Abstract
Objective
The objectives of this study were to identify early risk factors of circulatory assistance by venoarterial extracorporeal membrane oxygenation (VA-ECMO) on admission to intensive care unit (ICU) in population with severe cardiotoxic poisoning and to compare group characteristics with and without assistance.
Method
All patients included had severe clinical cardiotoxic poisoning, defined by hemodynamic failure requiring treatment with one or more catecholamines, in order to identify factors for circulatory assistance by multivariable logistic regression using bootstrap and supervised hierarchical classifiers methods. This single-center and retrospective study was carried out over 10 years from January 2012 to February 2022. We excluded all the patients with benign poisoning.
Results
One-hundred forty-six cases were enrolled, including 24 with VA-ECMO (16.4%) and 122 with conventional treatment (83.6%). In circulatory support group, mortality, pneumoniae rate, intensive care length of stay and duration of mechanical ventilation were higher than conventional treatment group. Predictive factors for VA-ECMO on admission were mean arterial pressure ≤ 60 mmHg, lactatemia > 4.1 mmol/L and left ventricular ejection fraction ≤ 20%. The estimation of the classification and regression tree analysis was 91.1%.
Conclusions
Patients admitted for severe cardiotoxic poisoning presenting with hypotension, hyperlactatemia, and left ventricular dysfunction were more frequently treated with extracorporeal life support, consistent with clinical severity. Early identification of such high-risk profiles is essential to prioritize timely referral to expert centers for optimal management.
Introduction
Intentional drug intoxication is a common reason for admission to the emergency department or intensive care unit (ICU). 1 Cardiovascular drugs are the fourth most common agent (7.06%) involved in adult poisoning consultations but above all the most fatal with a mortality rate close to 12.03%. 1 The occurrence of organ failure requires admission to ICU and shock is the most serious feature and worsens the prognosis.1,2 Its mechanism may involve direct cardiac toxicity, negative inotropic effect, high-grade conduction or ventricular rhythm disturbances, or systemic vasoplegia or more often be multifactorial. 3 Management is mainly symptomatic and depends on pharmacology, the supposed dose ingested and the clinical impact. Recently, the guidelines on therapeutic strategy for cardiotoxic poisoning have been updated by scientific societies. 4
Pharmacological treatment is generally sufficient to control shock.5,6 In some cases, the occurrence of refractory cardiogenic shock, persistent cardiac arrest or severe ventricular arrhythmia may require circulatory assistance while awaiting the restoration of satisfactory cardiac function.3,7 Patients experiencing cardiogenic shock may derive greater benefit from circulatory support compared to those with vasoplegic or hypovolemic shock. Veno-arterial extracorporeal membrane oxygenation (VA-ECMO) maintains systemic perfusion flow pending functional recovery and ensures tissue perfusion.2,8 The literature remains based literature review, on human case series and prospective randomized studies in animals, with a potential benefit on mortality if implantation is early.4,9–11 Thus, the level of evidence for the use of VA-ECMO remains low.
In French and American guidelines, experts suggest its use, compared with conventional treatment, to improve survival in cases of cardiac arrest or refractory cardiogenic shock.4,12,13 We hypothesize that delayed initiation of VA-ECMO in refractory cardiogenic shock may fail to reverse established multiorgan failure, ultimately resulting in increased mortality. Can we detect earlier patients requiring assistance by VA-ECMO after severe cardiotoxic poisoning and what criteria should be used?
In this context, the primary objective was to identify the early predictive factors, on admission to ICU, for circulatory assistance by VA-ECMO in cases of severe cardiotoxic poisoning. The secondary objectives were to compare patient characteristics, therapeutic management and to assess patient outcome with or without VA-ECMO.
