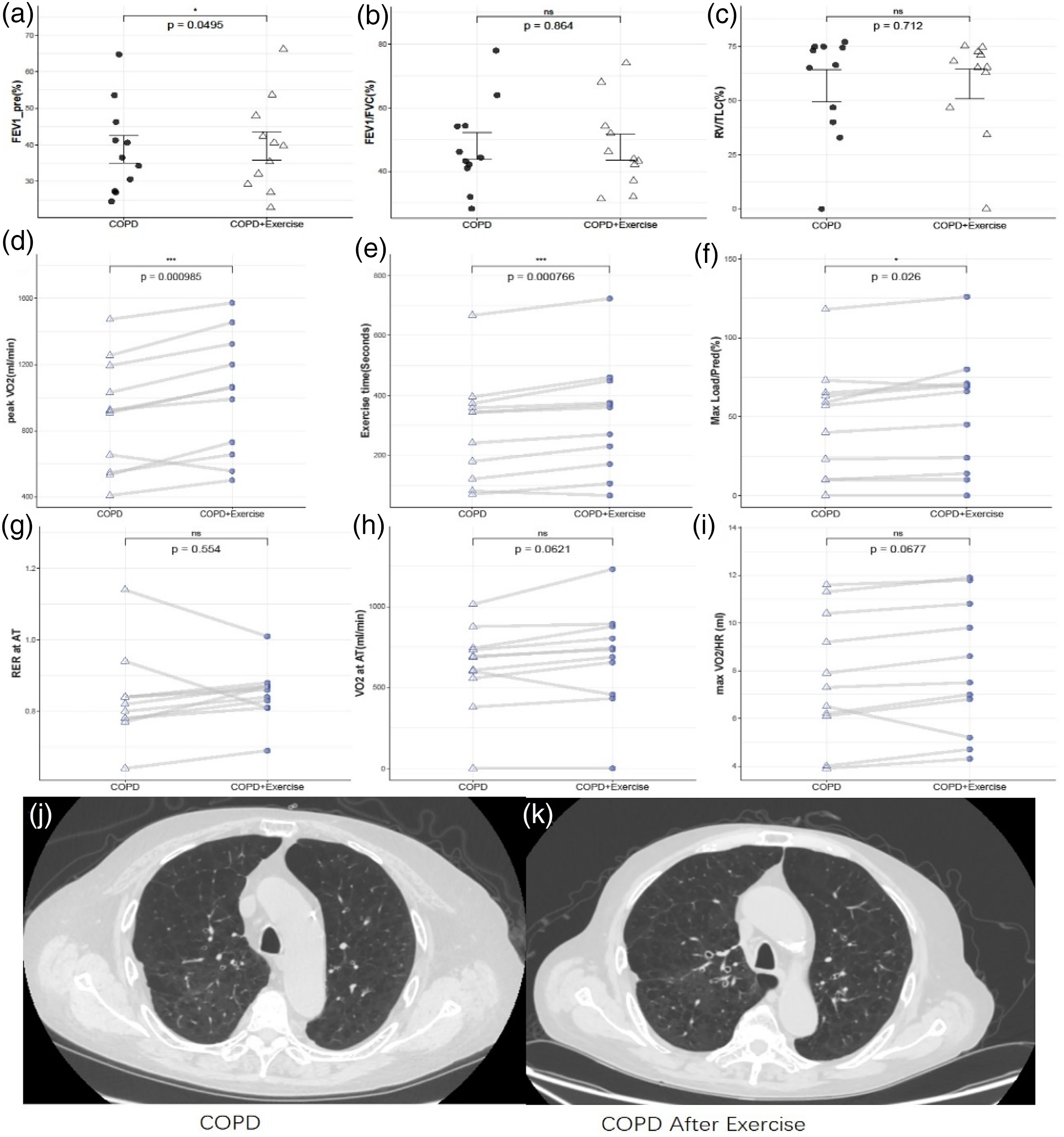
Changes of exercise endurance, circulation and metabolic function in patients with COPD after exercise training. (a–c) No significant improvement in pulmonary ventilation function before and after exercise. (d–f) A significant increase in exercise duration, endurance, and peak oxygen uptake in COPD patients after exercise. (g and h) A trend toward delayed anaerobic threshold in some patients. (i) A partial increase in oxygen uptake per stroke after exercise training. (j) Pre-exercise CT scan of a COPD patient, (k) post-exercise CT scan of the same patient. Panel j shows slightly reduced density of pulmonary vascular bronchial bundles, increased lung field radiolucency, and a slight decrease in localized cystic lucencies compared to Panel k, highlighting improvements in lung morphology and reduced inflammation after exercise.