Methods
We conducted a retrospective cohort study in Toulouse university hospital (France) from January 2012 to February 2022. This study adhered to the 1975 Declaration of Helsinki as revised in 2024 and its later amendments and approved by the Commission Nationale de l’Informatique et des Libertés (CNIL n°2088490v0, February 2015). The data were collected using the ICCA® software in accordance with the ethical standards of our hospital's institutional review board. The requirement for informed consent was waived due to the anonymization of all patient data. This observational study was designed and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 14
We included all adults admitted in ICU for severe poisoning with cardiotoxic drug, defined by cardiogenic or vasoplegic shock or severe arrhythmia requiring one or more catecholamines for hemodynamic support after optimal fluid resuscitation. The diagnosis was based on clinical, biological, and ultrasound data and/or continuous cardiac output monitoring methods. Cardiogenic shock was defined, according to the Society for Cardiovascular Angiography and Intervention guidelines, as reduced cardiac output with clinical and/or biological signs of hypoperfusion. 13 Vasoplegic shock is a circulatory failure characterized by low systemic vascular resistance responsible for tissue hypoperfusion, in the presence of normal or increased cardiac output. 15 Severe arrhythmia was defined as high grade conduction block (Mobitz second or third degree) or rhythm disorders (ventricular tachycardia or fibrillation) or membrane-stabilizing activity with QRS duration ≥ 120 ms responsible for hemodynamic failure. We excluded from the analysis all patients admitted for benign poisoning, characterized by the absence of shock and severe arrhythmia. These patients were admitted early to ICU because they reported a high intake of cardiotoxic drugs that could lead a cardiovascular collapse.
The outcomes were patients’ characteristics, agents involved in cardiovascular poisoning (beta-blockers, calcium channel blockers, renin angiotensin aldosterone system antagonists, antiarrhythmic agents, others). We collected the reason for admission in ICU, clinical data, treatment administered, ICU outcomes (mechanical ventilation, renal replacement therapy, VA-ECMO support, length of stay in ICU and in the hospital, and mortality in ICU).
The intoxication was characterized by: substance, quantity supposedly ingested, pharmacological class (with or without membrane-stabilizing activity), specific treatments initiated (antidotes, hemodynamic support) and clinical, electrocardiographic, echocardiographic, and biological repercussions.
VA-ECMO has been used exclusively in our regional center of expertise since 2012, with an experienced team consistently recruiting an average of 30 patients per year. Some aspects of ECMO management and data collection procedures in this study are aligned with institutional protocols previously described by Delmas et al. who investigated left atrial unloading in VA-ECMO patients at our center. 16 The VA-ECMO characteristics were detailed: indication, time of implantation in relation to the time of admission to hospital, clinical characteristics of the patient at the time of implantation, duration of support and technique complications. VA-ECMO was initiated, in accordance with the 2021 Extracorporeal Life Support Organization guidelines, within 6 h of the onset of drug-refractory cardiogenic shock with persistent hemodynamic collapse. 17 These indications and their management were based on local protocol and after multidisciplinary decision including cardiologists, intensivits, and cardiac surgeons. The VA-ECMO implantation in femoral vessels was performed with a modified Seldinger technique and addition of a systematic ipsilateral superficial femoral artery reperfusion. Flow was adjusted to target mean arterial pressure ≥ 65 mmHg. Dobutamine could be continued to maintain aortic valve opening. Anticoagulation was systematically started by intravenous unfractionated heparin (anti-Xa level target 0.3 to 0.6 unity/mL).
The distribution of continuous variables was evaluated by examining skewness and kurtosis coefficients. The study population was initially divided into two groups based on whether or not circulatory support was used. In a second step, the population of patients who received VA-ECMO support was specifically analyzed, with comparison between survivors and nonsurvivors. Results were expressed as median and interquartile for the quantitative variables, and as numbers and percentages (%) for the qualitative variables. Groups were compared using nonparametrical Mann–Whitney U test for continuous variables and Chi2 test or Fisher's exact test for qualitative variables as suitable. Survival rates were analyzed using the Kaplan–Meier method and the log-rank test. The discriminant value of the covariates of interest based on the VA-ECMO group was assessed by studying the receiver operating characteristic (ROC) curves and their associated areas under the curve (AUC). Binary logistic regression for the prediction of allocation to VA-ECMO therapy was performed adjusting for multiple covariates, using a backward elimination procedure which consists of including all the chosen variables then gradually eliminating those that were nonsignificant. Considering the limited number of patients requiring ECMO support, the accuracy and calibration of the model were assessed using 4000 bootstrap resamples for internal validation. We performed this validation by using the Hosmer–Lemeshow test (χ2 goodness of fit), the prediction percentage of the model, the Nagelkerke pseudo r-squared. The probabilities of the tested model were also evaluated by an ROC curve procedure. To better identify and visualize risk stratification for VA-ECMO requirement, a population partitioning using a segmentation tree was established by classification and regression trees (CART) method completed by study of independent variable importance. CART has been used extensively as an alternative to the classical linear and additive prediction models. Results are presented in tree form of a decision rule with a hierarchical sequential structure that can be easily understood and applied in clinical practice. The study was conducted on IBM® Corp. Released 2016. IBM SPSS Statistics for Windows, Version 24.0. Armonk, NY: IBM Corp. p < .05 was considered to be statistically significant.
Results
During study period, 222 patients were admitted on ICU for suspected severe cardiotoxic poisoning. Among the 146 patients were included in the final analysis, 24 (16.4%) received VA-ECMO support and 122 (83.6%) received medical conventional treatment. The inclusion flow chart and clinical characteristics are presented in Figure 1 and Table 1, respectively. The patients receiving VA-ECMO support had higher severity levels on SOFA and SAPS 2 scores. There were no differences in the medical and psychiatric histories. The primary reason for ICU admission was vasoplegic shock (n = 84, 57.5%), followed by disorders of consciousness (n = 46, 31.5%), and the presence of cardiogenic shock (n = 30, 20.5%) or the supposed ingested toxic dose (n = 30, 20.5%). Patients receiving VA-ECMO more often presented with cardiogenic shock (n = 14, 58.3% vs n = 16, 13.1%, p < .0001). Eighty percent (n = 117) of patients had ingested at least two toxic substances. Patients receiving VA-ECMO had taken more nondihydropyridine calcium channel blockers compared to patients with conventional treatment (n = 9, 37.5% vs n = 22, 18%, p = .0336). Among the cardiotoxic poisoning, the ingestion of calcium channel blockers was present in 73 cases (50%), 65 cases for beta-blockers (44.5%), and 16 cases of antiarrhythmic (flecainide, amiodarone, digoxin) (11%). The other drugs ingested and the characteristics of poisoning with cardiotoxic drugs are presented in Supplemental Tables S1 and S2 respectively.

Flow chart of study population.
Characteristics of patients admitted to ICU for severe poisoning with cardiotoxic drugs.

Abbreviations: VA-ECMO: venoarterial extracorporeal membrane oxygenation; ACE: angiotensin converting enzyme; ARB: angiotensin receptor blocker; BMI: body mass index; CCB: calcium channel blocker; DHP: dihydropyridine; HGCD: high grade conduction disorders; ICU: intensive care unit; SAPS II: Simplified Acute Physiology Score 2; SOFA: sequential organ failure assessment.
Data are expressed as median (interquartile range) and percentage, depending on the variable of interest.
The comparison of clinical and paraclinical data from admission and from Day 1 is presented in Table 2. Regarding the initial electrocardiographic and echocardiographic data, patients with VA-ECMO presented a sinus rhythm in 50% of cases (n = 12) compared to 88.5% (n = 108) in the non-ECMO group (p < .0001). The median QRS duration was 120 [80–170] ms in the ECMO group versus 80 [80–98] ms in the non-ECMO group (p = .0004). The minimum left ventricular ejection fraction (LVEF) was significantly lower in the VA-ECMO assisted group: 17.5 [10–50] % versus 55 [50–60] % (p < .0001) (Table 2).
Comparison of clinical and paraclinical data between group conventional treatment and VA-ECMO at admission and day 1.

Abbreviations: FiO2: fraction inspired oxygen; LVEF: left ventricular ejection function; MV: mechanical ventilation; PaO2: partial pressure of arterial oxygen; VA-ECMO: veino-arterial extracorporeal membrane oxygenation.
Data are expressed as median (interquartile range) and percentage, depending on the variable of interest.