Exercise training attenuates inflammation and modulates miRNA expression in COPD patients
To evaluate the levels of inflammatory factors, we performed ELISA on peripheral blood samples collected from COPD patients before and after the exercise intervention. The results revealed a significant decrease in the expression of inflammatory markers, including IL-17, IL-1β, and IL-6 (Figure 2(a) to (d)), following the exercise training, while no significant downregulation was observed in TNF-α levels. Additionally, we utilized high-throughput RNA sequencing to analyze the transcriptome of peripheral blood leukocytes. From the cohort, we selected three patients from these 11 COPD patients who showed remarkable improvement and compared their miRNA profiles before and after the exercise training to identify potential biomarkers and underlying mechanisms. Based on our screening criteria (log2 FoldChange ≥ 1.0 and p-values < 0.05), six out of the eight differentially expressed miRNAs (DE-miRNAs) were upregulated, while two were downregulated after exercise (Figure 2(f)). Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment (Figure 2(g)) and Gene Ontology (GO) analyses are presented in Supplemental Figure S1.

Exercise ameliorates COPD-induced lung injury by inhibiting inflammation and abnormal miRNA expression. (a–d) ELISA detection shows the expression of inflammatory factors IL-6, IL-17, TNF-α, and IL-1β. Data are expressed as means ± SD. (e) RT-qPCR detection shows the expression of miR-124-3p miRNA. Data are expressed as means ± SD. (d) Heat map detection shows that exercise results in abnormal expression of miRNAs, aPR means COPD patients after exercise, and bPR means COPD patients before exercise. (e) Gene ontology (GO) enrichment of different expressed miRNAs. Data are expressed as means ± SD.
Although our transcriptome sequencing did not detect miR-124-3p, RT-qPCR analysis revealed a significant increase in the expression of miR-124-3p in COPD patients following exercise, compared to pre-exercise levels (Figure 1(e)). These findings suggest that exercise training not only reduces systemic inflammation but also modulates specific miRNA expressions, which may contribute to the improvement of COPD symptoms. Building on these results, we next aimed to validate these mechanisms further using a mouse model of COPD, and to investigate whether miR-124-3p is involved in this process and how it functions in this context.
Exercise mitigates COPD-induced lung injury by inhibiting inflammation and modulating macrophage polarization in a murine model
To elucidate the mechanism by which exercise training suppresses the release of inflammatory factors, we measured the expression of key inflammatory markers (including TNF-α, IL-6, and IL-1β) in the lung tissue of cigarette smoke (CS)-induced COPD mice using ELISA. The results showed a significant upregulation of these inflammatory factors in COPD mice, while exercise training effectively reversed the COPD-induced inflammatory response (Figure 3(a) to (c)). Furthermore, RT-qPCR analysis revealed a significant reduction in the expression of miR-124-3p in lung tissue of COPD mice compared to the control group, with exercise intervention restoring its expression (Figure 3(d)). Immunofluorescence staining of lung tissue demonstrated an increase in the expression of iNOS, a marker of M1 macrophage polarization, in CS-induced COPD mice, which was significantly decreased after exercise intervention (Figure 3(e) and (f)). TUNEL staining results showed that exercise significantly reduced cell apoptosis in the lung tissue of COPD mice (Figure 3(g) and (h)). These findings suggest that exercise alleviates COPD-induced chronic lung damage by suppressing inflammation, regulating macrophage polarization, and reducing cell apoptosis. To further explore the underlying mechanisms, we next investigated the role of miR-124-3p-mediated competing endogenous RNA (ceRNA) networks in the protective effects of exercise against COPD.