At admission, patients who received VA-ECMO had more organ failures. Regarding the laboratory results on admission, patients assisted by VA-ECMO had higher levels of circulatory failure markers. They received more vascular filling, vasopressors, inotropes, and continuous renal replacement therapy (CRRT). Therapies administered on admission are described in Table 3, including more sodium bicarbonate, high dose insulin, electrical cardioversion, and electrosystolic stimulation in the VA-ECMO group.
Comparison of therapy and outcome between groups conventional treatment and VA-ECMO.
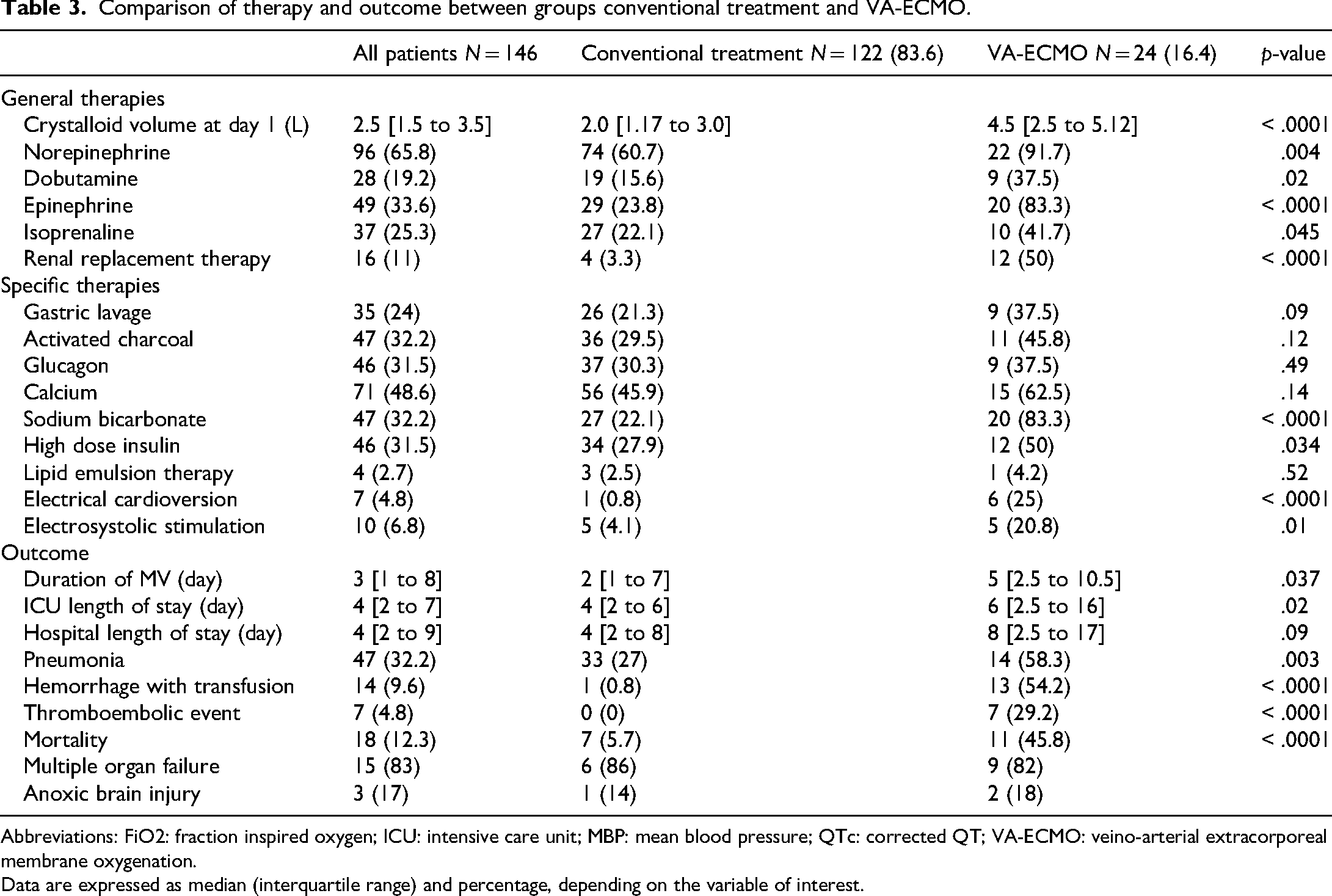
Abbreviations: FiO2: fraction inspired oxygen; ICU: intensive care unit; MBP: mean blood pressure; QTc: corrected QT; VA-ECMO: veino-arterial extracorporeal membrane oxygenation.