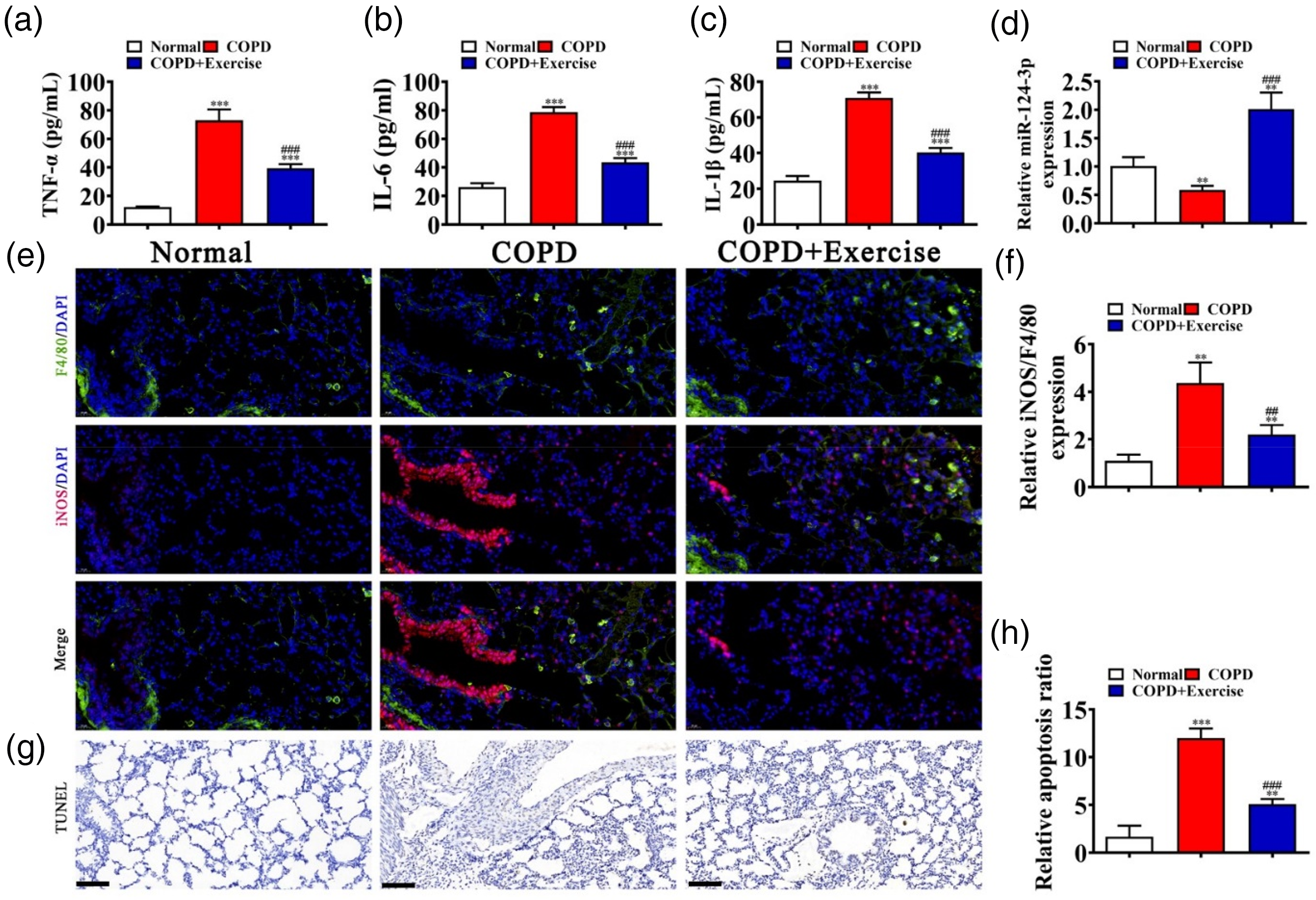
In vivo experimental detection shows that exercise ameliorates COPD-induced lung injury by inhibiting inflammation and regulating the macrophage phenotype. (a–c) ELISA detection shows the expression of inflammatory factors TNF-α, IL-6, and IL-1β. Data are expressed as means ± SD. (d) RT-qPCR detection shows the expression of miR-124-3p in lung tissues from control animals, in mice with COPD, and in exercised COPD mice. Data are expressed as means ± SD. (e and f) Immunofluorescence detection shows the macrophage phenotype as indicated by iNOS expression. Data are expressed as means ± SD. *** p < 0.001, ** p < 0.01 versus controls. ### p < 0.001 versus COPD. Scale bar: 20 μm. (g and h) TUNEL staining shows apoptosis in mouse lung tissues. Data are expressed as means ± SD. ** p < 0.01 versus controls. ## p < 0.01 versus COPD. Scale bar: 20 μm.