Data are expressed as median (interquartile range) and percentage, depending on the variable of interest.
Group supported by VA-ECMO had a higher mortality rate (n = 11, 45.8%) compared to another group (n = 7, 5.7%) (p < .0001). They also had longer mechanical ventilation duration and ICU stay, as well as more ventilator-associated pneumonias. Patients with VA-ECMO had more hemorrhagic complications requiring blood transfusion (n = 13, 54.2% vs n = 1, 0.8%, p < .0001) and thromboembolic events (n = 7, 29.2% vs n = 0 (0%), p < .0001 (Table 3).
In the subgroup with VA-ECMO (Supplemental Table S3), the survival rate was 54% (n = 13). There was no difference in the timing of VA-ECMO initiation between the survival (4.5 [1.7–13] hours) and death (15 [3.6–25.3] hours) groups (p = .18). Surviving patients had more dobutamine on VA-ECMO initiation (n = 4, 30.8% vs n = 0, 0%, p = .049), while there was no significant difference on mortality among patients receiving epinephrine (survivors n = 8, 61.5% vs dead n = 10, 90.9%, p = .11).
At Day 1 after admission, the improvement of organ failure was significantly correlated with survival, as demonstrated by correction of arterial pH in survivors (pH = 7.45 [7.37–7.51] vs 7.28 [7.2–7.38] in nonsurvivors, p = .012), correction of hyperlactatemia in survivors (1.7 [1.2–3.3] mmol/L vs 4.2 [3.0–12.0] mmol/L in nonsurvivors, p = .01), and of base excess in survivors (−3.6 [−8.4 to 0.6] mmol/L) vs −10.5 [−14.7 to −6] in nonsurvivors (p = .008) (Supplemental Table S3).
The multivariate analysis of covariates, on ICU admission, for VA-ECMO assistance were lactatemia >4.1 mmol/L (OR = 8.6, 95% CI [2.33–31.81], p = .0001), MAP ≤ 60 mmHg (OR = 5.37, 95% CI [1.39–20.74], p = .006) and LVEF ≤ 20% (OR = 20.50, 95% CI [5.08–82.77], p = .0001). AUC of the model was 0.91, 95% CI [0.85–0.96], p < .0001 (Table 4). The area under curves, sensitivity, specificity, positive and negative predictive values of the variable of interest are presented in Supplemental Table S4.
Multivariate analysis of risk factors for VA-ECMO (logistic regression with bootstraping—backward method).

Abbreviations: AUC: area under curve; CI: confidence interval; LVEF: left ventricular ejection fraction; MAP: mean arterial pressure; VA-ECMO: veino-arterial extracorporeal membrane oxygenation.
Note: Bootstrapa: Unless otherwise noted, bootstrap results are based on 4000 bootstrap samples.
Results of CART are shown in Figure 2. The estimation percentage of CART analysis was 91.1%. For example, patient admitted on ICU for severe cardiotoxic poisoning with MAP ≤ 60 mmHg, LVEF ≤ 20% and lactatemia > 4.1 mmol/L has 90% probability of being assisted by VA-ECMO. On the other hand, patient with MAP > 60 mmHg, LVEF > 20%, with lactate level on admission ≤ 4.1 mmol/L has probability of VA-ECMO implementation of 1.4%.

Partitioning of patients according VA-ECMO treatment according to their initial MAP (mmHg), lactatemia level (mmol/L), and their LVEF (%).
The Supplemental Figure S1 presented the normalized importance of independent variable. The variables ranked in decreasing order according to percentage of normalized importance were LVEF ≤ 20%, initial lactatemia > 4.1 mmol/L, initial MBP ≤ 60 mmHg and QRS duration >110 ms.
Discussion
This study investigated predictive factors of circulatory assistance by VA-ECMO in an ICU cohort over 10 years with severe cardiotoxic poisoning. VA-ECMO patients had higher incidence of clinically and biologically organ failures, severe electrocardiogram abnormalities, and received more catecholamine support and CRRT than conventional treatment patients. Group with VA-ECMO had longer durations of mechanical ventilation, ICU stays, and higher mortality rates compared to conventional treatment group. The improvement of organ failures with ECMO at 24 h after admission was associated with good prognosis. According to the multivariate model, the predictive factors, on ICU admission, of VA-ECMO were MAP ≤ 60 mmHg, LVEF ≤ 20% and initial hyperlactatemia >4.1 mmol/L. In addition, the CART analysis provided high probability of VA-ECMO when three predictive factors were present on ICU admission and with high predictability.
There is no consensus in the existing literature regarding severe cardiotoxic poisoning definition, despite its use in guidelines. 4 Some recent publications propose clinical criteria for identifying severe cases.3,4 Our definition excluded deliberately the notion of presumed ingested dose because some patients remained asymptomatic even though they reported significant intakes of cardiotoxic drugs. The reality of the intake is questioned.
Most studies of cardiotoxic poisonings consist of literature reviews, cohorts of only patients assisted with VA-ECMO support or case report.7,12,18–21 The originality of our study resided in our primary results presented using CART model and our cohort included only severe intoxications with hemodynamic failure with or without circulatory assistance. Consequently, intoxications with no significant clinical impact, suggesting little or no intake cardiotoxic drugs, were not included. This approach enabled us to study specifically the predicting factors of VA-ECMO in severe cardiotoxic poisoning.
On admission, VA-ECMO group was more severe, according to SAPS II and SOFA scores, had more multiple organ dysfunction and received more interventions such as EERC. The initial brain disorders observed could be attributed to hypoperfusion resulting from hemodynamic failure or multidrug intoxication, common in our study. Peripheral circulatory support is appropriate in cases of heart pump failure to restore organ perfusion.4,17 Initial hyperlactatemia is recognized as a reliable prognostic marker in severe intoxications, except in cases involving beta-blockers, where relatively low lactate levels may be observed despite severe hemodynamic failure. 22 Special attention should be given to such cases.
Deaths in conventional treatment group were few and were attributed to either noncardiac or cardiac reasons, and occurred in patients rejected for VA-ECMO because of their advanced age and/or comorbidities. Our VA-ECMO patients had a high mortality rate, 44.4% (eight deaths out of 18 patients) for refractory cardiogenic shock and 50% (three deaths out of six patients) for refractory cardiac arrest. These data are in line with the literature, with figures ranging from 18.5% to 53% for refractory cardiogenic shock and from 44% to 75% for refractory cardiac arrest.3,6,12,21,23 For ethical reasons, there are no randomized controlled studies on circulatory assistance in cardiotoxic poisoning cases. Only animal studies have demonstrated benefit of VA-ECMO on survival, such as Freedman and Larkin works on canine and porcine models intoxicated with lidocaine and amitriptyline.9,10 Masson et al. demonstrated, in a retrospective cohort, that VA-ECMO assistance was associated with mortality reduction. 24 VA-ECMO restores perfusion to the organs, especially liver and kidneys, facilitating clearance of cardiotoxic substance.
Experts suggest use of VA-ECMO in cases of refractory cardiogenic shock or cardiac arrest.3,4,25,26 However, there is no consensus on refractory cardiogenic shock definition, which can delay the initiation of VA-ECMO impacting the prognosis. The literature offers several definitions integrating various criteria and standards, limiting their comparability. Voicu et al.’s recent review suggests different indications for circulatory assistance depending on the molecule involved. 3 Megarbane and al. and Duburcq integrate notions of arterial hypotension, left ventricular dysfunction, catecholaminergic support and peripheral organ failure but with different thresholds.27,28 Within our cohort, VA-ECMO indication was established according to ELSO guidelines and after multidisciplinary consultation including intensivists, cardiologists, and cardiac surgeons. 17 Delayed initiation of circulatory support in cases of cardiogenic shock may impact the prognosis due to worsening of multivisceral lesions. In retrospective study, patients who survived on VA-ECMO were assisted earlier than those who died. 28 Our results are in the same direction, but are not significant, probably due to lack of power.