Identification of ERN1 as the downstream target of miR-124-3p via RAW264.7 cells
Accumulating research has confirmed that miR-124-3p can modulate mRNA expression.14–16 To explore the mechanism underlying the increased expression of miR-124-3p following exercise training, we utilized bioinformatics data (https://starbase.sysu.edu.cn/) to predict the downstream genes of miR-124-3p. ERN1 3'-UTR (encoding IRE1) was selected as the interacting target gene, as previous studies have shown that abnormal expression of ERN1 is associated with the progression of pulmonary disease and impairs the differentiation and cytokine secretion of TH17 cells. We then constructed a luciferase reporter vector in RAW264.7 cells, demonstrating that miR-124-3p decreased luciferase activity (Figure 4(a) and (b)), suggesting that ERN1 is a downstream target of miR-124-3p. Additional experiments revealed that miR-124-3p could interact with the 3'-UTR of ERN1 in mice (Figure 4(c) and (d)), further supporting that ERN1 is a downstream target of miR-124-3p. RT-qPCR analysis showed that miR-124-3p expression increased after transfection of miR-124-3p mimics into RAW cells. However, overexpression of ERN1 did not reverse the expression of miR-124-3p (Figure 4(e)), also indicating that ERN1 is a target gene of miR-124-3p. Furthermore, miR-124-3p mimic transfection in RAW cells led to a decrease in ERN1 expression (Figure 4(f)). Therefore, ERN1 has been confirmed as the downstream gene of miR-124-3p.

ERN1 is the downstream target of miR-124-3p. (a) Bioinformatics analysis predicted binding sites in 3'-UTR-ERN1 for miR-124-3p. The mutant (Mut) version of 3'-UTR-ERN1 is shown. (b) Relative luciferase activity was determined 48 hours after transfection of RAW 264.7 cells with miR-124-3p mimic/NC or ERN1 wild-type/Mut. Data are expressed as means ± SD. *** p < 0.01. (c) Prediction of miR-124-3p binding sites in the 3'-UTR of mouse ERN1. The mutant version of 3'-UTR-ERN1 is shown. (d) Relative luciferase activity 48 hours after transfection of RAW cells with miR-124-3p mimic/NC or 3'-UTR-ERN1 wild-type/Mut. Data are expressed as means ± SD. *** p < 0.001. (e and f) RT-qPCR detection shows the expression of miR-124-3p and ERN1 in Raw264.7 cells. Data are expressed as means ± SD. *** p < 0.001 versus NC. ### p < 0.001 versus mimic.
ERN1 and miR-124-3p regulate macrophage phenotype
To further elucidate the role of miR-124-3p in modulating the pro-inflammatory phenotype of macrophages in response to CS exposure, we conducted overexpression experiments in CSE-treated RAW264.7 cells. Consistent with our hypothesis, ELISA analysis revealed a decrease in the expression of inflammatory factors IL-6, TNF-α, and IL-1β upon miR-124-3p overexpression following CSE exposure. Conversely, overexpression of ERN1 led to an increase in the expression of these inflammatory factors (Figure 5(a) to (c)). Western blot analysis showed that iNOS was downregulated upon miR-124-3p mimic transfection. Conversely, overexpression of ERN1 led to an increase in iNOS expression (Figure 4(d)), further supporting these findings. Additionally, RT-qPCR analysis of blood leukocyte samples from COPD patients revealed a significantly higher relative expression of ERN1 compared to the normal control group. Although there was a trend toward a decrease in ERN1 expression after exercise, this change did not reach statistical significance (Figure 5(e)). Notably, the expression level of ERN1 showed a weak negative correlation with miR-124-3p expression, with a correlation coefficient of −0.11 (Figure 5(f)).

Upregulation of ERN1 reverses the inhibitory effect of miR-124-3p on the M1 macrophage phenotype and inflammatory factor expression induced by CSE. (a–c) ELISA detection shows the expression of inflammatory factors TNF-α, IL-6, and IL-1β. Data are expressed as means ± SD. ** p < 0.01, *** p < 0.001 versus CSE. # p < 0.05, ## p < 0.01, ### p < 0.001 versus CSE + mimic. (d) Western blot detection shows the expression of iNOS in Raw264.7 cells. Data are expressed as means ± SD. * p < 0.01 versus CSE. (e) RT-qPCR detection shows the expression of ERN1 in human blood leucocytes. Data are expressed as means ± SD. ** p < 0.01, *** p < 0.001 versus NC. CSE represents cigarette smoke extract stimulation; CSE + mimic indicates cigarette smoke extract stimulation combined with miR-124-3p overexpression; CSE + mimic + ERN1 refers to cigarette smoke extract stimulation combined with both miR-124-3p and ERN1 overexpression; NC represents the healthy control group; COPD represents patients with chronic obstructive pulmonary disease; COPD + exercise indicates COPD patients after exercise training.
Discussion
Exercise training has emerged as a crucial strategy in addressing the growing global challenges posed by aging and metabolic disorders, including COPD. Extensive research highlights the profound effects of exercise on anti-inflammatory, anti-infective, and immunometabolic functions.25–28 Our study demonstrates that exercise training significantly improves exercise tolerance and circulatory function in COPD patients while attenuating systemic inflammation and modulating miR-124-3p expression. In a murine model of COPD, exercise alleviated lung injury by suppressing inflammation and inhibiting the pro-inflammatory M1 macrophage phenotype. Furthermore, we identified ERN1 as a downstream target of miR-124-3p in RAW264.7 cells, emphasizing the role of the miR-124-3p/ERN1 axis in regulating inflammatory responses in macrophages and COPD patients. These findings further support the role of the miR-124-3p/ERN1 axis in mediating the anti-inflammatory effects of exercise in COPD.
The improvement in exercise tolerance and circulatory function observed in COPD patients following exercise training is consistent with previous studies demonstrating the beneficial effects of exercise on cardiorespiratory fitness and muscle function.23,24 Exercise training likely enhances oxygen utilization and delivery, thereby alleviating the symptoms of dyspnea and fatigue commonly experienced by COPD patients. Additionally, the attenuation of systemic inflammation by exercise training may contribute to the overall improvement in patient outcomes, as chronic inflammation is a key driver of COPD progression. Beyond direct cardiorespiratory enhancements, exercise ameliorates COPD through broader systemic effects. It offers significant anti-inflammatory benefits, 29 crucial given the systemic nature of inflammation in COPD. 30 Exercise can also induce beneficial shifts in the immune system, modulating immune cell populations and their inflammatory mediator production. 31 Furthermore, exercise promotes mitochondrial biogenesis and function,32–34 which can enhance cellular energy production and reduce oxidative stress, both of which are implicated in COPD pathogenesis. Moreover, exercise-induced exerkines, metabolic adaptations, and microRNA changes contribute to lung tissue repair and overall disease modulation, further supporting its role in COPD management.10–13
miR-124-3p is a highly conserved microRNA (miRNA) with a similar sequence across multiple species. In mammals, it is encoded by several loci, primarily located on chromosomes 8p23.1, 8q12.3, and 20q13.33. miR-124-3p is abundantly expressed in the nervous system, where it plays a crucial role in neuronal differentiation and microglial activation. Moreover, studies have shown that miR-124-3p exhibits dysregulated expression in various pulmonary diseases, including acute respiratory distress syndrome (ARDS), lung cancer, and pulmonary fibrosis. Although there is no consensus on the expression levels of miR-124-3p in lung tissue and peripheral blood, these findings suggest that miR-124-3p may play a significant role in the pathogenesis and progression of lung diseases.11,16 For example, previous studies have found that miR-124-3p suppresses NLRP3 inflammasome activation by targeting TRAF6 and can enhance M2 macrophage polarization. Our findings further highlight the central role of miR-124-3p in mediating anti-inflammatory effects. The upregulation of miR-124-3p likely suppresses the expression of iNOS, a key regulator of macrophage function. By targeting ERN1, miR-124-3p may inhibit the activation of pro-inflammatory signaling pathways, thereby promoting the polarization of macrophages toward the anti-inflammatory M2 phenotype, which is consistent with the conclusions of other studies.18,20,35,36 This phenotypic shift is critical for resolving inflammation and protecting lung tissue from injury in COPD.