Our patients requiring VA-ECMO were mainly composed of individuals poisoned by nondihydropyridine calcium channel blockers. These intoxications present a higher risk of circulatory assistance because of their prolonged half-life and their mixed effects, which can lead to vasoplegic and/or cardiogenic shock and conduction disorders. Various studies of patients on VA-ECMO confirm this finding.29,30
Serious poisoning by cardiotoxic substances received specific therapeutic management and resuscitation of the organs affected. International guidelines are limited and propose algorithms that may vary from one country to another.4,12,26 These recommendations have a low level of evidence, mainly derived from retrospective animal or human studies. It is suggested that patients be transferred to expert centers to ensure optimal management.
Our study has several limitations. Firstly, it was retrospective and took place over a 10-year period, which may lead to missing data and variations in the availability of information. The lack of standardization of VA-ECMO implementation criteria and advances in resuscitation techniques since 2012 may also introduce potential bias. There was no prior institutional experience with ECMO before 2012. Therefore, we acknowledge that experience gained over time, including improvements in patient selection, technical performance, and postcannulation management, may have influenced the results. Secondly, there was no uniform approach to ensuring consistent management of cardiotropic poisoning. Since this study, we have established a local protocol. Thirdly, as the only expert center in the region, patient admission times were variable, which may have contributed to delays in the start of treatment. Fourthly, the predictive factors of VA-ECMO were defined at a point in time: “on admission.” They didn't take account the clinical state evolution, including hemodynamic failure, after admission.
Conclusion
In this cohort, patients admitted for severe cardiotoxic poisoning presenting with hypotension, hyperlactatemia, and left ventricular dysfunction were more frequently treated with extracorporeal life support, consistent with clinical severity. These results would assist medical teams to detect as early as possible those patients most at risk of requiring circulatory assistance. It is recommended that patients be referred to an expert center as a priority, in order to optimize management and specific treatments administration, and to rapidly consider the VA-ECMO. Larger prospective multicenter studies are needed to confirm these results and to explore other factors likely to influence the decision to implant VA-ECMO in this population.
Supplemental Material
sj-docx-1-sci-10.1177_00368504251358951 - Supplemental material for Use of extracorporeal membrane oxygenation in severe cardiotoxic poisoning: Analysis of a cohort over 10 years
Supplemental material, sj-docx-1-sci-10.1177_00368504251358951 for Use of extracorporeal membrane oxygenation in severe cardiotoxic poisoning: Analysis of a cohort over 10 years by Baptiste Compagnon, Elsa Tardif, Vincent Pey, Bertrand Marcheix, François Labaste, Jean-Marie Conil, Vincent Minville and Fanny Vardon-Bounes in Science Progress
Footnotes
Acknowledgments
The authors would like to thank the clinicians, paramedics from the critical care unit, and Magali Centelles for her help in collecting the data.
ORCID iDs
Ethical considerations
This study adhered to the 1975 Declaration of Helsinki and its later amendments received ethical approval from the Commission Nationale de l’Informatique et des Libertés (CNIL n°2088490v0, February 2015). All patient information was deidentified and patient consent was not required. Patient data will not be shared with third parties.
Consent to participate
Consent to participate has been waived by the relevant Ethics Committee.
Authors contributions
BC and F.V.B. contributed equally; V.M. and F.V.B. supervised this work; BC, E.T., and V.P. participated in the study design and data acquisition; BC and F.V.B. participated in the drafting of the article; J.M.C. participated in statistical analysis; J.M.C., V.M., B.M., and F.L. provided feedback and improved the manuscript; B.M., V.M., and F.V.B. participated in supervision. All authors read and approved the final article. All authors read and had the opportunity to revise the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Baptiste Compagnon has full access to all data and accepts responsibility for the data integrity and the accuracy of its analysis.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