Through bioinformatics analysis and in vitro experiments, we identified ERN1 as a downstream target of miR-124-3p, establishing a mechanistic link between exercise training and inflammation modulation. ERN1, also known as IRE1α, is a key sensor of endoplasmic reticulum stress and plays a role in activating pro-inflammatory pathways such as NF-κB and JNK. Notably, ERN1 expression was elevated when M2 polarization markers were low. While ERN1 has been previously linked to inflammation and endoplasmic reticulum stress, its regulation by miR-124-3p in the context of exercise and COPD has not been explored. Beyond COPD, the miR-124-3p/ERN1 axis is involved in other inflammatory diseases such as neuroinflammation, spinal cord injury, rheumatoid arthritis, and inflammatory bowel disease.16,37,38 In these conditions, miR-124-3p reduces inflammation by targeting ERN1 and other downstream molecules like TRAF6, STAT3, and YY1, suggesting its broader therapeutic potential. These findings provide new insights into the molecular mechanisms underlying the anti-inflammatory effects of exercise and highlight the miR-124-3p/ERN1 axis as a potential therapeutic target for COPD management.
Several limitations of this study should be acknowledged. First, despite efforts to control for confounding, the observational nature and small sample size of this study may limit the generalizability of our findings. The correlation between miR-124-3p and ERN1 in peripheral blood leukocytes was weak, potentially attributable to the small sample size. Our findings are based on a combination of human and murine models, and further validation in larger clinical cohorts is necessary to confirm the translational significance of the miR-124-3p/ERN1 axis. Second, the extraction of total RNA in our high-throughput sequencing experiments was performed using TRIzol, which may have affected miRNA recovery, potentially leading to the absence of miR-124-3p in the transcriptomic data. However, we optimized the extraction method in subsequent PCR experiments to improve miRNA recovery. Third, while our study focused on miR-124-3p, exercise may exert anti-inflammatory effects through multiple molecular pathways, involving other miRNAs and downstream targets. Future research should explore the potential interactions between miR-124-3p and other miRNAs, as well as the role of additional downstream targets in mediating the beneficial effects of exercise training.
Conclusion
In conclusion, our pilot study suggests that exercise training may exert beneficial effects in COPD by upregulating miR-124-3p, suppressing ERN1, and modulating macrophage polarization. These preliminary findings may provide insight into the molecular mechanisms underlying exercise-induced anti-inflammatory effects and indicate the potential of the miR-124-3p/ERN1 axis as a therapeutic target. Furthermore, our results support the possible role of regular physical activity as a non-pharmacological intervention that may contribute to improved lung health and quality of life in COPD patients.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251360892 - Supplemental material for Exercise's protective role in chronic obstructive pulmonary disease via modulation of M1 macrophage phenotype through the miR-124-3p/ERN1 axis
Supplemental material, sj-docx-1-sci-10.1177_00368504251360892 for Exercise's protective role in chronic obstructive pulmonary disease via modulation of M1 macrophage phenotype through the miR-124-3p/ERN1 axis by Haizhu Zeng, Xiaoxiang Liu, Panpan Liu, Shuyin Jia, Guangyu Wei, Guo Chen and Lei Zhao in Science Progress
Footnotes
Acknowledgements
We thank Mr Chuangjian Duan for his assistance in conceptualizing and formatting the figures in our manuscript.
Ethical considerations and informed consent
The Ethics Committee of Shanghai Gongli Hospital approved the animal experiments.
Author contributions
HZZ and XXL contributed to the study equally. XXL completed the CT score and the assessment of pre- and post-exercise imaging changes for the patients. PPL and HZZ collected data and performed data analysis. All authors contributed to the interpretation of the data and the completion of figures and tables. All authors contributed to the drafting of the article and final approval of the submitted version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Discipline Construction project of Shanghai Pudong New Area Health Commission, Commission Pudong New Area health and family planning commission project, Shanghai Municipal Health and Family Planning Commission general Project (grant numbers PWZxq2022-12, PW2021A-20, PW2022A-02, and 202040191).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request. The high-throughput sequencing data can be downloaded from the GEO database (GSE235174)
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
